Genetic Disorders Detectable by Fetal MRI: A Review
Abstract
1. Introduction
2. Central Nervous System Disorders
2.1. Brain Malformations
2.2. Mitochondrial Disorders
3. Craniofacial Syndromes and Eye Syndromes
3.1. Craniofacial Syndrome
3.1.1. Malformations of Facial Bones
3.1.2. Malformations of Skull Bones
3.2. Eye Syndromes
3.3. Conclusions
4. Skeletal Dysplasia
5. Congenital Lung Malformations
6. Urinary System Genetic Disorders
6.1. Cystic Kidney Disease
6.2. Renal Agenesis
6.3. Renal Dysplasia
6.4. Lower Urinary Tract Obstruction (LUTO)
6.5. Conclusions
7. Fetal Proportional Analysis
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kingsmore, S.F.; Henderson, A.; Owen, M.J.; Clark, M.M.; Hansen, C.; Dimmock, D.; Chambers, C.D.; Jeliffe-Pawlowski, L.L.; Hobbs, C. Measurement of Genetic Diseases as a Cause of Mortality in Infants Receiving Whole Genome Sequencing. npj Genom. Med. 2020, 5, 49. [Google Scholar] [CrossRef]
- Bomback, M.; Everett, S.; Lyford, A.; Sahni, R.; Kim, F.; Baptiste, C.; Motelow, J.E.; Tolia, V.; Clark, R.; Dugoff, L.; et al. Genetic Disorders and Their Association with Morbidity and Mortality in Early Preterm Small for Gestational Age Infants. Am. J. Obstet. Gynecol. 2025, 232, 487.e1–487.e14. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. EMRO Genetic Diseases. Available online: https://www.emro.who.int/health-topics/genetic-diseases/ (accessed on 26 December 2025).
- Hengstschläger, M. Fetal Magnetic Resonance Imaging and Human Genetics. Eur. J. Radiol. 2006, 57, 312–315. [Google Scholar] [CrossRef] [PubMed]
- Wilson, L.; Whitby, E.H. The Value of Fetal Magnetic Resonance Imaging in Diagnosis of Congenital Anomalies of the Fetal Body: A Systematic Review and Meta-Analysis. BMC Med. Imaging 2024, 24, 111. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, P.D.; Bradburn, M.; Campbell, M.J.; Cooper, C.L.; Embleton, N.; Graham, R.; Hart, A.R.; Jarvis, D.; Kilby, M.D.; Lie, M.; et al. MRI in the Diagnosis of Fetal Developmental Brain Abnormalities: The MERIDIAN Diagnostic Accuracy Study. Health Technol. Assess. 2019, 23, 1–144. [Google Scholar] [CrossRef]
- Prayer, D.; Malinger, G.; De Catte, L.; De Keersmaecker, B.; Gonçalves, L.F.; Kasprian, G.; Laifer-Narin, S.; Lee, W.; Millischer, A.-E.; Platt, L.; et al. ISUOG Practice Guidelines (Updated): Performance of Fetal Magnetic Resonance Imaging. Ultrasound Obstet. Gynecol. 2023, 61, 278–287. [Google Scholar] [CrossRef]
- ACR–SPR Practice Parameter for the safe and optimal performance of fetal magnetic resonance imaging (MRI). Available online: https://gravitas.acr.org/PPTS/GetDocumentView?docId=89 (accessed on 26 December 2025).
- Papaioannou, G.; Klein, W.; Cassart, M.; Garel, C. Indications for Magnetic Resonance Imaging of the Fetal Central Nervous System: Recommendations from the European Society of Paediatric Radiology Fetal Task Force. Pediatr. Radiol. 2021, 51, 2105–2114. [Google Scholar] [CrossRef]
- Papaioannou, G.; Caro-Domínguez, P.; Klein, W.M.; Garel, C.; Cassart, M. Indications for Magnetic Resonance Imaging of the Fetal Body (Extra-Central Nervous System): Recommendations from the European Society of Paediatric Radiology Fetal Task Force. Pediatr. Radiol. 2023, 53, 297–312. [Google Scholar] [CrossRef]
- Doherty, D.; Millen, K.J.; Barkovich, A.J. Midbrain and Hindbrain Malformations: Advances in Clinical Diagnosis, Imaging, and Genetics. Lancet Neurol. 2013, 12, 381–393. [Google Scholar] [CrossRef]
- Melton, K.R.; Iulianella, A.; Trainor, P.A. Gene Expression and Regulation of Hindbrain and Spinal Cord Development. Front. Biosci. 2004, 9, 117–138. [Google Scholar] [CrossRef][Green Version]
- Huang, J.; Wah, I.Y.M.; Pooh, R.K.; Choy, K.W. Molecular Genetics in Fetal Neurology. Semin. Fetal Neonatal Med. 2012, 17, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Guimaraes, C.V.A.; Dahmoush, H.M. Imaging Phenotype Correlation with Molecular and Molecular Pathway Defects in Malformations of Cortical Development. Pediatr. Radiol. 2020, 50, 1974–1987. [Google Scholar] [CrossRef] [PubMed]
- Acrocallosal Syndrome: MedlinePlus Genetics. Available online: https://medlineplus.gov/genetics/condition/acrocallosal-syndrome/ (accessed on 26 December 2025).
