Frequency of TERT Promoter Mutations in Ameloblastoma: A Retrospective Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
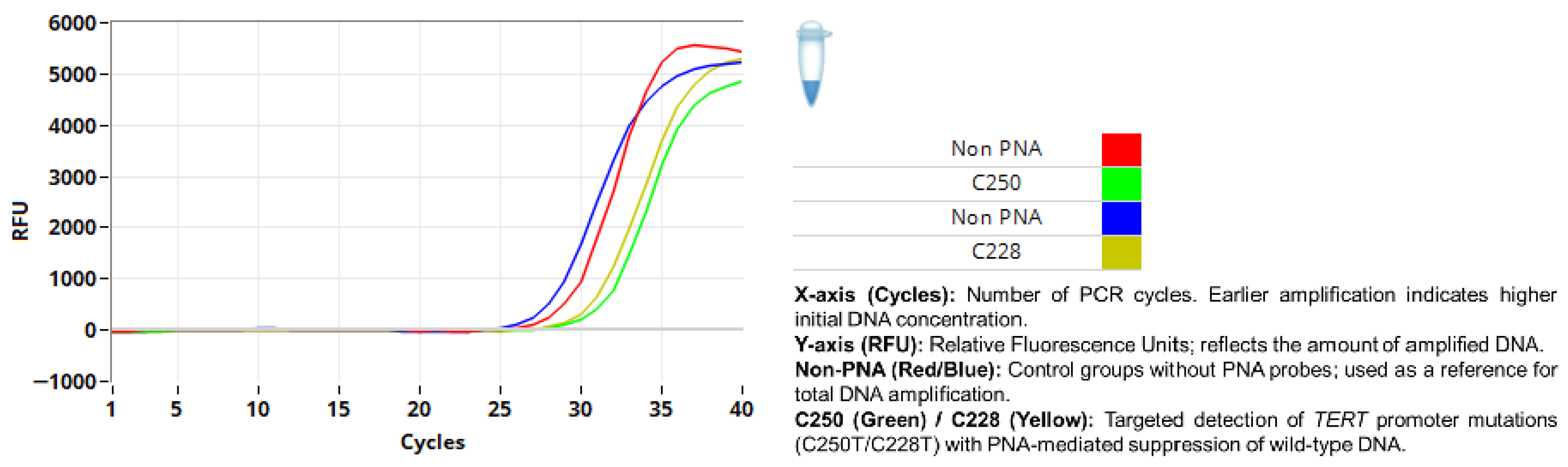
2.2. TERT Promoter Mutation Analyses
2.3. Statistical Analysis
3. Results
3.1. Clinical Characteristics of Patients
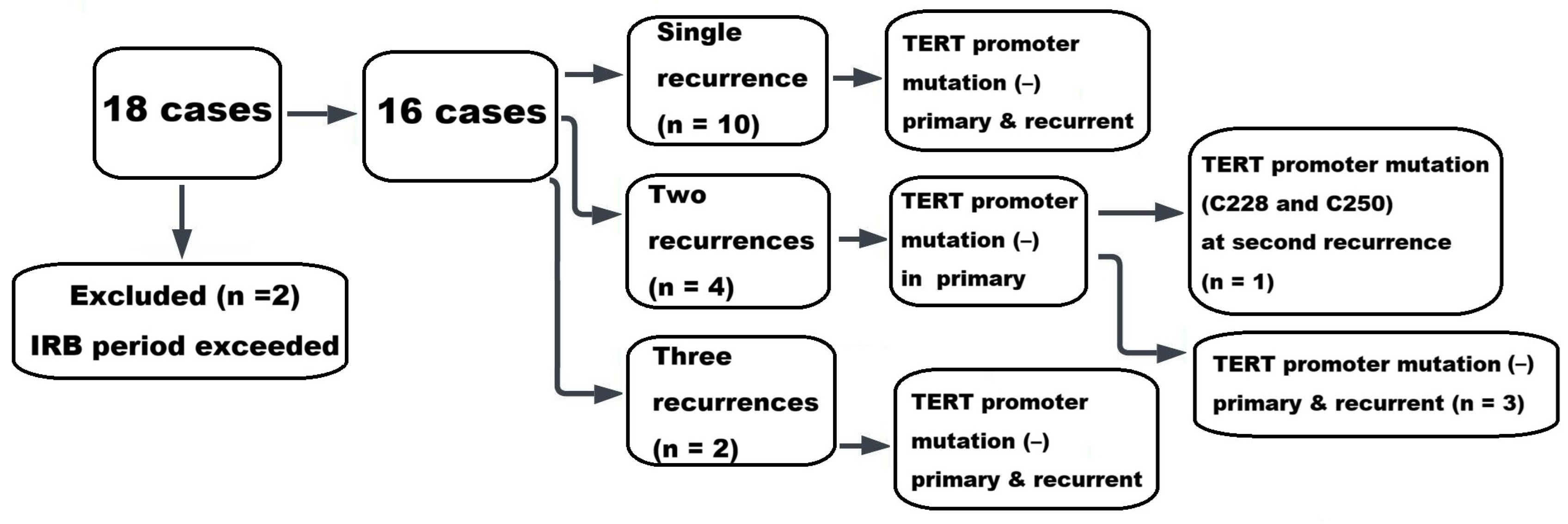
3.2. TERT Promoter Mutation Status in Ameloblastoma
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shay, J.W.; Wright, W.E. Role of Telomeres and Telomerase in Cancer. Semin. Cancer Biol. 2011, 21, 349–353. [Google Scholar] [CrossRef]
- De Lange, T. Shelterin: The Protein Complex that Shapes and Safeguards Human Telomeres. Genes Dev. 2005, 19, 2100–2110. [Google Scholar] [CrossRef]
- Yuan, X.; Larsson, C.; Xu, D. Mechanisms Underlying the Activation of TERT Transcription and Telomerase Activity in Human Cancer: Old Actors and New Players. Oncogene 2019, 38, 6172–6183. [Google Scholar] [CrossRef]
- Dratwa, M.; Wysoczańska, B.; Łacina, P.; Kubik, T.; Bogunia-Kubik, K. TERT—Regulation and Roles in Cancer Formation. Front. Immunol. 2020, 11, 589929. [Google Scholar] [CrossRef]
- Kumamoto, H.; Kinouchi, Y.; Ooya, K. Telomerase Activity and Telomerase Reverse Transcriptase (TERT) Expression in Ameloblastomas. J. Oral Pathol. Med. 2001, 30, 231–236. [Google Scholar] [CrossRef]
- Magalhães, M.C.S.V.; Felix, F.A.; Guimarães, L.M.; Dos Santos, J.N.; De Marco, L.A.; Gomez, R.S.; Gomes, C.C.; De Sousa, S.F. Interrogation of TERT Promoter Hotspot Mutations in Ameloblastoma and Ameloblastic Carcinoma. J. Oral Pathol. Med. 2023, 52, 271–275. [Google Scholar] [CrossRef]
- Tao, Q.; Lv, B.; Qiao, B.; Zheng, C.; Chen, Z. Immortalization of Ameloblastoma Cells via Reactivation of Telomerase Function: Phenotypic and Molecular Characteristics. Oral Oncol. 2009, 45, e239–e244. [Google Scholar] [CrossRef]
- Ajila, V.; Hegde, S. Ameloblastomas vs. Recurrent Ameloblastomas: A Systematic Review. J. Oral Med. Oral Surg. 2022, 28, 11. [Google Scholar] [CrossRef]
- Duarte-Andrade, F.F.; Vitório, J.G.; Pereira, T.D.S.F.; Gomes, C.C.; Gomez, R.S. A Review of the Molecular Profile of Benign and Malignant Odontogenic Lesions. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2020, 129, 357–368. [Google Scholar] [CrossRef]
