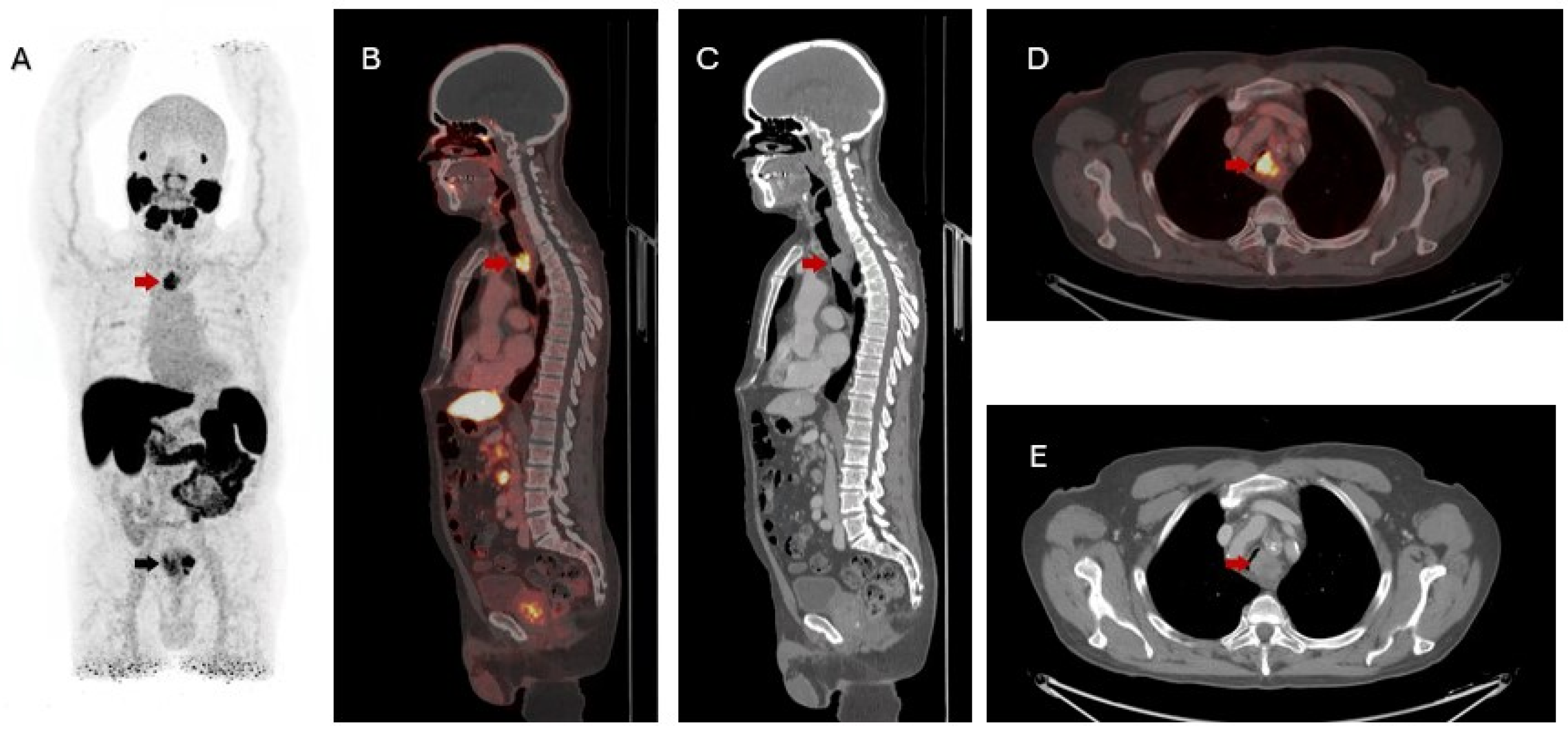
Pretherapeutic 18F-PSMA PET/CT Reveals Incidental Tracheal Epithelial–Myoepithelial Carcinoma
Abstract

Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kiess, A.P.; Banerjee, S.R.; Mease, R.C.; Rowe, S.P.; Rao, A.; Foss, C.A.; Chen, Y.; Yang, X.; Cho, S.Y.; Nimmagadda, S.; et al. Prostate-specific membrane antigen as a target for cancer imaging and therapy. Q. J. Nucl. Med. Mol. Imaging 2015, 59, 241–268. [Google Scholar] [PubMed]
- Wolf, P.; Freudenberg, N.; Bühler, P.; Alt, K.; Schultze-Seemann, W.; Wetterauer, U.; Elsässer-Beile, U. Three conformational antibodies specific for different PSMA epitopes are promising diagnostic and therapeutic tools for prostate cancer. Prostate 2010, 70, 562–569. [Google Scholar] [CrossRef] [PubMed]
- de Keizer, B.; Krijger, G.C.; Ververs, F.T.; van Es, R.J.J.; de Bree, R.; Willems, S. 68Ga-PSMA PET-CT Imaging of Metastatic Adenoid Cystic Carcinoma. Nucl. Med. Mol. Imaging 2017, 51, 360–361. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lutje, S.; Sauerwein, W.; Lauenstein, T.; Bockisch, A.; Poeppel, T.D. In Vivo Visualization of Prostate-Specific Membrane Antigen in Adenoid Cystic Carcinoma of the Salivary Gland. Clin. Nucl. Med. 2016, 41, 476–477. [Google Scholar] [CrossRef] [PubMed]
- Klein Nulent, T.J.W.; van Es, R.J.J.; Krijger, G.C.; de Bree, R.; Willems, S.M.; de Keizer, B. Prostate-specific membrane antigen PET imaging and immunohistochemistry in adenoid cystic carcinoma-a preliminary analysis. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1614–1621. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Gossili, F.; Fuentes-Martinez, N.; Høyer, C. Pretherapeutic 18F-PSMA PET/CT Reveals Incidental Tracheal Epithelial–Myoepithelial Carcinoma. Diagnostics 2026, 16, 883. https://doi.org/10.3390/diagnostics16060883
Gossili F, Fuentes-Martinez N, Høyer C. Pretherapeutic 18F-PSMA PET/CT Reveals Incidental Tracheal Epithelial–Myoepithelial Carcinoma. Diagnostics. 2026; 16(6):883. https://doi.org/10.3390/diagnostics16060883
Chicago/Turabian StyleGossili, Farid, Nelson Fuentes-Martinez, and Christian Høyer. 2026. "Pretherapeutic 18F-PSMA PET/CT Reveals Incidental Tracheal Epithelial–Myoepithelial Carcinoma" Diagnostics 16, no. 6: 883. https://doi.org/10.3390/diagnostics16060883
APA StyleGossili, F., Fuentes-Martinez, N., & Høyer, C. (2026). Pretherapeutic 18F-PSMA PET/CT Reveals Incidental Tracheal Epithelial–Myoepithelial Carcinoma. Diagnostics, 16(6), 883. https://doi.org/10.3390/diagnostics16060883