- Putoux, A.; Baas, D.; Paschaki, M.; Morlé, L.; Maire, C.; Attié-Bitach, T.; Thomas, S.; Durand, B. Altered GLI3 and FGF8 Signaling Underlies Acrocallosal Syndrome Phenotypes in Kif7 Depleted Mice. Hum. Mol. Genet. 2019, 28, 877–887. [Google Scholar] [CrossRef] [PubMed]
- Shimada, I.S.; Somatilaka, B.N.; Hwang, S.-H.; Anderson, A.G.; Shelton, J.M.; Rajaram, V.; Konopka, G.; Mukhopadhyay, S. Derepression of Sonic Hedgehog Signaling upon Gpr161 Deletion Unravels Forebrain and Ventricular Abnormalities. Dev. Biol. 2019, 450, 47–62. [Google Scholar] [CrossRef]
- Cheng, F.Y.; Fleming, J.T.; Chiang, C. Bergmann Glial Sonic Hedgehog Signaling Activity Is Required for Proper Cerebellar Cortical Expansion and Architecture. Dev. Biol. 2018, 440, 152–166. [Google Scholar] [CrossRef]
- De Luca, A.; Cerrato, V.; Fucà, E.; Parmigiani, E.; Buffo, A.; Leto, K. Sonic Hedgehog Patterning during Cerebellar Development. Cell Mol. Life Sci. 2016, 73, 291–303. [Google Scholar] [CrossRef]
- Fernandez, C.; Soulier, M.; Coulibaly, B.; Liprandi, A.; Benoit, B.; Giuliano, F.; Sigaudy, S.; Figarella-Branger, D.; Fallet-Bianco, C. Acrocallosal Syndrome in Fetus: Focus on Additional Brain Abnormalities. Acta Neuropathol. 2008, 115, 151–156. [Google Scholar] [CrossRef]
- Lerman-Sagie, T.; Pogledic, I.; Leibovitz, Z.; Malinger, G. A Practical Approach to Prenatal Diagnosis of Malformations of Cortical Development. Eur. J. Paediatr. Neurol. 2021, 34, 50–61. [Google Scholar] [CrossRef]
- Kyriakopoulou, V.; Vatansever, D.; Davidson, A.; Patkee, P.; Elkommos, S.; Chew, A.; Martinez-Biarge, M.; Hagberg, B.; Damodaram, M.; Allsop, J.; et al. Normative Biometry of the Fetal Brain Using Magnetic Resonance Imaging. Brain Struct. Funct. 2017, 222, 2295–2307. [Google Scholar] [CrossRef]
- Machado-Rivas, F.; Cortés-Albornoz, M.C.; Afacan, O.; Bedoya, M.A.; Calixto, C.; Choi, J.J.; Ruggiero, M.; Gholipour, A.; Jaimes, C. Fetal MRI at 3 T: Principles to Optimize Success. RadioGraphics 2023, 43, e220141. [Google Scholar] [CrossRef]
- Van den Veyver, I.B. Prenatally Diagnosed Developmental Abnormalities of the Central Nervous System and Genetic Syndromes: A Practical Review. Prenat. Diagn. 2019, 39, 666–678. [Google Scholar] [CrossRef] [PubMed]
- Khandelwal, A.; Aggarwal, A.; Sharma, A.; Malik, A.; Bose, A. Magnetic Resonance Imaging of Malformations of Cortical Development-A Comprehensive Review. World Neurosurg. 2022, 159, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, A.K.; Whitehead, M.T.; Kruszka, P.; Sanapo, L.; Yano, S.; Tanpaiboon, P.; Muenke, M.; Fraser, J.L.; du Plessis, A.J. Prenatal Diagnosis of Diencephalic-Mesencephalic Junction Dysplasia: Fetal Magnetic Resonance Imaging Phenotypes, Genetic Diagnoses, and Outcomes. Prenat. Diagn. 2021, 41, 778–790. [Google Scholar] [CrossRef] [PubMed]
- Fortin, O.; Christoffel, K.; Shoaib, A.B.; Venkatesan, C.; Cilli, K.; Schroeder, J.W.; Alves, C.; Ganetzky, R.D.; Fraser, J.L. Fetal Brain MRI Abnormalities in Pyruvate Dehydrogenase Complex Deficiency. Neurology 2024, 103, e209728. [Google Scholar] [CrossRef]
- Robinson, J.N.; Norwitz, E.R.; Mulkern, R.; Brown, S.A.; Rybicki, F.; Tempany, C.M. Prenatal Diagnosis of Pyruvate Dehydrogenase Deficiency Using Magnetic Resonance Imaging. Prenat. Diagn. 2001, 21, 1053–1056. [Google Scholar] [CrossRef]
- Natarajan, N.; Tully, H.M.; Chapman, T. Prenatal Presentation of Pyruvate Dehydrogenase Complex Deficiency. Pediatr. Radiol. 2016, 46, 1354–1357. [Google Scholar] [CrossRef]
