Clinical and Molecular Diagnostic Profiling of Vaginitis Using Multiplex Real-Time PCR: A Multicenter Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design, Participants, and Ethical Considerations
2.2. Clinical Data Collection and Definitions of Symptom Severity
2.3. Vaginal Sample Collection and pH Measurement
2.4. Multiplex Real-Time PCR Detection of Pathogen
2.5. Statistical Analysis
3. Results
3.1. Participant Demographics and Clinical Characteristics
3.2. Vaginal Pathogen Profile
3.3. Clinical Symptoms and Vaginal pH Associated with Pathogen Infection
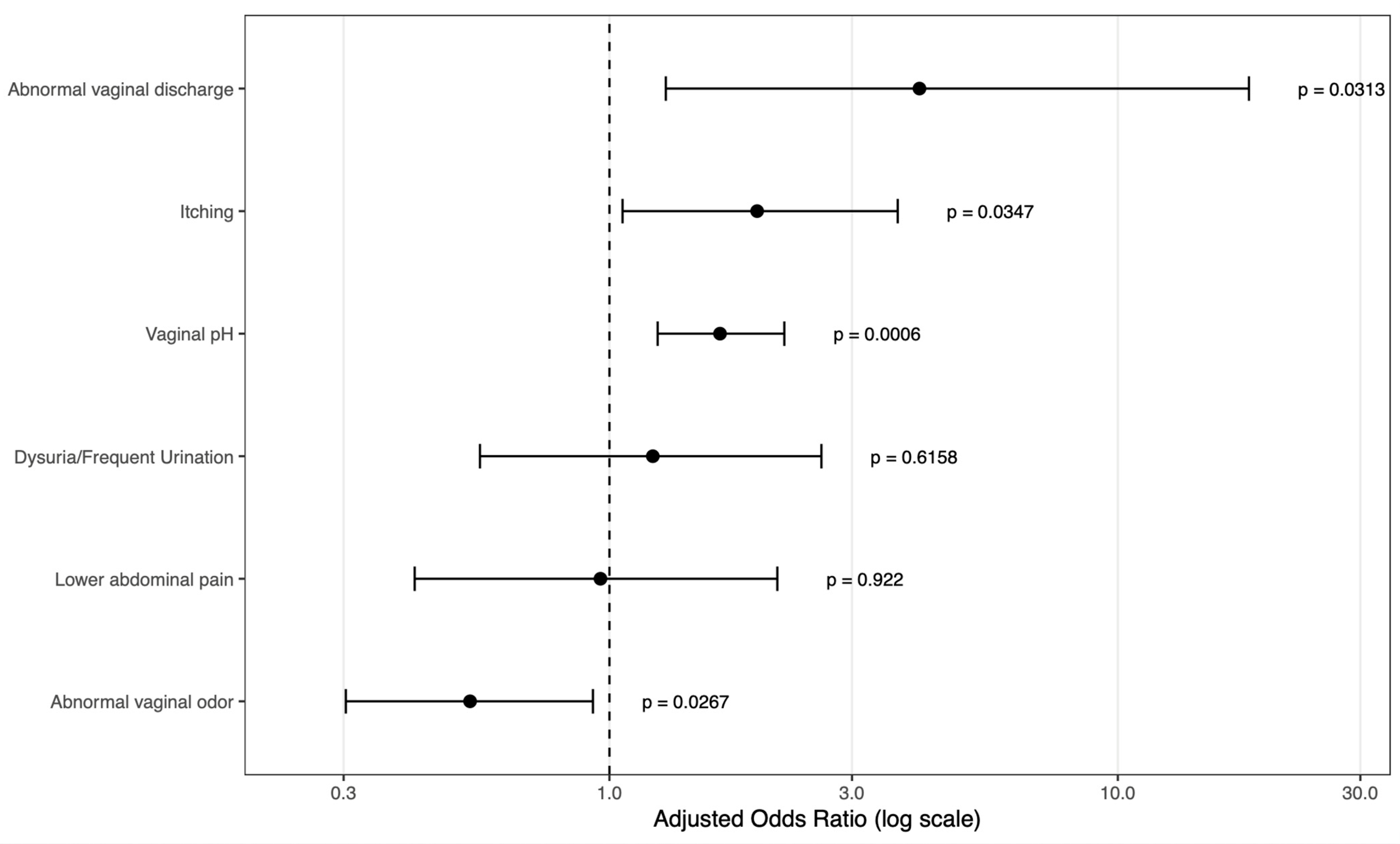
3.4. Clinical Symptoms, Vaginal pH, and Infection Profiles Associated with Study Site
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sobel, J.D.; Vempati, Y.S. Bacterial Vaginosis and Vulvovaginal Candidiasis Pathophysiologic Interrelationship. Microorganisms 2024, 12, 108. [Google Scholar] [CrossRef] [PubMed]
- Workowski, K.A. Sexually transmitted infections treatment guidelines. MMWR. Recomm. Rep. 2021, 70, 1–187. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, C.S.; Sobel, J.D. Current Treatment of Bacterial Vaginosis-Limitations and Need for Innovation. J. Infect. Dis. 2016, 214, S14–S20. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.; Wenlock, R.; Roper, T.; Butler, C.; Vera, J.H. Facilitators and barriers to point-of-care testing for sexually transmitted infections in low- and middle-income countries: A scoping review. BMC Infect. Dis. 2022, 22, 561. [Google Scholar] [CrossRef]
- Peebles, K.; Velloza, J.; Balkus, J.E.; McClelland, R.S.; Barnabas, R.V. High global burden and costs of bacterial vaginosis: A systematic review and meta-analysis. Sex. Transm. Dis. 2019, 46, 304–311. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for the Management of Symptomatic Sexually Transmitted Infections; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Kamga, Y.M.; Ngunde, J.P.; Akoachere, J.-F.K.T. Prevalence of bacterial vaginosis and associated risk factors in pregnant women receiving antenatal care at the Kumba Health District (KHD), Cameroon. BMC Pregnancy Childbirth 2019, 19, 166. [Google Scholar] [CrossRef]
- Go, V.F.; Quan, V.M.; Celentano, D.D.; Moulton, L.H.; Zenilman, J.M. Prevalence and risk factors for reproductive tract infections among women in rural Vietnam. Southeast Asian J. Trop. Med. Public Health 2006, 37, 185–189. [Google Scholar]
- Anh, P.K.; Khanh, N.T.; Ha, D.T.; Chien, D.T.; Thuc, P.T.; Luong, P.H.; Kilmarx, P.H.; Wongchotigul, V.; Kitayaporn, D.; Rowe, P.J. Prevalence of lower genital tract infection among women attending maternal and child health and family planning clinics in Hanoi, Vietnam. Southeast Asian J. Trop. Med. Public Health 2003, 34, 367–373. [Google Scholar]
