Use of Machine Learning in Predicting the Risk of Cirrhosis in Autoimmune Hepatitis Based on Clinical and Immunological Indicators
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
3.1. Patient Characteristics
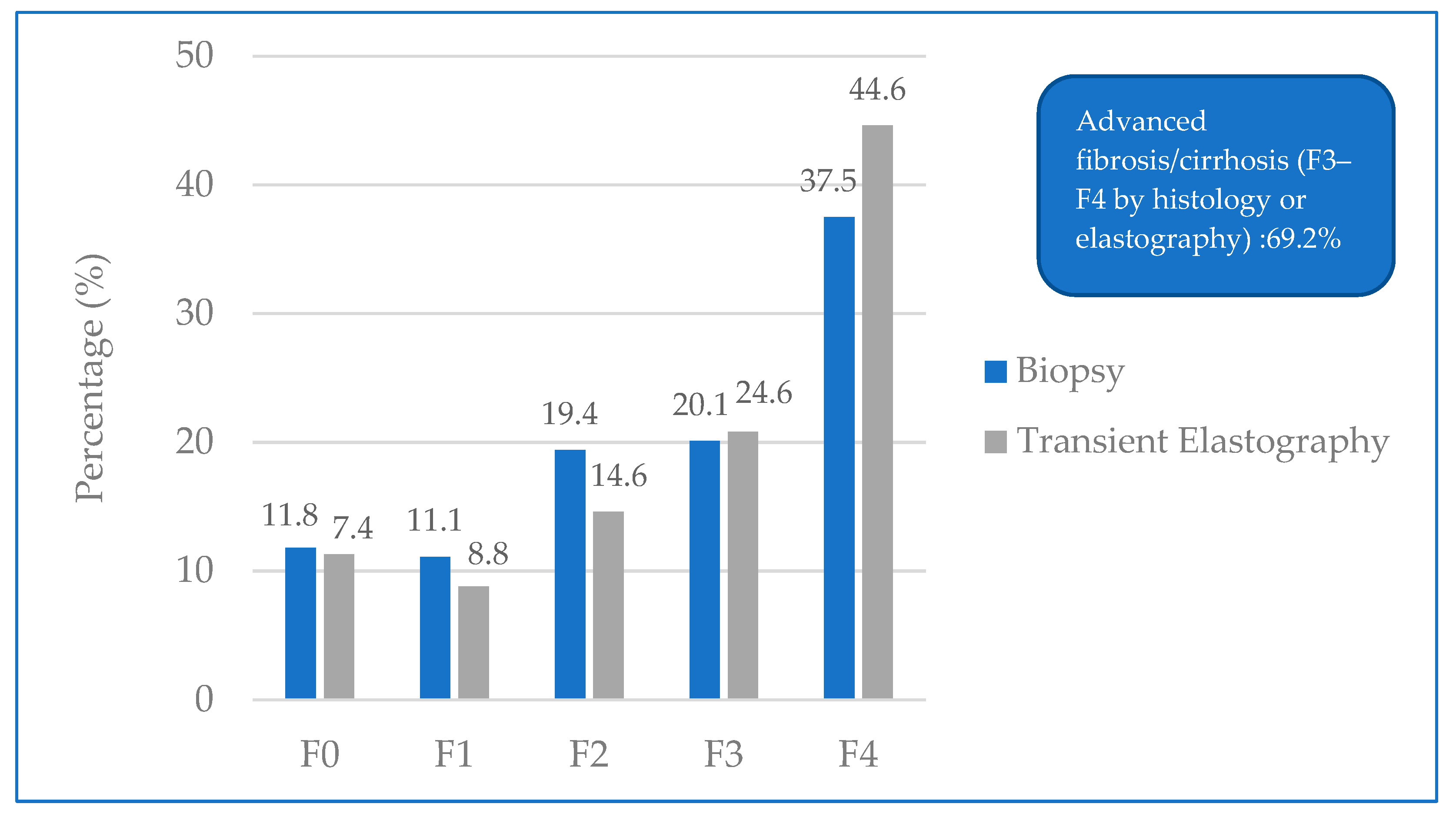
3.2. Fibrosis Assessment
3.3. Concomitant Diseases
3.4. Diagnostic Delay
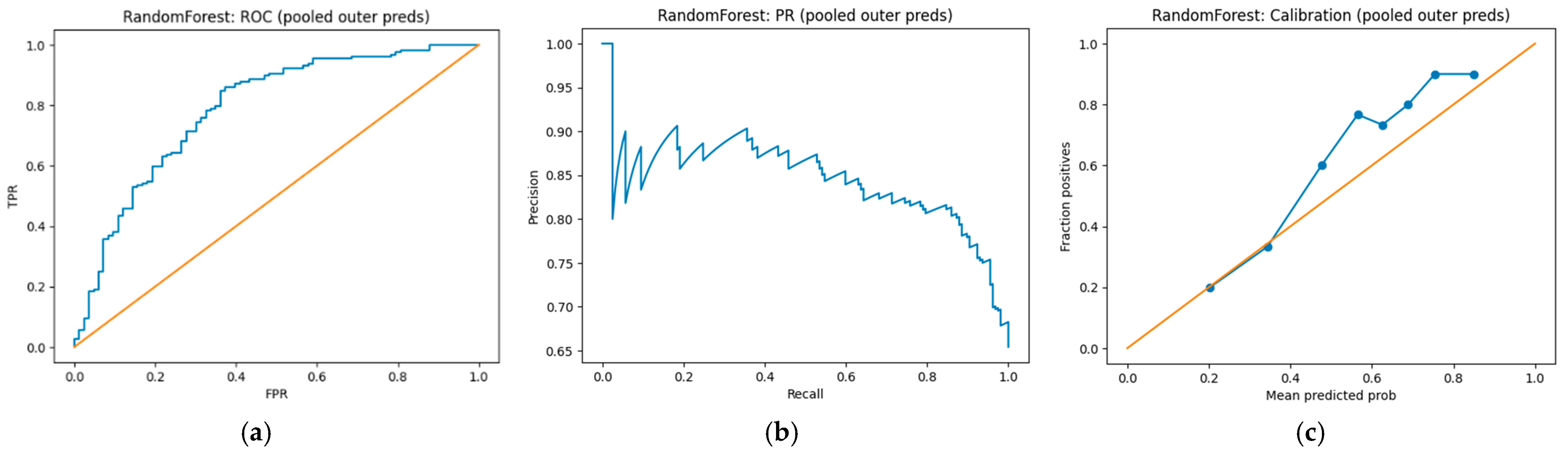
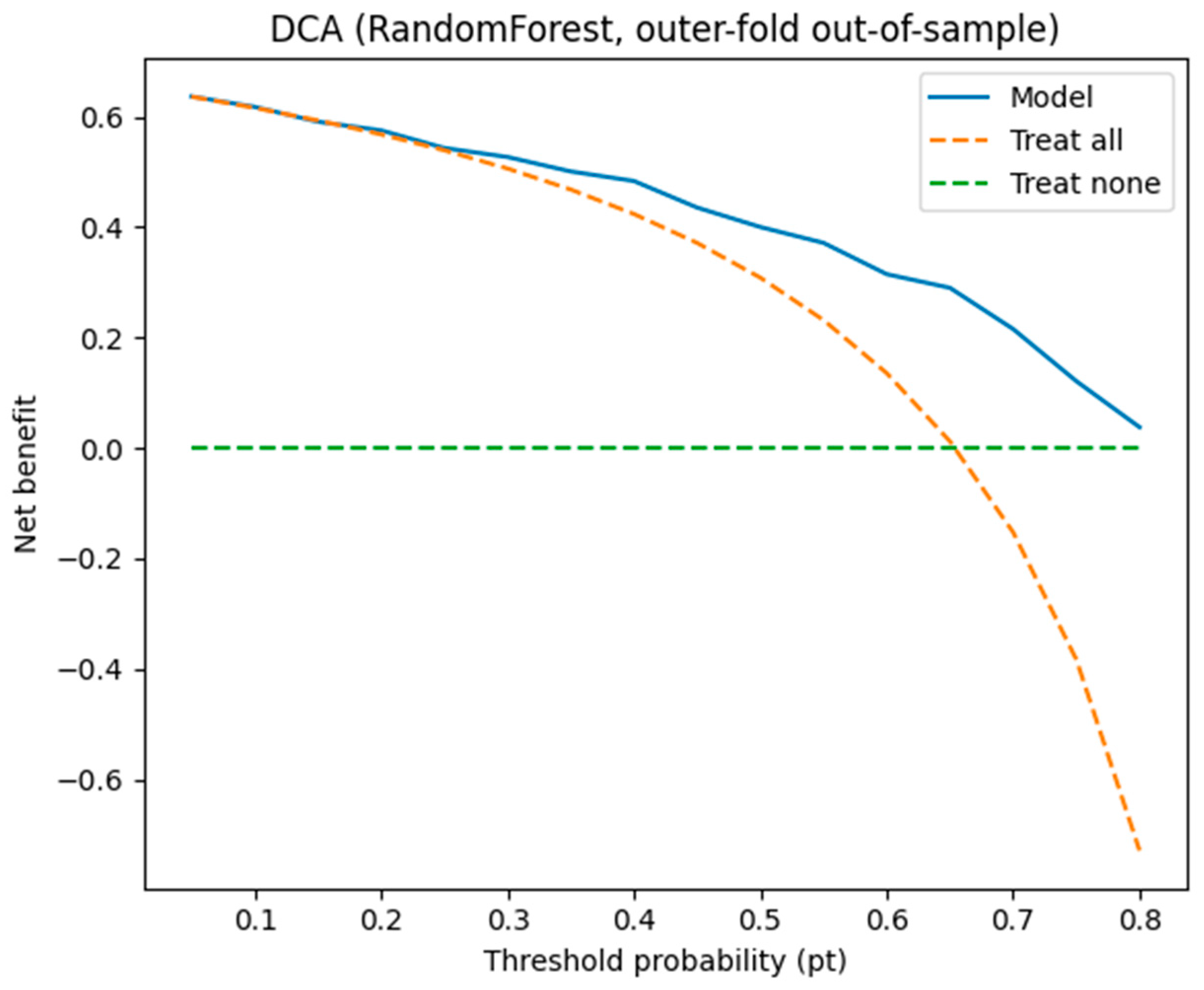
3.5. An Interpretable Machine Learning Model for Predicting Advanced Fibrosis
4. Discussion
4.1. Main Findings and Explanations
4.2. Machine Learning Provision
4.3. Future Directions and Implications
4.4. Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AFP | Alpha-fetoprotein |
| AIH | Autoimmune hepatitis |
| ALP | Alkaline phosphatase |
| ALT | Alanine aminotransferase |
| AMA | Antimitochondrial antibodies |
| ANA | Antinuclear antibodies |
| Anti-HCV | Antibodies to the hepatitis C virus |
| Anti-M2 | Anti-mitochondrial M2 antibodies |
| APRI | AST to Platelet Ratio Index |
| AST | Aspartate aminotransferase |
| BMI | Body mass index |
| COPD | Chronic obstructive pulmonary disease |
| CT | Computed tomography |
| Fib-4 | Fibrosis-4 score |
| GGT | Gamma-glutamyl transferase |
| HBsAg | Hepatitis B surface antigen |
| HIV | Human immunodeficiency virus |
| IAIHG | International Autoimmune Hepatitis Group |
| IgG | Immunoglobulin G |
| IgM | Immunoglobulin M |
| IQR | Interquartile range |
| IST | Immunosuppressive therapy |
| kPa | Kilopascal |
| LKM | Liver kidney microsomal antibodies |
| MASLD | Metabolic dysfunction-associated fatty liver disease |
| p-ANCA | Perinuclear anti-neutrophil cytoplasmic antibodies |
| PBC | Primary biliary cholangitis |
| PSC | Primary sclerosing cholangitis |
| SD | Standard deviation |
| ASMA/SMA | Anti–smooth muscle antibodies |
| TE | Transient elastography |
| UDCA | Ursodeoxycholic acid |
| ULN | Upper limit of normal |
References
