MiR-21 Is a Novel Diagnostic and Prognostic Circulating Biomarker in Pleural Mesothelioma
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Patient Material
2.2. Staging and Treatment
2.3. Determination of Circulating miR-21 Levels by qRT-PCR in Plasma
2.4. Detection of miR-21 in PM Tissues by In Situ Hybridization (ISH)
2.5. Measurement of SMRP in Serum
2.6. Statistical Analysis
3. Results
3.1. Patient Characteristics
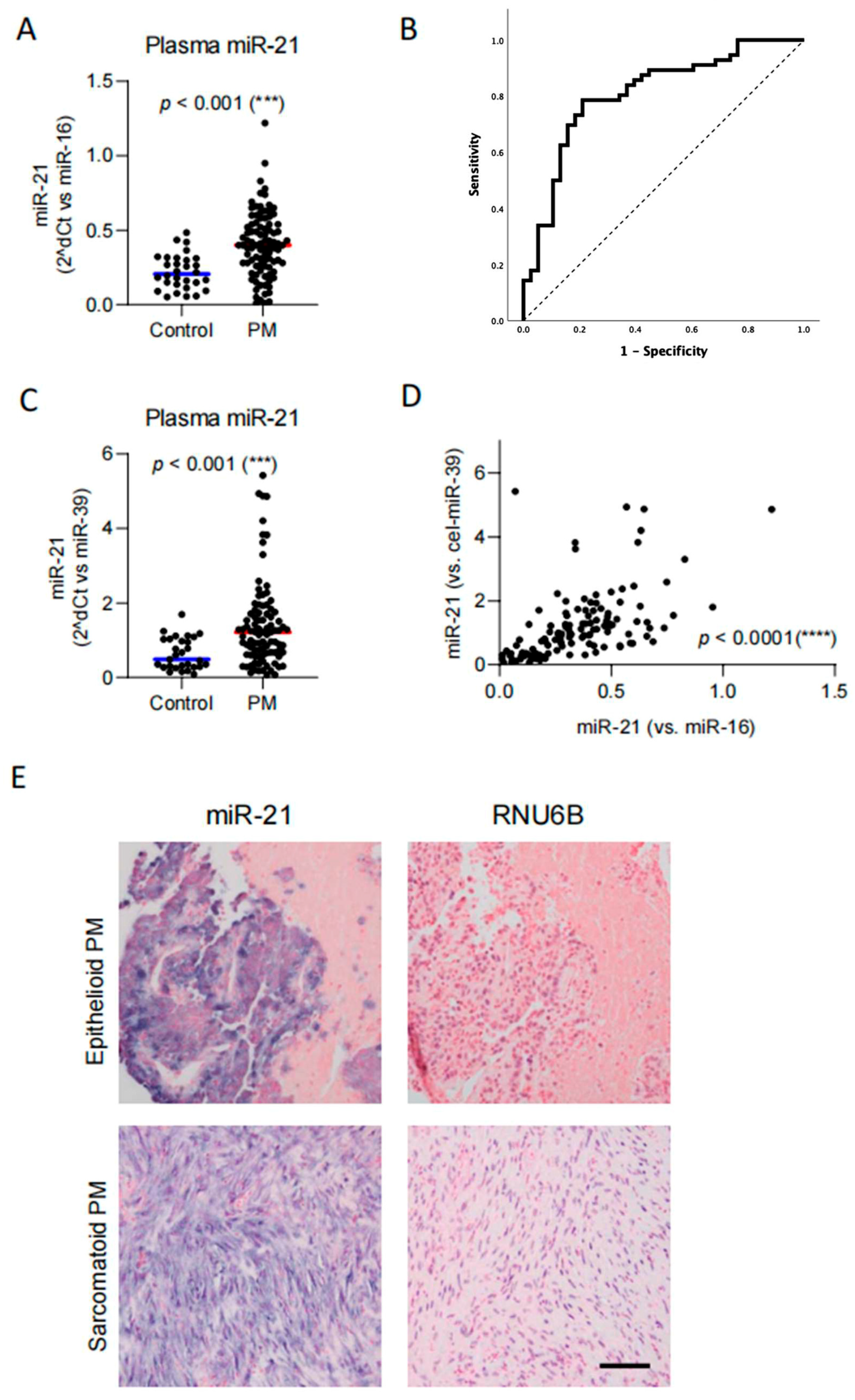
3.2. Circulating miR-21 Is Elevated in Patients with Pleural Mesothelioma
3.3. MiR-21 Is Present in Tumor Cells
3.4. Circulating miR-21 Levels Do Not Correlate with Histology, Disease Stage, Sex, Age, and SMRP
3.5. Circulating miR-21 Is an Independent Prognostic Factor in Pleural Mesothelioma
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kindler, H.L.; Rosenthal, A.; Giroux, D.J.; Nowak, A.K.; Bille, A.; Gill, R.R.; Pass, H.; Rice, D.; Ripley, R.T.; Wolf, A.; et al. The International Association for the Study of Lung Cancer Mesothelioma Staging Project: Proposals for the M Descriptors in the Forthcoming Ninth Edition of the TNM Classification for Pleural Mesothelioma. J. Thorac. Oncol. 2024, 19, 1564–1577. [Google Scholar] [CrossRef]
- Sofianidi, A.A.; Syrigos, N.K.; Blyth, K.G.; Charpidou, A.; Vathiotis, I.A. Breaking Through: Immunotherapy Innovations in Pleural Mesothelioma. Clin. Lung Cancer 2025, 26, 458–469. [Google Scholar] [CrossRef] [PubMed]
- Strbac, D.; Dolzan, V. Novel and Future Treatment Options in Mesothelioma: A Systematic Review. Int. J. Mol. Sci. 2022, 23, 1975. [Google Scholar] [CrossRef] [PubMed]
- Suraya, R.; Nagano, T.; Tachihara, M. Recent Advances in Mesothelioma Treatment: Immunotherapy, Advanced Cell Therapy, and Other Innovative Therapeutic Modalities. Cancers 2025, 17, 694. [Google Scholar] [CrossRef] [PubMed]
- Kindler, H.L.; Ismaila, N.; Bazhenova, L.; Chu, Q.; Churpek, J.E.; Dagogo-Jack, I.; Bryan, D.S.; Drazer, M.W.; Forde, P.; Husain, A.N.; et al. Treatment of Pleural Mesothelioma: ASCO Guideline Update. J. Clin. Oncol. 2025, 43, 1006–1038. [Google Scholar] [CrossRef]
- Sinn, K.; Mosleh, B.; Hoda, M.A. Malignant pleural mesothelioma: Recent developments. Curr. Opin. Oncol. 2021, 33, 80–86. [Google Scholar] [CrossRef]
- Robinson, B.W.; Creaney, J.; Lake, R.; Nowak, A.; Musk, A.W.; de Klerk, N.; Winzell, P.; Hellstrom, K.E.; Hellstrom, I. Mesothelin-family proteins and diagnosis of mesothelioma. Lancet 2003, 362, 1612–1616. [Google Scholar] [CrossRef]
- Hollevoet, K.; Reitsma, J.B.; Creaney, J.; Grigoriu, B.D.; Robinson, B.W.; Scherpereel, A.; Cristaudo, A.; Pass, H.I.; Nackaerts, K.; Rodriguez Portal, J.A.; et al. Serum mesothelin for diagnosing malignant pleural mesothelioma: An individual patient data meta-analysis. J. Clin. Oncol. 2012, 30, 1541–1549. [Google Scholar] [CrossRef]
- Johnen, G.; Gawrych, K.; Raiko, I.; Casjens, S.; Pesch, B.; Weber, D.G.; Taeger, D.; Lehnert, M.; Kollmeier, J.; Bauer, T.; et al. Calretinin as a blood-based biomarker for mesothelioma. BMC Cancer 2017, 17, 386. [Google Scholar] [CrossRef]
- Viscardi, G.; Di Natale, D.; Fasano, M.; Brambilla, M.; Lobefaro, R.; De Toma, A.; Galli, G. Circulating biomarkers in malignant pleural mesothelioma. Explor. Target. Antitumor Ther. 2020, 1, 434–451. [Google Scholar] [CrossRef]
- Sorino, C.; Mondoni, M.; Marchetti, G.; Agati, S.; Inchingolo, R.; Mei, F.; Flamini, S.; Lococo, F.; Feller-Kopman, D. Pleural Mesothelioma: Advances in Blood and Pleural Biomarkers. J. Clin. Med. 2023, 12, 7006. [Google Scholar] [CrossRef]
