Dermoscopy of Cutaneous Melanoma Metastases: A Comprehensive Literature Review
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Literature Review
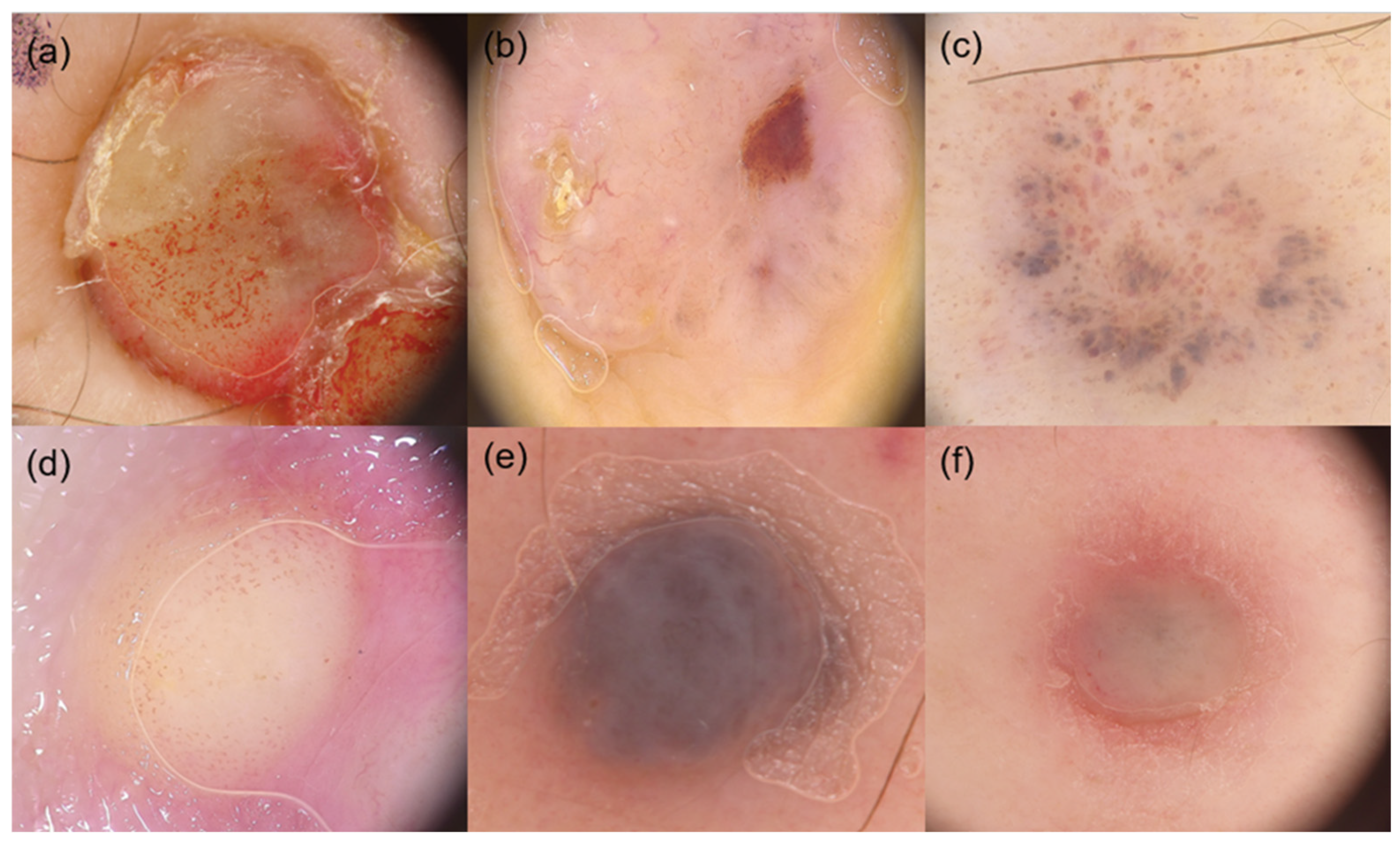
3.2. Global Dermoscopic Features and Pigmentation Patterns
3.3. Focal Dermoscopic Features
3.4. Dermoscopic Features of Vascular Structures
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schadendorf, D.; van Akkooi, A.C.J.; Berking, C.; Griewank, K.G.; Gutzmer, R.; Hauschild, A.; Stang, A.; Roesch, A.; Ugurel, S. Melanoma. Lancet 2018, 392, 971–984. [Google Scholar] [CrossRef]
- Scarfì, F.; Patrizi, A.; Veronesi, G.; Lambertini, M.; Tartari, F.; Mussi, M.; Melotti, B.; Dika, E. The role of topical imiquimod in melanoma cutaneous metastases: A critical review of the literature. Dermatol. Ther. 2020, 33, e14165. [Google Scholar] [CrossRef] [PubMed]
- Tasdogan, A.; Sullivan, R.J.; Katalinic, A.; Lebbe, C.; Whitaker, D.; Puig, S.; van de Poll-Franse, L.V.; Massi, D.; Schadendorf, D. Cutaneous melanoma. Nat. Rev. Dis. Primers 2025, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Hartman, R.I.; Lin, J.Y. Cutaneous Melanoma-A Review in Detection, Staging, and Management. Hematol. Oncol. Clin. N. Am. 2019, 33, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Bono, R.; Giampetruzzi, A.R.; Concolino, F.; Puddu, P.; Scoppola, A.; Sera, F.; Marchetti, P. Dermoscopic patterns of cutaneous melanoma metastases. Melanoma Res. 2004, 14, 367–373. [Google Scholar] [CrossRef]
- Turner, N.; Ware, O.; Bosenberg, M. Genetics of metastasis: Melanoma and other cancers. Clin. Exp. Metastasis 2018, 35, 379–391. [Google Scholar] [CrossRef]
- Pan, Y.; Haydon, A.M.; McLean, C.A.; McDonald, P.B.; Kelly, J.W. Prognosis associated with cutaneous melanoma metastases. Australas. J. Dermatol. 2015, 56, 25–28. [Google Scholar] [CrossRef]
- Avilés-Izquierdo, J.A.; Ciudad-Blanco, C.; Sánchez-Herrero, A.; Mateos-Mayo, A.; Nieto-Benito, L.M.; Rodríguez-Lomba, E. Dermoscopy of cutaneous melanoma metastases: A color-based pattern classification. J. Dermatol. 2019, 46, 564–569. [Google Scholar] [CrossRef]
- Simionescu, O.; Petrică, M.; Avram, A.M.; Costache, M.; Scurtu, L.G.; Tudorache, S.I.; Iorga, P.G.; Grigore, M. Dermoscopy of skin metastases in advanced cancer—Systemic (visceral, hematologic) and cutaneous. Front. Med. 2024, 11, 1445811. [Google Scholar] [CrossRef]
- Tiodorovic, D.; Stojkovic-Filipovic, J.; Marghoob, A.; Argenziano, G.; Puig, S.; Malvehy, J.; Tognetti, L.; Pietro, R.; Akay, B.N.; Zalaudek, I.; et al. Dermatoscopic patterns of cutaneous metastases: A multicentre cross-sectional study of the International Dermoscopy Society. J. Eur. Acad. Dermatol. Venereol. 2024, 38, 1432–1438. [Google Scholar] [CrossRef]