- Tamaru, S.; Kikuchi, A.; Takagi, K.; Okuno, J.; Ishikawa, K.; Imada, S.; Horikoshi, T.; Goto, Y.; Hirabayashi, S. A Case of Pyruvate Dehydrogenase E1α Subunit Deficiency with Antenatal Brain Dysgenesis Demonstrated by Prenatal Sonography and Magnetic Resonance Imaging. J. Clin. Ultrasound 2012, 40, 234–238. [Google Scholar] [CrossRef]
- Wada, N.; Matsuishi, T.; Nonaka, M.; Naito, E.; Yoshino, M. Pyruvate Dehydrogenase E1alpha Subunit Deficiency in a Female Patient: Evidence of Antenatal Origin of Brain Damage and Possible Etiology of Infantile Spasms. Brain Dev. 2004, 26, 57–60. [Google Scholar] [CrossRef]
- Egloff, C.; Eldin de Pecoulas, A.; Mechler, C.; Tassin, M.; Mairovitz, V.; Corrizi, F.; Dussaux, C.; Boutron, A.; Simon, I.; Guet, A.; et al. Prenatal Sonographic Description of Fetuses Affected by Pyruvate Dehydrogenase or Pyruvate Carboxylase Deficiency. Prenat. Diagn. 2018, 38, 607–616. [Google Scholar] [CrossRef]
- Savvidou, A.; Ivarsson, L.; Naess, K.; Eklund, E.A.; Lundgren, J.; Dahlin, M.; Frithiof, D.; Sofou, K.; Darin, N. Novel Imaging Findings in Pyruvate Dehydrogenase Complex (PDHc) Deficiency-Results from a Nationwide Population-Based Study. J. Inherit. Metab. Dis. 2022, 45, 248–263. [Google Scholar] [CrossRef]
- Barnerias, C.; Saudubray, J.-M.; Touati, G.; De Lonlay, P.; Dulac, O.; Ponsot, G.; Marsac, C.; Brivet, M.; Desguerre, I. Pyruvate Dehydrogenase Complex Deficiency: Four Neurological Phenotypes with Differing Pathogenesis. Dev. Med. Child. Neurol. 2010, 52, e1–e9. [Google Scholar] [CrossRef] [PubMed]
- Soares-Fernandes, J.P.; Teixeira-Gomes, R.; Cruz, R.; Ribeiro, M.; Magalhães, Z.; Rocha, J.F.; Leijser, L.M. Neonatal Pyruvate Dehydrogenase Deficiency Due to a R302H Mutation in the PDHA1 Gene: MRI Findings. Pediatr. Radiol. 2008, 38, 559–562. [Google Scholar] [CrossRef] [PubMed]
- Kirschner, R.E.; LaRossa, D. Cleft lip and palate. Otolaryngol. Clin. N. Am. 2000, 33, 1191–1215. [Google Scholar] [CrossRef] [PubMed]
- Stanier, P.; Moore, G.E. Genetics of cleft lip and palate: Syndromic genes contribute to the incidence of non-syndromic clefts. Hum. Mol. Genet. 2004, 13, R73–R81. [Google Scholar] [CrossRef]
- Hammond, N.L.; Dixon, M.J. Revisiting the embryogenesis of lip and palate development. Oral Dis. 2022, 28, 1306–1326. [Google Scholar] [CrossRef]
- Goswami, M.; Jangra, B.; Bhushan, U. Management of feeding Problem in a Patient with Cleft Lip/Palate. Int. J. Clin. Pediatr. Dent. 2016, 9, 143–145. [Google Scholar] [CrossRef]
- Venkatesh, R. Syndromes and anomalies associated with cleft. Indian J. Plast. Surg. 2009, 42, S51–S55. [Google Scholar] [CrossRef]
- Mailath-Pokorny, M.; Worda, C.; Krampl-Bettelheim, E.; Watzinger, F.; Brugger, P.C.; Prayer, D. What does magnetic resonance imaging add to the prenatal ultrasound diagnosis of facial clefts? Ultrasound Obstet. Gynecol. 2010, 36, 445–451. [Google Scholar] [CrossRef]
- Laifer-Narin, S.; Schlechtweg, K.; Lee, J.; Booker, W.; Miller, R.; Ayyala, R.S.; Imahiyerobo, T. A Comparison of Early Versus Late Prenatal Magnetic Resonance Imaging in the Diagnosis of Cleft Palate. Ann. Plast. Surg. 2019, 82, S242–S246. [Google Scholar] [CrossRef]
- van der Hoek-Snieders, H.E.M.; van den Heuvel, A.; van Os-Medendorp, H.; Kamalski, D.M.A. Diagnostic accuracy of fetal MRI to detect cleft palate: A meta-analysis. Eur. J. Pediatr. 2020, 179, 29–38. [Google Scholar] [CrossRef]
- Tian, M.; Xiao, L.; Jian, N.; Wei, X.; Liu, S.; Zhao, H.; Li, G.; Zhang, S.; Liang, W.; Lin, N.; et al. Accurate Diagnosis of Fetal Cleft Lip/Palate by Typical Signs of Magnetic Resonance Imaging. Prenat. Diagn. 2019, 39, 883–889. [Google Scholar] [CrossRef] [PubMed]
- Descamps, M.J.; Golding, S.J.; Sibley, J.; McIntyre, A.; Alvey, C.; Goodacre, T. MRI for definitive in utero diagnosis of cleft palate: A useful adjunct to antenatal care? Cleft Palate Craniofacial J. 2010, 47, 578–585. [Google Scholar] [CrossRef]