- Nguyen, M.H.; Kurtzhals, J.; Do, T.T.; Rasch, V. Reproductive tract infections in women seeking abortion in Vietnam. BMC Womens Health 2009, 9, 1. [Google Scholar] [CrossRef]
- Brown, H.; Drexler, M. Improving the diagnosis of vulvovaginitis: Perspectives to align practice, guidelines, and awareness. Popul. Health Manag. 2020, 23, S-3-S-12. [Google Scholar] [CrossRef]
- Gaydos, C.A.; Cartwright, C.P.; Colaninno, P.; Welsch, J.; Holden, J.; Ho, S.Y.; Webb, E.M.; Anderson, C.; Bertuzis, R.; Zhang, L. Performance of the Abbott Real time CT/NG for Detection of Chlamydia trachomatis and Neisseria gonorrhoeae. J. Clin. Microbiol. 2010, 48, 3236–3243. [Google Scholar] [CrossRef]
- Bui, H.T.V.; Bui, H.T.; Chu, S.V.; Nguyen, H.T.; Nguyen, A.T.V.; Truong, P.T.; Dang, T.T.H.; Nguyen, A.T.V. Simultaneous real-time PCR detection of nine prevalent sexually transmitted infections using a predesigned double-quenched TaqMan probe panel. PLoS ONE 2023, 18, e0282439. [Google Scholar] [CrossRef]
- Denning, D.W.; Kneale, M.; Sobel, J.D.; Rautemaa-Richardson, R. Global burden of recurrent vulvovaginal candidiasis: A systematic review. Lancet Infect. Dis. 2018, 18, e339–e347. [Google Scholar] [CrossRef]
- World Health Organization. Implementing the Global Health Sector Strategies on HIV, Viral Hepatitis and Sexually Transmitted Infections, 2022–2030: Report on Progress and Gaps 2024; World Health Organization: Geneva, Switzerland, 2024. [Google Scholar]
- Lewis, J.; Price, M.J.; Horner, P.J.; White, P.J. Genital Chlamydia trachomatis infections clear more slowly in men than women, but are less likely to become established. J. Infect. Dis. 2017, 216, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Herrero, M.; Broner, S.; Cruells, A.; Esteve, S.; Ferré, L.; Mendioroz, J.; Jané, M.; Ciruela, P. Epidemiology and antimicrobial resistance profile of Neisseria gonorrhoeae in Catalonia, Spain, 2016–2019. Eur. J. Clin. Microbiol. Infect. Dis. 2023, 42, 883–893. [Google Scholar] [CrossRef] [PubMed]
- Kissinger, P.J.; Gaydos, C.A.; Seña, A.C.; Scott McClelland, R.; Soper, D.; Secor, W.E.; Legendre, D.; Workowski, K.A.; Muzny, C.A. Diagnosis and management of Trichomonas vaginalis: Summary of evidence reviewed for the 2021 Centers for Disease Control and Prevention sexually transmitted infections treatment guidelines. Clin. Infect. Dis. 2022, 74, S152–S161. [Google Scholar] [CrossRef]
- Pereyre, S.; Caméléna, F.; Hénin, N.; Berçot, B.; Bébéar, C. Clinical performance of four multiplex real-time PCR kits detecting urogenital and sexually transmitted pathogens. Clin. Microbiol. Infect. 2022, 28, e733–e737. [Google Scholar] [CrossRef] [PubMed]
