Retrospective Analysis of 1168 Cases of Ovular Decidual Tissue from First-Trimester Abortions: Proposal for a Histopathological Diagnostic Framework
Abstract
1. Introduction
- •
- Inevitable loss due to an intrinsically abnormal gestation (e.g., abnormal karyotype);
- •
- Failure to maintain an otherwise normal gestation due to underlying maternal physiologic or structural problems;
- •
- Pathologic elimination of a normal gestation due to an active maternal disease process (e.g., massive perivillous fibrin deposition, chronic histiocytic intervillositis, chronic villitis and decidual vasculitis).
1.1. Development of the Placenta
First-Trimester Physiological Morphology of the Placenta
2. Materials and Methods
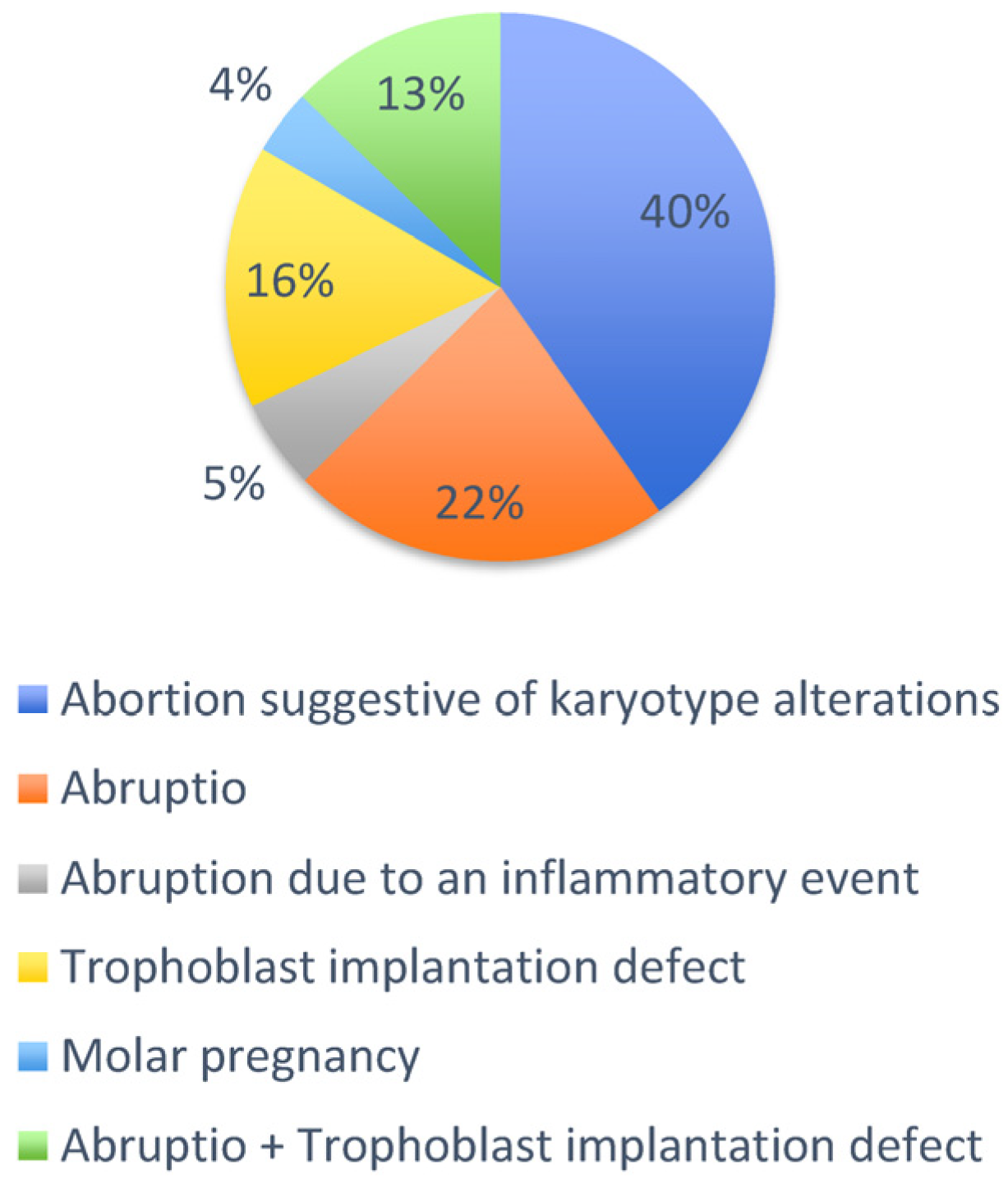
3. Results
3.1. Histological Features in EPL Following Different Causes
3.1.1. Abortion Due to Karyotype Alterations
Histology
3.1.2. Trophoblast Implantation Defect
Histology
3.1.3. Abruption
Histology
3.1.4. Blighted Ovum
Histology
3.1.5. Exuberant Placental Site
Histology
3.1.6. Partial Molar Pregnancy
Histology
3.1.7. Complete Molar Pregnancy
Histology
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| EPL | Early Pregnancy Loss |
| ART | Assisted Reproductive Technology |
| IVF | In Vitro Fertilization |
| ICSI | Intracytoplasmic Sperm Injection |
| H&E | Hematoxylin and Eosin |
| RBCs | Red Blood Cells |
| TID | Trophoblast Implantation Defects |
| GTD | Gestational Trophoblastic Disease |
| PMP | Partial Molar Pregnancy |
| CMP | Complete Molar Pregnancy |
| CMV | Cytomegalovirus |
References
- Fram, K.M. Histological analysis of the products of conception following first trimester abortion at Jordan University Hospital. Eur. J. Obstet. Gynecol. Reprod. Biol. 2002, 105, 147–149. [Google Scholar] [CrossRef] [PubMed]
- Shakhatreh, H.J.M.; Salih, A.J.; Aldrou, K.K.A.R.; Alazzam, F.A.F.; Issa, M.S.B. Medico-Legal Aspects of Abortion: Updates of the Literature. Med. Arch. 2022, 76, 373–376. [Google Scholar] [CrossRef] [PubMed]
- Essers, R.; Lebedev, I.N.; Kurg, A.; Fonova, E.A.; Stevens, S.J.C.; Koeck, R.M.; von Rango, U.; Brandts, L.; Deligiannis, S.P.; Nikitina, T.V.; et al. Prevalence of chromosomal alterations in first-trimester spontaneous pregnancy loss. Nat. Med. 2023, 29, 3233–3242. [Google Scholar] [CrossRef] [PubMed]
- Novais Nogueira Cardoso, R.M.A.; Nogueira Cardoso, P.L.N.; Azevedo, A.P.; Cadillá, J.S.; Oliveira Rodrigues Amorim, M.G.R.; Rocha Gomes, M.E.; Correia Pinto, J.M.N. First-trimester miscarriage: A histopathological classification proposal. Heliyon 2021, 7, e06359. [Google Scholar] [CrossRef]