- Dabadie, A.; Quarello, E.; Degardin, N.; Desbriere, R.; Heckenroth, H.; Sigaudy, S.; Gorincour, G. Added value of MRI for the prenatal diagnosis of isolated orofacial clefts and comparison with ultrasound. Diagn. Interv. Imaging 2016, 97, 915–921. [Google Scholar] [CrossRef] [PubMed]
- Rosen, H.; Chiou, G.J.; Stoler, J.M.; Mulliken, J.B.; Tarui, T.; Meara, J.G.; Estroff, J.A. Magnetic resonance imaging for detection of brain abnormalities in fetuses with cleft lip and/or cleft palate. Cleft Palate Craniofacial J. 2011, 48, 619–622. [Google Scholar] [CrossRef] [PubMed]
- Davalbhakta, A.; Hall, P.N. The impact of antenatal diagnosis on the effectiveness and timing of counselling for cleft lip and palate. Br. J. Plast. Surg. 2000, 53, 298–301. [Google Scholar] [CrossRef]
- Russell, K.A.; Allen, V.M.; MacDonald, M.E.; Smith, K.; Dodds, L. A population-based evaluation of antenatal diagnosis of orofacial clefts. Cleft Palate Craniofacial J. 2008, 45, 148–153. [Google Scholar] [CrossRef]
- Penny, C.; McGuire, C.; Bezuhly, M. A Systematic Review of Feeding Interventions for Infants with Cleft Palate. Cleft Palate Craniofacial J. 2022, 59, 1527–1536. [Google Scholar] [CrossRef]
- Walsh, K.; Foy, C. Surgical Repair of Cleft Lip: Comparison of Neonatal and Standard Timing in a Systematic Review. Cureus 2025, 17, e92858. [Google Scholar] [CrossRef]
- Jin, S.W.; Sim, K.B.; Kim, S.D. Development and Growth of the Normal Cranial Vault: An Embryologic Review. J. Korean Neurosurg. Soc. 2016, 59, 192–196. [Google Scholar] [CrossRef]
- Boulet, S.L.; Rasmussen, S.A.; Honein, M.A. A population-based study of craniosynostosis in metropolitan Atlanta, 1989–2003. Am. J. Med. Genet. A 2008, 146A, 984–991. [Google Scholar] [CrossRef]
- Johnson, D.; Wilkie, A.O. Craniosynostosis. Eur. J. Hum. Genet. 2011, 19, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Wilkie, A.O. Craniosynostosis: Genes and mechanisms. Hum. Mol. Genet. 1997, 6, 1647–1656. [Google Scholar] [CrossRef] [PubMed]
- Derderian, C.; Seaward, J. Syndromic craniosynostosis. Semin. Plast. Surg. 2012, 26, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Tiberio, F.; Salvati, M.; Polito, L.; Tisci, G.; Vita, A.; Parolini, O.; Massimi, L.; Di Pietro, L.; Ceci, P.; Tamburrini, G.; et al. Targeted Allele-Specific FGFR2 Knockdown via Human Recombinant Ferritin Nanoparticles for Personalized Treatment of Crouzon Syndrome. Mol. Ther. Nucleic Acids 2025, 36, 102427. [Google Scholar] [CrossRef]
- Kimonis, V.; Gold, J.A.; Hoffman, T.L.; Panchal, J.; Boyadjiev, S.A. Genetics of craniosynostosis. Semin. Pediatr. Neurol. 2007, 14, 150–161. [Google Scholar] [CrossRef]
- Kaushik, A.; Bhatia, H.; Sharma, N. Crouzon’s Syndrome: A Rare Genetic Disorder. Int. J. Clin. Pediatr. Dent. 2016, 9, 384–387. [Google Scholar] [CrossRef]
- Gupta, R.; Debnath, N.; Rawat, P.; Singh, N. Crouzon’s syndrome and its dentofacial features. BMJ Case Rep. 2023, 16, e258125. [Google Scholar] [CrossRef]
- Michael Cohen, M., Jr.; Kreiborg, S.; Lammer, E.J.; Cordero, J.F.; Mastroiacovo, P.; Erickson, J.D.; Roeper, P.; Martinez-Frias, M.L. Birth prevalence study of the Apert syndrome. Am. J. Med. Genet. 1992, 42, 655–659. [Google Scholar] [CrossRef]
- Stavropoulos, D.; Tarnow, P.; Mohlin, B.; Kahnberg, K.E.; Hagberg, C. Comparing patients with Apert and Crouzon syndromes—Clinical features and cranio-maxillofacial surgical reconstruction. Swed. Dent. J. 2012, 36, 25–34. [Google Scholar]