- Beyer, H.L.; Geschwindt, R.D.; Glover, C.L.; Tran, L.; Hellstrom, I.; Hellstrom, K.E.; Miller, M.C.; Verch, T.; Allard, W.J.; Pass, H.I.; et al. MESOMARK: A potential test for malignant pleural mesothelioma. Clin. Chem. 2007, 53, 666–672. [Google Scholar] [CrossRef]
- Grosso, F.; Mannucci, M.; Ugo, F.; Ferro, P.; Cassinari, M.; Vigani, A.; De Angelis, A.M.; Delfanti, S.; Lia, M.; Guaschino, R.; et al. Pilot Study to Evaluate Serum Soluble Mesothelin-Related Peptide (SMRP) as Marker for Clinical Monitoring of Pleural Mesothelioma (PM): Correlation with Modified RECIST Score. Diagnostics 2021, 11, 2015. [Google Scholar] [CrossRef] [PubMed]
- Burt, B.M.; Lee, H.S.; Lenge De Rosen, V.; Hamaji, M.; Groth, S.S.; Wheeler, T.M.; Sugarbaker, D.J. Soluble Mesothelin-Related Peptides to Monitor Recurrence After Resection of Pleural Mesothelioma. Ann. Thorac. Surg. 2017, 104, 1679–1687. [Google Scholar] [CrossRef]
- Arnold, D.T.; De Fonseka, D.; Hamilton, F.W.; Rahman, N.M.; Maskell, N.A. Prognostication and monitoring of mesothelioma using biomarkers: A systematic review. Br. J. Cancer 2017, 116, 731–741. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Croce, C.M. The role of MicroRNAs in human cancer. Signal Transduct. Target. Ther. 2016, 1, 15004. [Google Scholar] [CrossRef] [PubMed]
- Azimi, M.; Totonchi, M.; Ebrahimi, M. Determining The Role of MicroRNAs in Self-Renewal, Metastasis and Resistance to Drugs in Human Gastric Cancer Based on Data Mining Approaches: A Systematic Review. Cell J. 2022, 24, 1–6. [Google Scholar]
- Lamichhane, S.R.; Thachil, T.; De Ieso, P.; Gee, H.; Moss, S.A.; Milic, N. Prognostic Role of MicroRNAs in Human Non-Small-Cell Lung Cancer: A Systematic Review and Meta-Analysis. Dis. Markers 2018, 2018, 8309015. [Google Scholar] [CrossRef]
- Golhani, V.; Ray, S.K.; Mukherjee, S. Role of MicroRNAs and Long Non-Coding RNAs in Regulating Angiogenesis in Human Breast Cancer: A Molecular Medicine Perspective. Curr. Mol. Med. 2022, 22, 882–893. [Google Scholar] [CrossRef]
- Prasad, M.; Hamsa, D.; Fareed, M.; Karobari, M.I. An update on the molecular mechanisms underlying the progression of miR-21 in oral cancer. World J. Surg. Oncol. 2025, 23, 73. [Google Scholar] [CrossRef]
- De Santi, C.; Melaiu, O.; Bonotti, A.; Cascione, L.; Di Leva, G.; Foddis, R.; Cristaudo, A.; Lucchi, M.; Mora, M.; Truini, A.; et al. Deregulation of miRNAs in malignant pleural mesothelioma is associated with prognosis and suggests an alteration of cell metabolism. Sci. Rep. 2017, 7, 3140. [Google Scholar] [CrossRef]
- Lo Russo, G.; Tessari, A.; Capece, M.; Galli, G.; de Braud, F.; Garassino, M.C.; Palmieri, D. MicroRNAs for the Diagnosis and Management of Malignant Pleural Mesothelioma: A Literature Review. Front. Oncol. 2018, 8, 650. [Google Scholar] [CrossRef]
- Mozzoni, P.; Ampollini, L.; Goldoni, M.; Alinovi, R.; Tiseo, M.; Gnetti, L.; Carbognani, P.; Rusca, M.; Mutti, A.; Percesepe, A.; et al. MicroRNA Expression in Malignant Pleural Mesothelioma and Asbestosis: A Pilot Study. Dis. Markers 2017, 2017, 9645940. [Google Scholar] [CrossRef]
- Reid, G. MicroRNAs in mesothelioma: From tumour suppressors and biomarkers to therapeutic targets. J. Thorac. Dis. 2015, 7, 1031–1040. [Google Scholar]
- Dixit, S.; Choi, A.Y.; Singh, A.; Pittala, K.; Pruett, N.; Hoang, C.D. Promising therapeutic potential of tumor suppressor microRNAs for malignant pleural mesothelioma. J. Cancer Metastasis Treat. 2022, 8, 47. [Google Scholar] [CrossRef] [PubMed]