- Jauniaux, E.; Burton, G.J. Pathophysiology of histological changes in early pregnancy loss. Placenta 2005, 26, 114–123. [Google Scholar] [CrossRef]
- Pinar, M.H.; Gibbins, K.; He, M.; Kostadinov, S.; Silver, R. Early Pregnancy Losses: Review of Nomenclature, Histopathology, and Possible Etiologies. Fetal Pediatr. Pathol. 2018, 37, 191–209. [Google Scholar] [CrossRef]
- Alves, C.; Jenkins, S.M.; Rapp, A. Early Pregnancy Loss (Spontaneous Abortion). In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins—Gynecology. ACOG Practice Bulletin No. 200: Early Pregnancy Loss. Obstet. Gynecol. 2018, 132, e197–e207. [Google Scholar] [CrossRef]
- Genest, D.R.; Roberts, D.; Boyd, T.; Bieber, F.R. Fetoplacental histology as a predictor of karyotype: A controlled study of spontaneous first trimester abortions. Hum. Pathol. 1995, 26, 201–209. [Google Scholar] [CrossRef]
- Massalska, D.; Zimowski, J.G.; Bijok, J.; Pawelec, M.; Czubak-Barlik, M.; Jakiel, G.; Roszkowski, T. First trimester pregnancy loss: Clinical implications of genetic testing. J. Obstet. Gynaecol. Res. 2017, 43, 23–29. [Google Scholar] [CrossRef]
- Hu, L.; Du, J.; Lv, H.; Zhao, J.; Chen, M.; Wang, Y.; Wu, F.; Liu, F.; Chen, X.; Zhang, J.; et al. Influencing factors of pregnancy loss and survival probability of clinical pregnancies conceived through assisted reproductive technology. Reprod. Biol. Endocrinol. 2018, 16, 74. [Google Scholar] [CrossRef]
- Emami, F.; Eftekhar, M.; Jalaliani, S. Correlation between clinical and laboratory parameters and early pregnancy loss in assisted reproductive technology cycles: A cross-sectional study. Int. J. Reprod. Biomed. 2022, 20, 683–690, Erratum in Int. J. Reprod. Biomed. 2022, 20, 883. https://doi.org/10.18502/ijrm.v20i10.12272. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Niu, A.; Feng, X.; Yan, Y.; Chen, Y. The threshold effect of factors associated with spontaneous abortion in human-assisted reproductive technology. Sci. Rep. 2021, 11, 11368. [Google Scholar] [CrossRef] [PubMed]
- Pendina, A.A.; Efimova, O.A.; Chiryaeva, O.G.; Tikhonov, A.V.; Petrova, L.I.; Dudkina, V.S.; Sadik, N.A.; Fedorova, I.D.; Galembo, I.A.; Kuznetzova, T.V.; et al. A comparative cytogenetic study of miscarriages after IVF and natural conception in women aged under and over 35 years. J. Assist. Reprod. Genet. 2014, 31, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Ly, K.D.; Agarwal, A.; Nagy, Z.P. Preimplantation genetic screening: Does it help or hinder IVF treatment and what is the role of the embryo? J. Assist. Reprod. Genet. 2011, 28, 833–849. [Google Scholar] [CrossRef]
