Current Status and Future Perspectives of Wearable Technologies for Oral Health in Clinical Applications
Abstract
1. Introduction
2. Historical Development of Oral Wearable Devices
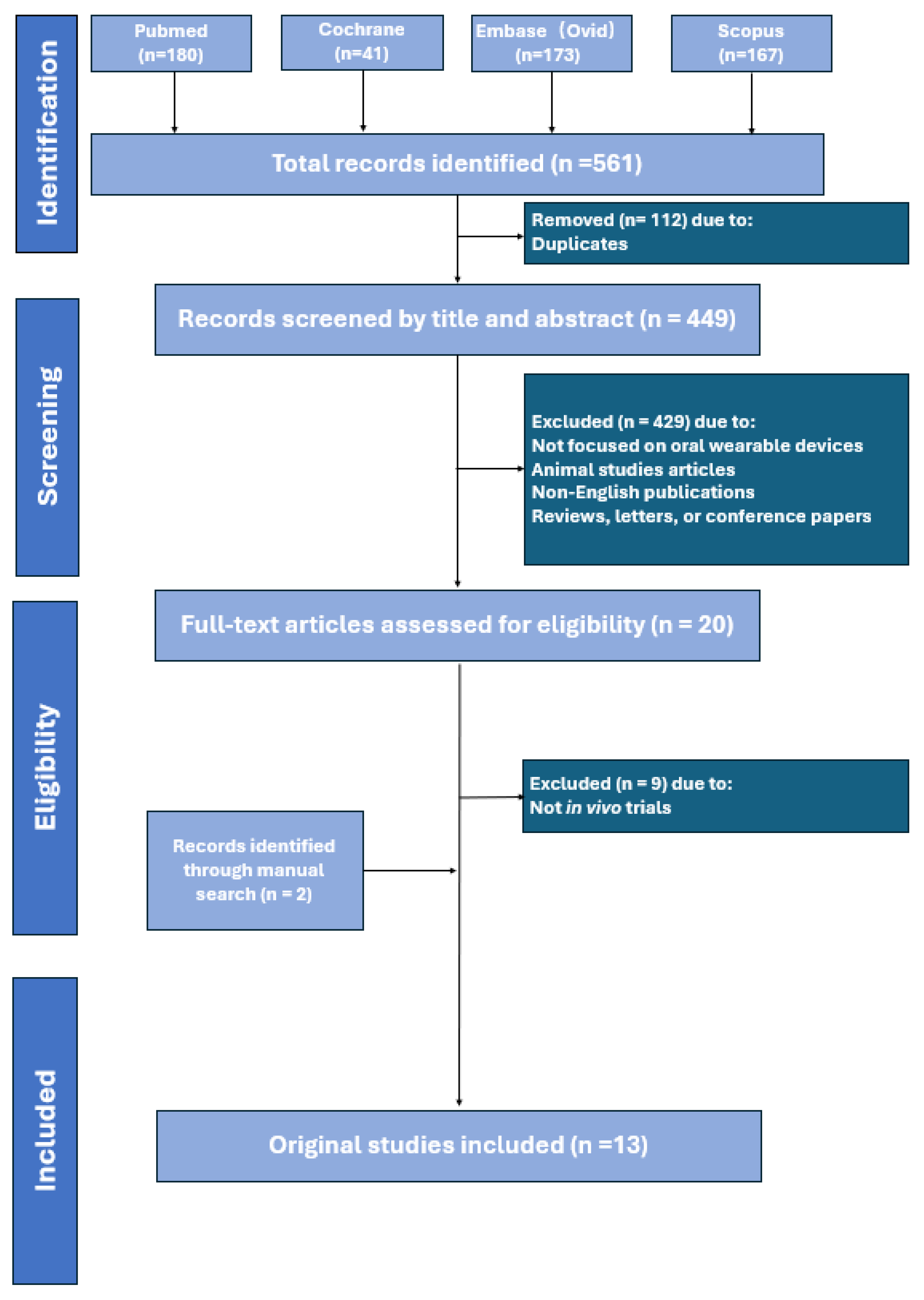
3. Methodology
4. Results
4.1. Diagnostic Applications
4.2. Therapeutic Applications
4.3. Monitoring Applications
4.4. Preventive Applications
5. Discussion
5.1. Clinical Significance of Oral Wearable Devices
5.2. Limitations and Challenges in the Development of Oral Wearable Devices
5.3. Future Perspectives on the Clinical Application of Oral Wearable Devices
5.4. Limitations of This Review
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AI | Artificial Intelligence |
| BAMS | Bruxist Activity Monitor System |
| DTA | Diagnostic Test Accuracy Study |
| H2S | Hydrogen Sulfide |
| MMP-8 | Matrix Metalloproteinase-8 |
| OCEBM | Oxford Centre for Evidence-Based Medicine |
| PEMF | Pulsed Electromagnetic Field |
| OSA | Sleep Apnea |
| RCT | Randomized Controlled Trial |
| SaO2 | Arterial Oxygen Saturation |
| VSCs | Volatile Sulfur Compounds |
| VR | Virtual Reality |
References
- Liu, Y.; Chen, L.; Hong, R.; Dong, X.; Sha, W.; Jin, Z.; Wu, T.; Chen, G. A survey of mastication evaluation: From traditional approach to artificial intelligence. Food Nutr. Health 2025, 2, 18. [Google Scholar] [CrossRef]
- González-Cañete, F.J.; Casilari, E. UMATBrush: A dataset of inertial signals of toothbrushing activities. Data Brief 2025, 62, 111980. [Google Scholar] [CrossRef]
- Jeon, S.; Kim, S.H.; Heo, G.; Heo, H.J.; Chae, S.Y.; Kwon, Y.W.; Lee, S.K.; Han, D.W.; Kim, H.J.; Kim, Y.H.; et al. A Wearable Electrochemical Biosensor for Salivary Detection of Periodontal Inflammation Biomarkers: Molecularly Imprinted Polymer Sensor with Deep Learning Integration. Adv. Sci. 2025, 12, e09658. [Google Scholar] [CrossRef]
- Pan, J.; Li, X.; Sun, R.; Xu, Y.; Shi, Z.; Dai, C.; Wen, H.; Han, R.P.S.; Ye, Q.; Zhang, F.; et al. Hydrogel-based radio frequency H2S sensor for in situ periodontitis monitoring and antibacterial treatment. Biosens. Bioelectron. 2024, 259, 116404. [Google Scholar] [CrossRef]
- Ozawa, A.; Suzuki, Y.; Okura, K.; Tajima, T.; Taniwaki, T.; Shibagaki, A.; Moreno, F.J.J.; Okawa, T.; Abe, S.; Shigemoto, S.; et al. Evaluation of sleep bruxism by three-dimensional analysis of the deformation of acrylic resin occlusal appliances. J. Prosthodont. Res. 2025, 70, 141–150. [Google Scholar] [CrossRef]
- Liu, Z.; Sun, T.; Chen, Y.; Mo, X.; Kao, H.; Chen, H.J. Integrated Multiplex Sensing Clear Aligner for In Situ Monitoring of Dental Enamel Demineralization. ACS Biomater. Sci. Eng. 2023, 9, 3680–3689. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Xiao, Y.; Xiong, P.; Zheng, P.; Wu, S.; Wang, X.; Yin, Y.; Fang, H.; Wang, C.; Lu, Y.; et al. Boosting Mechanoluminescence Performance in Doped CaZnOS by the Facile Self-Reduction Approach. Adv. Mater. 2025, 38, e11643. [Google Scholar] [CrossRef]
