Postmenopausal Enlargement of a Presumed Leiomyoma Revealing STUMP: A Diagnostic Pitfall with Important Clinical Implications—A Case Report
Abstract
1. Introduction
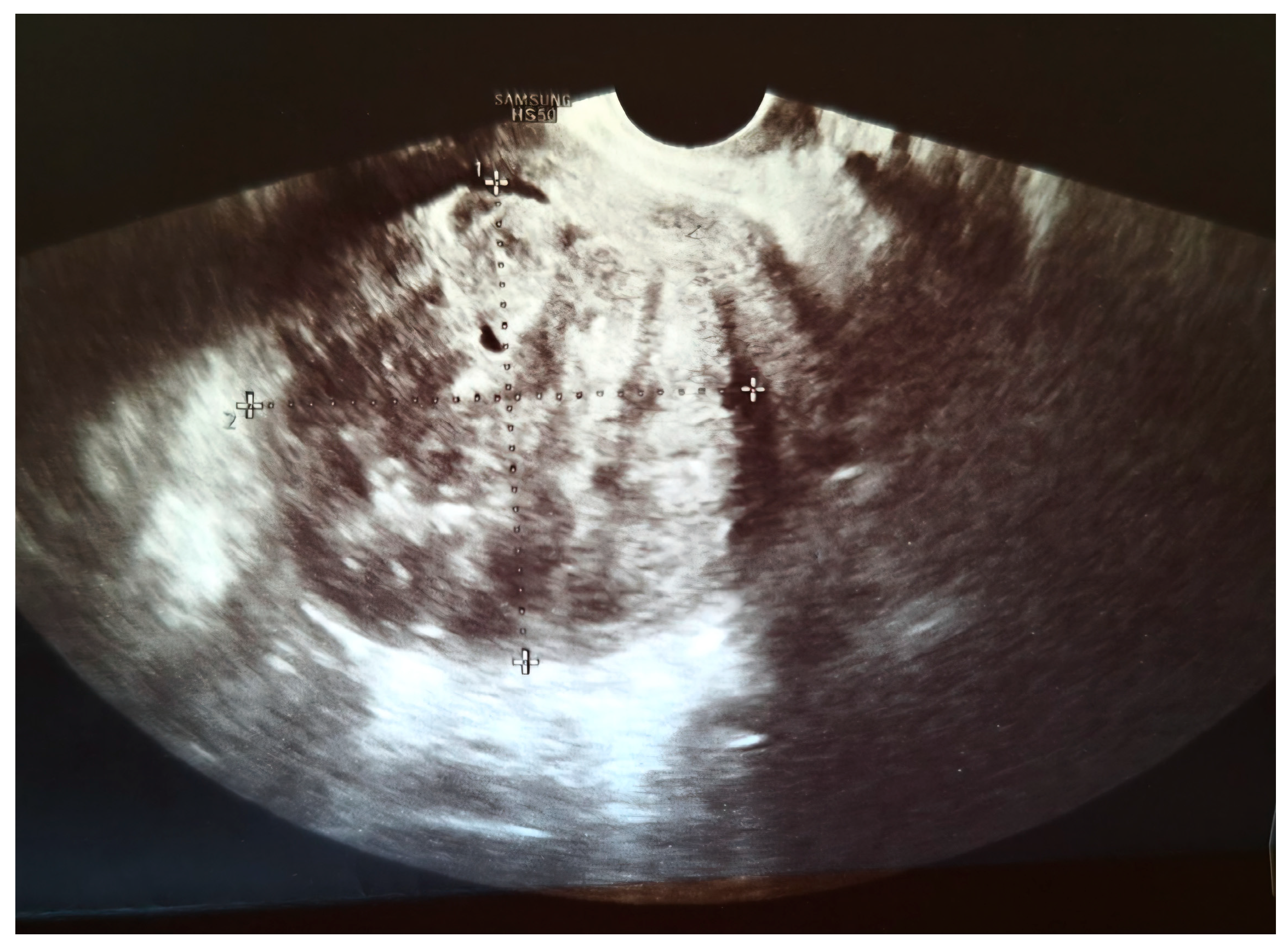
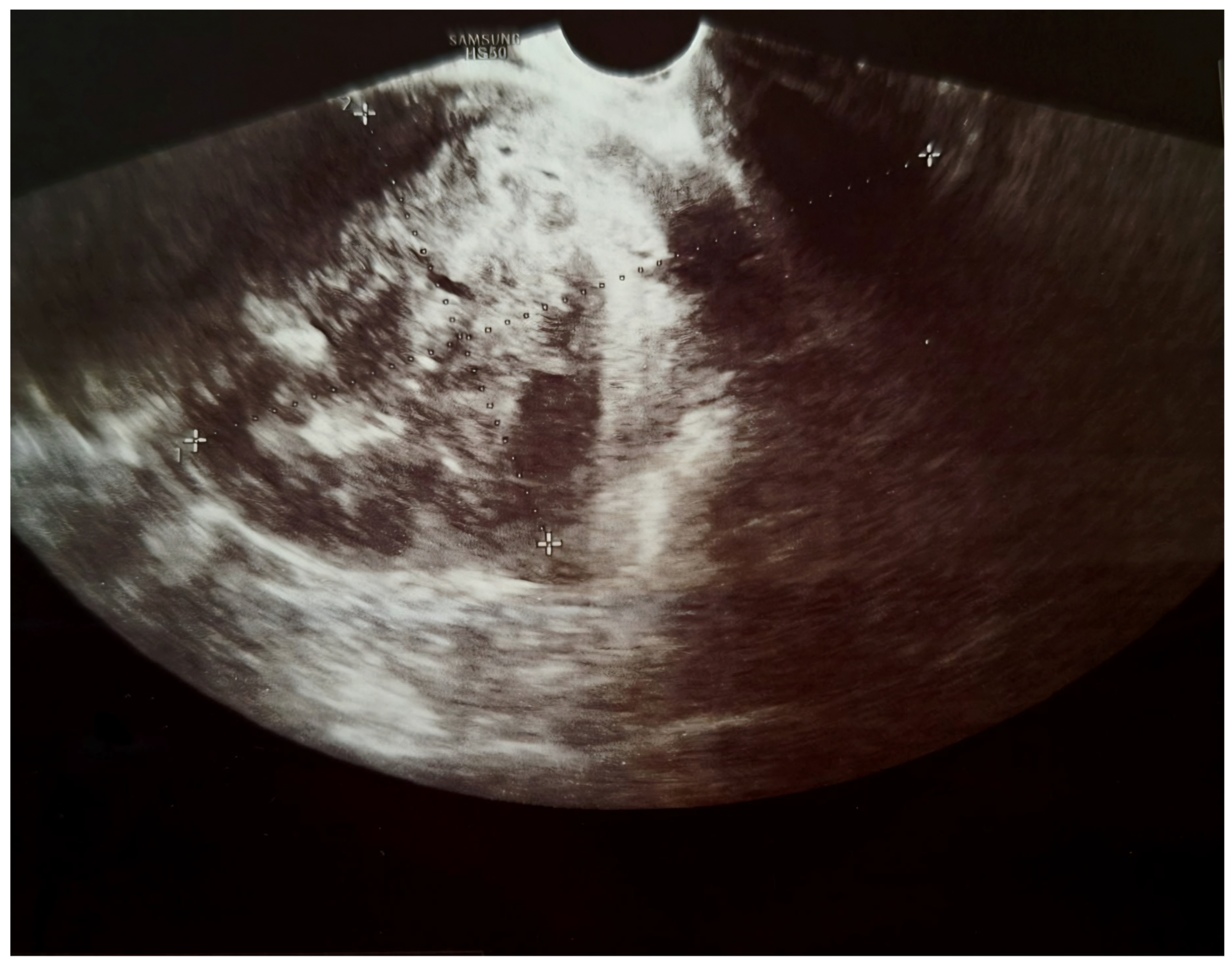
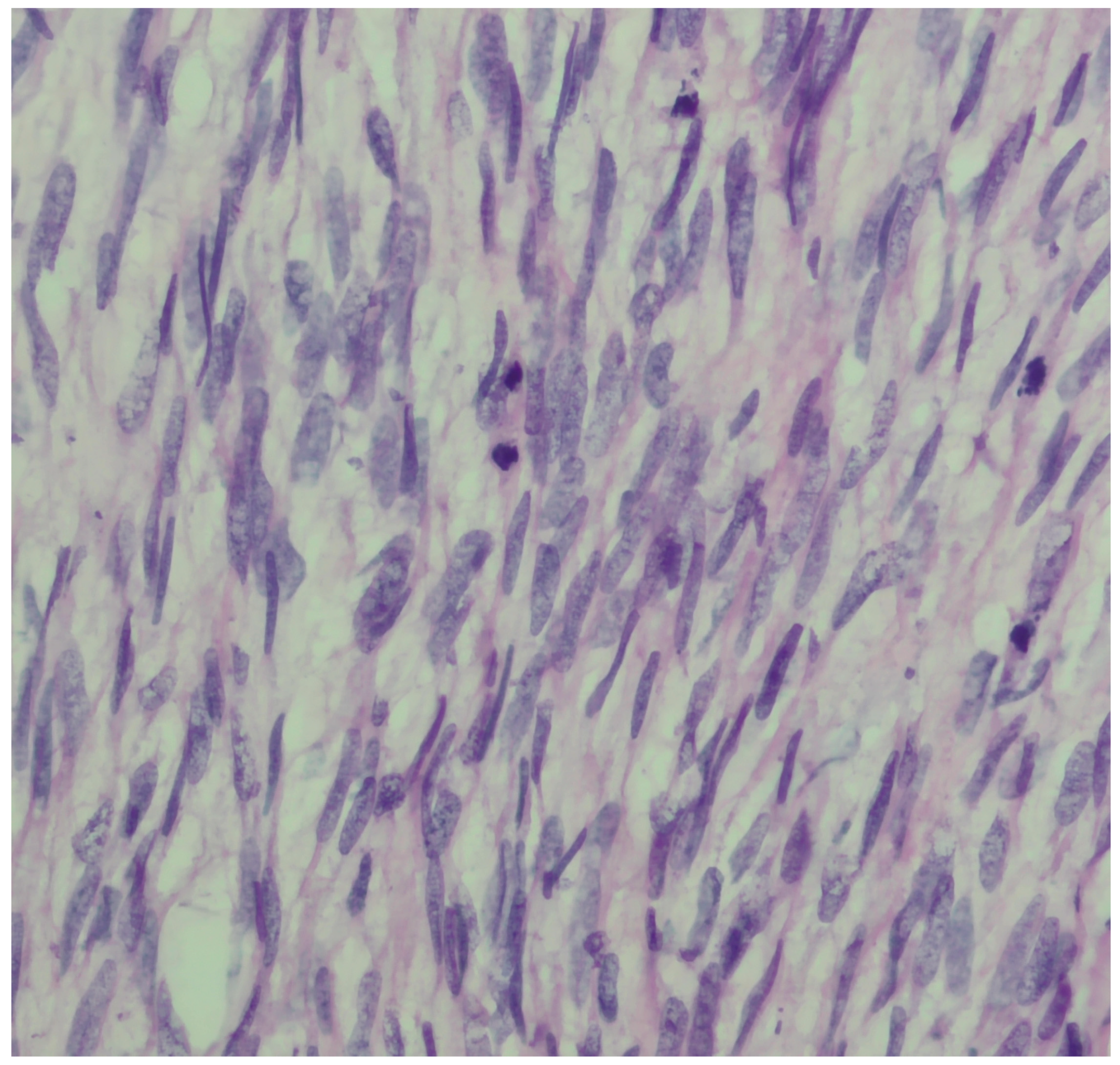
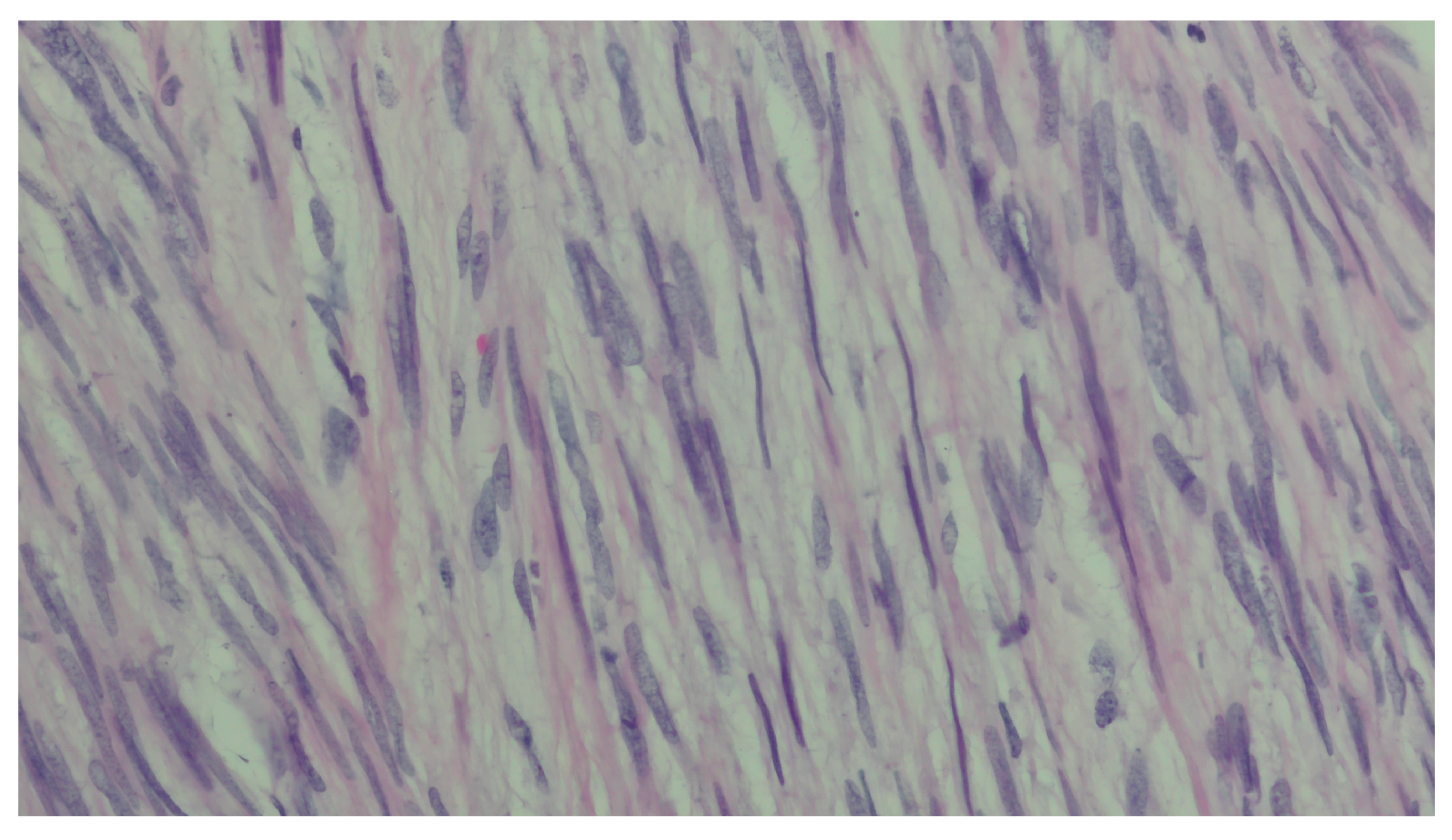
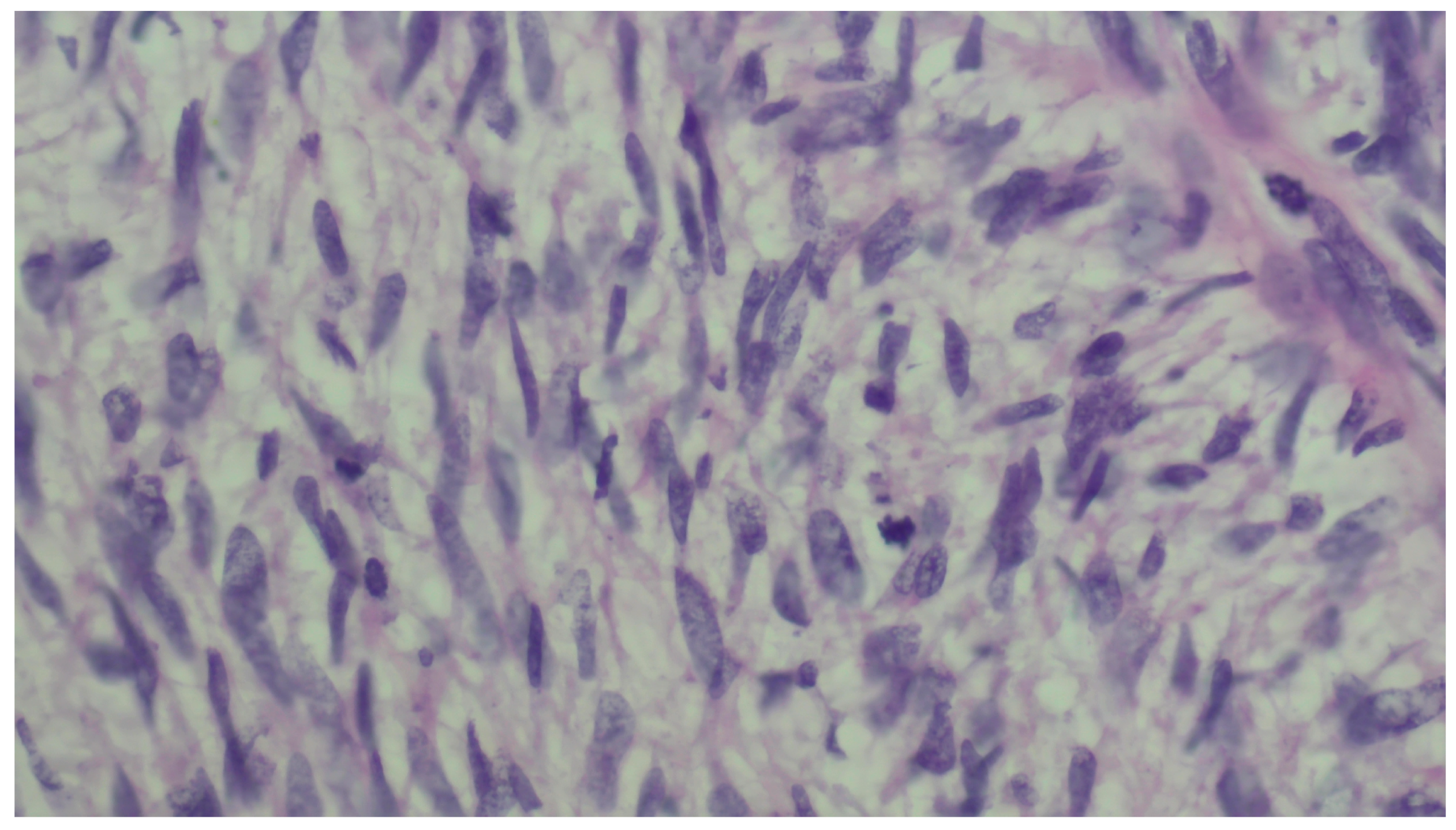
2. Case Presentation
3. Discussion
3.1. STUMP as a Diagnostic Gray Zone
3.2. Differential Diagnosis in Relation to Leiomyosarcoma
| Characteristic | Leiomyoma (LM) | STUMP | Leiomyosarcoma (LMS) |
|---|---|---|---|
| Cytological atypia | Minimal or absent | Moderate or focal, without diffuse severe atypia | Diffuse moderate–severe atypia |
| Mitotic index | <5/10 HPF | Most commonly <10/10 HPF (intermediate or discordant) | ≥10/10 HPF |
| Coagulative tumor necrosis | Absent | Focal or equivocal, without full LMS triad | Typically present |
| Growth pattern | Expansile, well-circumscribed | Most often well-circumscribed | Frequently infiltrative |
| Combination of criteria | Benign morphology | Discordant or borderline findings | ≥2 major LMS criteria |
| IHC: p16 | Negative/focal | Variable, patchy | Diffusely positive |
| IHC: p53 | Wild-type | Most commonly wild-type | Aberrant/mutant |
| Ki-67 | Low | Intermediate | High |
| Biological behavior | Benign | Unpredictable | Malignant |
| Risk of recurrence | Minimal | Low but real | High |
| Metastases | No | Rare, reported | Frequent |
| Definitive diagnosis | Histologically clear | Often postoperative | Histologically clear |
3.3. Postmenopausal STUMP—Clinical Importance and Specific Considerations
3.4. Diagnostic Pitfalls in Postmenopausal Uterine Masses
3.5. Treatment—Surgical Approach, Extent of Surgery, and the Role of Adjuvant Therapy