- Occhipinti, S.; Incognito, G.G.; Palumbo, M. The influence of the vaginal ecosystem on vaginitis, bacterial vaginosis, and sexually transmitted diseases: An epidemiological study and literature review. Arch. Gynecol. Obstet. 2025, 311, 347–353. [Google Scholar] [CrossRef]
- Vieira-Baptista, P.; Stockdale, C.K.; Sobel, J. (Eds.) International Society for the Study of Vulvovaginal Disease Recommendations for the Diagnosis and Treatment of Vaginitis; Admedic: Lisbon, Portugal, 2023; pp. 73–75. [Google Scholar] [CrossRef]
- Talan, D.A.; Gardner, O.B.; Faine, B.A. Women presenting to emergency departments with vaginitis should be offered single-dose empirical treatment. Acad. Emerg. Med. 2025, 32, 941–946. [Google Scholar] [CrossRef]
- Bujang, M.A.; Sa’at, N.; Sidik, T.; Joo, L.C. Sample Size Guidelines for Logistic Regression from Observational Studies with Large Population: Emphasis on the Accuracy Between Statistics and Parameters Based on Real Life Clinical Data. Malays. J. Med. Sci. 2018, 25, 122–130. [Google Scholar] [CrossRef]
- Chen, J.; Tse, J.; Shi, L.; Cheng, M.M.; Lillis, R.; Near, A.M. Real-world clinical burden of patients presenting with vaginitis symptoms in the United States. AJOG Glob. Rep. 2025, 5, 100504. [Google Scholar] [CrossRef]
- Mårdh, P.A.; Tchoudomirova, K.; Elshibly, S.; Hellberg, D. Symptoms and signs in single and mixed genital infections. Int. J. Gynaecol. Obstet. 1998, 63, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Eleutério, J., Jr.; Campaner, A.B.; de Carvalho, N.S. Diagnosis and treatment of infectious vaginitis: Proposal for a new algorithm. Front. Med. 2023, 10, 1040072. [Google Scholar] [CrossRef]
- Nkengasong, J.N.; Yao, K.; Onyebujoh, P. Laboratory medicine in low-income and middle-income countries: Progress and challenges. Lancet 2018, 391, 1873–1875. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, N.M.; Ramirez Hernandez, L.A.; Berman, D.; Morrill, S.; Gagneux, P.; Lewis, A.L. Social, microbial, and immune factors linking bacterial vaginosis and infectious diseases. J. Clin. Investig. 2025, 135, e184322. [Google Scholar] [CrossRef] [PubMed]
- Muzny, C.A.; Elnaggar, J.H.; Sousa, L.G.V.; Lima, Â.; Aaron, K.J.; Eastlund, I.C.; Graves, K.J.; Dong, C.; Van Gerwen, O.T.; Luo, M.; et al. Microbial interactions among Gardnerella, Prevotella and Fannyhessea prior to incident bacterial vaginosis: Protocol for a prospective, observational study. BMJ Open 2024, 14, e083516. [Google Scholar] [CrossRef]
- Muzny, C.A.; Cerca, N.; Elnaggar, J.H.; Taylor, C.M.; Sobel, J.D.; Van Der Pol, B. State of the Art for Diagnosis of Bacterial Vaginosis. J. Clin. Microbiol. 2023, 61, e00837-22. [Google Scholar] [CrossRef]