- Kochhar, S.; Assis, D.N.; Mack, C.; Izurieta, H.S.; Muratori, L.; Munoz, A.; Nordenberg, D.; Gidudu, J.F.; Blau, E.F.; Vierling, J.M. Au-toimmune hepatitis: Brighton Collaboration case definition and guidelines for data collection, analysis, and presentation of immunisation safety data. Vaccine 2024, 42, 1812–1825. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pape, S.; Snijders, R.J.A.L.M.; Gevers, T.J.G.; Chazouilleres, O.; Dalekos, G.N.; Hirschfield, G.M.; Lenzi, M.; Trauner, M.; Manns, M.P.; Vierling, J.M.; et al. Systematic review of response criteria and endpoints in autoimmune hepatitis by the International Autoimmune Hepatitis Group. J. Hepatol. 2022, 76, 841–849. [Google Scholar] [CrossRef] [PubMed]
- Shiffman, M.L. Autoimmune Hepatitis: Epidemiology, Subtypes, and Presentation. Clin. Liver Dis. 2024, 28, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Katsumi, T.; Ueno, Y. Epidemiology and surveillance of autoimmune hepatitis in Asia. Liver Int. 2022, 42, 2015–2022. [Google Scholar] [CrossRef] [PubMed]
- Heneghan, M.A.; Lohse, A.W. Update in clinical science: Autoimmune hepatitis. J. Hepatol. 2025, 82, 926–937. [Google Scholar] [CrossRef] [PubMed]
- Sucher, E.; Sucher, R.; Gradistanac, T.; Brandacher, G.; Schneeberger, S.; Berg, T. Autoimmune Hepatitis-Immunologically Triggered Liver Pathogenesis-Diagnostic and Therapeutic Strategies. J. Immunol. Res. 2019, 2019, 9437043. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Trivedi, P.J.; Hirschfield, G.M. Recent advances in clinical practice: Epidemiology of autoimmune liver diseases. Gut 2021, 70, 1989–2003. [Google Scholar] [CrossRef] [PubMed]
- Mroskowiak, A.; Suleja, A.; Stec, M.; Kuczmik, W.; Migacz, M.; Holecki, M. Autoimmune Hepatitis-Challenging Diagnosis. Medicina 2022, 58, 896. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Reau, N.S.; Lammert, C.S.; Weinberg, E.M. Autoimmune hepatitis: Current and future therapies. Hepatol. Commun. 2024, 8, e0458. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Enomoto, H.; Nishiguchi, S. Similarities and Differences in Autoimmune Hepatitis Epidemiology between East and West: Autoimmune Hepatitis in East Asia, Southeast Asia, and South Asia. Inflamm. Intest. Dis. 2017, 1, 150–158. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ronca, V.; Parente, A.; Lytvyak, E.; Hansen, B.E.; Hirschfield, G.; Bonder, A.; Ebadi, M.; Elwir, S.; Alsaed, M.; Milkiewicz, P.; et al. Recurrence of autoimmune hepatitis cholestatic variant syndromes after liver transplantation affects graft and patient survival. JHEP Rep. 2025, 7, 101332. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tanaka, A.; Ma, X.; Yokosuka, O.; Weltman, M.; You, H.; Amarapurkar, D.N.; Kim, Y.J.; Abbas, Z.; Payawal, D.A.; Chang, M.L.; et al. Autoimmune liver diseases in the Asia-Pacific region: Proceedings of APASL symposium on AIH and PBC 2016. Hepatol. Int. 2016, 10, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Cai, M.; Wang, G. Incidence and relative risk of colorectal cancer in autoimmune diseases: A global pooled-analysis with more than 91 million participants. Int. J. Surg. 2025, 111, 7236–7242. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sonthalia, N.; Rathi, P.M.; Jain, S.S.; Surude, R.G.; Mohite, A.R.; Pawar, S.V.; Contractor, Q. Natural History and Treatment Out-comes of Severe Autoimmune Hepatitis. J. Clin. Gastroenterol. 2017, 51, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Anand, L.; Choudhury, A.; Bihari, C.; Sharma, B.C.; Kumar, M.; Maiwall, R.; Siam Tan, S.; Shah, S.R.; Hamid, S.; Butt, A.S.; et al. Flare of Autoimmune Hepatitis Causing Acute on Chronic Liver Failure: Diagnosis and Response to Corticosteroid Therapy. Hepatology 2019, 70, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Hassen, H.; Dahmani, W.; Bedoui, G.; Sakly, S.; Souilem, E.; Ghdiri, Y.; Jaziri, H.; Ben Slema, A.; Brahem, A.; Elleuch, N.; et al. Triple autoimmune overlap syndrome of the liver: A rare case of concomitant PBC, PSC, and AIH. Future Sci. OA 2025, 11, 2601633. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jing, M.; Zhang, W.; Xia, M. Ulcerative colitis complicated by autoimmune hepatitis-primary biliary cholangitis-primary sclerosing cholangitis overlap syndrome. Rev. Esp. Enferm. Dig. 2022, 114, 765–766. [Google Scholar] [CrossRef] [PubMed]
- Suleimenova, M.; Abzaliyev, K.; Mansurova, M.; Abzaliyeva, S.; Kurmanova, A.; Tokhtakulinova, G.; Bugibayeva, A.; Sundetova, D.; Abdykassymova, M.; Sagalbayeva, U.; et al. A Predictive Model of Cardiovascular Aging by Clinical and Immunological Markers Using Machine Learning. Diagnostics 2025, 15, 850. [Google Scholar] [CrossRef]