- Reid, G.; Johnson, T.G.; van Zandwijk, N. Manipulating microRNAs for the Treatment of Malignant Pleural Mesothelioma: Past, Present and Future. Front. Oncol. 2020, 10, 105. [Google Scholar] [CrossRef] [PubMed]
- Balatti, V.; Maniero, S.; Ferracin, M.; Veronese, A.; Negrini, M.; Ferrocci, G.; Martini, F.; Tognon, M.G. MicroRNAs dysregulation in human malignant pleural mesothelioma. J. Thorac. Oncol. 2011, 6, 844–851. [Google Scholar] [CrossRef]
- Hashemi, M.; Mirdamadi, M.S.A.; Talebi, Y.; Khaniabad, N.; Banaei, G.; Daneii, P.; Gholami, S.; Ghorbani, A.; Tavakolpournegari, A.; Farsani, Z.M.; et al. Pre-clinical and clinical importance of miR-21 in human cancers: Tumorigenesis, therapy response, delivery approaches and targeting agents. Pharmacol. Res. 2023, 187, 106568. [Google Scholar] [CrossRef]
- Bautista-Sanchez, D.; Arriaga-Canon, C.; Pedroza-Torres, A.; De La Rosa-Velazquez, I.A.; Gonzalez-Barrios, R.; Contreras-Espinosa, L.; Montiel-Manriquez, R.; Castro-Hernandez, C.; Fragoso-Ontiveros, V.; Alvarez-Gomez, R.M.; et al. The Promising Role of miR-21 as a Cancer Biomarker and Its Importance in RNA-Based Therapeutics. Mol. Ther. Nucleic Acids 2020, 20, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.H.; Tsao, C.J. Emerging role of microRNA-21 in cancer. Biomed. Rep. 2016, 5, 395–402. [Google Scholar] [CrossRef]
- Sheedy, F.J. Turning 21: Induction of miR-21 as a Key Switch in the Inflammatory Response. Front. Immunol. 2015, 6, 19. [Google Scholar] [CrossRef]
- Sahraei, M.; Chaube, B.; Liu, Y.; Sun, J.; Kaplan, A.; Price, N.L.; Ding, W.; Oyaghire, S.; Garcia-Milian, R.; Mehta, S.; et al. Suppressing miR-21 activity in tumor-associated macrophages promotes an antitumor immune response. J. Clin. Investig. 2019, 129, 5518–5536. [Google Scholar] [CrossRef]
- Wang, S.; Wan, X.; Ruan, Q. The MicroRNA-21 in Autoimmune Diseases. Int. J. Mol. Sci. 2016, 17, 864. [Google Scholar] [CrossRef]
- Miyahara, N.; Benazzo, A.; Oberndorfer, F.; Iwasaki, A.; Laszlo, V.; Dome, B.; Hoda, M.A.; Jaksch, P.; Klepetko, W.; Hoetzenecker, K. MiR-21 in Lung Transplant Recipients With Chronic Lung Allograft Dysfunction. Transpl. Int. 2021, 35, 10184. [Google Scholar] [CrossRef]
- Rusch, V.W.; Chansky, K.; Kindler, H.L.; Nowak, A.K.; Pass, H.I.; Rice, D.C.; Shemanski, L.; Galateau-Salle, F.; McCaughan, B.C.; Nakano, T.; et al. The IASLC Mesothelioma Staging Project: Proposals for the M Descriptors and for Revision of the TNM Stage Groupings in the Forthcoming (Eighth) Edition of the TNM Classification for Mesothelioma. J. Thorac. Oncol. 2016, 11, 2112–2119. [Google Scholar] [CrossRef] [PubMed]
- Rice, D.; Chansky, K.; Nowak, A.; Pass, H.; Kindler, H.; Shemanski, L.; Opitz, I.; Call, S.; Hasegawa, S.; Kernstine, K.; et al. The IASLC Mesothelioma Staging Project: Proposals for Revisions of the N Descriptors in the Forthcoming Eighth Edition of the TNM Classification for Pleural Mesothelioma. J. Thorac. Oncol. 2016, 11, 2100–2111. [Google Scholar] [CrossRef] [PubMed]
- Nowak, A.K.; Chansky, K.; Rice, D.C.; Pass, H.I.; Kindler, H.L.; Shemanski, L.; Bille, A.; Rintoul, R.C.; Batirel, H.F.; Thomas, C.F.; et al. The IASLC Mesothelioma Staging Project: Proposals for Revisions of the T Descriptors in the Forthcoming Eighth Edition of the TNM Classification for Pleural Mesothelioma. J. Thorac. Oncol. 2016, 11, 2089–2099. [Google Scholar] [CrossRef]
- Pass, H.; Giroux, D.; Kennedy, C.; Ruffini, E.; Cangir, A.K.; Rice, D.; Asamura, H.; Waller, D.; Edwards, J.; Weder, W.; et al. The IASLC Mesothelioma Staging Project: Improving Staging of a Rare Disease Through International Participation. J. Thorac. Oncol. 2016, 11, 2082–2088. [Google Scholar] [CrossRef] [PubMed]