- Huang, Y.; Shen, Y.; Wei, X.; Feng, J.; Zhu, L. Predictive value of vaginal lactic acid bacteria changes on occurrence of HR-HPV-infected cervical intraepithelial neoplasia and construction and validation of nomogram model. BMC Cancer 2025, 25, 1212. [Google Scholar] [CrossRef]
- Kalia, N.; Singh, J.; Kaur, M. Immunopathology of Recurrent Vulvovaginal Infections: New Aspects and Research Directions. Front. Immunol. 2019, 10, 2034. [Google Scholar] [CrossRef]
- Pérez-Llantada, J.G.; Masero, A.R.; Lázaro-Carrasco, J.; Carballo, M.J.; Ridocci, F.; Sosa, M.; González, M.; Azumendi, P.; Palma, M.; Nieto, C. Single-pathogen and Mixed Vulvovaginal Infections Among Women of Reproductive Age Consulting Gynecologists: A Cross-Sectional Study. Int. J. Microbiol. Biotechnol. 2021, 6, 98–106. [Google Scholar] [CrossRef]
- Lin, Y.P.; Chen, W.C.; Cheng, C.M.; Shen, C.J. Vaginal pH Value for Clinical Diagnosis and Treatment of Common Vaginitis. Diagnostics 2021, 11, 1996. [Google Scholar] [CrossRef] [PubMed]
- Godha, K.; Tucker, K.M.; Biehl, C.; Archer, D.F.; Mirkin, S. Human vaginal pH and microbiota: An update. Gynecol. Endocrinol. 2018, 34, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.A.; Beasley, D.E.; Dunn, R.R.; Archie, E.A. Lactobacilli dominance and vaginal pH: Why is the human vaginal microbiome unique? Front. Microbiol. 2016, 7, 1936. [Google Scholar] [CrossRef] [PubMed]
- Swidsinski, S.; Moll, W.M.; Swidsinski, A. Bacterial Vaginosis-Vaginal Polymicrobial Biofilms and Dysbiosis. Dtsch. Arztebl. Int. 2023, 120, 347–354. [Google Scholar] [CrossRef]
- Srinivasan, S.; Fredricks, D.N. The human vaginal bacterial biota and bacterial vaginosis. Interdiscip. Perspect. Infect. Dis. 2008, 2008, 750479. [Google Scholar] [CrossRef]
- Schwebke, J.R.; Muzny, C.A.; Josey, W.E. Role of Gardnerella vaginalis in the pathogenesis of bacterial vaginosis: A conceptual model. J. Infect. Dis. 2014, 210, 338–343. [Google Scholar] [CrossRef]
- Van de Wijgert, J.; Jespers, V. The global health impact of vaginal dysbiosis. Res. Microbiol. 2017, 168, 859–864. [Google Scholar] [CrossRef]
- Cox, C.; Watt, A.P.; McKenna, J.P.; Coyle, P.V. Mycoplasma hominis and Gardnerella vaginalis display a significant synergistic relationship in bacterial vaginosis. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 481–487. [Google Scholar] [CrossRef]
- Castro, J.; Machado, D.; Cerca, N. Unveiling the role of Gardnerella vaginalis in polymicrobial bacterial vaginosis biofilms: The impact of other vaginal pathogens living as neighbors. ISME J. 2019, 13, 1306–1317. [Google Scholar] [CrossRef]
- Gaziano, R.; Sabbatini, S.; Monari, C. The interplay between Candida albicans, vaginal mucosa, host immunity and resident microbiota in health and disease: An overview and future perspectives. Microorganisms 2023, 11, 1211. [Google Scholar] [CrossRef]
