Cystatin C as a Renal Biomarker in Infants with Congenital Anomalies of the Kidney and Urinary Tract (CAKUT): A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Review Design and Rationale
2.2. Eligibility Criteria
2.2.1. Inclusion Criteria
- Neonates (0–28 days) or preterm infants up to 44 weeks postmenstrual age.
- Studies reporting serum and/or urinary cystatin C.
- Studies evaluating renal function, AKI, CAKUT, or CKD outcomes.
- Original research studies.
2.2.2. Exclusion Criteria
- Studies without cystatin C measurements.
- Non-neonatal populations.
- Non-renal outcomes.
- Reviews, editorials, letters, or animal studies.
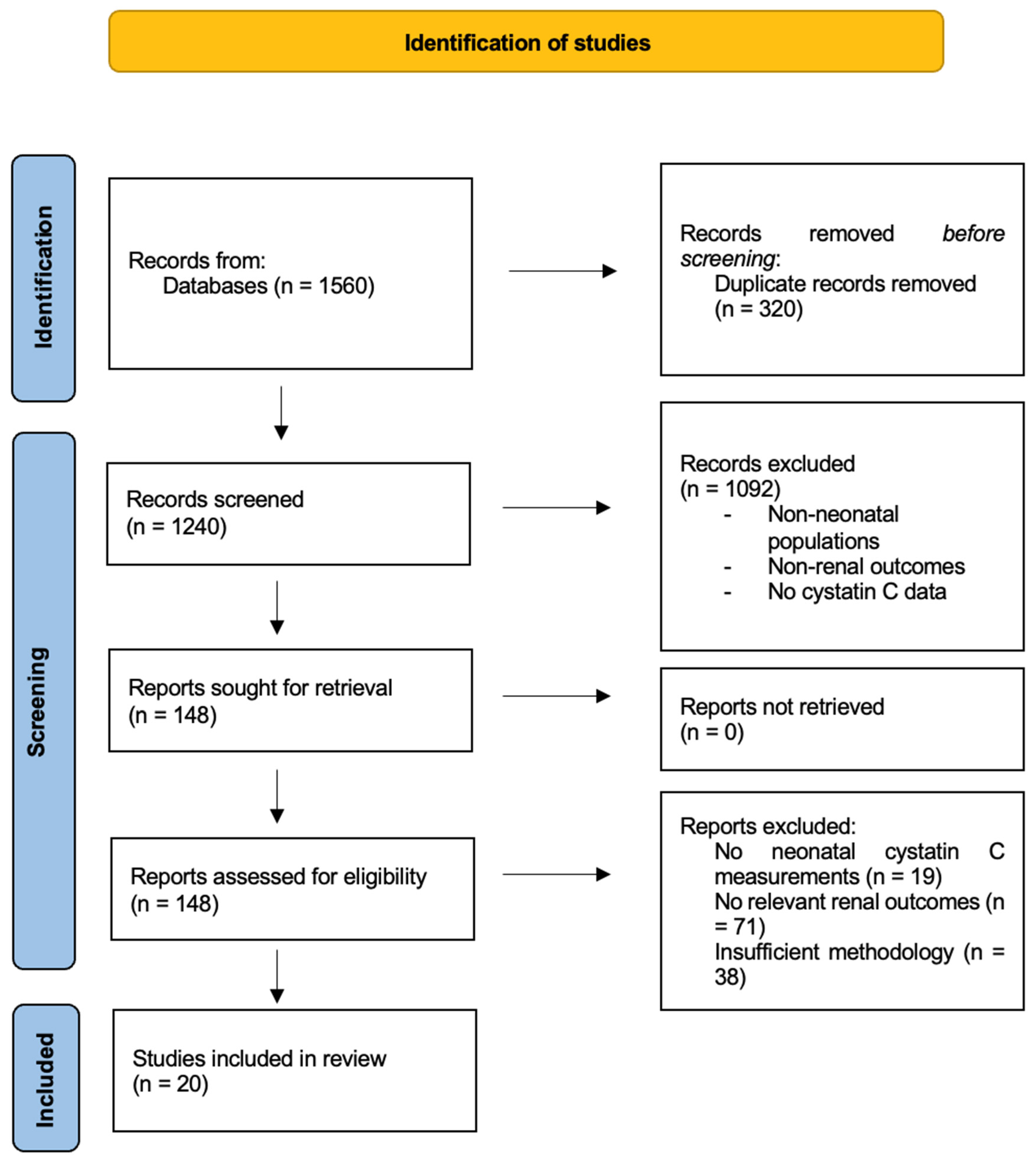
2.2.3. Identification and Screening of Studies
2.3. Data Extraction Process
2.4. Risk of Bias and Methodological Quality Assessment
2.5. Outcome Measures
2.6. Synthesis of Evidence
2.7. Protocol and Registration
3. Results
3.1. Included Studies
3.2. Serum Cystatin C in Healthy Neonates
3.3. Serum Cystatin C in Preterm Infants
3.4. Serum Cystatin C in Congenital Anomalies of the Kidney and Urinary Tract (CAKUT)
3.4.1. Pathophysiological Relevance of Cystatin C in CAKUT
3.4.2. Cystatin C Levels Correlate with CAKUT Severity
3.4.3. Prognostic Implications
3.5. Cystatin C in Neonatal Acute Kidney Injury (AKI)
3.5.1. Early Rise in Cystatin C Compared to Creatinine
3.5.2. Sensitivity and Specificity
3.5.3. Utility in High-Risk Neonatal Populations
3.6. Urinary Cystatin C as a Biomarker of Tubular Injury
3.7. Comparison with Other Renal Biomarkers
3.8. Cystatin C and Long-Term Renal Outcomes (CKD Risk)
4. Discussion
4.1. Limitations of the Review
4.2. Future Perspectives
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AKI | Acute Kidney Injury |
| CAKUT | Congenital Anomalies of the Kidney and Urinary Tract |
| CKD | Chronic Kidney Disease |
| eGFR | Estimated Glomerular Filtration Rate |
| ELBW | Extremely Low Birth Weight |
| GA | Gestational Age |
| GFR | Glomerular Filtration Rate |
| HIE | Hypoxic–Ischemic Encephalopathy |
| IL-18 | Interleukin-18 |
| KDIGO | Kidney Disease: Improving Global Outcomes |
| KIM-1 | Kidney Injury Molecule-1 |
| NGAL | Neutrophil Gelatinase-Associated Lipocalin |
| NICU | Neonatal Intensive Care Unit |
| PMA | Postmenstrual Age |
| RBF | Renal Blood Flow |
| RDS | Respiratory Distress Syndrome |
| VLBW | Very Low Birth Weight |
References
- Allegaert, K.; Mekahli, D.; van den Anker, J. Cystatin C in Newborns: A Promising Renal Biomarker in Search for Standardization and Validation. J. Matern.-Fetal Neonatal Med. 2015, 28, 1833–1838. [Google Scholar] [CrossRef]
- Bökenkamp, A.; Domanetzki, M.; Zinck, R.; Schumann, G.; Byrd, D.; Brodehl, J. Cystatin C—A New Marker of Glomerular Filtration Rate in Children Independent of Age and Height. Pediatrics 1998, 101, 875–881. [Google Scholar] [CrossRef]
- Selewski, D.T.; Charlton, J.R.; Jetton, J.G.; Guillet, R.; Mhanna, M.J.; Askenazi, D.J.; Kent, A.L. Neonatal Acute Kidney Injury. Pediatrics 2015, 136, e463–e473. [Google Scholar] [CrossRef] [PubMed]
- Allegaert, K.; Smits, A.; van Donge, T.; van den Anker, J.; Sarafidis, K.; Levtchenko, E.; Mekahli, D. Renal Precision Medicine in Neonates and Acute Kidney Injury: How to Convert a Cloud of Creatinine Observations to Support Clinical Decisions. Front. Pediatr. 2020, 8, 366. [Google Scholar] [CrossRef] [PubMed]