- Kirschner, M.B.; van Zandwijk, N.; Reid, G. Cell-free microRNAs: Potential biomarkers in need of standardized reporting. Front. Genet. 2013, 4, 56. [Google Scholar] [CrossRef]
- Valko, Z.; Megyesfalvi, Z.; Schwendenwein, A.; Lang, C.; Paku, S.; Barany, N.; Ferencz, B.; Horvath-Rozsas, A.; Kovacs, I.; Schlegl, E.; et al. Dual targeting of BCL-2 and MCL-1 in the presence of BAX breaks venetoclax resistance in human small cell lung cancer. Br. J. Cancer 2023, 128, 1850–1861. [Google Scholar] [CrossRef]
- Jorgensen, S.; Baker, A.; Moller, S.; Nielsen, B.S. Robust one-day in situ hybridization protocol for detection of microRNAs in paraffin samples using LNA probes. Methods 2010, 52, 375–381. [Google Scholar] [CrossRef]
- Medley, J.C.; Panzade, G.; Zinovyeva, A.Y. microRNA strand selection: Unwinding the rules. Wiley Interdiscip. Rev. RNA 2021, 12, e1627. [Google Scholar] [CrossRef]
- Qu, K.; Zhang, X.; Lin, T.; Liu, T.; Wang, Z.; Liu, S.; Zhou, L.; Wei, J.; Chang, H.; Li, K.; et al. Circulating miRNA-21-5p as a diagnostic biomarker for pancreatic cancer: Evidence from comprehensive miRNA expression profiling analysis and clinical validation. Sci. Rep. 2017, 7, 1692. [Google Scholar] [CrossRef]
- Karasek, P.; Gablo, N.; Hlavsa, J.; Kiss, I.; Vychytilova-Faltejskova, P.; Hermanova, M.; Kala, Z.; Slaby, O.; Prochazka, V. Pre-operative Plasma miR-21-5p Is a Sensitive Biomarker and Independent Prognostic Factor in Patients with Pancreatic Ductal Adenocarcinoma Undergoing Surgical Resection. Cancer Genom. Proteom. 2018, 15, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Donofrio, C.A.; Fioravanti, A.; Riccio, L.; Cominetti, M.; Cappelletti, M.R.; Longo, G.; Varotti, E.; Generali, D.; Servadei, F.; Grossi, I.; et al. Determination of longitudinal Circulating levels of miR-21-5p, miR-23b-3p and miR-34a-5p in plasma of patients with glioblastoma using droplet digital PCR. Neurosurg. Rev. 2025, 48, 592. [Google Scholar] [CrossRef]
- Burlacu, I.; Aschie, M.; Cozaru, G.C.; Deacu, M.; Vizireanu, G.M.; Mitroi, A.N.; Mitroi, A.F.; Brinzan, C.S. The miR-21-5p, miR-30c-5p, and miR-182-5p as Biomarkers in Clear Cell Renal Cell Carcinoma: A Southeastern Romanian Cohort Study. Genes 2025, 16, 650. [Google Scholar] [CrossRef]
- Schillebeeckx, E.; van Meerbeeck, J.P.; Lamote, K. Clinical utility of diagnostic biomarkers in malignant pleural mesothelioma: A systematic review and meta-analysis. Eur. Respir. Rev. 2021, 30, 210057. [Google Scholar] [CrossRef]
- Sun, H.H.; Vaynblat, A.; Pass, H.I. Diagnosis and prognosis-review of biomarkers for mesothelioma. Ann. Transl. Med. 2017, 5, 244. [Google Scholar] [CrossRef] [PubMed]
- Rhim, J.; Baek, W.; Seo, Y.; Kim, J.H. From Molecular Mechanisms to Therapeutics: Understanding MicroRNA-21 in Cancer. Cells 2022, 11, 2791. [Google Scholar] [CrossRef] [PubMed]
- Dillhoff, M.; Liu, J.; Frankel, W.; Croce, C.; Bloomston, M. MicroRNA-21 is overexpressed in pancreatic cancer and a potential predictor of survival. J. Gastrointest. Surg. 2008, 12, 2171–2176. [Google Scholar] [CrossRef]
- Lee, J.A.; Lee, H.Y.; Lee, E.S.; Kim, I.; Bae, J.W. Prognostic Implications of MicroRNA-21 Overexpression in Invasive Ductal Carcinomas of the Breast. J. Breast Cancer 2011, 14, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Farasati Far, B.; Vakili, K.; Fathi, M.; Yaghoobpoor, S.; Bhia, M.; Naimi-Jamal, M.R. The role of microRNA-21 (miR-21) in pathogenesis, diagnosis, and prognosis of gastrointestinal cancers: A review. Life Sci. 2023, 316, 121340. [Google Scholar] [CrossRef] [PubMed]