- Alenizi, A.S.; Al-Karawi, K.A. Machine Learning Approach for Liver Disease Prediction. In Proceedings of Tenth International Congress on Information and Communication Technology. ICICT 2025; Springer: Singapore, 2025; pp. 289–306. [Google Scholar] [CrossRef]
- Mostafa, F.; Hasan, E.; Williamson, M.; Khan, H. Statistical Machine Learning Approaches to Liver Disease Prediction. Livers 2021, 1, 294–312. [Google Scholar] [CrossRef]
- Sarkar, S.; Alurwar, A.; Ly, C.; Piao, C.; Donde, R.; Wang, C.J.; Meyers, F.J. A Machine Learning Model to Predict Risk for Hepatocellular Carcinoma in Patients With Metabolic Dysfunction-Associated Steatotic Liver Disease. Gastro Hep Adv. 2024, 3, 498–505. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mercado, L.A.; Gil-Lopez, F.; Chirila, R.M.; Harnois, D.M. Autoimmune Hepatitis: A Diagnostic and Therapeutic Overview. Diagnostics 2024, 14, 382. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, G.; Tanaka, A.; Zhao, H.; Jia, J.; Ma, X.; Harada, K.; Wang, F.S.; Wei, L.; Wang, Q.; Sun, Y.; et al. The Asian Pacific Association for the Study of the Liver clinical practice guidance: The diagnosis and management of patients with autoimmune hepatitis. Hepatol. Int. 2021, 15, 223–257. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Olivas, I.; Rodríguez-Tajes, S.; Londoño, M.C. Autoimmune hepatitis: Challenges and novelties. Med. Clin. 2022, 159, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Mieli-Vergani, G.; Vergani, D.; Czaja, A.J.; Manns, M.P.; Krawitt, E.L.; Vierling, J.M.; Lohse, A.W.; Montano-Loza, A.J. Autoimmune hepatitis. Nat. Rev. Dis. Primers 2018, 4, 18017. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Yang, Y. Advances in the Treatment of Autoimmune Hepatitis. J. Clin. Transl. Hepatol. 2024, 12, 878–885. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hussain, N.; Trivedi, P.J. The Inconvenient Truth of Primary Biliary Cholangitis/Autoimmune Hepatitis Overlap Syndrome. Clin. Liver Dis. 2022, 26, 657–680. [Google Scholar] [CrossRef] [PubMed]