- Michael Cohen, M., Jr. Apert, Crouzon, and Pfeiffer Syndromes. In Craniosynostoses: Molecular Genetics, Principles of Diagnosis, and Treatment; Muenke, M., Kress, W., Collmann, H., Solomon, B., Eds.; Karger: Basel, Switzerland, 2011; pp. 94–118. ISBN 978-3-8055-9594-0. [Google Scholar]
- Weber, B.; Schwabegger, A.H.; Vodopiutz, J.; Janecke, A.R.; Forstner, R.; Steiner, H. Prenatal diagnosis of apert syndrome with cloverleaf skull deformity using ultrasound, fetal magnetic resonance imaging and genetic analysis. Fetal Diagn. Ther. 2010, 27, 51–56. [Google Scholar] [CrossRef]
- Gorincour, G.; Rypens, F.; Grignon, A.; Garel, L.; Bortoluzzi, P.; Oligny, L.; Lemyre, E.; Duperron, L. Prenatal diagnosis of cloverleaf skull: Watch the hands! Fetal Diagn. Ther. 2005, 20, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Itoh, S.; Nojima, M.; Yoshida, K. Usefulness of magnetic resonance imaging for accurate diagnosis of Pfeiffer syndrome type II in utero. Fetal Diagn. Ther. 2006, 21, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Tonni, G.; Panteghini, M.; Rossi, A.; Baldi, M.; Magnani, C.; Ferrari, B.; Lituania, M. Craniosynostosis: Prenatal diagnosis by means of ultrasound and SSSE-MRI. Family series with report of neurodevelopmental outcome and review of the literature. Arch. Gynecol. Obstet. 2011, 283, 909–916. [Google Scholar] [CrossRef] [PubMed]
- Werner, H.; Castro, P.; Daltro, P.; Lopes, J.; Ribeiro, G.; Araujo Junior, E. Prenatal diagnosis of Apert syndrome using ultrasound, magnetic resonance imaging, and three-dimensional virtual/physical models: Three case series and literature review. Childs Nerv. Syst. 2018, 34, 1563–1571. [Google Scholar] [CrossRef]
- Fjortoft, M.I.; Sevely, A.; Boetto, S.; Kessler, S.; Sarramon, M.F.; Rolland, M. Prenatal diagnosis of craniosynostosis: Value of MR imaging. Neuroradiology 2007, 49, 515–521. [Google Scholar] [CrossRef]
- Rubio, E.I.; Blask, A.; Bulas, D.I. Ultrasound and MR imaging findings in prenatal diagnosis of craniosynostosis syndromes. Pediatr. Radiol. 2016, 46, 709–718. [Google Scholar] [CrossRef]
- Ketwaroo, P.D.; Robson, C.D.; Estroff, J.A. Prenatal Imaging of Craniosynostosis Syndromes. Semin. Ultrasound CT MR 2015, 36, 453–464. [Google Scholar] [CrossRef]
- Mak, A.S.L.; Leung, K.Y. Prenatal ultrasonography of craniofacial abnormalities. Ultrasonography 2019, 38, 13–24. [Google Scholar] [CrossRef]
- Nemec, S.F.; Kasprian, G.; Brugger, P.C.; Bettelheim, D.; Amann, G.; Nemec, U.; Rotmensch, S.; Graham, J.M.; Rimoin, D.L.; Lachman, R.S.; et al. Abnormalities of the Upper Extremities on Fetal Magnetic Resonance Imaging. Ultrasound Obstet. Gynecol. 2011, 38, 559–567. [Google Scholar] [CrossRef]
- Yoon, A.J.; Pham, B.N.; Dipple, K.M. Genetic Screening in Patients with Craniofacial Malformations. J. Pediatr. Genet. 2016, 5, 220–224. [Google Scholar] [CrossRef]
- Robson, C.D.; Barnewolt, C.E. MR imaging of fetal head and neck anomalies. Neuroimaging Clin. N. Am. 2004, 14, 273–291. [Google Scholar] [CrossRef]
- Kline-Fath, B.M.; Bulas, D.I.; Clauss, S.B. Fundamental and Advanced Fetal Imaging: Ultrasound and MRI, 2nd ed.; Wolters Kluwer: Philadelphia, PA, USA, 2020; pp. 550–626. ISBN 9781975117009. [Google Scholar]
- Fuhrmann, S. Eye morphogenesis and patterning of the optic vesicle. Curr. Top. Dev. Biol. 2010, 93, 61–84. [Google Scholar] [CrossRef]
- Llorente-Gonzalez, S.; Peralta-Calvo, J.; Abelairas-Gomez, J.M. Congenital anophthalmia and microphthalmia: Epidemiology and orbitofacial rehabilitation. Clin. Ophthalmol. 2011, 5, 1759–1765. [Google Scholar] [CrossRef] [PubMed]
- Stoll, C.; Dott, B.; Alembik, Y.; Roth, M.P. Associated anomalies in anophthalmia and microphthalmia. Eur. J. Med. Genet. 2024, 67, 104892. [Google Scholar] [CrossRef] [PubMed]