- Kirschner, M.B.; Cheng, Y.Y.; Armstrong, N.J.; Lin, R.C.; Kao, S.C.; Linton, A.; Klebe, S.; McCaughan, B.C.; van Zandwijk, N.; Reid, G. MiR-score: A novel 6-microRNA signature that predicts survival outcomes in patients with malignant pleural mesothelioma. Mol. Oncol. 2015, 9, 715–726. [Google Scholar] [CrossRef]
- Nicole, L.; Cappellesso, R.; Sanavia, T.; Guzzardo, V.; Fassina, A. MiR-21 over-expression and Programmed Cell Death 4 down-regulation features malignant pleural mesothelioma. Oncotarget 2018, 9, 17300–17308. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cappellesso, R.; Nicole, L.; Caroccia, B.; Guzzardo, V.; Ventura, L.; Fassan, M.; Fassina, A. Young investigator challenge: MicroRNA-21/MicroRNA-126 profiling as a novel tool for the diagnosis of malignant mesothelioma in pleural effusion cytology. Cancer Cytopathol. 2016, 124, 28–37. [Google Scholar] [CrossRef]
- Zhu, L.R.; Yuan, R.X.; Xia, X.B.; Wang, Y.; Zhu, Y.M.; Fi, L.; Li, J. Assessment of a panel of miRNAs in serum and pleural fluid for the differential diagnosis of malignant and benign pleural effusion. Cancer Biomark. 2022, 33, 71–82. [Google Scholar] [CrossRef]
- He, Y.; Zhang, L.; Cheng, G.; Yuan, R.; Zhuang, Y.; Zhang, D.; Zhou, D.; Xu, X. Upregulation of circulating miR-21 is associated with poor prognosis of nasopharyngeal carcinoma. Int. J. Clin. Exp. Pathol. 2017, 10, 7362–7368. [Google Scholar]
- Gkioka, A.I.; Tsota, M.; Koudouna, A.; Gkiokas, A.; Mitropoulou, C.A.; Palaiokrassa, A.; Alexandropoulos, A.; Papadatou-Gigante, M.; Bartzi, V.; Tryfou, T.M.; et al. Circulating miR-16 and miR-21 Levels in Multiple Myeloma: Prognostic Significance of Survival and Response to Lenalidomide Treatment. Int. J. Mol. Sci. 2024, 25, 6065. [Google Scholar] [CrossRef]
- Ye, T.T.; Yang, Y.L.; Liu, X.Y.; Ji, Q.Q.; Pan, Y.F.; Xiang, Y.Q. Prognostic value of circulating microRNA-21 in digestive system cancers: A meta-analysis. Int. J. Clin. Exp. Med. 2014, 7, 873–878. [Google Scholar]
- Gao, Y.; Dai, M.; Liu, H.; He, W.; Lin, S.; Yuan, T.; Chen, H.; Dai, S. Diagnostic value of circulating miR-21: An update meta-analysis in various cancers and validation in endometrial cancer. Oncotarget 2016, 7, 68894–68908. [Google Scholar] [CrossRef]
- Xu, F.; Xu, L.; Wang, M.; An, G.; Feng, G. The accuracy of circulating microRNA-21 in the diagnosis of colorectal cancer: A systematic review and meta-analysis. Colorectal Dis. 2015, 17, O100–O107. [Google Scholar] [CrossRef]
- Asangani, I.A.; Rasheed, S.A.; Nikolova, D.A.; Leupold, J.H.; Colburn, N.H.; Post, S.; Allgayer, H. MicroRNA-21 (miR-21) post-transcriptionally downregulates tumor suppressor Pdcd4 and stimulates invasion, intravasation and metastasis in colorectal cancer. Oncogene 2008, 27, 2128–2136. [Google Scholar] [CrossRef]
- Xu, P.; Zhu, Y.; Sun, B.; Xiao, Z. Colorectal cancer characterization and therapeutic target prediction based on microRNA expression profile. Sci. Rep. 2016, 6, 20616. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, P.; Ju, H.; Pesta, M.; Kulda, V.; Jin, W.; Cai, M.; Liu, C.; Wu, H.; Xu, J.; et al. Diagnostic and prognostic value of microRNA-21 in colorectal cancer: An original study and individual participant data meta-analysis. Cancer Epidemiol. Biomarkers Prev. 2014, 23, 2783–2792. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, Q. The expression and clinical significance of circulating microRNA-21 in serum of five solid tumors. J. Cancer Res. Clin. Oncol. 2012, 138, 1659–1666. [Google Scholar] [CrossRef]
