Diagnosis of Tuberculous Meningitis: Integrating Clinical Assessment and Molecular Diagnostics
Abstract
1. Introduction
2. Conventional Diagnosis of Tuberculous Meningitis
2.1. Initial Clinical Assessment and Diagnostic Challenges
2.2. Clinical Features, Neuroimaging, and Uniform Case Definition in Tuberculous Meningitis
2.3. Alternative Clinical Models for Differential Diagnosis
| Model | Intended Use | Cut-Offs/Categories | Key Advantages and Limitations | Development/ Validation Limitations | Best-Use Settings |
|---|---|---|---|---|---|
| Marais et al. (2010) [27] (Lancet consensus score) | Uniform case definition for diagnostic standardization in research and clinical reporting. | Definite: microbiological confirmation. Probable: ≥12 points (with neuroimaging available) or ≥10 points (without neuroimaging). Possible: 6–11 points (with neuroimaging) or 6–9 points (without neuroimaging). Not TBM: <6 points. | Advantages: Widely adopted; integrates clinical features, CSF, neuroimaging, and evidence of extrapulmonary TB. Limitations: Limited usefulness for immediate bedside decision-making. | Country: multinational. Model type: case definition. Sample size: NA. Performance (AuROC): NA. External validation: NA. | Clinical research, epidemiology, and comparative diagnostic studies. |
| Thwaites’ Diagnostic Score (Sulaiman et al. 2020 [31]) | Initial triage. Differential diagnosis: TBM vs. acute bacterial meningitis (ABM). | ≤4 points suggests TBM. >4 points suggests BM. | Advantages: Simple bedside application. Limitations: Lower performance in subacute/chronic meningitis; poor discrimination in PLWH and children; does not incorporate molecular testing. | Country: multinational. Model type: clinical score. Sample size: 395 (162 TBM). Performance (AuROC): <0.5. External validation: Yes (multicenter revalidation). | Resource-limited settings; early triage. |
| Yang et al. (2020) [32] | Differential diagnosis: TBM vs. BM. | TDI: ≥0 suggests TBM. | Advantages: Better discrimination than traditional scores; easy to apply. Limitations: Not evaluated in PLWH; single-country cohort; no external validation. | Country: China. Model type: clinical score. Sample size: 103 (58 TBM). Performance (AuROC): NR. External validation: No. | Tertiary-care centers. |
| Luo et al. (2021) [33] | Differential diagnosis: TBM vs. bacterial meningitis (BM). | Cut-off ≈ 0.54 suggests TBM. | Advantages: Incorporates an immunological biomarker; high performance with external validation. Limitations: Requires T-SPOT; the study did not include non-bacterial meningitis etiologies. | Country: China. Model type: logistic model. Sample size: 279 (115 TBM/164 BM). Performance (AuROC): 0.949 (derivation)/0.923 (external validation). External validation: Yes (independent cohort). | Centers with access to immunology testing. |
| Wen et al. (2022) [34] | Differential diagnosis: TBM vs. BM. | Clinical score (TDI): TDI ≥ 7 classifies as TBM. TDI < 7 classifies as BM. | Advantages: User-friendly model; strong internal performance. Limitations: Limited external validation; retrospective study. | Country: China. Model type: clinical score. Sample size: 380 (114 TBM). Performance (AuROC): 0.954. External validation: Yes (limited). | Referral hospitals |
| Krongsut et al. (2025) [35] | Multivariable clinical prediction model (web-based calculator available) Differential diagnosis: TBM vs. BM. | Provides an estimated probability of TBM with low, intermediate, and high-risk categories | Advantages: Very high discriminatory performance. Web-based calculator for real-time risk stratification and bedside clinical decision support. Limitations: Single-center study; lacks external validation. | Country: Thailand. Model type: web-based model. Sample size: 377 (142 TBM). Performance (AuROC): 0.978. External validation: NA. | Hospitals in TB-endemic areas; resource-limited settings. |
| Lee et al. (2018) [36] | Differential diagnosis: TBM vs. VM. | ≥4 points suggest TBM. | Advantages: Good performance; feasible bedside use. Limitations: Restricted to TBM vs. VM; no external validation. | Country: South Korea. Model type: clinical score. Sample size: NA. Performance (AuROC): 0.901. External validation: No. | General hospitals. |
| Lu et al. (2021) [30] | Differential diagnosis: TBM vs. BM, VM, and cryptococcal meningitis. | ≥5 points suggest TBM. | Advantages: Integrates clinical data, CSF, and neuroimaging; prospective validation. Limitations: Requires MRI; not evaluated in PLWH; lacks international validation. | Country: China. Model type: nomogram. Sample size: 382 (113 TBM). Performance (AuROC): 0.923 (derivation)/0.884 (prospective validation). External validation: Yes. | Tertiary-care centers with access to neuroimaging. |