- Guimarães, L.M.; Coura, B.P.; Gomez, R.S.; Gomes, C.C. The Molecular Pathology of Odontogenic Tumors: Expanding the Spectrum of MAPK Pathway Driven Tumors. Front. Oral Health 2021, 2, 740788. [Google Scholar] [CrossRef]
- Tun, K.M.; Lapthanasupkul, P.; Iamaroon, A.; Thosaporn, W.; Klanrit, P.; Kintarak, S.; Thanasan, S.; Srimaneekarn, N.; Kitkumthorn, N. A Multi-Center Cross-Sectional Investigation of BRAF V600E Mutation in Ameloblastoma. PeerJ 2025, 13, e19137. [Google Scholar] [CrossRef]
- Silveira, F.; Schuch, L.; Pereira-Prado, V.; Sicco, E.; Almeda-Ojeda, O.; Molina-Frechero, N.; Paparella, M.; Villarroel-Dorrego, M.; Hernadez, M.; Bologna-Molina, R. Frequency of BRAF V600E Immunoexpression in Ameloblastomas: A Multi-Institutional Analysis of 86 Cases in Latin America and Comprehensive Review of the Literature. Med. Oral 2024, 29, e509–e516. [Google Scholar] [CrossRef]
- Derakhshan, S.; Aminishakib, P.; Karimi, A.; Saffar, H.; Abdollahi, A.; Mohammadpour, H.; Kharazi Fard, M.; Memarha, A. High Frequency of BRAF V600E Mutation in Iranian Population Ameloblastomas. Med. Oral 2020, 25, e502–e507. [Google Scholar] [CrossRef]
- Mamat Yusof, M.N.; Ch’ng, E.S.; Radhiah Abdul Rahman, N. BRAF V600E Mutation in Ameloblastoma: A Systematic Review and Meta-Analysis. Cancers 2022, 14, 5593. [Google Scholar] [CrossRef]
- Gomes, C.C. Recurrent Driver Mutations in Benign Tumors. Mutat. Res./Rev. Mutat. Res. 2022, 789, 108412. [Google Scholar] [CrossRef]
- Gomes, C.C.; De Sousa, S.F.; Gomez, R.S. Craniopharyngiomas and Odontogenic Tumors Mimic Normal Odontogenesis and Share Genetic Mutations, Histopathologic Features, and Molecular Pathways Activation. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2019, 127, 231–236. [Google Scholar] [CrossRef]
- Wu, Y.; Shi, L.; Zhao, Y.; Chen, P.; Cui, R.; Ji, M.; He, N.; Wang, M.; Li, G.; Hou, P. Synergistic Activation of Mutant TERT Promoter by Sp1 and GABPA in BRAF V600E-Driven Human Cancers. npj Precis. Oncol. 2021, 5, 3. [Google Scholar] [CrossRef]
- Tavallaee, M.; Steiner, D.F.; Zehnder, J.L.; Folkins, A.K.; Karam, A.K. Coexistence of BRAF V600E and TERT Promoter Mutations in Low-Grade Serous Carcinoma of Ovary Recurring as Carcinosarcoma in a Lymph Node: Report of a Case. Int. J. Gynecol. Pathol. 2019, 38, 386–392. [Google Scholar] [CrossRef]