- Correa, L.P.; Marzano, A.C.S.; Filha, R.S.; Magalhães, R.C.; Simoes-e-Silva, A.C. Biomarkers of Renal Function in Preterm Neonates at 72 h and 3 weeks of Life. J. Pediatr. 2021, 97, 508–513. [Google Scholar] [CrossRef] [PubMed]
- Thongprayoon, C.; Cheungpasitporn, W.; Kashani, K. Serum Creatinine Level, a Surrogate of Muscle Mass, Predicts Mortality in Critically Ill Patients. J. Thorac. Dis. 2016, 8, E305–E311. [Google Scholar] [CrossRef]
- Ryan, D.; Sutherland, M.R.; Flores, T.J.; Kent, A.L.; Dahlstrom, J.E.; Puelles, V.G.; Bertram, J.F.; McMahon, A.P.; Little, M.H.; Moore, L.; et al. Development of the Human Fetal Kidney from Mid to Late Gestation in Male and Female Infants. eBioMedicine 2018, 27, 275–283. [Google Scholar] [CrossRef]
- Chew, J.S.C.; Saleem, M.; Florkowski, C.M.; George, P.M. Cystatin C—A Paradigm of Evidence Based Laboratory Medicine. Clin. Biochem. Rev. 2008, 29, 47–62. [Google Scholar]
- Plebani, M.; Mussap, M.; Bertelli, L.; Moggi, G.; Ruzzante, N.; Fanos, V.; Cataldi, L. Determination of blood cystatin C in pregnant women during labor and in their newborns. Pediatr. Medica Chir. Med. Surg. Pediatr. 1997, 19, 325–329. [Google Scholar]
- Menendez-Castro, C.; Nitz, D.; Cordasic, N.; Jordan, J.; Bäuerle, T.; Fahlbusch, F.B.; Rascher, W.; Hilgers, K.F.; Hartner, A. Neonatal Nephron Loss during Active Nephrogenesis–Detrimental Impact with Long-Term Renal Consequences. Sci. Rep. 2018, 8, 4542. [Google Scholar] [CrossRef]
- Fukunaga, S.; Fujita, Y. Low Glomerular Number at Birth Can Lead to the Development of Chronic Kidney Disease. Front. Endocrinol. 2023, 14, 1120801. [Google Scholar] [CrossRef]
- Nielsen, C.; Andersson Hall, U.; Lindh, C.; Ekström, U.; Xu, Y.; Li, Y.; Holmäng, A.; Jakobsson, K. Pregnancy-Induced Changes in Serum Concentrations of Perfluoroalkyl Substances and the Influence of Kidney Function. Environ. Health 2020, 19, 80. [Google Scholar] [CrossRef] [PubMed]
- Murty, M.S.N.; Sharma, U.K.; Pandey, V.B.; Kankare, S.B. Serum Cystatin C as a Marker of Renal Function in Detection of Early Acute Kidney Injury. Indian J. Nephrol. 2013, 23, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Nada, A.; Bonachea, E.M.; Askenazi, D.J. Acute Kidney Injury in the Fetus and Neonate. Semin. Fetal Neonatal Med. 2017, 22, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Koyner, J.L.; Bennett, M.R.; Worcester, E.M.; Ma, Q.; Raman, J.; Jeevanandam, V.; Kasza, K.E.; O’Connor, M.F.; Konczal, D.J.; Trevino, S.; et al. Urinary Cystatin C as an Early Biomarker of Acute Kidney Injury Following Adult Cardiothoracic Surgery. Kidney Int. 2008, 74, 1059–1069. [Google Scholar] [CrossRef]
- Whiting, P.F.; Rutjes, A.W.S.; Westwood, M.E.; Mallett, S.; Deeks, J.J.; Reitsma, J.B.; Leeflang, M.M.G.; Sterne, J.A.C.; Bossuyt, P.M.M.; QUADAS-2 Group. QUADAS-2: A Revised Tool for the Quality Assessment of Diagnostic Accuracy Studies. Ann. Intern. Med. 2011, 155, 529–536. [Google Scholar] [CrossRef]
- Gualdi-Russo, E.; Zaccagni, L. The Newcastle–Ottawa Scale for Assessing the Quality of Studies in Systematic Reviews. Publications 2026, 14, 4. [Google Scholar] [CrossRef]
- Coleman, C.; Tambay Perez, A.; Selewski, D.T.; Steflik, H.J. Neonatal Acute Kidney Injury. Front. Pediatr. 2022, 10, 842544. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Novo, A.C.d.A.C.F.; Sadeck, L.D.S.R.; Okay, T.S.; Leone, C.R. Longitudinal Study of Cystatin C in Healthy Term Newborns. Clinics 2011, 66, 217–220. [Google Scholar] [CrossRef]