3.6. Recurrence and Metastatic Potential
3.7. Immunohistochemical and Molecular Markers
3.8. Clinical Implications and the Role of the Presented Case
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| LM | Leiomyoma |
| LMS | Leiomyosarcoma |
| STUMP | Smooth muscle tumor of uncertain malignant potential |
| HPF | High-power field |
| IHC | Immunohistochemistry |
| ER | Estrogen receptor |
| PR | Progesterone receptor |
| KI-67 | Proliferation index |
| MSCT | Multislice computed tomography |
| H&E | Hematoxylin and eosin |
References
- WHO Classification of Tumours Editorial Board. Female Genital Tumours, 5th ed.; International Agency for Research on Cancer: Lyon, France, 2020. [Google Scholar]
- Ettore, C.; Incognito, G.G.; Gulino, F.A.; Russo, E.; Cannone, F.; Ettore, G. Uterine Smooth Muscle Tumor of Uncertain Malignant Potential: A Retrospective, Monocentric Cohort Study. Surgeries 2023, 4, 412–419. [Google Scholar] [CrossRef]
- Rizzo, A.; Ricci, A.D.; Saponara, M.; De Leo, A.; Perrone, A.M.; De Iaco, P.; Pantaleo, M.A.; Nannini, M. Recurrent uterine smooth-muscle tumors of uncertain malignant potential (STUMP): State of the art. Anticancer Res. 2020, 40, 1229–1238. [Google Scholar] [CrossRef] [PubMed]
- Borella, F.; Cosma, S.; Ferraioli, D.; Ray-Coquard, I.; Chopin, N.; Meeus, P.; Cockenpot, V.; Valabrega, G.; Scotto, G.; Turinetto, M.; et al. Clinical and Histopathological Predictors of Recurrence in Uterine Smooth Muscle Tumor of Uncertain Malignant Potential (STUMP): A Multicenter Retrospective Cohort Study of Tertiary Centers. Ann. Surg. Oncol. 2022, 29, 8302–8314. [Google Scholar] [CrossRef]
- Liu, L.; Xiao, Z.; Li, Z.; Zheng, J.; Xu, X.; Zhou, H. Uterine smooth muscle tumors of uncertain malignant potential: A 13-year retrospective study. Front. Oncol. 2024, 14, 1458968. [Google Scholar] [CrossRef]