- Benyas, D.; Sobel, J.D. Mixed Vaginitis Due to Bacterial Vaginosis and Candidiasis. J. Low. Genit. Tract. Dis. 2022, 26, 68–70. [Google Scholar] [CrossRef]
- Jun, L.; Wan, X.; Zhang, D.; Zheng, Y.; Chen, X.; Mi, L.; Xiao, B. Mixed vaginal infection status in women infected with Trichomonas vaginalis: Comparison of microscopy method and metagenomic sequencing analysis. Front. Cell. Infect. Microbiol. 2025, 15, 1638464. [Google Scholar] [CrossRef]
- Enwuru, C.A.; Enwuru, V.N. Pathogenesis and specific diagnosis of bacterial vaginosis, vaginitis, and flagellate/coinfection in women: An expert review. J. Microbiol. Infect. Dis. 2025, 15, 128–141. [Google Scholar] [CrossRef]
- Lu, S.; Li, Z.; Chen, X.; Chen, F.; Yao, H.; Sun, X.; Cheng, Y.; Wang, L.; Dai, P. Vaginal microbiota molecular profiling and diagnostic performance of artificial intelligence-assisted multiplex PCR testing in women with bacterial vaginosis: A single-center experience. Front. Cell. Infect. Microbiol. 2024, 14, 1377225. [Google Scholar] [CrossRef]

| Characteristic | Total (N = 289) | Characteristic | Total (N = 289) |
|---|---|---|---|
| Age of participants | Genital hygiene practices n (%) | ||
| Mean ± SD | 31.01 ± 9.60 | Vaginal douching | 41 (14.19) |
| 18–45 n (%) | 264 (91.35) | Use of cleaning solution | 225 (77.85) |
| 46–60 n (%) | 25 (8.65) | Clinical symptom n (%) | |
| Region n (%) | Abnormal vaginal odor | 124 (42.91) | |
| Urban | 169 (58.48) | Abnormal vaginal discharge | 257 (88.93) |
| Rural | 120 (41.52) | Itching | 188 (65.05) |
| Marital status n (%) | Dysuria/Frequent Urination | 39 (13.49) | |
| Single | 118 (40.83) | Lower abdominal pain | 37 (12.80) |
| Married | 166 (57.44) | Symptom burden n (%) | |
| Divorced | 4 (1.38) | Single symptom | 37 (12.80) |
| Number of births n (%) | 2 symptoms | 104 (35.99) | |
| No childbirth | 128 (44.29) | ≥3 symptoms | 131 (45.33) |
| 1 child | 39 (13.49) | Vaginal pH n (%) | |
| ≥2 children | 122 (42.21) | 3.8–4.5 | 61 (21.11) |
| History of HPV injection n (%) | 51 (17.65) | >4.5 | 228 (78.89) |
| Infection Category | Total (N = 289) | Infection Category | Total (N = 289) |
|---|---|---|---|
| Overall pathogen prevalence | G. vaginalis, M. genitalium | 2 (0.69) | |
| Bacterial Vaginosis n (%) | G. vaginalis, C. albicans | 46 (15.92) | |
| Gardnerella vaginalis | 260 (89.97) | G. vaginalis, C. trachomatis | 8 (2.77) |
| Mycoplasma hominis | 28 (9.69) | G. vaginalis, HSV-1 | 1 (0.35) |
| Vulvovaginal Candidiasis n (%) | T. vaginalis, M. hominis | 1 (0.35) | |
| Candida albicans | 63 (21.80) | 3-pathogen infection | 16 (5.54) |
| STIs n (%) | G. vaginalis, C. albicans, M. hominis | 7 (2.42) | |
| Chlamydia trachomatis | 16 (5.54) | G. vaginalis, C. albicans, C. trachomatis | 1 (0.35) |