- Costa, J.; Ortiz-Ibañez, K.; Salerni, G.; Borges, V.; Carrera, C.; Puig, S.; Malvehy, J. Dermoscopic patterns of melanoma metastases: Interobserver consistency and accuracy for metastasis recognition. Br. J. Dermatol. 2013, 169, 91–99. [Google Scholar] [CrossRef]
- Jones, O.T.; Jurascheck, L.; van Melle, M.; Hickman, S.; Burrows, N.; Hall, P.; Emery, J.; Walter, F. Dermoscopy for melanoma detection and triage in primary care: A systematic review. BMJ Open 2019, 9, e027529. [Google Scholar] [CrossRef]
- Ankad, B.S.; Behera, B.; Lallas, A.; Akay, B.N.; Bhat, Y.J.; Chauhan, P.; Enechukwu, N.A.; Geller, S.; Jha, A.K.; Kaliyadan, F.; et al. International Dermoscopy Society (IDS) criteria for skin tumors: Validation for skin of color through a Delphi expert consensus. Dermatol. Pract. Concept. 2023, 13, e2023067. [Google Scholar] [CrossRef] [PubMed]
- Virgili, A.; Zampino, M.R.; Corazza, M. Primary vulvar melanoma with satellite metastasis: Dermoscopic findings. Dermatology 2004, 208, 145–148. [Google Scholar] [CrossRef]
- Minagawa, A.; Koga, H.; Sakaizawa, K.; Sano, K.; Saida, T. Dermoscopic and histopathological findings of polymorphous vessels in amelanotic cutaneous metastasis of pigmented cutaneous melanoma. Br. J. Dermatol. 2009, 160, 1134–1136. [Google Scholar] [CrossRef]
- Contreras-Steyls, M.; Herrera-Acosta, E.; Moyano, B.; Herrera, E. Primary melanoma with multiple skin metastases. Actas Dermosifiliogr. 2011, 102, 226–229. [Google Scholar] [CrossRef]
- Jaimes, N.; Halpern, J.A.; Puig, S.; Malvehy, J.; Myskowski, P.L.; Braun, R.P.; Marghoob, A.A. Dermoscopy: An aid to the detection of amelanotic cutaneous melanoma metastases. Dermatol. Surg. 2012, 38, 1437–1444. [Google Scholar] [CrossRef]
- Duman, N.; Şahin, S.; Özaygen, G.E.; Gököz, Ö. Dermoscopy of satellite metastasis of balloon cell melanoma. J. Am. Acad. Dermatol. 2014, 71, e11–e12. [Google Scholar] [CrossRef] [PubMed]
- Hoelt, P.; Dalle, S.; Thomas, L. Cutaneous metastases of melanoma. Ann. Dermatol. Venereol. 2014, 141, 640–642. [Google Scholar] [CrossRef]
- Perrot, J.L.; Labeille, B.; Habougit, C.; Douchet, C.; Tognetti, L.; Cambazard, F.; Cinotti, E.; Groupe D’imagerie Cutanée Non Invasive (ICNI) de la Société Française de Dermatologie. The role of reflectance confocal microscopy in the diagnosis of cutaneous melanoma metastasis. Ann. Dermatol. Venereol. 2016, 143, 863–865. [Google Scholar] [CrossRef] [PubMed]