- Dall’Asta, A.; Gizzo, S.; Musarò, A.; Quaranta, M.; Noventa, M.; Migliavacca, C.; Sozzi, G.; Monica, M.; Mautone, D.; Berretta, R. Uterine smooth muscle tumors of uncertain malignant potential (STUMP): Pathology, follow-up and recurrence. Int. J. Clin. Exp. Pathol. 2014, 7, 8136–8142. [Google Scholar]
- Bell, S.W.; Kempson, R.L.; Hendrickson, M.R. Problematic uterine smooth muscle neoplasms. A clinicopathologic study of 213 cases. Am. J. Surg. Pathol. 1994, 18, 535–558. [Google Scholar] [CrossRef] [PubMed]
- Di Giuseppe, J.; Grelloni, C.; Giuliani, L.; Delli Carpini, G.; Giannella, L.; Ciavattini, A. Recurrence of uterine smooth muscle tumor of uncertain malignant potential: A systematic review of the literature. Cancers 2022, 14, 2323. [Google Scholar] [CrossRef]
- Liu, H.-T.; Wong, C.-N.; Wong, C.-N.; Liu, F.-S. Uterine smooth muscle tumor of uncertain malignant potential: A review of current knowledge. Taiwan. J. Obstet. Gynecol. 2022, 61, 935–940. [Google Scholar] [CrossRef]
- Jovanovic, L.; Milenkovic, S.; Janjic, T.; Andric, L.; Sosic-Jurjevic, B.; Dokic, M. Myxoid pattern in smooth muscle tumor of uncertain malignant potential (STUMP) is a significant prognostic parameter with difficult diagnostic dilemmas. Eur. J. Gynaecol. Oncol. 2025, 46, 66–71. [Google Scholar] [CrossRef]
- Guntupalli, S.R.; Ramirez, P.T.; Anderson, M.L.; Milam, M.R.; Bodurka, D.C.; Malpica, A. Uterine smooth muscle tumor of uncertain malignant potential: A retrospective analysis. Gynecol. Oncol. 2009, 113, 324–326. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Aminzadeh, P.; Alibrahim, E.; Dobrotwir, A.; Paul, E.; Goergen, S. Multiparametric MR evaluation of uterine leiomyosarcoma and STUMP versus leiomyoma in symptomatic women planned for high frequency focussed ultrasound: Accuracy of imaging parameters and interobserver agreement for identification of malignancy. Br. J. Radiol. 2021, 94, 20200483. [Google Scholar] [CrossRef]
- Jena, S.; Sinha, N.K.; Mishra, M.; Singh, V.; Upadhyay, A.K. Uterine Smooth Muscle Tumor of Undetermined Malignant Potential (STUMP): A Diagnostic Challenge. Cureus 2024, 16, e73115. [Google Scholar] [CrossRef]
- Bucuri, C.E.; Ciortea, R.; Malutan, A.M.; Oprea, V.; Toma, M.; Roman, M.P.; Ormindean, C.M.; Nati, I.; Suciu, V.; Mihu, D. Smooth Muscle Tumor of Uncertain Malignant Potential (STUMP): A Systematic Review of the Literature in the Last 20 Years. Curr. Oncol. 2024, 31, 5242–5254. [Google Scholar] [CrossRef]
- Huo, L.; Wang, D.; Wang, W.; Cao, D.; Yang, J.; Wu, M.; Yang, J.; Xiang, Y. Oncologic and reproductive outcomes of uterine smooth muscle tumor of uncertain malignant potential: A single-center retrospective study of 67 cases. Front. Oncol. 2020, 10, 647. [Google Scholar] [CrossRef] [PubMed]
- Richtarova, A.; Boudova, B.; Dundr, P.; Lisa, Z.; Hlinecka, K.; Zizka, Z.; Fruhauf, F.; Kuzel, D.; Slama, J.; Mara, M. Uterine smooth muscle tumors with uncertain malignant potential: Analysis following fertility-saving procedures. Int. J. Gynecol. Cancer 2023, 33, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Bosoteanu, M.; Deacu, M.; Voda, R.I.; Orasanu, C.I.; Aschie, M.; Vlad, S.E.; Penciu, R.C.; Chirila, S.I. Five-Year Retrospective Study of Uterine STUMP and Leiomyosarcoma. Clin. Pract. 2022, 12, 897–907. [Google Scholar] [CrossRef]