- Gabler, L.; Lötsch, D.; Kirchhofer, D.; Van Schoonhoven, S.; Schmidt, H.M.; Mayr, L.; Pirker, C.; Neumayer, K.; Dinhof, C.; Kastler, L.; et al. TERT Expression Is Susceptible to BRAF and ETS-Factor Inhibition in BRAF V600E/TERT Promoter Double-Mutated Glioma. Acta Neuropathol. Commun. 2019, 7, 128. [Google Scholar] [CrossRef]
- Li, Y.; Cheng, H.S.; Chng, W.J.; Tergaonkar, V. Activation of Mutant TERT Promoter by RAS-ERK Signaling Is a Key Step in Malignant Progression of BRAF-Mutant Human Melanomas. Proc. Natl. Acad. Sci. USA 2016, 113, 14402–14407. [Google Scholar] [CrossRef]
- Sumida, T.; Sogawa, K.; Hamakawa, H.; Sugita, A.; Tanioka, H.; Ueda, N. Detection of Telomerase Activity in Oral Lesions. J. Oral Pathol. Med. 1998, 27, 111–115. [Google Scholar] [CrossRef]




| Variable | Category | Recur (+) | Recur (−) | p-Value |
|---|---|---|---|---|
| Sex, n (%) | Male | 11 (61) | 7 (39) | 0.03 |
| Female | 7 (25) | 21 (75) | ||
| Age, years | Range (median) | 10–79 (46.5) | 8–77 (23) | 0.2 |
| Location, n (%) | Mandible | 13 (33) | 26 (67) | 0.09 |
| Maxilla | 5 (71) | 2 (29) | ||
| Tumor size, cm | Range (median) | 1.0–7.0 (2.5) | 0.9–9.6 (3.5) | 0.3 |
| Case | Recurrence Pattern | Episode | TERT Status | Tissue Availability/ Analysis |
|---|---|---|---|---|
| 1 | Recurrent | Primary | Wild type | Available/Successful |
| 1st recurrence | Wild type | Available/Successful | ||
| 2nd recurrence | C228T + C250T | Available/Successful | ||
| 2 | Non-recurrent | Primary | C250T | Available/Successful |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kim, M.-s.; Son, S.-A.; Choi, S.-Y. Frequency of TERT Promoter Mutations in Ameloblastoma: A Retrospective Study. Diagnostics 2026, 16, 1078. https://doi.org/10.3390/diagnostics16071078
Kim M-s, Son S-A, Choi S-Y. Frequency of TERT Promoter Mutations in Ameloblastoma: A Retrospective Study. Diagnostics. 2026; 16(7):1078. https://doi.org/10.3390/diagnostics16071078
Chicago/Turabian StyleKim, Mee-seon, Shin-Ah Son, and So-Young Choi. 2026. "Frequency of TERT Promoter Mutations in Ameloblastoma: A Retrospective Study" Diagnostics 16, no. 7: 1078. https://doi.org/10.3390/diagnostics16071078
APA StyleKim, M.-s., Son, S.-A., & Choi, S.-Y. (2026). Frequency of TERT Promoter Mutations in Ameloblastoma: A Retrospective Study. Diagnostics, 16(7), 1078. https://doi.org/10.3390/diagnostics16071078