- Ribero, S.; Marra, E.; Tomasini, C.; Fierro, M.; Bombonato, C.; Longo, C. Confocal microscopy and dermoscopy for the monitoring of BRAF inhibitor therapy of melanoma skin metastases. Br. J. Dermatol. 2017, 176, 1101–1102. [Google Scholar] [CrossRef] [PubMed]
- Mazzella, C.; Costa, C.; Cappello, M.; Scalvenzi, M. Difficult to diagnose small cutaneous melanoma metastases mimicking angiomas: Utility of dermoscopy. Int. J. Dermatol. 2018, 57, 1085–1087. [Google Scholar] [CrossRef] [PubMed]
- Santini, D.; Tonini, G.; Vincenzi, B.; Murace, R.; Ferranti, G.; Baldi, A. Skin lesions in melanoma and Kaposi’s sarcoma. J. Clin. Oncol. 2002, 20, 1411–1412. [Google Scholar] [CrossRef]
- Paganelli, A.; Longo, C.; Pampena, R.; Piana, S.; Borsari, S. Early diagnosis of skin melanoma metastasis by means of dermoscopy and confocal microscopy. JAMA Dermatol. 2018, 154, 1482–1485. [Google Scholar] [CrossRef]
- Pertusi, G.; Miglino, B.; Tiberio, R.; Veronese, F.; Giorgione, R.; Gattoni, M.; Colombo, E. Different dermoscopic patterns of cutaneous melanoma metastases in the same patient. G. Ital. Dermatol. Venereol. 2017, 152, 321–322. [Google Scholar] [CrossRef] [PubMed]
- Kostaki, M.; Plaka, M.; Moustaki, M.; Befon, A.; Champsas, G.; Kypreou, K.; Chardalia, V.; Chasapi, V.; Polydorou, D.; Stratigos, A. Cutaneous melanoma metastases: Clinical and dermoscopic findings. J. Eur. Acad. Dermatol. Venereol. 2023, 37, 941–944. [Google Scholar] [CrossRef]
- Chernoff, K.A.; Marghoob, A.A.; Lacouture, M.E.; Deng, L.; Busam, K.J.; Myskowski, P.L. Dermoscopic findings in cutaneous metastases. JAMA Dermatol. 2014, 150, 429. [Google Scholar] [CrossRef]
- Álvarez-Chinchilla, P.; Encabo-Duran, B.; Poveda, I.; Planelles, M.; Bañuls, J. Cutaneous metastases of melanoma presenting as sudden haematomas. Clin. Exp. Dermatol. 2018, 43, 852–854. [Google Scholar] [CrossRef]
- Farnetani, F.; Manfredini, M.; Longhitano, S.; Chester, J.; Shaniko, K.; Cinotti, E.; Mazzoni, L.; Venturini, M.; Manganoni, A.; Longo, C.; et al. Morphological classification of melanoma metastasis with reflectance confocal microscopy. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 676–685. [Google Scholar] [CrossRef] [PubMed]
- Lugano, R.; Ramachandran, M.; Dimberg, A. Tumor angiogenesis: Causes, consequences, challenges and opportunities. Cell. Mol. Life Sci. 2020, 77, 1745–1770. [Google Scholar] [CrossRef]
- Hashemi, G.; Dight, J.; Khosrotehrani, K.; Sormani, L. Melanoma tumour vascularization. Cancers 2022, 14, 4216. [Google Scholar] [CrossRef] [PubMed]