- Cotrino, I.; Carosso, A.; Macchi, C.; Baima Poma, C.; Cosma, S.; Ribotta, M.; Viora, E.; Sciarrone, A.; Borella, F.; Zola, P. Ultrasound and clinical characteristics of uterine smooth muscle tumors of uncertain malignant potential (STUMPs). Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 251, 167–172. [Google Scholar] [CrossRef]
- Tinelli, A.; D’Oria, O.; Civino, E.; Morciano, A.; Hashmi, A.A.; Baldini, G.M.; Stefanovic, R.; Malvasi, A.; Pecorella, G. Smooth Muscle Tumor of Uncertain Malignant Potential (STUMP): A Comprehensive Multidisciplinary Update. Medicina 2023, 59, 1371. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mäkinen, N.; Kämpjärvi, K.; Frizzell, N.; Bützow, R.; Vahteristo, P. Characterization of MED12, HMGA2, and FH alterations reveals molecular variability in uterine smooth muscle tumors. Mol. Cancer 2017, 16, 101. [Google Scholar] [CrossRef]
- Cherkaoui, A.; Atfi, F.; Gotni, A.; Boufetal, H.; Mahdaoui, S.; Samouh, N. Uterine Smooth Muscle Tumor of Uncertain Malignant Potential (STUMP): About a Case. Int. J. Surg. Case Rep. 2024, 117, 109556. [Google Scholar] [CrossRef]
- Ciccarone, F.; Biscione, A.; Robba, E.; Pasciuto, T.; Giannarelli, D.; Gui, B.; Manfredi, R.; Ferrandina, G.; Romualdi, D.; Moro, F.; et al. A Clinical Ultrasound Algorithm to Identify Uterine Sarcoma and Smooth Muscle Tumors of Uncertain Malignant Potential in Patients with Myometrial Lesions: The MYometrial Lesion UltrasouNd And mRi Study. Am. J. Obstet. Gynecol. 2025, 232, 108.e1–108.e22. [Google Scholar] [CrossRef]
- Şahin, H.; Karataş, F.; Çoban, G.; Özen, Ö.; Erdem, Ö.; Onan, M.A.; Ayhan, A. Uterine Smooth Muscle Tumor of Uncertain Malignant Potential: Fertility and Clinical Outcomes. J. Gynecol. Oncol. 2019, 30, e54. [Google Scholar] [CrossRef]