- Williamson, K.A.; FitzPatrick, D.R. The genetic architecture of microphthalmia, anophthalmia and coloboma. Eur. J. Med. Genet. 2014, 57, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Searle, A.; Shetty, P.; Melov, S.J.; Alahakoon, T.I. Prenatal diagnosis and implications of microphthalmia and anophthalmia with a review of current ultrasound guidelines: Two case reports. J. Med. Case Rep. 2018, 12, 250. [Google Scholar] [CrossRef]
- Burns, N.S.; Iyer, R.S.; Robinson, A.J.; Chapman, T. Diagnostic imaging of fetal and pediatric orbital abnormalities. AJR Am. J. Roentgenol. 2013, 201, W797–W808. [Google Scholar] [CrossRef]
- Unger, S.; Ferreira, C.R.; Mortier, G.R.; Ali, H.; Bertola, D.R.; Calder, A.; Cohn, D.H.; Cormier-Daire, V.; Girisha, K.M.; Hall, C.; et al. Nosology of Genetic Skeletal Disorders: 2023 Revision. Am. J. Med. Genet. A 2023, 191, 1164–1209. [Google Scholar] [CrossRef]
- Gilligan, L.A.; Calvo-Garcia, M.A.; Weaver, K.N.; Kline-Fath, B.M. Fetal Magnetic Resonance Imaging of Skeletal Dysplasias. Pediatr. Radiol. 2020, 50, 224–233. [Google Scholar] [CrossRef]
- Orioli, I.M.; Castilla, E.E.; Barbosa-Neto, J.G. The Birth Prevalence Rates for the Skeletal Dysplasias. J. Med. Genet. 1986, 23, 328–332. [Google Scholar] [CrossRef]
- Warman, M.L.; Cormier-Daire, V.; Hall, C.; Krakow, D.; Lachman, R.; LeMerrer, M.; Mortier, G.; Mundlos, S.; Nishimura, G.; Rimoin, D.L.; et al. Nosology and Classification of Genetic Skeletal Disorders: 2010 Revision. Am. J. Med. Genet. Pt A 2011, 155, 943–968. [Google Scholar] [CrossRef] [PubMed]
- Cole, W.G. Skeletal Dysplasias. In Genetics of Bone Biology and Skeletal Disease; Thakker, R.V., Whyte, M.P., Eisman, J.A., Igarashi, T., Eds.; Academic Press: London, UK, 2013; pp. 325–336. ISBN 9780123878298. [Google Scholar] [CrossRef]
- Bahado-Singh, R.O.; Raymond, M. The 20-Week Scan: Beyond Biometry and Anatomy. Clin. Obstet. Gynecol. 2007, 50, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Krakow, D.; Lachman, R.S.; Rimoin, D.L. Guidelines for the Prenatal Diagnosis of Fetal Skeletal Dysplasias. Genet. Med. 2009, 11, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J.; Henderson, A.; Campbell, S. The Fetal Femur/Foot Length Ratio: A New Parameter to Assess Dysplastic Limb Reduction. Obstet. Gynecol. 1988, 72, 181–184. [Google Scholar]
- Teele, R.L. A Guide to the Recognition of Skeletal Disorders in the Fetus. Pediatr. Radiol. 2006, 36, 473–484. [Google Scholar] [CrossRef]
- Yazici, Z.; Kline-Fath, B.M.; Laor, T.; Tinkle, B.T. Fetal MR Imaging of Kniest Dysplasia. Pediatr. Radiol. 2010, 40, 348–352. [Google Scholar] [CrossRef]
- Dovjak, G. Prenatally Diagnosed Hereditary Chondrodysplasia Punctata in The Early Second Trimester with Fetal MRI and Postmortem Correlation. Ann. Case Rep. 2020, 14, 348. [Google Scholar] [CrossRef]
- Matsubara, Y.; Higaki, T.; Tani, C.; Kamioka, S.; Harada, K.; Aoyama, H.; Nakamura, Y.; Akita, T.; Awai, K. Demonstration of Human Fetal Bone Morphology with MR Imaging: A Preliminary Study. Magn. Reson. Med. Sci. 2020, 19, 310–317. [Google Scholar] [CrossRef]
- Fink, A.M.; Hingston, T.; Sampson, A.; Ng, J.; Palma-Dias, R. Malformation of the Fetal Brain in Thanatophoric Dysplasia: US and MRI Findings. Pediatr. Radiol. 2010, 40, S134–S137. [Google Scholar] [CrossRef]
- Hecht, J.T.; Bodensteiner, J.B.; Butler, I.J. Neurologic Manifestations of Achondroplasia. Handb. Clin. Neurol. 2014, 119, 551–563. [Google Scholar] [CrossRef]