- Papachristou, P.; Söderholm, M.; Pallon, J.; Taloyan, M.; Polesie, S.; Paoli, J.; Anderson, C.D.; Falk, M. Evaluation of an artificial intelligence-based decision support for the detection of cutaneous melanoma in primary care: A prospective real-life clinical trial. Br. J. Dermatol. 2024, 191, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Lan, J.; Wen, J.; Cao, S.; Yin, T.; Jiang, B.; Lou, Y.; Zhu, J.; An, X.; Suo, H.; Li, D.; et al. Diagnostic accuracy of dermoscopy and reflectance confocal microscopy for amelanotic melanoma. Br. J. Dermatol. 2020, 183, 210–219. [Google Scholar] [CrossRef] [PubMed]

| Global CMM Features | Focal CMM Features | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N° of Lesions | Homogeneous Main Color Pigmentation | Amelanotic | Vascular | Saccular | Nevus Like Pattern * | Angioma-Like | Black Dots or Globules Irregular Distributed | Light Brown Halo | Peripheral Gray Dots | Crystalline Structures | Peripheral Erythema | Lacune Like Areas | ||||||
| Brown or Gray | Brown or Black | Whitish or Gray | Blue | Pink or Red | Two or More Colors | |||||||||||||
| Pizzichetta et al., [23] | 4 | - | - | - | - | - | 1 | - | - | - | 3 | - | - | - | - | - | - | - |
| Bono et al., [5] | 130 | 51 | 21 | - | 16 | 42 | 5 | 42 | - | - | - | - | - | 53 | 33 | - | 16 | - |
| Virgili et al., [14] | 1 | - | - | 1 | - | - | - | - | - | - | - | - | 1 | - | - | - | - | - |
| Minagawa et al., [15] | 1 | - | - | - | - | - | - | 1 | - | - | - | - | - | - | - | - | - | - |
| Contreras-Steyls et al., [16] | 1 | - | 1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Jaimes et al., [17] | 47 | - | - | - | - | - | - | 47 | - | - | - | - | - | - | - | - | - | - |
| Costa et al., [11] | 71 | - | 11 | - | 22 | - | - | - | 14 | - | - | 9 | 15 | - | - | - | - | 8 |
| Duman et al., [18] | 1 | - | - | - | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - |
| Hoelt et al., [19] | 3 | 1 | - | - | 1 | - | 1 | - | - | - | - | - | - | - | - | - | - | - |
| Perrot et al., [20] | 1 | - | - | - | - | - | 1 | - | - | - | - | - | - | - | - | - | - | - |
| Ribero et al., [21] | 1 | - | - | - | 1 | - | - | - | - | - | - | - | - | - | 1 | 1 | - | |
| Pertusi et al., [25] | 8 | 2 | - | - | 2 | - | - | 2 | - | 1 | 5 | - | 2 | 1 | 1 | - | 1 | - |
| Álvarez-Chinchilla et al., [28] | 11 | - | - | - | - | - | 11 | - | - | - | - | - | - | - | - | - | - | - |
| Mazzella et al., [22] | 2 | - | 1 | - | - | - | - | - | - | - | - | - | 1 | - | - | - | - | - |
| Paganelli et al., [24] | 1 | - | 1 | - | - | - | - | - | - | - | - | - | 1 | - | - | - | - | - |
| Avilés-Izquierdo et al., [8] | 150 | - | 37 | - | 34 | 42 | 37 | - | - | - | - | - | 37 | 53 | 51 | 48 | 15 | 13 |
| Kostaki et al., [26] | 42 | - | - | - | - | - | 13 | 12 | - | 7 | - | - | - | - | - | - | - | - |