- Leone Roberti Maggiore, U.; Fanfani, F.; Capasso, I.; Perrone, E.; Parisi, G.; Zannoni, G.F.; Falcone, F.; Di Giovanni, A.; Malzoni, M.; Perrone, A.M.; et al. Fertility-Sparing vs. Hysterectomy for Uterine STUMP: A Pragmatic Clinical Study. Eur. J. Cancer 2026, 236, 116260. [Google Scholar] [CrossRef]
- Giannini, A.; Golia D’Augè, T.; Bogani, G.; Laganà, A.S.; Chiantera, V.; Vizza, E.; Muzii, L.; Di Donato, V. Uterine Sarcomas: A Critical Review of the Literature. Eur. J. Obstet. Gynecol. Reprod. Biol. 2023, 287, 166–170. [Google Scholar] [CrossRef]
- Garg, M.; Rajanbabu, A.; Nair, I.R. Smooth Muscle Tumors of Uncertain Malignant Potential or Atypical Leiomyomas: A Long-Term Evaluation of Surgical Outcomes and Clinicopathological Features. Eur. J. Obstet. Gynecol. Reprod. Biol. 2024, 301, 201–205. [Google Scholar] [CrossRef]
- Sanada, S.; Ushijima, K.; Yanai, H.; Mikami, Y.; Ohishi, Y.; Kobayashi, H.; Tashiro, H.; Mikami, M.; Miyamoto, S.; Katabuchi, H. A Critical Review of “Uterine Leiomyoma” with Subsequent Recurrence or Metastasis: A Multicenter Study of 62 Cases. J. Obstet. Gynaecol. Res. 2022, 48, 3242–3251. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Laury, A.L.; Nucci, M.R.; Quade, B.J. Predictors of Adverse Outcome in Uterine Smooth Muscle Tumours of Uncertain Malignant Potential (STUMP): A Clinicopathological Analysis of 22 Cases with a Proposal for the Inclusion of Additional Histological Parameters. Histopathology 2018, 73, 284–298. [Google Scholar] [CrossRef] [PubMed]
- Lapresa-Alcalde, M.V.; Ruiz-Navarro, M.J.; Sancho de Salas, M.; Cubo, A.M. A Review and Follow-Up of Uterine Smooth Muscle Tumours of Uncertain Malignant Potential (STUMP): A Case Series and Literature Review. Diseases 2023, 11, 99. [Google Scholar] [CrossRef] [PubMed]