- Finney, H.; Newman, D.J.; Thakkar, H.; Fell, J.M.E.; Price, C.P. Reference Ranges for Plasma Cystatin C and Creatinine Measurements in Premature Infants, Neonates, and Older Children. Arch. Dis. Child. 2000, 82, 71–75. [Google Scholar] [CrossRef]
- Harmoinen, A.; Ylinen, E.; Ala-Houhala, M.; Janas, M.; Kaila, M.; Kouri, T. Reference Intervals for Cystatin C in Pre- and Full-Term Infants and Children. Pediatr. Nephrol. 2000, 15, 105–108. [Google Scholar] [CrossRef]
- Strevens, H.; Wide-Swensson, D.; Torffvit, O.; Grubb, A. Serum Cystatin C for Assessment of Glomerular Filtration Rate in Pregnant and Non-Pregnant Women. Indications of Altered Filtration Process in Pregnancy. Scand. J. Clin. Lab. Investig. 2002, 62, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Treiber, M.; Pečovnik Balon, B.; Gorenjak, M. A New Serum Cystatin C Formula for Estimating Glomerular Filtration Rate in Newborns. Pediatr. Nephrol. 2015, 30, 1297–1305. [Google Scholar] [CrossRef] [PubMed]
- Renganathan, A.; Warner, B.B.; Tarr, P.I.; Dharnidharka, V.R. The Progression of Serum Cystatin C Concentrations within the First Month of Life after Preterm Birth-a Worldwide Systematic Review. Pediatr. Nephrol. 2021, 36, 1709–1718. [Google Scholar] [CrossRef] [PubMed]
- Cataldi, L.; Mussap, M.; Bertelli, L.; Ruzzante, N.; Fanos, V.; Plebani, M. Cystatin C in Healthy Women at Term Pregnancy and in Their Infant Newborns: Relationship between Maternal and Neonatal Serum Levels and Reference Values. Am. J. Perinatol. 1999, 16, 287–295. [Google Scholar] [CrossRef]
- Kandasamy, Y.; Smith, R.; Wright, I.M.R. Measuring Cystatin C to Determine Renal Function in Neonates. Pediatr. Crit. Care Med. 2013, 14, 318–322. [Google Scholar] [CrossRef]
- Zaffanello, M.; Franchini, M.; Fanos, V. Is Serum Cystatin-C a Suitable Marker of Renal Function in Children? Ann. Clin. Lab. Sci. 2007, 37, 233–240. [Google Scholar]
- Tomotaki, S.; Toyoshima, K.; Shimokaze, T.; Shibasaki, J.; Nagafuchi, H. Association between Cord Blood Cystatin C Levels and Early Mortality of Neonates with Congenital Abnormalities of the Kidney and Urinary Tract: A Single-Center, Retrospective Cohort Study. Pediatr. Nephrol. 2017, 32, 2089–2095. [Google Scholar] [CrossRef]
- Parvex, P.; Combescure, C.; Rodriguez, M.; Girardin, E. Is Cystatin C a Promising Marker of Renal Function, at Birth, in Neonates Prenatally Diagnosed with Congenital Kidney Anomalies? Nephrol. Dial. Transplant. 2012, 27, 3477–3482. [Google Scholar] [CrossRef][Green Version]
- Steflea, R.M.; Stroescu, R.; Gafencu, M.; Stoicescu, E.R.; Isac, R.; Olariu, I.-C.; Micsescu-Olah, A.M.; Susa, S.R.; Murariu, M.; Doros, G. A Pilot Comparative Study between Creatinine- and Cystatin-C-Based Equations to Estimate GFR and Kidney Ultrasound Percentiles in Children with Congenital Anomalies of the Kidney and Urinary Tract. Diagnostics 2024, 14, 994. [Google Scholar] [CrossRef]
- Dorum, S.; Silfeler, I.; Dorum, B.A.; Silfeler, D.B.; Canbak, Y.; Say, A. Reference Values of Serum Cystatin-C for Full-Term and Preterm Neonates in Istanbul. Indian J. Pediatr. 2012, 79, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Guignard, J.P.; Torrado, A.; Da Cunha, O.; Gautier, E. Glomerular Filtration Rate in the First Three Weeks of Life. J. Pediatr. 1975, 87, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Elmas, A.T.; Tabel, Y.; Elmas, O.N. Serum Cystatin C Predicts Acute Kidney Injury in Preterm Neonates with Respiratory Distress Syndrome. Pediatr. Nephrol. 2013, 28, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Askenazi, D.J.; Montesanti, A.; Hunley, H.; Koralkar, R.; Pawar, P.; Shuaib, F.; Liwo, A.; Devarajan, P.; Ambalavanan, N. Urine Biomarkers Predict Acute Kidney Injury and Mortality in Very Low Birth Weight Infants. J. Pediatr. 2011, 159, 907–912.e1. [Google Scholar] [CrossRef]