- Allison, J.L.; Schust, D.J. Recurrent first trimester pregnancy loss: Revised definitions and novel causes. Curr. Opin. Endocrinol. Diabetes Obes. 2009, 16, 446–450. [Google Scholar] [CrossRef]
- Giacometti, C.; Bellan, E.; Ambrosi, A.; Dei Tos, A.P.; Cassaro, M.; Ludwig, K. “While there is p57, there is hope.” The past and the present of diagnosis in first trimester abortions: Diagnostic dilemmas and algorithmic approaches. A review. Placenta 2021, 116, 31–37. [Google Scholar] [CrossRef]
- Cindrova-Davies, T.; Sferruzzi-Perri, A.N. Human placental development and function. Semin. Cell Dev. Biol. 2022, 131, 66–77. [Google Scholar] [CrossRef]
- Faye-Petersen, O.M.; Heller, D.S.; Joshi, V.V. Handbook of Placental Pathology, 2nd ed.; Taylor and Fracis: Abingdon, UK, 2006. [Google Scholar]
- Heil, J.R.; Bordoni, B. Embryology, Umbilical Cord. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Queremel Milani, D.A.; Tadi, P. Genetics, Chromosome Abnormalities. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Fritz, R.; Jain, C.; Armant, D.R. Cell signaling in trophoblast-uterine communication. Int. J. Dev. Biol. 2014, 58, 261–271. [Google Scholar] [CrossRef]
- Atkinson, A.L.; Santolaya-Forgas, J.; Blitzer, D.N.; Santolaya, J.L.; Matta, P.; Canterino, J.; Oyelese, Y. Risk factors for perinatal mortality in patients admitted to the hospital with the diagnosis of placental abruption. J. Matern. Fetal Neonatal Med. 2015, 28, 594–597. [Google Scholar] [CrossRef]
- Schneider, E.; Kinzler, W.L. Placental Abruption: Pathophysiology, Diagnosis, and Management. Clin. Obstet. Gynecol. 2025, 68, 98–104. [Google Scholar] [CrossRef]
- Tikkanen, M. Placental abruption: Epidemiology, risk factors and consequences. Acta Obstet. Gynecol. Scand. 2011, 90, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Chaudhry, K.; Tafti, D.; Carlson, K.; Siccardi, M.A. Anembryonic Pregnancy. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Yeasmin, S.; Nakayama, K.; Katagiri, A.; Ishikawa, M.; Iida, K.; Nakayama, N.; Miyazaki, K. Exaggerated placental site mimicking placental site trophoblastic tumor: Case report and literature review. Eur. J. Gynaecol. Oncol. 2010, 31, 586–589. [Google Scholar]
- Al Ghadeer, H.A.; Al Kishi, N.; Algurini, K.H.; Albesher, A.B.; AlGhadeer, M.R.; Alsalman, A.A.; Bubshait, A.A.; Alkishi, B.M. Partial Molar Pregnancy With Normal Karyotype. Cureus 2022, 14, e30934. [Google Scholar] [CrossRef]