| Liu et al. (2023) [37] | Differential diagnosis: TBM vs. BM and VM. | Probabilistic nomogram: Approx. score 22–24 indicates intermediate probability (~50%). Approx. score 27–30 indicates high probability (~80–90%). | Advantages: Excellent discrimination (AuROC > 0.95); integrates clinical and neuroimaging predictors. Limitations: Does not incorporate molecular diagnostics; validated within one country. | Country: China. Model type: nomogram. Sample size: 322 (102 TBM). Performance (AuROC): 0.962. External validation: No. | High TB-burden settings. |
| Author, Year of Publication | Marais, 2010 [27] | Thwaites’ Score, 2002 [21] | Yang, 2020 [32] | Luo, 2021 [33] | Wen, 2022 [34] | Krongsut, 2025 [35] | Lee, 2018 [36] | Lu, 2021 [30] | Liu, 2023 [37] |
|---|---|---|---|---|---|---|---|---|---|
| Study characteristics | |||||||||
| Sample Size (TBM events) | NA | 395 (162) | 103 (58) | 279 (115) | 380 (114) | 377 (142) | NA | 382 (113) | 322 (102) |
| Key Predictors Included | |||||||||
| cough ≥ 2 weeks | − | − | + | − | − | − | − | − | − |
| disease duration | + | + | + | − | + | + | − | + | + |
| impaired consciousness | + | − | − | − | + | + | − | + | |
| Weight loss | − | − | − | − | − | + | − | − | + |
| cerebral infarction (imaging) | + | − | − | − | − | + | − | + | + |
| Serum sodium | − | − | + | − | − | − | + | + | − |
| CSF neutrophil percentage | − | + | + | − | − | − | − | + | − |
| CSF lymphocyte percentage | + | + | − | + | − | + | − | − | + |
| CSF glucose | + | − | − | − | − | + | − | − | + |
| CSF proteins | + | − | − | − | + | + | + | − | − |
| CSF chloride | − | − | − | − | + | − | − | − | − |
| immunological biomarkers | − | − | − | + | − | − | − | − | − |
| chest X-ray suggestive of TB | + | − | − | − | − | − | − | − | − |
| evidence of extrapulmonary TB | + | − | − | − | − | − | − | + | − |
2.4. Cytochemical Determinants and Biomarkers in CSF
| Biomarker | Typical Findings in TBM | Cut-Offs | Clinical Value | Main Limitations | References |
|---|---|---|---|---|---|
| Cytochemical parameters | |||||
| CSF glucose | Decreased (<2.2 mmol/L or CSF/serum ratio < 40–50%) | <30–40 mg/dL (depending on study and population) | Supports initial diagnostic suspicion. Moderate sensitivity and variable specificity. | Low specificity. Overlaps with other meningitis etiologies. May be normal in early TBM or in PLWH. Inferior diagnostic performance compared with CSF lactate. | [38,46] |
| CSF Lactate | Markedly elevated, especially in definite TBM | >5.5 mmol/L | Supports initial diagnostic suspicion (strong biomarker). Metabolic biomarker with high sensitivity and good specificity for definite TBM. Useful as a rapid and accessible test, including point-of-care methods. | Not specific for TBM. Also elevated in bacterial meningitis, cryptococcal meningitis, cerebral ischemia, seizures, and subarachnoid hemorrhage. Lower performance in probable/possible TBM. | [16,38,43] |
| CSF proteins | Elevated (>1 g/L, frequently 1–3 g/L) | >1 g/L (Supports diagnostic orientation) | Supports diagnosis in subacute/chronic meningitis. Reinforces suspicion of TBM versus viral meningitis. Associated with higher risk of complications (hydrocephalus, vasculitis) | Elevation is not exclusive. Increased in other etiologies such as fungal meningitis, bacterial meningitis, meningeal carcinomatosis, and neurosarcoidosis. | [27,46] |
| Cytochemical parameters | |||||
| Lymphocytic pleocytosis | 10–500 cells/µL; >50% lymphocytes | >10 cells/µL with lymphocytic predominance (supports diagnostic orientation) | Supports diagnosis in subacute/chronic meningitis. | Overlap with viral and fungal meningitis. May be neutrophilic or mild in early TBM or in PLWH. Low standalone discriminative power. | [27,46] |
| Erythrocyte count | Generally low. May be mildly elevated in complicated TBM | >6.5 erythrocytes/µL (cohort- specific) | Auxiliary finding/indicator of complexity. May reflect meningeal vascular involvement. | Poor discriminative utility. Low specificity; influenced by traumatic lumbar puncture and other causes of bleeding. No universal cut-off. | [46] |
| Enzymatic biomarkers | |||||
| ADA | Higher CSF ADA activity in TBM compared with non-TB meningitis (relative increase). | 5–10 U/L (cut-offs used across different studies; no standardized threshold). | Complementary enzymatic biomarker Good overall performance. Accessible and available. | No universal cut-off; performance influenced by immune status (e.g., PLWH) and coinfections. | [39,40,41,42] |
| Immunological biomarkers | |||||