- Refat, N.H.; Fathy, M.A.; Hafez, A.H.A.A.; Sabra, M.A.; Hamed, E.A.; Abdelraheem, Y.F. Early Predictor of Acute Kidney Injury in Newborns with Perinatal Asphyxia. J. Child Sci. 2023, 13, e104–e112. [Google Scholar] [CrossRef]
- Koralkar, R.; Ambalavanan, N.; Levitan, E.B.; McGwin, G.; Goldstein, S.; Askenazi, D. Acute Kidney Injury Reduces Survival in Very Low Birth Weight Infants. Pediatr. Res. 2011, 69, 354–358. [Google Scholar] [CrossRef]
- Hingorani, S.R.; Schmicker, R.H.; Halloran, B.; Brophy, P.; Heagerty, P.J.; Juul, S.; Goldstein, S.L.; Askenazi, D.; Thomas, B.; Elhassan, N.; et al. Association Between Urinary Biomarkers and CKD in Extremely Low Gestational Age Neonates. Am. J. Kidney Dis. 2024, 83, 497–507. [Google Scholar] [CrossRef]
- Allegaert, K. A Mechanistic Reflection on the Relationship between Maternal and Neonatal Serum Creatinine Values at Delivery. Clin. Exp. Nephrol. 2024, 28, 832–833. [Google Scholar] [CrossRef]
- Lee, J.-H.; Hahn, W.-H.; Ahn, J.; Chang, J.-Y.; Bae, C.-W. Serum Cystatin C during 30 Postnatal Days Is Dependent on the Postconceptional Age in Neonates. Pediatr. Nephrol. 2013, 28, 1073–1078. [Google Scholar] [CrossRef]
- Helin, I.; Axenram, M.; Grubb, A. Serum Cystatin C as a Determinant of Glomerular Filtration Rate in Children. Clin. Nephrol. 1998, 49, 221–225. [Google Scholar] [PubMed]
- Tong, C.; Liu, Y.; Wu, Y.; Li, Q.; Wu, Y.; Wang, L.; Chen, Y. Gestational and Age-Specific Cystatin C Reference Intervals for Newborns. Am. J. Perinatol. 2022, 39, 1654–1658. [Google Scholar] [CrossRef]
- van Donge, T.; Allegaert, K.; Gotta, V.; Smits, A.; Levtchenko, E.; Mekahli, D.; van den Anker, J.; Pfister, M. Characterizing Dynamics of Serum Creatinine and Creatinine Clearance in Extremely Low Birth Weight Neonates during the First 6 Weeks of Life. Pediatr. Nephrol. 2021, 36, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Smeets, N.J.L.; IntHout, J.; van der Burgh, M.J.P.; Schwartz, G.J.; Schreuder, M.F.; de Wildt, S.N. Maturation of GFR in Term-Born Neonates: An Individual Participant Data Meta-Analysis. J. Am. Soc. Nephrol. 2022, 33, 1277–1292. [Google Scholar] [CrossRef] [PubMed]
- Gubhaju, L.; Sutherland, M.R.; Horne, R.S.C.; Medhurst, A.; Kent, A.L.; Ramsden, A.; Moore, L.; Singh, G.; Hoy, W.E.; Black, M.J. Assessment of Renal Functional Maturation and Injury in Preterm Neonates during the First Month of Life. Am. J. Physiol.-Ren. Physiol. 2014, 307, F149–F158. [Google Scholar] [CrossRef]
- Bennett, M.R.; Nehus, E.; Haffner, C.; Ma, Q.; Devarajan, P. Pediatric Reference Ranges for Acute Kidney Injury Biomarkers. Pediatr. Nephrol. 2014, 30, 677–685. [Google Scholar] [CrossRef]
- Abitbol, C.L.; Rodriguez, M.M. The Long-Term Renal and Cardiovascular Consequences of Prematurity. Nat. Rev. Nephrol. 2012, 8, 265–274. [Google Scholar] [CrossRef]
- Schreuder, M.; Delemarre-van de Waal, H.; van Wijk, A. Consequences of Intrauterine Growth Restriction for the Kidney. Kidney Blood Press. Res. 2006, 29, 108–125. [Google Scholar] [CrossRef]
- Branagan, A.; Costigan, C.S.; Stack, M.; Slagle, C.; Molloy, E.J. Management of Acute Kidney Injury in Extremely Low Birth Weight Infants. Front. Pediatr. 2022, 10, 867715. [Google Scholar] [CrossRef]