- Yamada, H.; Suzuki, K.; Fujii, R.; Kawado, M.; Hashimoto, S.; Watanabe, Y.; Iso, H.; Fujino, Y.; Wakai, K.; Tamakoshi, A. Circulating miR-21, miR-29a, and miR-126 are associated with premature death risk due to cancer and cardiovascular disease: The JACC Study. Sci. Rep. 2021, 11, 5298. [Google Scholar] [CrossRef]
- Chen, H.; Liu, H.; Zou, H.; Chen, R.; Dou, Y.; Sheng, S.; Dai, S.; Ai, J.; Melson, J.; Kittles, R.A.; et al. Evaluation of Plasma miR-21 and miR-152 as Diagnostic Biomarkers for Common Types of Human Cancers. J. Cancer 2016, 7, 490–499. [Google Scholar] [CrossRef]
- Anwar, S.L.; Sari, D.N.I.; Kartika, A.I.; Fitria, M.S.; Tanjung, D.S.; Rakhmina, D.; Wardana, T.; Astuti, I.; Haryana, S.M.; Aryandono, T. Upregulation of Circulating MiR-21 Expression as a Potential Biomarker for Therapeutic Monitoring and Clinical Outcome in Breast Cancer. Asian Pac. J. Cancer Prev. 2019, 20, 1223–1228. [Google Scholar] [CrossRef]
- Chi, L.H.; Cross, R.S.N.; Redvers, R.P.; Davis, M.; Hediyeh-Zadeh, S.; Mathivanan, S.; Samuel, M.; Lucas, E.C.; Mouchemore, K.; Gregory, P.A.; et al. MicroRNA-21 is immunosuppressive and pro-metastatic via separate mechanisms. Oncogenesis 2022, 11, 38. [Google Scholar] [CrossRef] [PubMed]
- Jinling, W.; Sijing, S.; Jie, Z.; Guinian, W. Prognostic value of circulating microRNA-21 for breast cancer: A systematic review and meta-analysis. Artif. Cells Nanomed. Biotechnol. 2017, 45, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Liu, C.; Xie, Y.; Tang, M.; Luo, G.; Chen, X.; Tian, L.; Yu, X. Lung Cancer Cells Derived Circulating miR-21 Promotes Differentiation of Monocytes into Osteoclasts. Onco Targets Ther. 2020, 13, 2643–2656. [Google Scholar] [CrossRef]
- Pop-Bica, C.; Pintea, S.; Magdo, L.; Cojocneanu, R.; Gulei, D.; Ferracin, M.; Berindan-Neagoe, I. The Clinical Utility of miR-21 and let-7 in Non-small Cell Lung Cancer (NSCLC). A Systematic Review and Meta-Analysis. Front. Oncol. 2020, 10, 516850. [Google Scholar] [CrossRef]
- Wang, W.; Li, X.; Liu, C.; Zhang, X.; Wu, Y.; Diao, M.; Tan, S.; Huang, S.; Cheng, Y.; You, T. MicroRNA-21 as a diagnostic and prognostic biomarker of lung cancer: A systematic review and meta-analysis. Biosci. Rep. 2022, 42, BSR20211653. [Google Scholar] [CrossRef] [PubMed]
- Soliman, S.E.; Abdelaleem, A.H.; Alhanafy, A.M.; Ibrahem, R.A.L.; Elhaded, A.S.A.; Assar, M.F.A. Circulating miR-21-5p and miR-126-3p: Diagnostic, prognostic value, and multivariate analysis in non-small-cell lung cancer. Mol. Biol. Rep. 2021, 48, 2543–2552. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Mao, F.; Shen, T.; Luo, Q.; Ding, Z.; Qian, L.; Huang, J. Plasma miR-145, miR-20a, miR-21 and miR-223 as novel biomarkers for screening early-stage non-small cell lung cancer. Oncol. Lett. 2017, 13, 669–676. [Google Scholar] [CrossRef]
- Silvia Lima, R.Q.D.; Vasconcelos, C.F.M.; Gomes, J.P.A.; Bezerra de Menezes, E.D.S.; de Oliveira Silva, B.; Montenegro, C.; Paiva Junior, S.S.L.; Pereira, M.C. miRNA-21, an oncomiR that regulates cell proliferation, migration, invasion and therapy response in lung cancer. Pathol. Res. Pract. 2024, 263, 155601. [Google Scholar] [CrossRef]
- Yamada, A.; Horimatsu, T.; Okugawa, Y.; Nishida, N.; Honjo, H.; Ida, H.; Kou, T.; Kusaka, T.; Sasaki, Y.; Yagi, M.; et al. Serum miR-21, miR-29a, and miR-125b Are Promising Biomarkers for the Early Detection of Colorectal Neoplasia. Clin. Cancer Res. 2015, 21, 4234–4242. [Google Scholar] [CrossRef]
- Shan, L.; Ji, Q.; Cheng, G.; Xia, J.; Liu, D.; Wu, C.; Zhu, B.; Ding, Y. Diagnostic value of circulating miR-21 for colorectal cancer: A meta-analysis. Cancer Biomark. 2015, 15, 47–56. [Google Scholar] [CrossRef]