| FN-γ | Higher CSF IFN-γ levels compared with non-TB meningitis (relative increase) | Not standardized (SMD 0.45–1.73 across etiological comparisons). | Complementary use. Good discriminatory performance versus cryptococcal, viral, and bacterial meningitis; supports inclusion in multimarker diagnostic panels. | No validated cut-off; high inter-study heterogeneity and assay-dependent variability. | [42,45] |
| VEGF-A | Higher VEGF-A concentrations in CSF and plasma compared with non-TB meningitis (relative increase). | Not standardized. | Complementary use. Moderately consistent evidence; improves diagnostic performance when incorporated into multimarker models, particularly in combination with inflammatory cytokines. | No validated clinical cut-off; evidence derived mainly from selected cohorts and exploratory analyses; limited external validation. | [42] |
| TB Ag/PHA ratio (derived from T-SPOT) | Higher TB antigen to phytohaemagglutinin (TB Ag/PHA) ratio in TBM compared with bacterial meningitis (relative increase). | Not standardized. | Complementary use. Supports differential diagnosis between TBM and bacterial meningitis in adults when IGRA-based testing is available. | Requires availability of IGRA (T-SPOT). Not specifically validated for TBM. Limited utility in PLWH due to impaired T-cell responses. | [42] |
| Immunological biomarkers | |||||
| VEGF-A + IFN-γ ± MPO | Concurrent relative increase in multiple host-response biomarkers, reflecting combined inflammatory and vascular activation. | Not defined. | Complementary use. Improves diagnostic discrimination between TBM and bacterial meningitis when incorporated into multivariable models, as reflected by increased AUC. | Limited external validation; lack of standardized analytical platforms and reproducible cut-off values restrict clinical implementation. | [42] |
| Pathogen-derived markers | |||||
| LAM (FujiLAM > AlereLAM) | Positive in TBM, particularly in people living with HIV | Qualitative | Complementary rule-in test High specificity. Useful as a confirmatory assay in PLWH. | Low and variable sensitivity. Limited performance in HIV-negative individuals. | [16,44] |
2.5. Conventional Microbiological Methods: Scope and Limitations
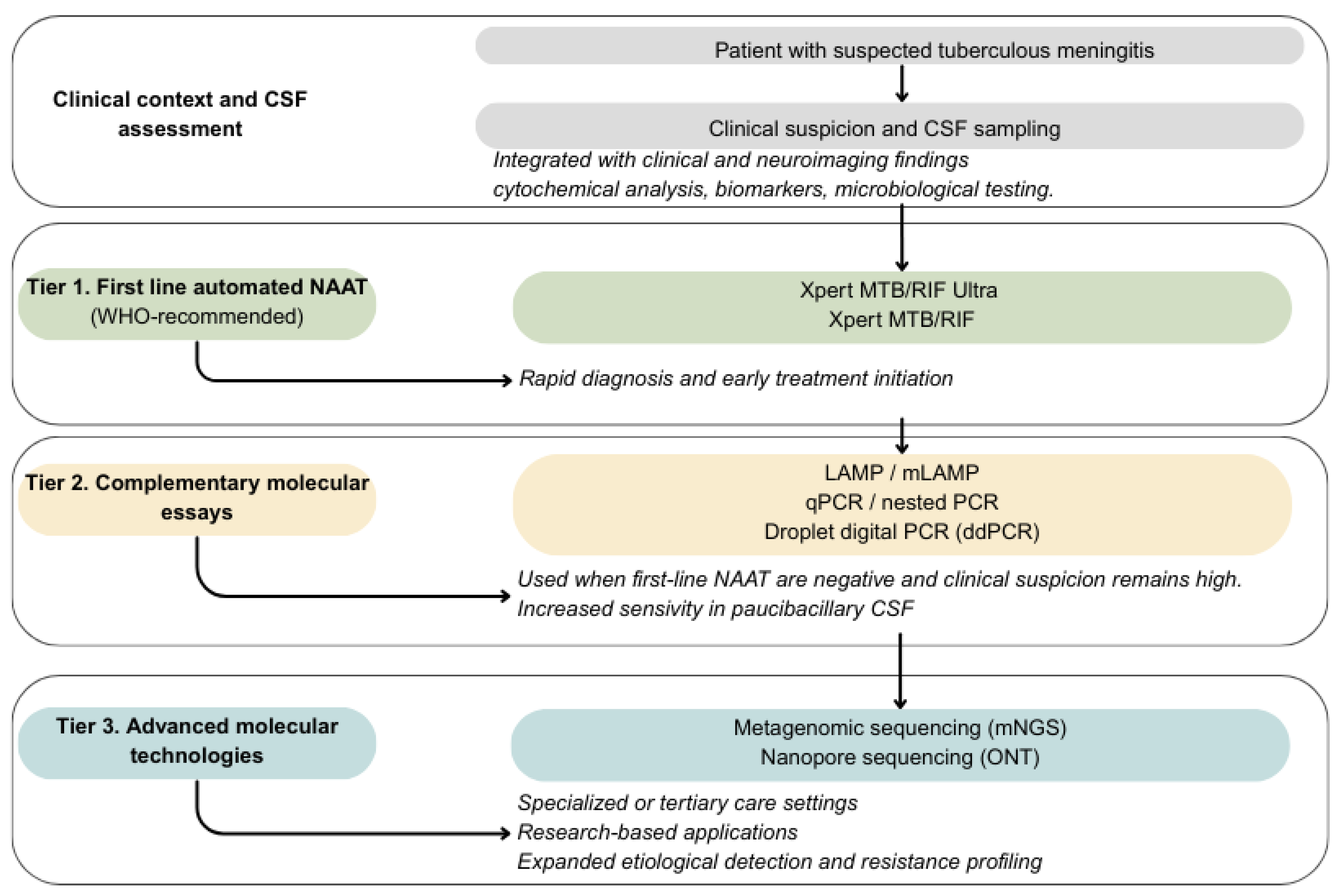
3. Molecular Diagnosis: Emerging Technologies, Clinical Application, and Perspectives
3.1. First-Line Molecular Assays (Automated NAAT)
3.2. Complementary and Emerging Molecular Assays
| Test Characteristics and Performance | Advantages/ Disadvantages | Level of Recommendation | Key Observations | References |
|---|---|---|---|---|
| Xpert MTB/RIF Cartridge-based PCR Target: rpoB Sens: 13–100% Spec: (≥98%). | Advantages: Automated, rapid (~2 h), detects rifampicin (RIF) resistance; high specificity. Disadvantages: Highly variable sensitivity in probable/possible TBM; requires basic infrastructure and continuous power supply; cartridge cost. | First-line test (WHO/IDSA) as the initial NAAT for TBM; a negative result does not exclude TBM. |
| [40,47,51,53,54,56,57,59,60,64,69] |
| Xpert MTB/RIF Ultra Multicopy PCR Target: IS6110/IS1081 Sens: ~35–88% Spec: 91–100% | Advantages: Higher sensitivity in paucibacillary settings; useful in pediatric populations; high specificity; detects RIF resistance. Disadvantages: Reduced sensitivity in “possible” TBM categories; trace results require careful interpretation; same platform and consumable requirements as Xpert MTB/RIF. | Preferred initial test for TBM across all groups (including PLWH and children); recommended by WHO as the NAAT of choice. |
| [56,58,61,62,63,64] |
| TrueNat MTB/MTB Plus Portable PCR Target: nrdB Sens: 83.75% Espc: 88.75% | Advantages: Portable; low infrastructure requirements. Disadvantages: Limited evidence in CSF; lack of robust validation against culture or composite reference standards (CRS). | Under evaluation; potential alternative in resource-limited settings where Xpert/Ultra are unavailable; does not replace first-line NAAT. |
| [25,70] |
| Alerta MTB qPCR (MTB Q-PCR) Multicopy insertion sequence Target: IS6110 Sens: ~81% Spec: NR | Advantages: High sensitivity in people living with HIV; performance superior to several NAATs on the same cohort; useful when bacillary load is high. Disadvantages: Specificity not reported; evaluated only against another NAAT (MTBDRplus); limited availability; not automated. | Second-line and complementary test in PLWH, within sequential diagnostic algorithms; not intended as a standalone assay. |
| [47] |
| PCR CapitalBio (RT-PCR) Conventional PCR Target: IS6110 Sens: 48% Spec: 100% | Advantages: Useful for TBM confirmation (especially in PLWH); short turnaround time (~3 h); technology available in many reference laboratories. Disadvantages: Intermediate sensitivity, particularly in paucibacillary cases; requires molecular infrastructure and trained personnel; slower than cartridge-based assays | Second-line/confirmatory test when Xpert/Ultra are unavailable or when additional testing is required. |