- Stocchero, M.; Gobbato, L.; De Biagi, M.; Bressan, E.; Sivolella, S. Pulsed electromagnetic fields for postoperative pain: A randomized controlled clinical trial in patients undergoing mandibular third molar extraction. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2015, 119, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, M.; Jain, S.; Ranjan, M.; Hassan, S.; Prakash, N.; Kumar, D.; Kumar, A.; Singh, S. Artificial intelligence tools in dentistry: A systematic review on their application and outcomes. Cureus 2025, 17, e85062. [Google Scholar] [CrossRef]
- Thompson, F.C.; Brudevold, F. A micro-antimony electrode designed for intraoral pH measurements in man and small experimental animals. J. Dent. Res. 1954, 33, 849–853. [Google Scholar] [PubMed]
- Graf, H.; Mühlemann, H.R. Oral telemetry of fluoride ion activity. Arch. Oral Biol. 1969, 14, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Badea, M.; Tiwari, S.; Marty, J.L. Wearable Biosensors: An Alternative and Practical Approach in Healthcare and Disease Monitoring. Molecules 2021, 26, 748. [Google Scholar] [CrossRef]
- Smith, A.A.; Li, R.; Tse, Z.T.H. Reshaping healthcare with wearable biosensors. Sci. Rep. 2023, 13, 4998. [Google Scholar] [CrossRef]
- Kurosawa, M.; Taniguchi, K.; Momose, H.; Sakaguchi, M.; Kamijo, M.; Nishikawa, A. Simultaneous Measurement of Ear Canal Movement, Electromyography of the Masseter Muscle and Occlusal Force for Earphone-Type Occlusal Force Estimation Device Development. Sensors 2019, 19, 3441. [Google Scholar] [CrossRef] [PubMed]
- Mannoor, M.S.; Tao, H.; Clayton, J.D.; Sengupta, A.; Kaplan, D.L.; Naik, R.R.; Verma, N.; Omenetto, F.G.; McAlpine, M.C. Graphene-based wireless bacteria detection on tooth enamel. Nat. Commun. 2012, 3, 763. [Google Scholar] [CrossRef]
- Trusculescu, L.M.; Pitic, D.E.; Sălcudean, A.; Popovici, R.A.; Forna, N.; Badoiu, S.C.; Enache, A.; Enasoni, S.; Kiș, A.; Cosoroabă, R.M.; et al. Virtual Reality as a Non-Pharmacological Aid for Reducing Anxiety in Pediatric Dental Procedures. Children 2025, 12, 930. [Google Scholar] [CrossRef]
- Jabbaripour, S.; Saien, S.; Heidari, R.; Erfanian, R.; Amali, A. Performance of Wrist-Worn Pulse Oximeter for the Screening of Obstructive Sleep Apnea. Iran. J. Otorhinolaryngol. 2024, 36, 603–612. [Google Scholar] [CrossRef]
- Wang, C.; Kumar, T.S.; De Raedt, W.; Camps, G.; Hallez, H.; Vanrumste, B. Eating Speed Measurement Using Wrist-Worn IMU Sensors Towards Free-Living Environments. IEEE J. Biomed. Health Inform. 2024, 28, 5816–5828. [Google Scholar] [CrossRef]
- Snow, C.; Pun, M.; Magnison-Benoit, S.; Tripp, T.R.; Clarke, A.; Berghmans, S.; Hansen, B.; Transfiguracion, L.; Di Simone, G.; Lai, M.L.; et al. A Buccal Mucosal Reflectance Oximeter Accurately Measures Arterial Oxyhemoglobin Saturation and Pulse Rate. Am. J. Respir. Crit. Care Med. 2025, 211, A4758. [Google Scholar] [CrossRef]