- Vanpée, M.; Blennow, M.; Linné, T.; Herin, P.; Aperia, A. Renal Function in Very Low Birth Weight Infants: Normal Maturity Reached during Early Childhood. J. Pediatr. 1992, 121, 784–788. [Google Scholar] [CrossRef]
- Kastl, J.T. Renal Function in the Fetus and Neonate–the Creatinine Enigma. Semin. Fetal Neonatal Med. 2017, 22, 83–89. [Google Scholar] [CrossRef]
- Shankaran, M.; Scottoline, B.; Imam, F.; Garton, K.; Lohr, J.; Nyangau, E.; Mohammed, H.; Hellerstein, M.; Evans, W.J. Skeletal Muscle Mass Measured Noninvasively by D3-Creatine Dilution in Children and Term Infants. J. Pediatr. Clin. Pract. 2025, 17, 200158. [Google Scholar] [CrossRef]
- Ataei, N.; Bazargani, B.; Ameli, S.; Madani, A.; Javadilarijani, F.; Moghtaderi, M.; Abbasi, A.; Shams, S.; Ataei, F. Early Detection of Acute Kidney Injury by Serum Cystatin C in Critically Ill Children. Pediatr. Nephrol. 2014, 29, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Starr, M.C.; Charlton, J.R.; Guillet, R.; Reidy, K.; Tipple, T.E.; Jetton, J.G.; Kent, A.L.; Abitbol, C.L.; Ambalavanan, N.; Mhanna, M.J.; et al. Advances in Neonatal Acute Kidney Injury. Pediatrics 2021, 148, e2021051220. [Google Scholar] [CrossRef] [PubMed]
- Askenazi, D.J.; Ambalavanan, N.; Goldstein, S.L. Acute Kidney Injury in Critically Ill Newborns: What Do We Know? What Do We Need to Learn? Pediatr. Nephrol. 2009, 24, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Uy, N.; Reidy, K. Developmental Genetics and Congenital Anomalies of the Kidney and Urinary Tract. J. Pediatr. Genet. 2016, 5, 51–60. [Google Scholar] [CrossRef]
- Sanna-Cherchi, S.; Westland, R.; Ghiggeri, G.M.; Gharavi, A.G. Genetic Basis of Human Congenital Anomalies of the Kidney and Urinary Tract. J. Clin. Investig. 2018, 128, 4–15. [Google Scholar] [CrossRef]
- Chevalier, R.L. CAKUT: A Pediatric and Evolutionary Perspective on the Leading Cause of CKD in Childhood. Pediatr. Rep. 2023, 15, 143–153. [Google Scholar] [CrossRef]
- Matsell, D.G.; Catapang, M.; Becknell, B. Predicting Outcomes in Children with Congenital Anomalies of the Kidney and Urinary Tract. Pediatr. Nephrol. 2023, 38, 3407–3415. [Google Scholar] [CrossRef]
- Shokeir, A.A. Role of Urinary Biomarkers in the Diagnosis of Congenital Upper Urinary Tract Obstruction. Indian J. Urol. 2008, 24, 313–319. [Google Scholar] [CrossRef]
- Gnech, M.; Hoen, L.; Radmayr, C.; Burgu, B.; Bogaert, G.; O’Kelly, F.; Castagnetti, M.; Quaedackers, J.; Silay, M.S.; Kennedy, U.; et al. Congenital Lower Urinary Tract Obstruction: Update and Summary of the European Association of Urology and European Society for Paediatric Urology Guidelines. Eur. Urol. Focus 2025, 11, 519–527. [Google Scholar] [CrossRef]
- Lange-Sperandio, B.; Anders, H.-J.; Stehr, M.; Chevalier, R.L.; Klaus, R. Congenital Anomalies of the Kidney and Urinary Tract: A Continuum of Care. Semin. Nephrol. 2023, 43, 151433. [Google Scholar] [CrossRef] [PubMed]
- Phua, Y.L.; Ho, J. Renal Dysplasia in the Neonate. Curr. Opin. Pediatr. 2016, 28, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Humphreys, J.D.; Jain, A.; Khan, A.M.; Findley, T.O.; Romero-Lopez, M.; Saroukhani, S.; Swinford, R.D.; Rysavy, M.A. Urinary Biomarkers of Acute Kidney Injury in Neonates < 25 Weeks’ Gestation: A Pilot Study. Pediatr. Nephrol. 2026, 41, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Calderon-Margalit, R.; Efron, G.; Pleniceanu, O.; Tzur, D.; Stern-Zimmer, M.; Afek, A.; Erlich, T.; Derazne, E.; Kark, J.D.; Keinan-Boker, L.; et al. Congenital Anomalies of the Kidney and Urinary Tract and Adulthood Risk of Urinary Tract Cancer. Kidney Int. Rep. 2021, 6, 946–952. [Google Scholar] [CrossRef]