| [69] |
| ddPCR (IS6110-ddPCR) Digital PCR Target: IS6110–gyrB Sens: 25–73% Espc: 97% | Advantages: Very high analytical sensitivity compared with conventional qPCR; high specificity in paucibacillary settings. Disadvantages: Costly and complex; requires specialized equipment and basic bioinformatics; limited availability. | Advanced second-line test. Use in reference centers when automated NAATs are negative and clinical suspicion persists. |
| [71] |
| LAMP (TB-LAMP) Amplificación isotérmica Target: IS6110, sdaA Sens: ~53–70% Spec: 66–78% | Advantages: Rapid technique (<1 h); does not require a thermocycler; feasible in resource-limited settings. Disadvantages: Lower specificity compared with Xpert; higher risk of post-amplification contamination; performance highly dependent on reference standard and CSF volume. | Alternative in very resource-limited settings, such as remote or rural areas when cartridge-based NAATs are unavailable. |
| [47,67] |
| MLAMP multitarget Isothermal amplification Target: IS6110, IS1081, sdaA Sens: ≥88% Spec: 100% | Advantages: Sensitivity comparable to Xpert Ultra in some series; useful in PLWH (~80%); short processing time. Disadvantages: Does not detect drug resistance; limited evidence; risk of contamination. | Promising/under evaluation as a high-performance tool in resource-limited settings; additional validation required. |
| [63] |
| GenoType MTBDRplus Reverse hybridization (line probe assay) Target: rpoB, katG, inhA Sens: 76% (PLWH with suspected TBM) Spec: NR | Advantages: Detects MTB and resistance mutations to rifampicin (RIF) and isoniazid (INH). Disadvantages: Requires specialized infrastructure and trained personnel; turnaround time 2–8 h; limited sensitivity in CSF with low bacillary load. | Complementary test when Xpert/Ultra are negative and clinical suspicion persists. |
| [47] |
| ProbeTec ET (SDA) Strand displacement amplification Target: IS6110. Sens: 50% Spec: 100% | Advantages: High specificity in CSF; short analysis time (<3 h); lower technical complexity than conventional PCR. Disadvantages: Low sensitivity in CSF; not optimized for CSF samples; inferior performance compared with automated NAATs. | Restricted use/legacy platform in laboratories with existing infrastructure; not recommended as a new investment. |
| [55] |
| Metagenomic next-generation sequencing (mNGS) Short-read sequencing Sens: 61–62% Definitive: 70–85%. Probable/possible: 7–63% Spec: ≥98% | Advantages: Broad “pan-pathogen” detection; identifies M. tuberculosis and alternative pathogens; high specificity in paucibacillary CSF. Disadvantages: High cost; requires sequencing platforms, bioinformatics expertise, and strict contamination control. | Advanced/tertiary diagnostic tool mainly for research or specialized reference centers. |
| [68,72,73,74,75] |
| ONT Real-time long-read sequencing Sens: ~78% Spec: 100% | Advantages: Real-time results (<6 h); increased specificity; enables strain typing and resistance profiling; higher sensitivity than culture and Xpert in some series. Disadvantages: Platform still under development; limited standardization; requires specialized infrastructure and expertise; high cost. | Advanced/research-based tool. Use restricted to specialized or research centers. |
| [77] |
3.3. Clinical Integration of Molecular Assays and Diagnostic Perspectives
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Tuberculosis Report 2023; World Health Organization: Geneva, Switzerland, 2023; Available online: https://www.who.int/teams/global-programme-on-tuberculosis-and-lung-health/tb-reports/global-tuberculosis-report-2023 (accessed on 15 December 2025).
- Donovan, J.; Cresswell, F.V.; Tucker, E.W.; Angharad, G.; Rohlwink, U.K.; Huynh, J.; Solomons, R.; James, A.; Bahr, N.C.; Van Laarhoven, A.; et al. A clinical practice guideline for tuberculous meningitis. Lancet Infect. Dis. 2026, 26, E96–E111. [Google Scholar] [CrossRef]
- (OPS) Organización Panamericana de la Salud. Tuberculosis en las Américas. Informe Regional 2020. Tuberculosis en las Américas. Informe Regional 2020. Washington, DC, USA. 2022. Available online: https://iris.paho.org/items/3fb890a5-79a4-47b4-9496-54a3f7457246 (accessed on 15 December 2025).
- Bailey, T.; Philips, J. Tuberculosis. In Tratado de Medicina Interna, 27th ed.; Goldman Lee, M., Cooney, K., Eds.; Elsevier: Barcelona Spain, 2024; pp. 2031–2044. Available online: https://www.clinicalkey.es/ (accessed on 15 December 2025).