- Flores-Ramirez, B.; Oreggioni, J.; Angeles-Medina, F.; Kreiner, M.; Pacheco-Guerrero, N.; Morales-Gonzalez, J.; Fernandez, I.; Suaste-Gomez, E. Bruxist Activity Monitor System (BAMS): An instrumental approach tool in the assessment of Bruxism. In 2021 43rd Annual International Conference of the IEEE Engineering in Medicine and Biology Society; IEEE: New York, NY, USA, 2021; pp. 1399–1402. [Google Scholar] [CrossRef]
- Feng, H.; Song, W.; Li, R.; Yang, L.; Chen, X.; Guo, J.; Liao, X.; Ni, L.; Zhu, Z.; Chen, J.; et al. A Fully Integrated Orthodontic Aligner with Force Sensing Ability for Machine Learning-Assisted Diagnosis. Adv. Sci. 2025, 12, e2411187. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, J.; Zeng, Y.J.; Xu, T. Janus Wettable Mask with Integrated Ratiometric Fluorescent Probe for Comfortable Exhaled H2S Gas Sensing. ACS Sens. 2024, 9, 6638–6645. [Google Scholar] [CrossRef]
- Li, X.; Luo, C.; Fu, Q.; Zhou, C.; Ruelas, M.; Wang, Y.; He, J.; Wang, Y.; Zhang, Y.S.; Zhou, J. A Transparent, Wearable Fluorescent Mouthguard for High-Sensitive Visualization and Accurate Localization of Hidden Dental Lesion Sites. Adv. Mater. 2020, 32, e2000060. [Google Scholar] [CrossRef]
- Ma, D.; Chen, B.; Li, Y.; Pang, X.; Fu, Q.; Xiao, Z.; Shi, Z.; Li, X.; Luo, C.; Zhou, Z.K.; et al. Au@Ag Nanorods-PDMS Wearable Mouthguard as a Visualized Detection Platform for Screening Dental Caries and Periodontal Diseases. Adv. Healthc. Mater. 2022, 11, e2102682. [Google Scholar] [CrossRef]
- Matsumoto, H.; Iitani, K.; Mitsubayashi, K.; Moriyama, K. Real-Time Continuous Tongue Pressure Measurement with Mouthguard-Type Pressure-Sensing Device. Orthod. Craniofac. Res. 2025, 28, S215–S220. [Google Scholar] [CrossRef]
- Sedghi, L.; DiMassa, V.; Harrington, A.; Lynch, S.V.; Kapila, Y.L. The oral microbiome: Role of key organisms and complex networks in oral health and disease. Periodontol. 2000 2021, 87, 107–131. [Google Scholar] [CrossRef] [PubMed]
- Faran Ali, S.M.; Tanwir, F. Oral microbial habitat a dynamic entity. J. Oral Biol. Craniofac. Res. 2012, 2, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Raphael, K.G.; Janal, M.N.; Sirois, D.A.; Dubrovsky, B.; Klausner, J.J.; Krieger, A.C.; Lavigne, G.J. Validity of self-reported sleep bruxism among myofascial temporomandibular disorder patients and controls. J. Oral Rehabil. 2015, 42, 751–758. [Google Scholar] [CrossRef]
- Lobbezoo, F.; Ahlberg, J.; Raphael, K.G.; Wetselaar, P.; Glaros, A.G.; Kato, T.; Santiago, V.; Winocur, E.; De Laat, A.; De Leeuw, R.; et al. International consensus on the assessment of bruxism: Report of a work in progress. J. Oral Rehabil. 2018, 45, 837–844. [Google Scholar] [CrossRef]
- Tabi-Amankwa, M.; Bawua, S.A.; Amankwah, I.; Boamah, M.O.; Otoo, D.D.; Owusu, R. Economic cost of oral conditions to patients attending the dental department of the Korle Bu Teaching Hospital, Ghana. Health Sci. Investig. J. 2025, 7, 1123–1131. [Google Scholar]