- Cruzado, L.B.; González, E.P.; Martos, Z.M.; Guitarte, C.B.; Asencio, M.G.; Lagares, S.L.; Patón, M.M.; Padilla, J.P. Serum Cystatin C Levels in Preterm Newborns in Our Setting: Correlation with Serum Creatinine and Preterm Pathologies. Nefrol. (Engl. Ed.) 2015, 35, 296–303. [Google Scholar] [CrossRef]
- Xu, X.; Nie, S.; Xu, H.; Liu, B.; Weng, J.; Chen, C.; Liu, H.; Yang, Q.; Li, H.; Kong, Y.; et al. Detecting Neonatal AKI by Serum Cystatin C. J. Am. Soc. Nephrol. 2023, 34, 1253–1263. [Google Scholar] [CrossRef]
- Yang, H.; Lin, C.; Zhuang, C.; Chen, J.; Jia, Y.; Shi, H.; Zhuang, C. Serum Cystatin C as a Predictor of Acute Kidney Injury in Neonates: A Meta-Analysis. J. Pediatr. 2022, 98, 230–240. [Google Scholar] [CrossRef]
- Leghrouz, B.; Kaddourah, A. Impact of Acute Kidney Injury on Critically Ill Children and Neonates. Front. Pediatr. 2021, 9, 635631. [Google Scholar] [CrossRef]
- Totapally, A.; Bridges, B.C.; Selewski, D.T.; Zivick, E.E. Managing the Kidney–The Role of Continuous Renal Replacement Therapy in Neonatal and Pediatric ECMO. Semin. Pediatr. Surg. 2023, 32, 151332. [Google Scholar] [CrossRef]
- Turner, M.J.; Rumpel, J.A.; Spray, B.J.; Stence, N.; Neuberger, I.; Frymoyer, A.; Chock, V.Y.; Courtney, S.; Gist, K. Urine Biomarkers of Acute Kidney Injury and Association with Brain MRI Abnormalities in Neonatal Hypoxic-Ischemic Encephalopathy. J. Perinatol. 2024, 44, 1203–1207. [Google Scholar] [CrossRef]
- Benoit, S.W.; Ciccia, E.A.; Devarajan, P. Cystatin C as a Biomarker of Chronic Kidney Disease: Latest Developments. Expert. Rev. Mol. Diagn. 2020, 20, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.-F. Cystatin C as a Potential Biomarker for Neonatal Infants. Pediatr. Neonatol. 2016, 57, 163–164. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, Y.; Zheng, X.; Yang, Z.; Deng, K.; Fu, H.; Huang, L. Cystatin C for Predicting Acute Kidney Injury in Critically Ill Children with Bacterial Infections: A Retrospective Cohort Study. BMJ Paediatr. Open 2026, 10, e004152. [Google Scholar] [CrossRef] [PubMed]
- Simsek, A.; Tugcu, V.; Tasci, A.I. New Biomarkers for the Quick Detection of Acute Kidney Injury. ISRN Nephrol. 2012, 2013, 394582. [Google Scholar] [CrossRef]
- Virzì, G.M.; Morisi, N.; Paulo, C.O.; Clementi, A.; Ronco, C.; Zanella, M. Neutrophil Gelatinase-Associated Lipocalin: Biological Aspects and Potential Diagnostic Use in Acute Kidney Injury. J. Clin. Med. 2025, 14, 1570. [Google Scholar] [CrossRef]
- Eldegwi, M.; Hassan, S.; Saadoun, M.; Ahmed, H.; Reyad, H.; Elnahal, A. The Role of Serum Neutrophil Gelatinase-Associated Lipocalin (NGAL) in Detecting Acute Kidney Injury in Preterm Neonates Exposed to Nephrotoxic Drugs. BMC Pediatr. 2025, 26, 63. [Google Scholar] [CrossRef]
- Dokousli, V.; Gkiourtzis, N.; Stoimeni, A.; Samourkasidou, D.; Makedou, K.; Tsakalidis, C.; Koliakos, G.; Tramma, D. Early Detection of Kidney Impairment in School-Aged Children Born Very Preterm: A Parallel Use of Traditional and Modern Biomarkers. Pediatr. Nephrol. 2026, 41, 423–436. [Google Scholar] [CrossRef]
- Mussap, M.; Noto, A. Renal Disorders in Pregnancy. J. Lab. Precis. Med. 2020, 5, 18. [Google Scholar] [CrossRef]