- Foppiano Palacios, C.; Saleeb, P.G. Challenges in the diagnosis of tuberculous meningitis. J. Clin. Tuberc. Other Mycobact. Dis. 2020, 20, 100164. [Google Scholar] [CrossRef]
- Khatiwada, A.; Sharada, K.C.; Jha, A.; Singh, S.S. A case of tubercular meningoencephalitis with transverse myelitis. Radiol. Case Rep. 2024, 19, 3555–3560. [Google Scholar] [CrossRef]
- Lourens, R.; Singh, G.; Arendse, T.; Thwaites, G.; Rohlwink, U. Tuberculous Meningitis Across the Lifespan. J. Infect. Dis. 2025, 231, 1101–1111. [Google Scholar] [CrossRef]
- Dodd, P.J.; Osman, M.; Cresswell, F.V.; Stadelman, A.M.; Lan, N.H.; Thuong, N.T.T.; Muzyamba, M.; Glaser, L.; Dlamini, S.S.; Seddon, J.A. The global burden of tuberculous meningitis in adults: A modelling study. PLoS Glob. Public Health 2021, 1, e0000069. [Google Scholar] [CrossRef]
- Oo, N.; Agrawal, D.K. Management Strategies of Tuberculous Meningitis. Arch. Intern. Med. Res. 2025, 8, 48–58. [Google Scholar] [CrossRef]
- Preez, K.; Jenkins, H.E.; Martinez, L.; Chiang, S.S.; Dlamini, S.S.; Kobe, J.; Graham, S.M.; Starke, J.R.; Seddon, J.A. Global burden of tuberculous meningitis in children aged 0–14 years in 2019: A mathematical modelling study. Lancet Glob. Health 2025, 13, e59–e68. [Google Scholar]
- Chen, X.; Wei, J.; Zhang, M.; Su, B.; Ren, M.; Cai, M.; Zhang, Y.; Zhang, T. Prevalence, incidence, and case fatality of tuberculous meningitis in adults living with HIV: A systematic review and meta-analysis. BMC Public Health 2024, 24, 2145. [Google Scholar] [CrossRef] [PubMed]
- Stadelman, A.M.; Ellis, J.; Samuels, T.H.A.; Mutengesa, E.; Dobbin, J.; Ssebambulidde, K.; Rutakingirwa, M.K.; Tugume, L.; Boulware, D.R.; Grint, D.; et al. Treatment outcomes in adult tuberculous meningitis: A systematic review and meta-analysis. Open Forum Infect. Dis. 2020, 7, ofaa257. [Google Scholar] [CrossRef]
- Igbokwe, V.; Ruby, L.C.; Sultanli, A.; Bélard, S. Post-tuberculosis sequelae in children and adolescents: A systematic review. Lancet Infect. Dis. 2023, 23, e138–e150. [Google Scholar] [CrossRef]
- Ngo, C.C.; Katoh, S.; Hasebe, F.; Dhoubhadel, B.G.; Hiraoka, T.; Hamaguchi, S.; Le, A.T.K.; Nguyen, A.T.H.; Dang, A.D.; Smith, C.; et al. Characteristics and biomarkers of patients with central nervous system infection admitted to a referral hospital in Northern Vietnam. Trop. Med. Health 2021, 49, 42. [Google Scholar] [CrossRef]
- Rindi, L. Rapid Molecular Diagnosis of Extra-Pulmonary Tuberculosis by Xpert/RIF Ultra. Front. Microbiol. 2022, 13, 817661. [Google Scholar] [CrossRef]
- Lu, W.; Li, Y.; Deng, J.; Su, Y.; Fan, H.; Huang, K. Advancements in the identification and utilization of cerebrospinal fluid immunological biomarkers for the diagnosis of tuberculous meningitis. Microb. Pathog. 2025, 206, 107826. [Google Scholar] [CrossRef]
- Cao, W.F.; Leng, E.L.; Liu, S.M.; Zhou, Y.L.; Luo, C.Q.; Xiang, Z.B.; Cai, W.; Rao, W.; Hu, F.; Zhang, P.; et al. Recent advances in microbiological and molecular biological detection techniques of tuberculous meningitis. Front. Microbiol. 2023, 14, 1202752. [Google Scholar] [CrossRef]
- Swinkels, H.M.; Jilani, T.N.; Tobin, E.H. Tuberculosis Prevention, Control, and Elimination; StatPearls: Tampa, FL, USA, 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK513246/ (accessed on 15 December 2025).
- Elbehiry, A.; Marzouk, E.; Edrees, H.M.; AlShaqi, R.; Ellethy, A.T.; Alzaben, F.; Anagreyyah, S.; Algarni, A.; Almuhaydili, K.; Alotaibi, I.; et al. Advancing the fight against tuberculosis: Integrating innovation and public health in diagnosis, treatment, vaccine development, and implementation science. Front. Med. 2025, 12, 1596579. [Google Scholar] [CrossRef]
- Ssebambulidde, K.; Gakuru, J.; Ellis, J.; Cresswell, F.V.; Bahr, N.C. Improving Technology to Diagnose Tuberculous Meningitis: Are We There Yet? Front. Neurol. 2022, 13, 892224. [Google Scholar]
- Thwaites, G.E.; Chau, T.T.H.; Stepniewska, K.; Phu, N.H.; Chuong, L.V.; Sinh, D.X.; White, N.; Parry, C.; Farrar, J. Diagnosis of adult tuberculous meningitis by use of clinical and laboratory features. Lancet 2002, 360, 1287–1292. [Google Scholar] [CrossRef]
- Lewinsohn, D.M.; Leonard, M.K.; Lobue, P.A.; Cohn, D.L.; Daley, C.L.; Desmond, E.; Keane, J.; Lewinsohn, D.A.; Loeffler, A.M.; Mazurek, G.H.; et al. Official American Thoracic Society/Infectious Diseases Society of America/Centers for Disease Control and Prevention Clinical Practice Guidelines: Diagnosis of Tuberculosis in Adults and Children. Clin. Infect. Dis. 2017, 64, e1–e33. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Guidelines on Meningitis Diagnosis, Treatment and Care. 2025. Available online: https://www.who.int/publications/i/item/9789240108042 (accessed on 15 December 2025).