- Dai, C.; Shi, Z.; Xu, Y.; Su, L.; Li, X.; Deng, P.; Wen, H.; Wang, J.; Ye, Q.; Han, R.P.S.; et al. Wearable Multifunctional Hydrogel for Oral Microenvironment Visualized Sensing Coupled with Sonodynamic Bacterial Elimination and Tooth Whitening. Adv. Healthc. Mater. 2025, 14, e2401269. [Google Scholar] [CrossRef]
- Cheah, L.A.; Bai, J.; Gonzalez, J.A.; Gilbert, J.M.; Ell, S.R.; Green, P.D.; Moore, R.K. Preliminary evaluation of a silent speech interface based on intra-oral magnetic sensing. In Proceedings of the International Conference on Biomedical Electronics and Devices; SciTePress: Setúbal, Portugal, 2016; pp. 108–116. [Google Scholar]
- Yip, M. Ultra-Low-Power Circuits and Systems for Wearable and Implantable Medical Devices. Ph.D. Thesis, Massachusetts Institute of Technology, Cambridge, MA, USA, 2013. [Google Scholar]
- Kim, J.; Imani, S.; de Araujo, W.R.; Warchall, J.; Valdés-Ramírez, G.; Paixão, T.R.; Mercier, P.P.; Wang, J. Wearable salivary uric acid mouthguard biosensor with integrated wireless electronics. Biosens. Bioelectron. 2015, 74, 1061–1068. [Google Scholar] [CrossRef] [PubMed]
- Al-Hamad, K.A.; Asiri, A.; Al-Qahtani, A.M.; Alotaibi, S.; Almalki, A. Development and in-vitro validation of an intraoral wearable biofeedback system for bruxism management. Front. Bioeng. Biotechnol. 2025, 13, 1572970. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Ghovanloo, M. Wireless Communication of Intraoral Devices and Its Optimal Frequency Selection. IEEE Trans. Microw. Theory Tech. 2014, 62, 3205–3215. [Google Scholar] [CrossRef]
- Vallejo, M.; Recas, J.; Del Valle, P.G.; Ayala, J.L. Accurate Human Tissue Characterization for Energy-Efficient Wireless On-Body Communications. Sensors 2013, 13, 7546–7569. [Google Scholar]
- Wang, J.; Gao, X. Opportunities and challenges in wearable ultrasound for oral health. Transl. Dent. Res. 2025, 1, 100052. [Google Scholar] [CrossRef]
- Bodine, K.; Gemperle, F. Effects of functionality on perceived comfort of wearables. In Proceedings of the Seventh IEEE International Symposium on Wearable Computers, White Plains, NY, USA, 21–23 October 2003; pp. 57–60. [Google Scholar]
- McGrath, C.; Chau, C.W.R.; Molina, G.F. Monitoring oral health remotely: Ethical considerations when using AI among vulnerable populations. Front. Oral Health 2025, 6, 1587630. [Google Scholar] [CrossRef]
- Kaplan, B. Seeing through health information technology: The need for transparency in software, algorithms, data privacy, and regulation. J. Law Biosci. 2020, 7, lsaa062. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Miao, D.; Li, R.; Liu, Y.; Li, X. Cache-Based Privacy Protection Scheme for Continuous Location Query. Entropy 2023, 25, 201. [Google Scholar] [CrossRef]
- Li, T.; Sahu, A.K.; Talwalkar, A.; Smith, V. Federated Learning: Challenges, Methods, and Future Directions. IEEE Signal Process. Mag. 2020, 37, 50–60. [Google Scholar] [CrossRef]