- Mehollin-Ray, A.R.; Stover, S.; Cassady, C.I.; Zhang, B.; Calvo-Garcia, M.; Kline-Fath, B. Using MRI-Derived Observed-to-Expected Total Fetal Lung Volume to Predict Lethality in Fetal Skeletal Dysplasia. Pediatr. Radiol. 2024, 54, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Payette, K.; Geiger, J.; Zellner, M.; Steger, C.; Kellenberger, C.J.; Tuura, R.; Kottke, R.; Jakab, A. Lung Volume Segmentation in Fetal MRI: Super-Resolution Reconstructions Improve Inter-Rater Reliability. Eur. Radiol. Exp. 2025, 9, 88. [Google Scholar] [CrossRef] [PubMed]
- Berceanu, C.; Gheonea, I.A.; Vlădăreanu, S.; Cîrstoiu, M.M.; Vlădăreanu, R.; Mehedinţu, C.; Berceanu, S.; Ciortea, R.; Brătilă, E. Ultrasound and MRI Comprehensive Approach in Prenatal Diagnosis of Fetal Osteochondrodysplasias. Cases Series. Med. Ultrason. 2017, 19, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Baran, K.; Skrzynska, K.; Czyrek, A.A.; Wittek, A.; Krowarsch, D.; Szlachcic, A.; Zakrzewska, M.; Chudzian, J. Fibroblast Growth Factors in Lung Development and Regeneration: Mechanisms and Therapeutic Potential. Cells 2025, 14, 1256. [Google Scholar] [CrossRef]
- Schittny, J.C. Development of the Lung. Cell Tissue Res. 2017, 367, 427–444. [Google Scholar] [CrossRef]
- Nikolić, M.Z.; Sun, D.; Rawlins, E.L. Human Lung Development: Recent Progress and New Challenges. Development 2018, 145, dev163485. [Google Scholar] [CrossRef]
- Pederiva, F.; Rothenberg, S.S.; Hall, N.; Ijsselstijn, H.; Wong, K.K.Y.; von der Thüsen, J.; Ciet, P.; Achiron, R.; Pio d’Adamo, A.; Schnater, J.M. Congenital Lung Malformations. Nat. Rev. Dis. Primers 2023, 9, 60. [Google Scholar] [CrossRef]
- Leblanc, C.; Baron, M.; Desselas, E.; Phan, M.H.; Rybak, A.; Thouvenin, G.; Lauby, C.; Irtan, S. Congenital Pulmonary Airway Malformations: State-of-the-Art Review for Pediatrician’s Use. Eur. J. Pediatr. 2017, 176, 1559–1571. [Google Scholar] [CrossRef]
- Correia-Pinto, J.; Gonzaga, S.; Huang, Y.; Rottier, R. Congenital Lung Lesions—Underlying Molecular Mechanisms. Semin. Pediatr. Surg. 2010, 19, 171–179. [Google Scholar] [CrossRef]
- Morrisey, E.E.; Hogan, B.L.M. Preparing for the First Breath: Genetic and Cellular Mechanisms in Lung Development. Dev. Cell 2010, 18, 8–23. [Google Scholar] [CrossRef]
- Annunziata, F.; Bush, A.; Borgia, F.; Raimondi, F.; Montella, S.; Poeta, M.; Borrelli, M.; Santamaria, F. Congenital Lung Malformations: Unresolved Issues and Unanswered Questions. Front. Pediatr. 2019, 7, 239. [Google Scholar] [CrossRef] [PubMed]
- Cass, D.L.; Olutoye, O.O.; Cassady, C.I.; Moise, K.J.; Johnson, A.; Papanna, R.; Lazar, D.A.; Ayres, N.A.; Belleza-Bascon, B. Prenatal Diagnosis and Outcome of Fetal Lung Masses. J. Pediatr. Surg. 2011, 46, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Sintim-Damoa, A.; Cohen, H.L. Fetal Imaging of Congenital Lung Lesions with Postnatal Correlation. Pediatr. Radiol. 2022, 52, 1921–1934. [Google Scholar] [CrossRef] [PubMed]
- Pacharn, P.; Kline-Fath, B.; Calvo-Garcia, M.; Linam, L.E.; Rubio, E.I.; Salisbury, S.; Brody, A.S. Congenital Lung Lesions: Prenatal MRI and Postnatal Findings. Pediatr. Radiol. 2013, 43, 1136–1143. [Google Scholar] [CrossRef]
- Adams, N.C.; Victoria, T.; Oliver, E.R.; Moldenhauer, J.S.; Adzick, N.S.; Colleran, G.C. Fetal Ultrasound and Magnetic Resonance Imaging: A Primer on How to Interpret Prenatal Lung Lesions. Pediatr. Radiol. 2020, 50, 1839–1854. [Google Scholar] [CrossRef]
- Whitby, E.; Gaunt, T. Fetal Lung MRI and Features Predicting Post-Natal Outcome: A Scoping Review of the Current Literature. Br. J. Radiol. 2023, 96, 20220344. [Google Scholar] [CrossRef]
- Mileto, A.; Itani, M.; Katz, D.S.; Siebert, J.R.; Dighe, M.K.; Dubinsky, T.J.; Moshiri, M. Fetal Urinary Tract Anomalies: Review of Pathophysiology, Imaging, and Management. Am. J. Roentgenol. 2018, 210, 1010–1021. [Google Scholar] [CrossRef]