- Thwaites, G.; Fisher, M.; Hemingway, C.; Scott, G.; Solomon, T.; Innes, J. British Infection Society guidelines for the diagnosis and treatment of tuberculosis of the central nervous system in adults and children. J. Infect. 2009, 59, 167–187. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Consolidated Guidelines on Tuberculosis Module 3: Diagnosis; WHO Press: Geneva, Switzerland, 2025; pp. 1–184. [Google Scholar]
- Takwoingi, Y.; Whitworth, H.; Rees-Roberts, M.; Badhan, A.; Partlett, C.; Green, N.; Boakye, A.; Lambie, H.; Marongiu, L.; Jit, M.; et al. Interferon gamma release assays for Diagnostic Evaluation of Active tuberculosis (IDEA): Test accuracy study and economic evaluation. Health Technol. Assess 2019, 23, 1–152. [Google Scholar]
- Marais, S.; Thwaites, G.; Schoeman, J.F.; Török, M.E.; Misra, U.K.; Prasad, K.; Donald, P.R.; Wilkinson, R.J.; Marais, B.J. Tuberculous meningitis: A uniform case definition for use in clinical research. Lancet Infect. Dis. 2010, 10, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Daniel, B.D.; Selladurai, E.; Balaji, S.; Venkatesan, A.; Venkatesan, M.; Giridharan, P.; Shanmugam, S.; Natrajan, S.; Karunaianantham, R.; Kandasamy, D.; et al. Clinical and diagnostic features of central nervous system tuberculosis in Indian children—A descriptive study. Ther. Adv. Infect. Dis. 2024, 11, 20499361241274251. [Google Scholar] [CrossRef]
- Ma, Q.; Yi, Y.; Liu, T.; Wen, X.; Shan, F.; Feng, F.; Yan, Q.; Shen, J.; Yang, G.; Shi, Y. MRI-based radiomics signature for identification of invisible basal cisterns changes in tuberculous meningitis: A preliminary multicenter study. Eur. Radiol. 2022, 32, 8659–8669. [Google Scholar] [CrossRef]
- Lu, Y.; Ma, C.; Chen, R.; Hu, Z.; Yao, H.; Zhang, Q.; Yan, Q.; Shen, J.; Yang, G.; Shi, Y. Development and validation of a new scoring system for the early diagnosis of tuberculous meningitis in adults. Diagn. Microbiol. Infect. Dis. 2021, 101, 115393. [Google Scholar] [CrossRef]
- Sulaiman, T.; Medi, S.; Erdem, H.; Senbayrak, S.; Ozturk-Engin, D.; Inan, A.; Civljak, R.; Nechifor, M.; Akbulut, A.; Crisan, A.; et al. The diagnostic utility of the “Thwaites’ system” and “lancet consensus scoring system” in tuberculous vs. non-tuberculous subacute and chronic meningitis: Multicenter analysis of 395 adult patients. BMC Infect. Dis. 2020, 20, 20499361241274251. [Google Scholar] [CrossRef]
- Yang, Y.; Qu, X.H.; Zhang, K.N.; Wu, X.M.; Wang, X.R.; Wen, A.; Li, L.-J. A Diagnostic Formula for Discrimination of Tuberculous and Bacterial Meningitis Using Clinical and Laboratory Features. Front. Cell. Infect. Microbiol. 2020, 9, 448. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Xue, Y.; Lin, Q.; Mao, L.; Tang, G.; Song, H.; Liu, W.; Wu, S.; Liu, W.; Zhou, Y.; et al. Diagnostic Model for Discrimination Between Tuberculous Meningitis and Bacterial Meningitis. Front. Immunol. 2021, 12, 731876. [Google Scholar] [CrossRef] [PubMed]
- Wen, A.; Liu, S.M.; Cao, W.F.; Zhou, Y.L.; Luo, C.Q.; Xiang, Z.B.; Hu, F.; Zhang, P.; Leng, E.-L. A New Scoring System to Differentially Diagnose and Distinguish Tuberculous Meningitis and Bacterial Meningitis in South China. Front. Neurol. 2022, 13, 830969. [Google Scholar] [CrossRef]
- Krongsut, S.; Srithanabout, P.; Pansaksiri, S.; Na-Ek, N. A new diagnostic model for the early differentiation of tuberculous and bacterial meningitis to guide timely diagnosis and treatment: Model development and internal validation. J. Infect. Public Health 2025, 18, 102984. [Google Scholar] [CrossRef]
- Lee, S.A.; Kim, S.W.; Chang, H.H.; Jung, H.; Kim, Y.; Hwang, S.; Yang, X. New Scoring System Tb Meningitis. J. Korean Med. Sci. 2018, 33, 1–11. [Google Scholar]
- Liu, Q.; Cao, M.; Shao, N.; Qin, Y.; Liu, L.; Zhang, Q.; Yang, X. Development and validation of a new model for the early diagnosis of tuberculous meningitis in adults based on simple clinical and laboratory parameters. BMC Infect. Dis. 2023, 23, 901. [Google Scholar] [CrossRef]
- Nuwagira, E.; Huppler Hullsiek, K.; Jjunju, S.; Rutakingirwa, M.; Kasibante, J.; Tadeo, K.K.; Kagimu, E.; Tugume, L. Diagnostic and Prognostic Value of Cerebrospinal Fluid Lactate and Glucose in HIV-Associated Tuberculosis Meningitis. Microbiol. Spectr. 2022, 10, e0161822. [Google Scholar] [CrossRef] [PubMed]
- Pormohammad, A.; Riahi, S.M.; Nasiri, M.J.; Fallah, F.; Aghazadeh, M.; Doustdar, F.; Pouriran, R. Diagnostic test accuracy of adenosine deaminase for tuberculous meningitis: A systematic review and meta-analysis. J. Infect. 2017, 74, 545–554. [Google Scholar] [CrossRef]
- Manyelo, C.; Solomons, R.S.; Gerhard, W.; Chengou, N. Tuberculous Meningitis: Pathogenesis, Immune Responses, Diagnostic Challenges, and the Potential of Biomarker-Based Approaches. J. Clin. Microbiol. 2021, 59, 10–1128. [Google Scholar] [CrossRef]
- Ekermans, P.; Dusé, A.; George, J. The dubious value of cerebrospinal fluid adenosine deaminase measurement for the diagnosis of tuberculous meningitis. BMC Infect. Dis. 2017, 17, 6–8. [Google Scholar] [CrossRef]
- Isaiah, A.R.; Williams, M.; Loots, D.T.; Williams, A.A.; Chegou, N.N.; van Furth, M.T.; Mason, S. Potential biomarkers for tuberculous meningitis diagnosis using metabolomics and proteomics: A systematic review. BMC Infect. Dis. 2025, 25, 1298. [Google Scholar] [CrossRef] [PubMed]