- To, U.; Silveira, M. Overlap Syndrome of Autoimmune Hepatitis and Primary Biliary Cholangitis. Clin. Liver Dis. 2018, 22, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Sohal, A.; Nikzad, N.; Kowdley, K.V. Overlap syndromes in autoimmune liver disease: A review. Transl. Gastroenterol. Hepatol. 2025, 10, 33. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Joshita, S.; Yoshizawa, K.; Umemura, T.; Ohira, H.; Takahashi, A.; Harada, K.; Hiep, N.C.; Tsuneyama, K.; Kage, M.; Nakano, M.; et al. Clinical features of autoimmune hepatitis with acute presentation: A Japanese nationwide survey. J. Gastroenterol. 2018, 53, 1079–1088. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Wu, G.Y. Autoimmune Hepatitis Associated with Other Autoimmune Diseases: A Critical Review. J. Clin. Transl. Hepatol. 2025, 13, 869–877. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zachou, K.; Azariadis, K.; Lytvyak, E.; Snijders, R.J.A.L.M.; Takahashi, A.; Gatselis, N.K.; Robles, M.; Andrade, R.J.; Schramm, C.; Lohse, A.W.; et al. Treatment responses and outcomes in patients with autoimmune hepatitis and concomitant features of non-alcoholic fat-ty liver disease. JHEP Rep. 2023, 5, 100778. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]




| Characteristic | N (%) |
|---|---|
| Age, years | 49.3 ± 14.3 (median 51; IQR 39–61) |
| Female sex | 209 (87.1%) |
| BMI, kg/m2 | 25.1 ± 4.5 (median 24.8; IQR 21.9–27.9) |
| Overweight/obesity † | 117 (48.8%) |
| Disease duration at diagnosis | |
| Duration, months | 36.3 ± 44.8 (median 24; IQR 12–48) |
| ≤12 months | 98 (40.8%) |
| 13–24 months | 42 (17.5%) |
| >24 months | 100 (41.7%) |
| Presenting symptoms | |
| Jaundice | 52 (21.7%) |
| Pruritus | 87 (36.2%) |
| Right upper quadrant pain | 71 (29.6%) |
| Weakness/fatigue | 154 (64.2%) |
| Weight loss | 13 (5.4%) |
| Variable | N (%) |
|---|---|
| ANA positive | 206 (85.8%) |
| ASMA positive | 14 (5.8%) |
| AMA positive (any) | 110 (45.8%) |
| Anti-mitochondrial M2 (AMA-M2) positive | 42 (17.5%) |
| Elevated IgG | 184 (76.7%) |
| Elevated IgM | 65 (27.1%) |
| Elevated γ-globulins | 171 (71.2%) |
| Variable | β (Coefficient) | SE | OR | 95% CI | p-Value |
|---|---|---|---|---|---|
| Advanced fibrosis/cirrhosis (≥F3) | 0.7232 | 0.2997 | 2.06 | 1.15–3.71 | 0.0158 |