- Nazer, L.H.; Zatarah, R.; Waldrip, S.; Ke, J.X.C.; Moukheiber, M.; Khanna, A.K.; Hicklen, R.S.; Moukheiber, L.; Moukheiber, D.; Ma, H.; et al. Bias in artificial intelligence algorithms and recommendations for mitigation. PLOS Digit. Health 2023, 2, e0000278. [Google Scholar] [CrossRef]
- Gupte, T.; Nitave, T.; Gobburu, J. Regulatory landscape of accelerated approval pathways for medical devices in the United States and the European Union. Front. Med. Technol. 2025, 7, 1586070. [Google Scholar] [CrossRef]
- CenterWatch. FDA Guidance on AI-Enabled Devices: Transparency, Bias, Lifecycle Oversight. Available online: https://www.centerwatch.com/insights/fda-guidance-on-ai-enabled-devices-transparency-bias-lifecycle-oversight (accessed on 12 March 2026).
- U.S. Food and Drug Administration (FDA). 21 CFR Part 872 Subpart F—Dental Devices: Auto Titration Device for Oral Appliances (§ 872.5571). Available online: https://www.ecfr.gov/current/title-21/chapter-I/subchapter-H/part-872/subpart-F (accessed on 15 March 2026).
- Zhang, M.; Chu, R.; Liu, C.; Zhang, S.; Ren, X. Navigating the AI tide: Challenges, opportunities, and future directions for early-career dermatologists. Front. Med. 2025, 12, 1684035. [Google Scholar] [CrossRef]
- Li, G. Looking forward to a better 2023 and beyond. J. Orthop. Transl. 2022, 37, A1–A2. [Google Scholar] [CrossRef]
- Serra, P.A. Biosensors for Health, Environment and Biosecurity; IntechOpen: Rijeka, Croatia, 2011. [Google Scholar] [CrossRef]
- An, B.W.; Shin, J.H.; Kim, S.-Y.; Kim, J.; Ji, S.; Park, J.; Lee, Y.; Jang, J.; Park, Y.-G.; Cho, E.; et al. Smart Sensor Systems for Wearable Electronic Devices. Polymers 2017, 9, 303. [Google Scholar] [CrossRef]
- Chou, J.; Du, N.; Ou, T.; Floriano, P.N.; Christodoulides, N.; McDevitt, J.T. Hot embossed polyethylene through-hole chips for bead-based microfluidicdevices. Biosens. Bioelectron. 2013, 42, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Sikka, N.; Gupta, S.; Arya, L.; Bala, S.; Arya, V.; Prajapat, J. Assessment of Knowledge, Attitude, Practice, and Barriers to the Use of Teledentistry by Dental Professionals of India: A Cross-sectional Survey. Int. J. Clin. Pediatr. Dent. 2025, 18, 1051–1057. [Google Scholar] [CrossRef]
- Andronic, A.; Maniu, G.; Birlutiu, V.; Popa, M. Utilization Patterns and Implementation Barriers in Adoption of Teledentistry Within Romanian Dental Practice. Healthcare 2025, 13, 3176. [Google Scholar] [CrossRef] [PubMed]
- Adeoye, J.; Su, Y.X. Artificial intelligence in salivary biomarker discovery and validation for oral diseases. Oral Dis. 2024, 30, 23–37. [Google Scholar]
- Fahim, M.; Sharma, V.; Duong, T.Q. A Wearable-based Preventive Model to Promote Oral Health Through Personalized Notification. In 2022 44th Annual International Conference of the IEEE Engineering in Medicine & Biology Society; IEEE: New York, NY, USA, 2022; pp. 4282–4285. [Google Scholar] [CrossRef]