- Ferraro, A.; Kontos, C.K.; Boni, T.; Bantounas, I.; Siakouli, D.; Kosmidou, V.; Vlassi, M.; Spyridakis, Y.; Tsipras, I.; Zografos, G.; et al. Epigenetic regulation of miR-21 in colorectal cancer: ITGB4 as a novel miR-21 target and a three-gene network (miR-21-ITGBeta4-PDCD4) as predictor of metastatic tumor potential. Epigenetics 2014, 9, 129–141. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Liu, D.; Guan, S.; Dong, M. Diagnostic role of circulating MiR-21 in colorectal cancer: A update meta-analysis. Ann. Med. 2021, 53, 87–102. [Google Scholar] [CrossRef]
- Yu, W.; Wang, Z.; Shen, L.I.; Wei, Q. Circulating microRNA-21 as a potential diagnostic marker for colorectal cancer: A meta-analysis. Mol. Clin. Oncol. 2016, 4, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Abue, M.; Yokoyama, M.; Shibuya, R.; Tamai, K.; Yamaguchi, K.; Sato, I.; Tanaka, N.; Hamada, S.; Shimosegawa, T.; Sugamura, K.; et al. Circulating miR-483-3p and miR-21 is highly expressed in plasma of pancreatic cancer. Int. J. Oncol. 2015, 46, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Gao, P.; Wang, Y.; Wang, X. Blood-Derived microRNAs for Pancreatic Cancer Diagnosis: A Narrative Review and Meta-Analysis. Front. Physiol. 2018, 9, 685. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, L.; Carugno, M.; Mensi, C.; Pesatori, A.C. Circulating Epigenetic Biomarkers in Malignant Pleural Mesothelioma: State of the Art and critical Evaluation. Front. Oncol. 2020, 10, 445. [Google Scholar] [CrossRef]
- Cavallari, I.; Urso, L.; Sharova, E.; Pasello, G.; Ciminale, V. Liquid Biopsy in Malignant Pleural Mesothelioma: State of the Art, Pitfalls, and Perspectives. Front. Oncol. 2019, 9, 740. [Google Scholar] [CrossRef]
- Paajanen, J.; Ilonen, I.; Lauri, H.; Jarvinen, T.; Sutinen, E.; Ollila, H.; Rouvinen, E.; Lemstrom, K.; Rasanen, J.; Ritvos, O.; et al. Elevated Circulating Activin A Levels in Patients With Malignant Pleural Mesothelioma Are Related to Cancer Cachexia and Reduced Response to Platinum-based Chemotherapy. Clin. Lung Cancer 2020, 21, e142–e150. [Google Scholar] [CrossRef]
- Hoda, M.A.; Dong, Y.; Rozsas, A.; Klikovits, T.; Laszlo, V.; Ghanim, B.; Stockhammer, P.; Ozsvar, J.; Jakopovic, M.; Samarzija, M.; et al. Circulating activin A is a novel prognostic biomarker in malignant pleural mesothelioma—A multi-institutional study. Eur. J. Cancer 2016, 63, 64–73. [Google Scholar] [CrossRef]
- Ghanim, B.; Hoda, M.A.; Klikovits, T.; Winter, M.P.; Alimohammadi, A.; Grusch, M.; Dome, B.; Arns, M.; Schenk, P.; Jakopovic, M.; et al. Circulating fibrinogen is a prognostic and predictive biomarker in malignant pleural mesothelioma. Br. J. Cancer 2014, 110, 984–990. [Google Scholar] [CrossRef]
- Klikovits, T.; Stockhammer, P.; Laszlo, V.; Dong, Y.; Hoda, M.A.; Ghanim, B.; Opitz, I.; Frauenfelder, T.; Nguyen-Kim, T.D.L.; Weder, W.; et al. Circulating complement component 4d (C4d) correlates with tumor volume, chemotherapeutic response and survival in patients with malignant pleural mesothelioma. Sci. Rep. 2017, 7, 16456. [Google Scholar] [CrossRef]


| Demographics | Study Cohort (n = 94) | Low miR-21 (n = 47) | High miR-21 (n = 47) | p-Value |
|---|---|---|---|---|
| Age, years, median, IQR | 64 (56–64) | 64 (55–71) | 65 (58–71) | 0.856 |
| Sex | 0.614 | |||
| Male | 74 (79%) | 38 (81%) | 36 (77%) | |
| Female | 20 (21%) | 9 (19%) | 11 (23%) | |
| Histology | 0.441 | |||
| Epithelioid | 75 (80%) | 39 (83%) | 36 (77%) | |
| Non-epithelioid | 19 (20%) | 8 (17%) | 11 (23%) | |