- Momeni-Boroujeni, A.; Yousefi, E.; Balakrishnan, R.; Riviere, S.; Kertowidjojo, E.; Hensley, M.L.; Ladanyi, M.; Ellenson, L.H.; Chiang, S. Molecular-Based Immunohistochemical Algorithm for Uterine Leiomyosarcoma Diagnosis. Mod. Pathol. 2023, 36, 100084. [Google Scholar] [CrossRef]
- Travaglino, A.; Raffone, A.; Gencarelli, A.; Neola, D.; Oliviero, D.A.; Alfano, R.; Campanino, M.R.; Cariati, F.; Zullo, F.; Mollo, A.; et al. p53, p16 and Ki67 as Immunohistochemical Prognostic Markers in Uterine Smooth Muscle Tumors of Uncertain Malignant Potential (STUMP). Pathol. Res. Pract. 2021, 226, 153592. [Google Scholar] [CrossRef]




| Parameter | Result | Reference Range | Unit |
|---|---|---|---|
| Leukocytes | 8.6 | 4.0–10.0 | ×109/L |
| Neutrophils | 63.9 | 50.0–70.0 | % |
| Lymphocytes | 25.5 | 25.0–40.0 | % |
| Monocytes | 7.4 | 2.0–6.0 | % |
| Eosinophils | 2.6 | 2.0–4.0 | % |
| Basophils | 0.6 | <1.0 | % |
| Erythrocytes | 4.42 | 3.80–5.80 | ×1012/L |
| Hemoglobin | 142 | 115–160 | g/L |
| Hematocrit | 0.413 | 0.370–0.470 | L/L |
| MCV | 93.5 | 80–100 | fL |
| MCH | 32.2 | 27–32 | Pg |
| MCHC | 344 | 320–360 | g/L |
| Platelets | 276 | 150–500 | ×109/L |
| CRP | 3.12 | <5.0 | mg/L |
| AST | 17 | <31 | U/L |
| ALT | 16 | <34 | U/L |
| LDH | 190 | <247 | U/L |
| Total bilirubin | 7.4 | 2–21 | µmol/L |
| Creatinine | 53 | 58–96 | µmol/L |
| Urea | 4.5 | 2.8–7.2 | mmol/L |
| Total proteins | 59.6 | 66–83 | g/L |
| Albumin | 38.3 | 34–53 | g/L |
| Time from Surgery | Clinical Examination | Pelvic Imaging (US/MRI) | Thoracic Imaging | Additional Considerations |
|---|---|---|---|---|
| 0–24 months | Every 6 months | Every 6 months | Annually | Higher vigilance in cases with necrosis, higher mitotic index, or fertility-sparing surgery |
| 25–60 months | Annually | Annually | Annually | Consider MRI if ultrasound findings are equivocal |