- Poplin, V.; Boulware, D.R.; Bahr, N.C. Methods for rapid diagnosis of meningitis etiology in adults. Biomark. Med. 2020, 14, 459–479. [Google Scholar] [CrossRef] [PubMed]
- Quinn, C.M.; Kagimu, E.; Okirworth, M.; Bangdiwala, A.S.; Mugumya, G.; Ramachandran, P.S.; Wilson, M.R.; Meya, D.B.; Cresswell, F.V.; Bahr, N.C. Fujifilm SILVAMP TB LAM Assay on Cerebrospinal Fluid for the Detection of Tuberculous Meningitis in Adults with Human Immunodeficiency Virus. Clin. Infect. Dis. 2021, 73, e3428–e3434. [Google Scholar] [CrossRef]
- Saghazadeh, A.; Rezaei, N. Central Inflammatory Cytokines in Tuberculous Meningitis: A Systematic Review and Meta-analysis. J. Interf. Cytokine Res. 2022, 42, 95–107. [Google Scholar] [CrossRef]
- Hueda-Zavaleta, M.; Gómez de la Torre, J.C.; Barletta-Carrillo, C.; Tapia-Sequeiros, G.; Flores, C.; Piscoche, C.; Miranda, C.; Mendoza, A.; Sánchez-Tito, M.; Benites-Zapata, V.A. Cytochemical analysis of cerebrospinal fluid in tuberculous meningitis versus other etiologies. PLoS ONE 2025, 20, e0318398. [Google Scholar] [CrossRef]
- Mitha, M.; Pillay, M.; Moodley, J.Y.; Balakrishna, Y.; Abbai, N.; Bhagwan, S.; Dangor, Z.; Bhigjee, A.I. Laboratory diagnosis of tuberculous meningitis in human immunodeficiency virus–seropositive patients: Correlation with the uniform case definition. S. Afr. J. Infect. Dis. 2020, 35, 5. [Google Scholar] [CrossRef] [PubMed]
- Stadelman, A.M.; Ssebambulidde, K.; Buller, A.; Tugume, L.; Bakker, C.J.; Boulware, D.R.; Bahr, N.C. Cerebrospinal fluid AFB smear in adults with tuberculous meningitis: A systematic review and diagnostic test accuracy meta-analysis. Tuberculosis 2022, 135, 102230. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Zhang, W.; Xiong, Y.; Wang, Y.; Yu, Q.; Ma, Y.; Xie, Y. Comparative performance of microbiological methods for the detection of tuberculous meningitis pathogens in cerebrospinal fluid. Diagn. Microbiol. Infect. Dis. 2023, 107, 116025. [Google Scholar] [CrossRef] [PubMed]
- Lin, F. Tuberculous meningitis diagnosis and treatment: Classic approaches and high-throughput pathways. Front. Immunol. 2024, 15, 1543009. [Google Scholar] [CrossRef]
- Chen, Y.Z.; Sun, L.C.; Wen, Y.H.; Li, Z.W.; Fan, S.J.; Tan, H.K.; Qiu, M.; Pan, Z.-Y.; Li, Q.; Zhao, Y.-Z.; et al. Pooled analysis of the Xpert MTB/RIF assay for diagnosing tuberculous meningitis. Biosci. Rep. 2020, 40, BSR20191312. [Google Scholar] [CrossRef]
- Anand, R.P.; Tandon, R.; Jha, S. Clinical, Laboratory and Radiological Correlates of Xpert MTB/RIF Assay Study in Cerebrospinal Fluid in CNS Tuberculosis. Neurol. India 2024, 72, 546–552. [Google Scholar] [CrossRef]
- Winichakoon, P.; Watcharasaksilp, K.; Butphet, S.; Wongworapat, K.; Pantip, C.; Khamnoi, P.; Supparatpinyo, K.; Salee, P. Sequential testing with Xpert MTB/RIF assay for diagnosis of tuberculous meningitis in Maharaj Nakorn Chiang Mai University Hospital. Sci. Rep. 2025, 15, 3675. [Google Scholar] [CrossRef]
- Patel, S.; Dadheech, M.; Maurya, A.K.; Singh, J.; Purwar, S.; Rai, N.; Sarawagi, R.; Joshi, A.; Khadanga, S. Assessment of the Diagnostic Utility of GeneXpert Mycobacterium tuberculosis/Rifampicin (MTB/RIF) Assay in the Suspected Cases of Tuberculous Meningitis. Cureus 2023, 15, 2–9. [Google Scholar]
- Mokaddas, E.; Ahmad, S.; Eldeen, H. Performance comparison of GeneXpert MTB/RIF and ProbeTec et tests for rapid molecular diagnosis of extrapulmonary tuberculosis in a low TB/MDR-TB incidence country. Med. Princ. Pract. 2021, 30, 277–284. [Google Scholar] [CrossRef]
- Rahman, S.M.M.; Nasrin, R.; Kabir, S.; Kabir, F.; Rahman, A.M.R.; Uddin, M.K.M.; Islam, A.S.M.I.; Khatun, R.; Ahmed, S.; Alam Mondal, B.; et al. Performance of Xpert MTB/RIF ultra for the diagnosis of tuberculous meningitis in children using cerebrospinal fluid. Sci. Rep. 2025, 15, 13060. [Google Scholar] [CrossRef]
- Hernandez, A.V.; de Laurentis, L.; Souza, I.; Pessanha, M.; Thota, P.; Roman, Y.M.; Barboza-Meca, J.; Boulware, D.R.; Vidal, J.E. Diagnostic accuracy of Xpert MTB/RIF for tuberculous meningitis: Systematic review and meta-analysis. Trop. Med. Int. Health 2021, 26, 122–132. [Google Scholar] [CrossRef]
- Huang, M.; Wang, G.; Sun, Q.; Jiang, G.; Li, W.; Ding, Z.; Jia, H.; Gao, M.; Huang, H.; Li, Q. Diagnostic accuracy of Xpert MTB/RIF Ultra for tuberculous meningitis in a clinical practice setting of China. Diagn. Microbiol. Infect. Dis. 2021, 100, 122–132. [Google Scholar] [CrossRef]
- de Almeida, S.M.; Kussen, G.M.B.; Cogo, L.; Carvalho, J.H.; Nogueira, K. Diagnostic characteristics of Xpert MTB/RIF assay for the diagnosis of tuberculous meningitis and rifampicin resistance in Southern Brazil. Arq. Neuropsiquiatr. 2020, 78, 700–707. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Forsman, L.D.; Wang, S.; Wang, S.; Shao, G.; Xiong, H.; Bao, Z.; Hu, Y. The diagnostic performance of GeneXpert MTB/RIF in tuberculosis meningitis: A multicentre accuracy study. Diagn. Microbiol. Infect. Dis. 2024, 109, 116277. [Google Scholar] [PubMed]
- Shen, Y.; Yu, G.; Zhao, W.; Lang, Y. Efficacy of Xpert MTB/RIF Ultra in diagnosing tuberculosis meningitis. Medicine 2021, 100, e26778. [Google Scholar] [CrossRef]