| Age (years) | −0.0071 | 0.0098 | 0.99 | 0.97–1.01 | 0.4719 |
| Female sex | −0.5054 | 0.4170 | 0.60 | 0.27–1.37 | 0.2255 |
| Constant | 0.6697 | 0.5851 | — | — | 0.2524 |
| Operating Point | Threshold (Mean ± SD) | Sensitivity | Specificity | PPV | NPV |
|---|---|---|---|---|---|
| Fixed 0.5 | 0.500 ± 0.000 | 0.848 ± 0.045 | 0.557 ± 0.184 | 0.789 ± 0.071 | 0.656 ± 0.040 |
| Youden-optimal | 0.591 ± 0.044 | 0.766 ± 0.153 | 0.701 ± 0.122 | 0.834 ± 0.045 | 0.636 ± 0.105 |
| Sensitivity ≥ 0.85 | 0.474 ± 0.030 | 0.867 ± 0.033 | 0.533 ± 0.165 | 0.782 ± 0.067 | 0.676 ± 0.025 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ashimova, N.; Raissova, A.; Kuantay, E.; Khozhakhmedova, M.; Aldabergenova, N.; Suleimenova, M.; Abzaliyev, K.; Nargiza, N.; Kulmanbetov, R.; Nersesov, A. Use of Machine Learning in Predicting the Risk of Cirrhosis in Autoimmune Hepatitis Based on Clinical and Immunological Indicators. Diagnostics 2026, 16, 974. https://doi.org/10.3390/diagnostics16070974
Ashimova N, Raissova A, Kuantay E, Khozhakhmedova M, Aldabergenova N, Suleimenova M, Abzaliyev K, Nargiza N, Kulmanbetov R, Nersesov A. Use of Machine Learning in Predicting the Risk of Cirrhosis in Autoimmune Hepatitis Based on Clinical and Immunological Indicators. Diagnostics. 2026; 16(7):974. https://doi.org/10.3390/diagnostics16070974
Chicago/Turabian StyleAshimova, Nazugum, Aigul Raissova, Elmira Kuantay, Moldir Khozhakhmedova, Nurgul Aldabergenova, Madina Suleimenova, Kuat Abzaliyev, Nassyrova Nargiza, Ruslan Kulmanbetov, and Alexander Nersesov. 2026. "Use of Machine Learning in Predicting the Risk of Cirrhosis in Autoimmune Hepatitis Based on Clinical and Immunological Indicators" Diagnostics 16, no. 7: 974. https://doi.org/10.3390/diagnostics16070974
APA StyleAshimova, N., Raissova, A., Kuantay, E., Khozhakhmedova, M., Aldabergenova, N., Suleimenova, M., Abzaliyev, K., Nargiza, N., Kulmanbetov, R., & Nersesov, A. (2026). Use of Machine Learning in Predicting the Risk of Cirrhosis in Autoimmune Hepatitis Based on Clinical and Immunological Indicators. Diagnostics, 16(7), 974. https://doi.org/10.3390/diagnostics16070974