- Wang, L.; Pan, P.; Ma, H.; He, C.; Qin, Z.; He, W.; Huang, J.; Tan, S.; Meng, D.; Wei, H.; et al. Malformations of Cortical Development: Fetal Imaging and Genetics. Mol. Genet. Genom. Med. 2024, 12, e2440. [Google Scholar] [CrossRef]
- Chalouhi, G.E.; Millischer, A.; Mahallati, H.; Siauve, N.; Melbourne, A.; Grevent, D.; Vinit, N.; Heidet, L.; Aigrain, Y.; Ville, Y.; et al. The Use of Fetal MRI for Renal and Urogenital Tract Anomalies. Prenat. Diagn. 2020, 40, 100–109. [Google Scholar] [CrossRef]
- Manganaro, L.; Capuani, S.; Gennarini, M.; Miceli, V.; Ninkova, R.; Balba, I.; Galea, N.; Cupertino, A.; Maiuro, A.; Ercolani, G.; et al. Fetal MRI: What’s New? A Short Review. Eur. Radiol. Exp. 2023, 7, 41. [Google Scholar] [CrossRef]
- Cassart, M.; Massez, A.; Metens, T.; Rypens, F.; Lambot, M.A.; Hall, M.; Avni, F.E. Complementary Role of MRI After Sonography in Assessing Bilateral Urinary Tract Anomalies in the Fetus. Am. J. Roentgenol. 2004, 182, 689–695. [Google Scholar] [CrossRef]
- Walker, E.Y.X.; Winyard, P.; Marlais, M. Congenital Anomalies of the Kidney and Urinary Tract: Antenatal Diagnosis, Management and Counselling of Families. Pediatr. Nephrol. 2024, 39, 1065–1075. [Google Scholar] [CrossRef]
- Capone, V.; Persico, N.; Berrettini, A.; Decramer, S.; De Marco, E.A.; De Palma, D.; Familiari, A.; Feitz, W.; Herthelius, M.; Kazlauskas, V.; et al. Definition, Diagnosis and Management of Fetal Lower Urinary Tract Obstruction: Consensus of the ERKNet CAKUT-Obstructive Uropathy Work Group. Nat. Rev. Urol. 2022, 19, 295–303. [Google Scholar] [CrossRef]
- Falcon, O.; Cavoretto, P.; Peralta, C.F.; Csapo, B.; Nicolaides, K.H. Fetal Head-to-Trunk Volume Ratio in Chromosomally Abnormal Fetuses at 11 + 0 to 13 + 6 Weeks of Gestation. Ultrasound Obstet. Gynecol. 2005, 26, 755–760. [Google Scholar] [CrossRef]


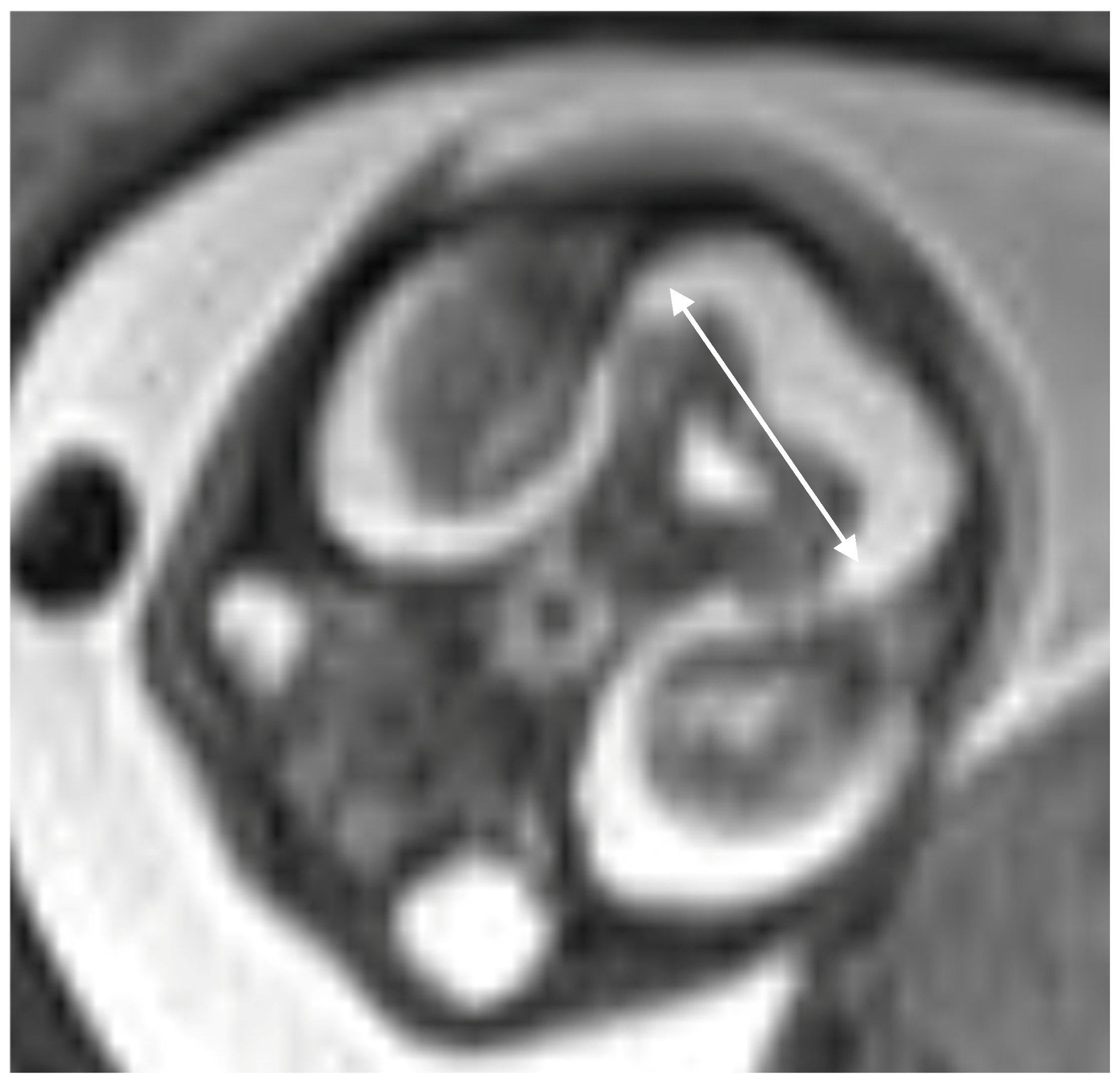





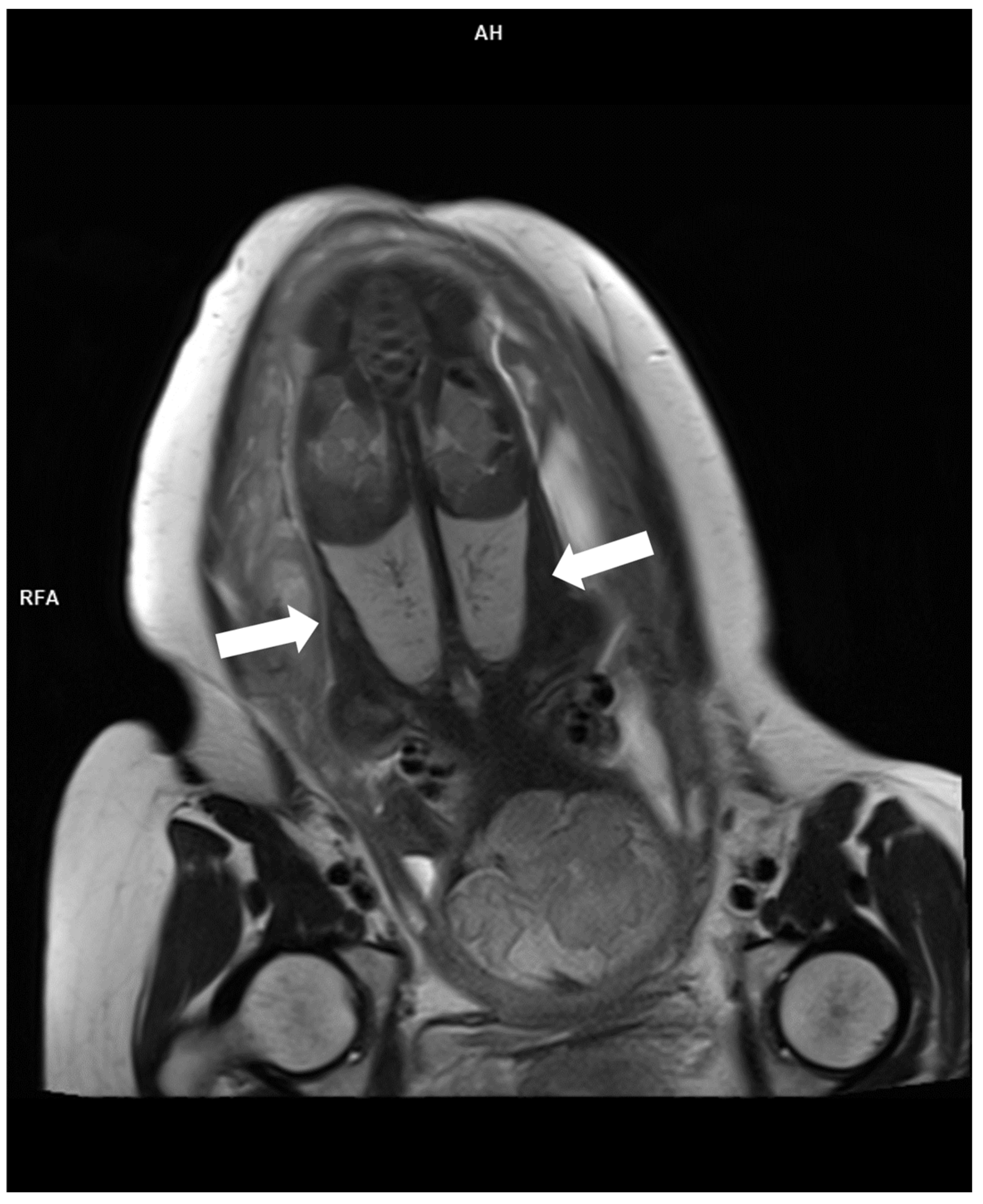


| Feature | ARPKD | ADPKD |
|---|---|---|
| Inheritance | Autosomal recessive | Autosomal dominant |
| Main gene(s) | PKHD1 | PKD1, PKD2 |
| Typical gestation for detection | Mid–late 2nd trimester | Late 2nd–3rd trimester |
| Kidney size | Markedly enlarged, smooth contour | Enlarged, sometimes asymmetric |
| Parenchymal signal | Diffuse T2 hyperintensity with fine microcystic texture | Heterogeneous parenchyma with discrete macrocysts in cortex/medulla |
| Macroscopic cysts on MRI | Absent or minimal | Common and often numerous |
| Hepatic findings | Possible biliary ectasia, congenital hepatic fibrosis | Usually absent prenatally |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wong, K.C.; Chow, T.H.; Cheung, C.; Chan, J.P.K.; Lee, J.C.Y.; Kan, E.Y.L.; Chu, W.C.W. Genetic Disorders Detectable by Fetal MRI: A Review. Diagnostics 2026, 16, 1112. https://doi.org/10.3390/diagnostics16071112
Wong KC, Chow TH, Cheung C, Chan JPK, Lee JCY, Kan EYL, Chu WCW. Genetic Disorders Detectable by Fetal MRI: A Review. Diagnostics. 2026; 16(7):1112. https://doi.org/10.3390/diagnostics16071112
Chicago/Turabian StyleWong, Kwok Chun, Tsz Ho Chow, Claudia Cheung, Joyce Pui Kwan Chan, Jonan Chun Yin Lee, Elaine Yee Ling Kan, and Winnie Chiu Wing Chu. 2026. "Genetic Disorders Detectable by Fetal MRI: A Review" Diagnostics 16, no. 7: 1112. https://doi.org/10.3390/diagnostics16071112
APA StyleWong, K. C., Chow, T. H., Cheung, C., Chan, J. P. K., Lee, J. C. Y., Kan, E. Y. L., & Chu, W. C. W. (2026). Genetic Disorders Detectable by Fetal MRI: A Review. Diagnostics, 16(7), 1112. https://doi.org/10.3390/diagnostics16071112