| Stage | 0.527 | |||
| Early | 37 (39%) | 17 (36%) | 20 (43%) | |
| Late | 57 (61%) | 30 (64%) | 27 (57%) | |
| Treatment | 0.265 | |||
| MMT | 48 (51%) | 22 (47%) | 26 (55%) | |
| CHT +/− RTH | 37 (39%) | 22 (47%) | 15 (32%) | |
| BSC | 9 (10%) | 3 (6%) | 6 (13%) |
| Demographics | Study Cohort (n = 84) | Low SMRP (n = 42) | High SMRP (n = 42) | p-Value |
|---|---|---|---|---|
| Age, years, median, IQR | 64 (55–71) | 63 (54–69) | 66 (57–75) | 0.513 |
| Sex | 0.917 | |||
| Male | 66 (79%) | 33 (79%) | 33 (79%) | |
| Female | 18 (21%) | 9 (21%) | 9 (21%) | |
| Histology | 0.578 | |||
| Epithelioid | 68 (81%) | 33 (79%) | 35 (83%) | |
| Non-epithelioid | 16 (19%) | 9 (21%) | 7 (17%) | |
| Stage | 0.046 | |||
| Early | 35 (42%) | 22 (52%) | 13 (31%) | |
| Late | 49 (58%) | 20 (48%) | 29 (69%) | |
| Treatment | 0.194 | |||
| MMT | 42 (50%) | 25 (60%) | 17 (41%) | |
| CHT +/− RTH | 33 (39%) | 14 (33%) | 19 (45%) | |
| BSC | 9 (11%) | 3 (7%) | 6 (14%) |
| Variables | Univariable | Multivariable | ||||||
|---|---|---|---|---|---|---|---|---|
| n = 84 | OS (CI) | p-Value | HR | 95% CI | p-Value | HR | 95% CI | |
| Age | 0.487 | 1.18 | 0.74–1.90 | 0.512 | 1.01 | 0.59–1.71 | ||
| >65 | 41 | 15.6 (11.1–20.1) | ||||||
| <65 | 43 | 18.5 (14.1–22.9) | ||||||
| Sex | 0.357 | 1.33 | 0.72–2.44 | 0.935 | 1.01 | 0.53–1.92 | ||
| Male | 66 | 17.9 (13.9–21.9) | ||||||
| Female | 18 | 16.2 (10.8–21.6) | ||||||
| Histology | 0.008 | 2.25 | 1.21–4.19 | 0.024 | 2.09 | 1.11–3.95 | ||
| Epithelioid | 68 | 18.5 (14.9–22.2) | ||||||
| Non-epithelioid | 16 | 5.4 (0.0–16.0) | ||||||
| MiR-21 levels | 0.041 | 1.64 | 1.10–2.64 | <0.001 | 3.12 | 1.78–5.47 | ||
| High | 42 | 12.4 (10.2–14.6) | ||||||
| Low | 42 | 24.3 (18.3–29.9) | ||||||
| SMRP levels | 0.032 | 1.55 | 0.96–2.49 | 0.090 | 1.58 | 0.93–2.66 | ||
| High | 42 | 12.4 (6.7–18.1) | ||||||
| Low | 42 | 19.5 (10.2–28.8) | ||||||
| Stage | <0.001 | 2.94 | 1.77–4.89 | 0.068 | 2.79 | 1.25–6.26 | ||
| Early | 35 | 24.2 (22.1–26.3) | ||||||
| Late | 49 | 11.8 (8.1–15.6) | ||||||
| Treatment | <0.001 | 1.96 | 1.17–3.29 | <0.001 | 2.37 | 1.18–4.78 | ||
| MMT | 42 | 24.0 (18.4–30.0) | ||||||
| CTH +/− RTH | 33 | 13.2 (9.2–17.2) | ||||||
| BSC | 9 | 2.8 (2.0–3.5) | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mosleh, B.; Dong, Y.; Lang, E.; Klikovits, T.; Sinn, K.; Kao, S.; Jakopovic, M.; Aigner, C.; Hegedüs, B.; Baldes, N.; et al. MiR-21 Is a Novel Diagnostic and Prognostic Circulating Biomarker in Pleural Mesothelioma. Diagnostics 2026, 16, 1142. https://doi.org/10.3390/diagnostics16081142
Mosleh B, Dong Y, Lang E, Klikovits T, Sinn K, Kao S, Jakopovic M, Aigner C, Hegedüs B, Baldes N, et al. MiR-21 Is a Novel Diagnostic and Prognostic Circulating Biomarker in Pleural Mesothelioma. Diagnostics. 2026; 16(8):1142. https://doi.org/10.3390/diagnostics16081142
Chicago/Turabian StyleMosleh, Berta, Yawen Dong, Elisabeth Lang, Thomas Klikovits, Katharina Sinn, Steven Kao, Marko Jakopovic, Clemens Aigner, Balazs Hegedüs, Natalie Baldes, and et al. 2026. "MiR-21 Is a Novel Diagnostic and Prognostic Circulating Biomarker in Pleural Mesothelioma" Diagnostics 16, no. 8: 1142. https://doi.org/10.3390/diagnostics16081142
APA StyleMosleh, B., Dong, Y., Lang, E., Klikovits, T., Sinn, K., Kao, S., Jakopovic, M., Aigner, C., Hegedüs, B., Baldes, N., Bölükbas, S., Dome, B., Hoda, M. A., Laszlo, V., Grusch, M., & Schelch, K. (2026). MiR-21 Is a Novel Diagnostic and Prognostic Circulating Biomarker in Pleural Mesothelioma. Diagnostics, 16(8), 1142. https://doi.org/10.3390/diagnostics16081142