- Gubhaju, L.; Sutherland, M.R.; Black, M.J. Preterm Birth and the Kidney: Implications for Long-Term Renal Health. Reprod. Sci. 2011, 18, 322–333. [Google Scholar] [CrossRef]
- Deffrennes, S.; Rayyan, M.; Fidlers, T.; van den Heuvel, L.; Levtchenko, E.; Arcolino, F.O. Impact of Preterm Birth on Kidney Health and Development. Front. Med. 2024, 11, 1363097. [Google Scholar] [CrossRef]
- Khwaja, A. KDIGO Clinical Practice Guidelines for Acute Kidney Injury. Nephron Clin. Pract. 2012, 120, c179–c184. [Google Scholar] [CrossRef]
- Zappitelli, M.; Ambalavanan, N.; Askenazi, D.J.; Moxey-Mims, M.M.; Kimmel, P.L.; Star, R.A.; Abitbol, C.L.; Brophy, P.D.; Hidalgo, G.; Hanna, M.; et al. Developing a Neonatal Acute Kidney Injury Research Definition: A Report from the NIDDK Neonatal AKI Workshop. Pediatr. Res. 2017, 82, 569–573. [Google Scholar] [CrossRef]
- Inker, L.A.; Schmid, C.H.; Tighiouart, H.; Eckfeldt, J.H.; Feldman, H.I.; Greene, T.; Kusek, J.W.; Manzi, J.; Van Lente, F.; Zhang, Y.L.; et al. Estimating Glomerular Filtration Rate from Serum Creatinine and Cystatin C. N. Engl. J. Med. 2012, 367, 20–29. [Google Scholar] [CrossRef]
| Study/Year | Population | Design | N | Biomarker (s) | Timing of Measurement | Key Outcomes | Comments/Limitations |
|---|---|---|---|---|---|---|---|
| Ferreira Novo et al. [20] | Healthy term neonates | Prospective longitudinal | 82 | Serum cystatin C | Birth, days 3–28 | Predictable postnatal decline; independent of maternal creatinine | Single-center study |
| Finney et al. [21] | Term and preterm neonates | Prospective reference study | 97 | Serum cystatin C, serum creatinine | Birth to day 10 | Gestational age specific reference ranges; cystatin C more reliable than creatinine | Few extremely preterm infants |
| Harmoinen et al. [22] | Term and preterm neonates | Prospective | 84 | Serum cystatin C | Birth to day 5 | Higher cystatin C in preterm infants; correlates with renal maturation | No long-term outcomes |
| Strevens et al. [23] | Healthy term neonates | Prospective | 50 | Serum cystatin C | Birth to day 7 | Decline parallels postnatal GFR maturation | No renal pathology |
| Treiber et al. [24] | Term and preterm neonates | Prospective methodological study | 80 | Serum cystatin C | Birth and early neonatal period | Developed and validated a cystatin C—based formula for estimating GFR in newborns | Single-center cohort; limited external validation |
| Renganathan et al. [25] | Term and preterm neonates | Prospective | 68 | Serum cystatin C | Birth to 1 month | Demonstrated gestational age—dependent cystatin C trajectories during the first postnatal month | Single-center study |
| Cataldi et al. [26] | Healthy term neonates | Prospective | 45 | Serum cystatin C | Cord blood | Demonstrated lack of placental transfer | Small cohort |
| Kandasamy et al. [27] | NICU neonates | Prospective | 58 | Serum cystatin C | First week of life | Earlier renal dysfunction detection than creatinine | Mixed clinical diagnoses |
| Zaffanello et al. [28] | Neonates and children | Observational | 70 | Serum cystatin C | Neonatal period | Reliable marker independent of muscle mass | Mixed pediatric population |
| Tomotaki et al. [29] | Neonates with CAKUT | Prospective | 61 | Serum cystatin C | Cord blood | Elevated cystatin C predicted early mortality | Short follow-up |
| Parvex et al. [30] | Neonates with CAKUT | Prospective | 49 | Serum cystatin C | Birth to day 7 | Correlated with renal dysplasia severity | No long-term outcomes |