- Wang, H.C.; Li, J.H.; Lin, Y.C.; Lin, C.Y.; Liu, C.P.; Lin, T.H.; Chan, C.T.; Hsieh, C.Y. Machine Learning-Based Toothbrushing Region Recognition Using Smart Toothbrush Holder and Wearable Sensors. Biosensors 2025, 15, 798. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.; Yeo, W.-H. Advances in Energy Harvesting Technologies for Wearable Devices. Micromachines 2024, 15, 884. [Google Scholar] [CrossRef]
- Bhide, A.; Ganguly, A.; Parupudi, T.; Ramasamy, M.; Muthukumar, S.; Prasad, S. Next-Generation Continuous Metabolite Sensing toward Emerging Sensor Needs. ACS Omega 2021, 6, 6031–6040. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Islam, S.; Kim, H.-E.; Korostoff, J.; Blatz, M.B.; Hwang, G.; Kim, A. Human Oral Motion-Powered Smart Dental Implant (SDI) for In Situ Ambulatory Photo-biomodulation Therapy. Adv. Healthc. Mater. 2020, 9, 2000658. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Lu, Y.; Shen, S.; Xu, Y.; Shu, C.; Wu, Y.; Lv, J.; Li, X.; Yan, Z.; An, Z.; et al. Wearable battery-free theranostic dental patch for wireless intraoral sensing and drug delivery. npj Flex. Electron. 2022, 6, 49. [Google Scholar] [CrossRef]
| Reference | Study Design | Tool | Score | Grade |
|---|---|---|---|---|
| Stocchero et al. [8] | RCT | RoB_2.0 | 5/5 | High |
| Trusculescu et al. [16] | RCT | RoB_2.0 | 4/5 | Moderate |
| Jabbaripour et al. [17] | DTA | JBI | 8/9 | High |
| Wang et al. [18] | Diagnostic Cohort Study | JBI | 8/9 | High |
| Snow et al. [19] | Diagnostic Cohort Study | JBI | 9/9 | High |
| Flores-Ramirez et al. [20] | Diagnostic Cohort Study | JBI | 6/10 | Moderate |
| González-Cañete et al. [2] | Case Series | JBI | 4/10 | Low |
| Kurosawa et al. [14] | Case Series | JBI | 6/10 | Moderate |
| Feng et al. [21] | Case Series | JBI | 7/10 | Moderate |
| Zhang et al. [22] | Case Series | JBI | 6/10 | Moderate |
| Li et al. [23] | Case Series | JBI | 5/10 | Low |
| Ma et al. [24] | Case Series | JBI | 5/10 | Low |
| Matsumoto et al. [25] | Case Series | JBI | 6/10 | Moderate |
| Clinical Function | Reference | Clinical Application | Application Site | Key Parameters | Level (OCEBM) |
|---|---|---|---|---|---|
| Diagnostic Application | Li et al. [23] | Caries Localization | Teeth | VSCs | 4 |
| Ma et al. [24] | Caries & Periodontal Disease | Teeth | H2S | 4 | |
| Zhang et al. [22] | Halitosis & Caries | Face (Integrated Mask) | H2S | 4 | |
| Snow et al. [19] | Sleep Apnea (OSA) Diagnosis | Buccal Mucosa | SaO2 | 2b | |
| Jabbaripour et al. [17] | Sleep Apnea (OSA) Diagnosis | Wrist (Wristband) | Pulse Oximetry and Heart Rate | 2b | |
| Feng et al. [21] | Malocclusion Etiology | Dentition (Clear Aligner) | Occlusal Force, Malocclusion Classification | 4 | |
| Flores-Ramirez et al. [20] | Bruxism | Mandibular Molars (Intraoral Appliance) | Occlusal Events, Duration, Intervals | 2b | |
| Therapeutic Applications | Stocchero et al. [8] | Post-surgical Rehabilitation | Postoperative Site (Wearable Patch) | Wound Healing, Pain Reduction | 1b |
| Trusculescu et al. [16] | Pediatric Dental Anxiety | Head (VR Headset) | Stress Reduction | 1b | |