- Pradhan, N.N.; Paradkar, M.S.; Kagal, A.; Valvi, C.; Kinikar, A.; Khwaja, S.; Dhage, R.; Chandane, J.; Ithape, M.; Bendre, M.; et al. Performance of Xpert® MTB/RIF and Xpert® Ultra for the diagnosis of tuberculous meningitis in children. Int. J. Tuberc. Lung Dis. 2022, 26, 317–325. [Google Scholar] [CrossRef]
- Yadav, B.; Sharma, M.; Singla, N.; Shree, R.; Goyal, M.; Modi, T.; Sharma, A.; Sharma, A.; Sharma, N.; Ray, P.; et al. Molecular diagnosis of Tuberculous meningitis: SdaA-based multi-targeted LAMP and GeneXpert Ultra. Tuberculosis 2023, 140, 102339. [Google Scholar] [CrossRef] [PubMed]
- Kaur, H.; Guglani, V.; Singhal, L.; Randev, S.; Kumar, P.; Gupta, V. The new Xpert Mycobacterium tuberculosis/rifampicin (MTB/Rif) Ultra assay in comparison to Xpert MTB/Rif assay for diagnosis of tuberculosis in children and adolescents. J. Trop. Pediatr. 2024, 70, fmad046. [Google Scholar] [CrossRef]
- Yang, N.; Liu, Z. Advancements and applications of loop-mediated isothermal amplification technology: A comprehensive overview. Front. Microbiol. 2024, 15, 1406632. [Google Scholar] [CrossRef]
- Mousavi-sagharchi, S.M.A.; Afrazeh, E.; Seyyedian-nikjeh, S.F. New insight in molecular detection of Mycobacterium tuberculosis. AMB Express 2024, 14, 74. [Google Scholar]
- Chauhan, V.S.; Jorwal, P.; Singh, B.K.; Nischal, N.; Soneja, M.; Vyas, S.; Vishnu, V.Y.; Sethi, P.; Ranjan, P.; Wig, N. Evaluation of Loop-Mediated Isothermal Amplification Assay in Diagnosing Tuberculous Meningitis. Cureus 2024, 16, e57490. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, R.; Liu, L.; Qi, T.; Wang, Z.; Song, W.; Tang, Y.; Sun, J.; Liu, D.; Lin, Y.; et al. Clinical usefulness of metagenomic next-generation sequencing for the diagnosis of central nervous system infection in people living with HIV. Int. J. Infect. Dis. 2021, 107, 139–144. [Google Scholar] [CrossRef]
- Sun, L.; Yao, L.; Fu, G.; Lin, L.; Zhu, E.; Huang, J. A comparison of the accuracy of the CapitalBio Mycobacterium real-time polymerase chain reaction and the Xpert MTB/RIF assay for the diagnosis of tuberculous meningitis. Int. J. Infect. Dis. 2021, 104, 92–96. [Google Scholar] [CrossRef]
- Nanda, S.; Bansal, M.K.; Singh, P.; Shrivastav, A.K.; Malav, M.K.; Prakash, C. Evaluation of the Cerebrospinal Fluid (CSF)-Truenat Assay: A Novel Chip-Based Test in the Diagnosis and Management of Tubercular Meningitis at a Tertiary Care Hospital. Cureus 2024, 16, e74522. [Google Scholar] [CrossRef]
- Li, Z.; Pan, L.; Lyu, L.; Li, J.; Jia, H.; Du, B.; Sun, Q.; Zhang, Z. Diagnostic accuracy of droplet digital PCR analysis of cerebrospinal fluid for tuberculous meningitis in adult patients. Clin. Microbiol. Infect. 2020, 26, 213–219. [Google Scholar] [CrossRef]
- Xiang, Z.B.; Leng, E.L.; Cao, W.F.; Liu, S.M.; Zhou, Y.L.; Luo, C.Q.; Hu, F.; Wen, A. A systematic review and meta-analysis of the diagnostic accuracy of metagenomic next-generation sequencing for diagnosing tuberculous meningitis. Front. Immunol. 2023, 14, 1223675. [Google Scholar] [CrossRef]
- Yu, G.; Zhao, W.; Shen, Y.; Zhu, P.; Zheng, H. Metagenomic next generation sequencing for the diagnosis of tuberculosis meningitis: A systematic review and meta-analysis. PLoS ONE 2020, 15, e0243161. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Sun, W.; Lu, Z.; Fan, L. Metagenomic Next-Generation Sequencing (mNGS) in cerebrospinal fluid for rapid diagnosis of Tuberculosis meningitis in HIV-negative population. Int. J. Infect. Dis. 2020, 96, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, Y.; Liu, X.; Li, W.; Fu, H.; Liu, X.; Zhang, X.; Zhou, X.; Yang, B.; Yao, J.; et al. Comparative diagnostic utility of metagenomic next-generation sequencing, GeneXpert, modified Ziehl–Neelsen staining, and culture using cerebrospinal fluid for tuberculous meningitis: A multi-center, retrospective study in China. J. Clin. Lab. Anal. 2022, 36, e24307. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Lu, Z.; Yan, L. Clinical efficacy of metagenomic next-generation sequencing for rapid detection of Mycobacterium tuberculosis in smear-negative extrapulmonary specimens in a high tuberculosis burden area. Int. J. Infect. Dis. 2021, 103, 91–96. [Google Scholar] [CrossRef]
- Zhou, L.; Zou, X.; Yong, Y.; Hu, Q. Using cerebrospinal fluid nanopore sequencing assay to diagnose tuberculous meningitis: A retrospective cohort study in China. BMJ Open 2024, 14, e080904. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Leiva-Ordoñez, J.E.; Quintero, B. Diagnosis of Tuberculous Meningitis: Integrating Clinical Assessment and Molecular Diagnostics. Diagnostics 2026, 16, 552. https://doi.org/10.3390/diagnostics16040552
Leiva-Ordoñez JE, Quintero B. Diagnosis of Tuberculous Meningitis: Integrating Clinical Assessment and Molecular Diagnostics. Diagnostics. 2026; 16(4):552. https://doi.org/10.3390/diagnostics16040552
Chicago/Turabian StyleLeiva-Ordoñez, Jorge E., and Beatriz Quintero. 2026. "Diagnosis of Tuberculous Meningitis: Integrating Clinical Assessment and Molecular Diagnostics" Diagnostics 16, no. 4: 552. https://doi.org/10.3390/diagnostics16040552
APA StyleLeiva-Ordoñez, J. E., & Quintero, B. (2026). Diagnosis of Tuberculous Meningitis: Integrating Clinical Assessment and Molecular Diagnostics. Diagnostics, 16(4), 552. https://doi.org/10.3390/diagnostics16040552