| Steflea et al. [31] | Infants with CAKUT | Diagnostic accuracy study | 92 | Cystatin C—based eGFR, creatinine-based eGFR | Neonatal period | Cystatin C—eGFR superior to creatinine | Single assay platform |
| Dorum et al. [32] | Term and preterm neonates | Prospective observational study | 90 | Serum cystatin C | Birth to early neonatal period | Established gestational-age specific serum cystatin C reference values | Single center; no long-term renal outcomes |
| Guignard et al. [33] | Preterm neonates | Physiologic cohort | 44 | Serum cystatin C, serum creatinine | Birth to day 14 | Demonstrated creatinine unreliability in early life | Not outcome-focused |
| Elmas AT et al. [34] | Preterm neonates with RDS | Prospective observational study | 60 | Serum and urinary cystatin C | First 72 h of life | Elevated serum cystatin C levels predicted development of AKI before serum creatinine | Single center study; limited sample size |
| Askenazi et al. [35] | Extremely low birth weight infants | Prospective | 120 | Urinary cystatin C | Days 1–14 | Predicted tubular injury and CKD risk | Urine collection variability |
| Refat NH et al. [36] | Neonates with HIE | Prospective observational study | 80 | Serum cystatin C, serum creatinine | First 72 h of life | Serum cystatin C rose significantly earlier than creatinine; cystatin C demonstrated higher sensitivity for early AKI prediction following perinatal asphyxia | Single center study; short term follow-up |
| Selewski et al. [3] | NICU neonates | Cohort | 250 | Serum cystatin C, serum creatinine | Daily | Improved AKI detection and prognosis | No urinary biomarkers |
| Koralkar et al. [37] | Preterm infants | Prospective follow-up | 72 | Serum cystatin C | Neonatal period and 2-year follow-up | Neonatal cystatin C predicted CKD traits | Underpowered for CKD endpoints |
| Hingorani SR et al. [38] | Extremely low birth weight infants | Prospective cohort | 327 | Urinary cystatin C, NGAL, IL-18, KIM-1 | Neonatal period and longitudinal follow-up | Elevated urinary cystatin C associated with reduced eGFR and development of chronic kidney disease at follow-up | Biomarkers not measured serially in all infants; CKD outcomes assessed later in childhood |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Dobre, M.; Jura, A.M.C.; Stroescu, R.; Popescu, D.E.; David, V.L. Cystatin C as a Renal Biomarker in Infants with Congenital Anomalies of the Kidney and Urinary Tract (CAKUT): A Systematic Review. Diagnostics 2026, 16, 1115. https://doi.org/10.3390/diagnostics16081115
Dobre M, Jura AMC, Stroescu R, Popescu DE, David VL. Cystatin C as a Renal Biomarker in Infants with Congenital Anomalies of the Kidney and Urinary Tract (CAKUT): A Systematic Review. Diagnostics. 2026; 16(8):1115. https://doi.org/10.3390/diagnostics16081115
Chicago/Turabian StyleDobre, Mihaela, Ana Maria Cristina Jura, Ramona Stroescu, Daniela Eugenia Popescu, and Vlad Laurentiu David. 2026. "Cystatin C as a Renal Biomarker in Infants with Congenital Anomalies of the Kidney and Urinary Tract (CAKUT): A Systematic Review" Diagnostics 16, no. 8: 1115. https://doi.org/10.3390/diagnostics16081115
APA StyleDobre, M., Jura, A. M. C., Stroescu, R., Popescu, D. E., & David, V. L. (2026). Cystatin C as a Renal Biomarker in Infants with Congenital Anomalies of the Kidney and Urinary Tract (CAKUT): A Systematic Review. Diagnostics, 16(8), 1115. https://doi.org/10.3390/diagnostics16081115