| Monitoring Applications | Kurosawa et al. [14] | Occlusal Force Monitoring | External Ear Canal (Earphone-type) | Dynamic Changes in Ear Canal Shape | 4 |
| Matsumoto et al. [25] | Malocclusion Etiology | Lingual of Maxillary Incisors (Mouth Guard) | Tongue Pressure During Swallowing | 4 | |
| Wang et al. [18] | Dietary & Health Monitoring | Wrist (Wearable Motion Sensor) | Chewing Actions, Eating Speed, Duration | 2b | |
| Preventive Applications | González-Cañete et al. [2] | Oral Hygiene Management | Wrist (Smartwatch) | Brushing Compliance and Frequency | 4 |
| Reference | Study Design | Level (OCEBM) | Functional Category | Clinical Readiness | Subjects & Sample Size |
|---|---|---|---|---|---|
| Stocchero et al. [8] | RCT * | 1b | Therapeutics | Validated Device | 114 patients |
| Trusculescu et al. [16] | RCT | 1b | Therapeutics | Validated Device | 120 patients |
| Snow et al. [19] | Diagnostic Cohort Study | 2b | Diagnosis | Validated Device | 12 volunteers |
| Jabbaripour et al. [17] | DTA * | 2b | Diagnosis | Validated Device | 102 patients |
| Flores-Ramirez et al. [20] | Diagnostic Cohort Study | 2b | Diagnosis | Validated Device | 1 volunteer |
| Wang et al. [18] | Diagnostic Cohort Study | 2b | Monitoring | Validated Device | 61 volunteers |
| González-Cañete et al. [2] | Case Series | 4 | Prevention | Experimental Prototype | 10 volunteers |
| Kurosawa et al. [14] | Case Series | 4 | Monitoring | Experimental Prototype | 5 healthy subjects |
| Li et al. [23] | Case Series | 4 | Diagnosis | Experimental Prototype | 8 volunteers |
| Ma et al. [24] | Case Series | 4 | Diagnosis | Experimental Prototype | 9 volunteers |
| Zhang et al. [22]. | Case Series | 4 | Diagnosis | Experimental Prototype | 2 volunteers |
| Feng et al. [21] | Case Series | 4 | Diagnosis | Experimental Prototype | 5 volunteers |
| Matsumoto et al. [25] | Case Series | 4 | Monitoring | Experimental Prototype | 1 volunteer |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Li, Y.; Wang, M.; Qiu, S.; Chen, J.; Wang, F. Current Status and Future Perspectives of Wearable Technologies for Oral Health in Clinical Applications. Diagnostics 2026, 16, 1015. https://doi.org/10.3390/diagnostics16071015
Li Y, Wang M, Qiu S, Chen J, Wang F. Current Status and Future Perspectives of Wearable Technologies for Oral Health in Clinical Applications. Diagnostics. 2026; 16(7):1015. https://doi.org/10.3390/diagnostics16071015
Chicago/Turabian StyleLi, Yao, Mu Wang, Siqi Qiu, Jinyan Chen, and Feng Wang. 2026. "Current Status and Future Perspectives of Wearable Technologies for Oral Health in Clinical Applications" Diagnostics 16, no. 7: 1015. https://doi.org/10.3390/diagnostics16071015
APA StyleLi, Y., Wang, M., Qiu, S., Chen, J., & Wang, F. (2026). Current Status and Future Perspectives of Wearable Technologies for Oral Health in Clinical Applications. Diagnostics, 16(7), 1015. https://doi.org/10.3390/diagnostics16071015