| Simionescu et al., [9] | 715 | 42 | 66 | 139 | 162 | 71 | - | - | - | - | 137 | - | 46 | - | - | - | - | - |
| Tiodorovic et al., [10] | 158 | - | - | 104 | - | - | - | - | - | - | - | - | - | - | - | - | - | 2 |
| Vessels Dermoscopic Structures | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Linear | Glomerular | Dotted | Serpentine | Milky Red Areas | Hairpin | Corkscrew-Winding | Arborizing | Curved | Two or More Vascular Patterns | |
| Bono et al., [5] | - | - | - | - | - | - | 34 | - | - | 25 |
| Virgili et al., [14] | 1 | - | - | - | - | - | - | - | - | - |
| Minagawa et al., [15] | - | - | - | - | - | - | - | - | - | 1 |
| Jaimes et al., [17] | - | 14 | 7 | 21 | 2 | 11 | 9 | 8 | - | 2 |
| Costa et al., [11] | - | - | - | - | - | - | 5 | 1 | - | 7 |
| Chernoff et al., [27] | - | - | - | - | - | - | - | - | - | 1 |
| Duman et al., [18] | 1 | - | - | - | - | 1 | - | 1 | - | |
| Hoelt et al., [19] | - | - | - | - | 1 | - | 1 | - | - | 1 |
| Pertusi et. al., [25] | 3 | - | 1 | - | - | - | - | 1 | - | 1 |
| Avilés-Izquierdo et al., [8] | 40 | 18 | 15 | 9 | 7 | 6 | 3 | 3 | - | 50 |
| Kostaki et al., [26] | - | - | - | - | - | - | 4 | - | - | 2 |
| Simionescu et al., [9] | 18 | - | 33 | 2 | 1 | 8 | 5 | 2 | 10 | - |
| Tiodorovic et al., [10] | - | - | - | 104 | - | - | - | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
D’Onghia, M.; Agueci, S.; Scotti, B.; Falcinelli, F.; Lo Conte, S.; Cartocci, A.; Cortez, C.D.; Dika, E.; Tognetti, L.; Rubegni, P.; et al. Dermoscopy of Cutaneous Melanoma Metastases: A Comprehensive Literature Review. Diagnostics 2026, 16, 738. https://doi.org/10.3390/diagnostics16050738
D’Onghia M, Agueci S, Scotti B, Falcinelli F, Lo Conte S, Cartocci A, Cortez CD, Dika E, Tognetti L, Rubegni P, et al. Dermoscopy of Cutaneous Melanoma Metastases: A Comprehensive Literature Review. Diagnostics. 2026; 16(5):738. https://doi.org/10.3390/diagnostics16050738
Chicago/Turabian StyleD’Onghia, Martina, Serena Agueci, Biagio Scotti, Francesca Falcinelli, Sofia Lo Conte, Alessandra Cartocci, Christian Dorado Cortez, Emi Dika, Linda Tognetti, Pietro Rubegni, and et al. 2026. "Dermoscopy of Cutaneous Melanoma Metastases: A Comprehensive Literature Review" Diagnostics 16, no. 5: 738. https://doi.org/10.3390/diagnostics16050738
APA StyleD’Onghia, M., Agueci, S., Scotti, B., Falcinelli, F., Lo Conte, S., Cartocci, A., Cortez, C. D., Dika, E., Tognetti, L., Rubegni, P., Perrot, J., & Cinotti, E. (2026). Dermoscopy of Cutaneous Melanoma Metastases: A Comprehensive Literature Review. Diagnostics, 16(5), 738. https://doi.org/10.3390/diagnostics16050738