- Sileo, F.G.; Giuliani, G.A.; Facchinetti, F.; Contu, G.; Chiossi, G.; Bertucci, E. Complete hydatidiform mole in higher-order multiple pregnancies. Minerva Obstet. Gynecol. 2022, 74, 308–313. [Google Scholar] [CrossRef]
- Soper, J.T.; Mutch, D.G.; Schink, J.C.; American College of Obstetricians and Gynecologists. Diagnosis and treatment of gestational trophoblastic disease: ACOG Practice Bulletin No. 53. Gynecol. Oncol. 2004, 93, 575–585. [Google Scholar] [CrossRef]



| Possible Cause of Miscarriage | Pathology Report | Comment |
|---|---|---|
| Trophoblast implantation defect | Normal villi, poor trophoblast invasion | Suggestive of implantation failure |
| Karyotype alterations | Edematous, avascular villi with incisions and pseudo-cystic degeneration of the trophoblast. | Suggestive, not conclusive- genetic testing needed |
| Blind ovum | Edematous villi, no fetal elements | Consistent with anembryonic pregnancy |
| Abruption | Ischemic villi, blood, fibrin, thrombi in decidua | Consistent with placenta abruption. |
| Inflammatory abruption | Intervillous blood, inflammation | Indicates secondary inflammatory cause |
| Exuberant placental site | Hydropic villi, myometrium fibers, extravillous trophoblast | Requires follow-up |
| Partial molar pregnancy | Stromal cisterns, focal non-polar proliferation, pseudo-cystic degeneration. p57+ | Suggestive of PMP |
| Complete molar pregnancy | Cystic villi, atypical trophoblast. p57− | Diagnostic of CMP |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Nardi, E.; Arena, V. Retrospective Analysis of 1168 Cases of Ovular Decidual Tissue from First-Trimester Abortions: Proposal for a Histopathological Diagnostic Framework. Diagnostics 2026, 16, 1128. https://doi.org/10.3390/diagnostics16081128
Nardi E, Arena V. Retrospective Analysis of 1168 Cases of Ovular Decidual Tissue from First-Trimester Abortions: Proposal for a Histopathological Diagnostic Framework. Diagnostics. 2026; 16(8):1128. https://doi.org/10.3390/diagnostics16081128
Chicago/Turabian StyleNardi, Eleonora, and Vincenzo Arena. 2026. "Retrospective Analysis of 1168 Cases of Ovular Decidual Tissue from First-Trimester Abortions: Proposal for a Histopathological Diagnostic Framework" Diagnostics 16, no. 8: 1128. https://doi.org/10.3390/diagnostics16081128
APA StyleNardi, E., & Arena, V. (2026). Retrospective Analysis of 1168 Cases of Ovular Decidual Tissue from First-Trimester Abortions: Proposal for a Histopathological Diagnostic Framework. Diagnostics, 16(8), 1128. https://doi.org/10.3390/diagnostics16081128