| Mycoplasma genitalium | 5 (1.73) | G. vaginalis, C. albicans, T. vaginalis | 1 (0.35) |
| Trichomonas vaginalis | 2 (0.69) | G. vaginalis, C. albicans, M. genitalium | 1 (0.35) |
| Neisseria gonorrhoeae | 3 (1.04) | G. vaginalis, C. trachomatis, M. hominis | 2 (0.69) |
| Herpes simplex virus (HSV-1&2) | 4 (1.38) | G. vaginalis, C. trachomatis, N. gonorrhoeae | 1 (0.35) |
| Pathogen infection patterns | G. vaginalis, M. hominis, M. genitalium | 1 (0.35) | |
| Single-pathogen infection n (%) | 180 (62.28) | G. vaginalis, M. hominis, HSV-1 | 1 (0.35) |
| G. vaginalis | 173 (59.86) | G. vaginalis, M. hominis, N. gonorrhoeae | 1 (0.35) |
| M. hominis | 2 (0.69) | 4-pathogen infection | 3 (1.04) |
| C. albicans | 4 (1.38) | G. vaginalis, C. albicans, C. trachomatis, M. hominis | 1 (0.35) |
| HSV-1 | 1 (0.35) | G. vaginalis, C. albicans, C. trachomatis, M. genitalium | 1 (0.35) |
| Multi-pathogen infection n (%) | 88 (30.45) | G. vaginalis, C. trachomatis, M. hominis, N. gonorrhoeae | 1 (0.35) |
| 2-pathogen infection | 68 (23.53) | 5-pathogen infection | 1 (0.35) |
| G. vaginalis, M. hominis | 10 (3.46) | G. vaginalis, C. albicans, C. trachomatis, M. hominis, HSV-2 | 1 (0.35) |
| Factor | Single Infection (N = 180) | Multi Infections (N = 88) | OR (95% CI) | p Value | Adjusted p Value |
|---|---|---|---|---|---|
| Clinical sign or symptom n (%) | |||||
| Abnormal vaginal odor | 108 (60.00) | 43 (48.86) | 0.637 (0.3785–1.066) | 0.0843 a | 0.1405 |
| Abnormal vaginal discharge | 151 (83.89) | 85 (96.59) | 5.442 (1.709–17.36) | 0.0022 b | 0.011 |
| Itching | 108 (60.00) | 67 (76.14) | 2.127 (1.212–3.722) | 0.0092 a | 0.023 |
| Dysuria/Frequent Urination | 22 (12.22) | 15 (17.05) | 1.476 (0.7215–3.001) | 0.2824 a | 0.353 |
| Lower abdominal pain | 23 (12.78) | 12 (13.64) | 1.078 (0.5118–2.298) | 0.8447 a | 0.8447 |
| Symptom burden n (%) | |||||
| Single symptom | 24 (13.33) | 6 (6.82) | 0.4756 (0.1959–1.203) | 0.1121 a | 0.1716 |
| 2 symptoms | 58 (32.22) | 37 (42.05) | 1.526 (0.7170–1.995) | 0.1144 a | 0.1716 |
| ≥3 symptoms | 82 (45.56) | 44 (50.00) | 1.195 (0.6396–1.829) | 0.4936 a | 0.4936 |
| Vaginal pH n (%) | |||||
| 3.8–4.5 | 52 (28.89) | 7 (7.95) | 4.701 (2.110–11.26) | <0.0001 b | |
| >4.5 | 128 (71.11) | 81 (92.05) |
| Factor | Bac Ninh CDC (N = 128) | Hanoi OHG (N = 161) | OR (95% CI) | p Value | Adjusted p Value |
|---|---|---|---|---|---|
| Clinical sign or symptom n (%) | |||||
| Abnormal vaginal odor | 91 (71.09) | 64 (39.75) | 0.2683 (0.1626–0.4395) | 0.0001 a | 0.00025 |
| Abnormal vaginal discharge | 103 (80.47) | 154 (95.65) | 5.34 (2.301–13.53) | 0.0001 a | 0.00025 |
| Itching | 80 (62.50) | 108 (67.08) | 1.223 (0.7453–2.002) | 0.4172 a | 0.4172 |