| >5 years | Individualized | Individualized | If clinically indicated | Long-term follow-up advised due to reported late recurrences |
| At any time if symptoms occur | Immediate evaluation | Targeted imaging | Targeted imaging | Assess for local recurrence or distant metastasis (lungs most common site) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Rakic, N.; Ivanovic, S.; Ivanovic, M.; Tulic, L.; Milincic, M.; Dosev, T.; Jovic, N.; Arsenijevic, N.; Joksimovic Jovic, J. Postmenopausal Enlargement of a Presumed Leiomyoma Revealing STUMP: A Diagnostic Pitfall with Important Clinical Implications—A Case Report. Diagnostics 2026, 16, 1075. https://doi.org/10.3390/diagnostics16071075
Rakic N, Ivanovic S, Ivanovic M, Tulic L, Milincic M, Dosev T, Jovic N, Arsenijevic N, Joksimovic Jovic J. Postmenopausal Enlargement of a Presumed Leiomyoma Revealing STUMP: A Diagnostic Pitfall with Important Clinical Implications—A Case Report. Diagnostics. 2026; 16(7):1075. https://doi.org/10.3390/diagnostics16071075
Chicago/Turabian StyleRakic, Nenad, Stefan Ivanovic, Milica Ivanovic, Lidija Tulic, Milos Milincic, Tatjana Dosev, Nikola Jovic, Neda Arsenijevic, and Jovana Joksimovic Jovic. 2026. "Postmenopausal Enlargement of a Presumed Leiomyoma Revealing STUMP: A Diagnostic Pitfall with Important Clinical Implications—A Case Report" Diagnostics 16, no. 7: 1075. https://doi.org/10.3390/diagnostics16071075
APA StyleRakic, N., Ivanovic, S., Ivanovic, M., Tulic, L., Milincic, M., Dosev, T., Jovic, N., Arsenijevic, N., & Joksimovic Jovic, J. (2026). Postmenopausal Enlargement of a Presumed Leiomyoma Revealing STUMP: A Diagnostic Pitfall with Important Clinical Implications—A Case Report. Diagnostics, 16(7), 1075. https://doi.org/10.3390/diagnostics16071075