| Dysuria/Frequent Urination | 13 (10.16) | 26 (16.15) | 1.704 (0.8644–3.415) | 0.1386 a | 0.17325 |
| Lower abdominal pain | 12 (9.38) | 25 (15.53) | 1.777 (0.8711–3.628) | 0.1199 a | 0.17325 |
| Symptom burden n (%) | |||||
| Single symptom | 12 (9.38) | 25 (15.53) | 1.777 (0.8711–3.628) | 0.1199 a | 0.17985 |
| 2 symptoms | 37 (28.91) | 67 (41.61) | 1.753 (1.067–2.871) | 0.0254 a | 0.0762 |
| ≥3 symptoms | 65 (50.78) | 66 (40.99) | 0.6734 (0.4253–1.09) | 0.9669 a | 0.9669 |
| Vaginal pH n (%) | |||||
| 3.8–4.5 | 51 (39.84) | 10 (6.21) | 10.00 (4.905–20.07) | <0.0001 b | |
| >4.5 | 77 (60.16) | 151 (93.79) | |||
| Infections | |||||
| Bacterial Vaginosis (BV) n (%) | |||||
| Gardnerella vaginalis | 128 (100.00) | 132 (81.99) | 0.00 (0.000–0.1222) | <0.0001 b | |
| Mycoplasma hominis | 2 (1.56) | 26 (16.15) | 13.10 (3.495–56.71) | <0.0001 b | |
| Vulvovaginal Candidiasis (VVC) n (%) | |||||
| Candida albicans | 9 (7.03) | 54 (33.54) | 0.1499 (0.07102–0.3122) | <0.0001 a | |
| Other STIs n (%) | 0 (0.00) | 30 (18.63) | 0.00 (0.000–0.1169) c | <0.0001 b | |
| Chlamydia trachomatis | 0 (0.00) | 16 (9.94) | 0.00 (0.000–0.2645) c | <0.0001 b | |
| Single or multi-infections n (%) | |||||
| Single infection | 117 (91.41) | 63 (39.13) | 16.55 (8.289–32.92) | <0.0001 a | |
| Multi-infections | 11 (8.59) | 77 (47.83) | 9.75 (4.915–19.31) | <0.0001 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mai, H.T.; Nguyen, C.C.; Vo, H.T.N.; Nguyen, T.T.B.; Pham, T.T.; Ngo, H.T.; Ngo, X.T.; Bui, A.T.P.; Ta, H.T.K.; Nguyen, A.T.V. Clinical and Molecular Diagnostic Profiling of Vaginitis Using Multiplex Real-Time PCR: A Multicenter Study. Diagnostics 2026, 16, 783. https://doi.org/10.3390/diagnostics16050783
Mai HT, Nguyen CC, Vo HTN, Nguyen TTB, Pham TT, Ngo HT, Ngo XT, Bui ATP, Ta HTK, Nguyen ATV. Clinical and Molecular Diagnostic Profiling of Vaginitis Using Multiplex Real-Time PCR: A Multicenter Study. Diagnostics. 2026; 16(5):783. https://doi.org/10.3390/diagnostics16050783
Chicago/Turabian StyleMai, Hung Trong, Chuong Canh Nguyen, Hao Thi Ngoc Vo, Thuy Thi Bich Nguyen, Trang Thi Pham, Hong Thi Ngo, Xuan Thi Ngo, Anh Thi Phuong Bui, Hue Thi Kim Ta, and Anh Thi Van Nguyen. 2026. "Clinical and Molecular Diagnostic Profiling of Vaginitis Using Multiplex Real-Time PCR: A Multicenter Study" Diagnostics 16, no. 5: 783. https://doi.org/10.3390/diagnostics16050783
APA StyleMai, H. T., Nguyen, C. C., Vo, H. T. N., Nguyen, T. T. B., Pham, T. T., Ngo, H. T., Ngo, X. T., Bui, A. T. P., Ta, H. T. K., & Nguyen, A. T. V. (2026). Clinical and Molecular Diagnostic Profiling of Vaginitis Using Multiplex Real-Time PCR: A Multicenter Study. Diagnostics, 16(5), 783. https://doi.org/10.3390/diagnostics16050783






