Imaging Evaluation for Jaw Deformities: Diagnostic Workup and Pre-Treatment Imaging Checklist for Orthognathic Surgery
Abstract
1. Introduction
2. Materials and Methods
2.1. Objectives of Preoperative Diagnosis for Jaw Deformities
- To obtain an accurate assessment of the current condition of the jaw deformity, including the assessment of growth potential, prior to the preoperative evaluation;
- To determine an appropriate treatment plan for correction;
- To identify potential complications that may interfere with the planned procedures.
2.2. Overview of Preoperative Imaging Diagnosis for Jaw Deformities
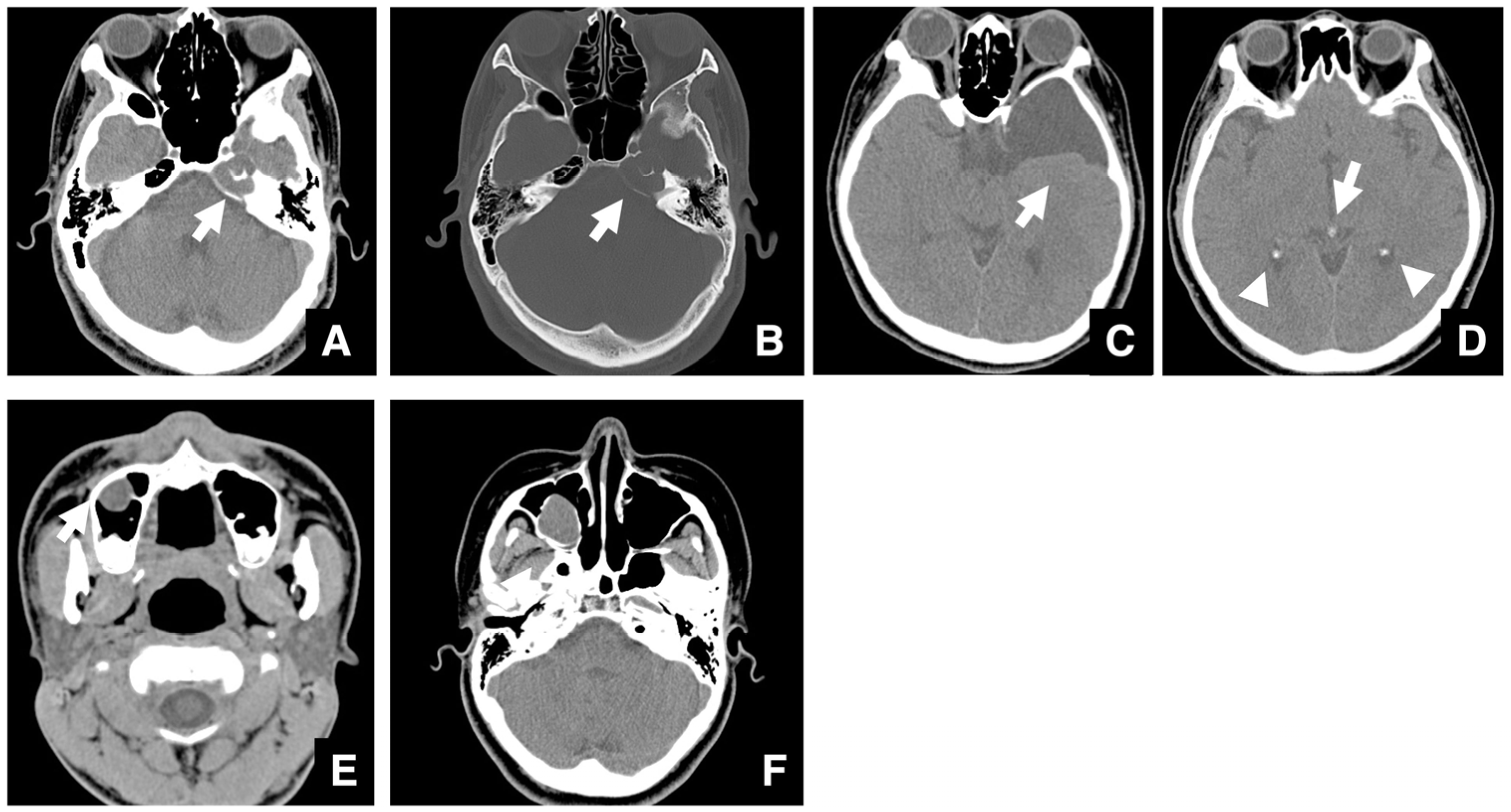
2.3. Diseases That Take Priority over the Chief Complaint in Imaging Evaluation for Jaw Deformities
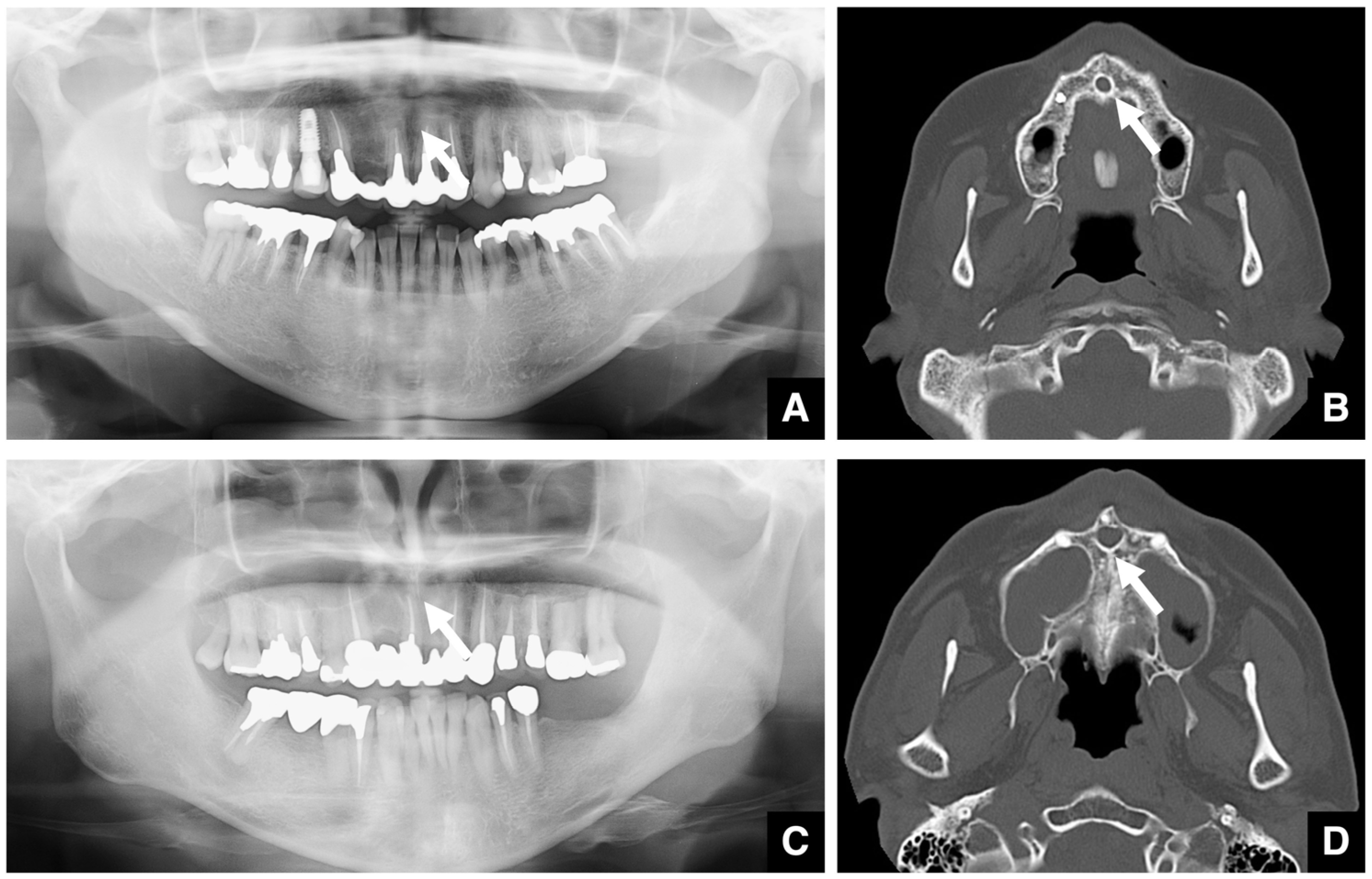
2.4. Imaging Diagnosis Focused on the Chief Complaint in Jaw Deformities
2.5. Confounding Factors/Limitations
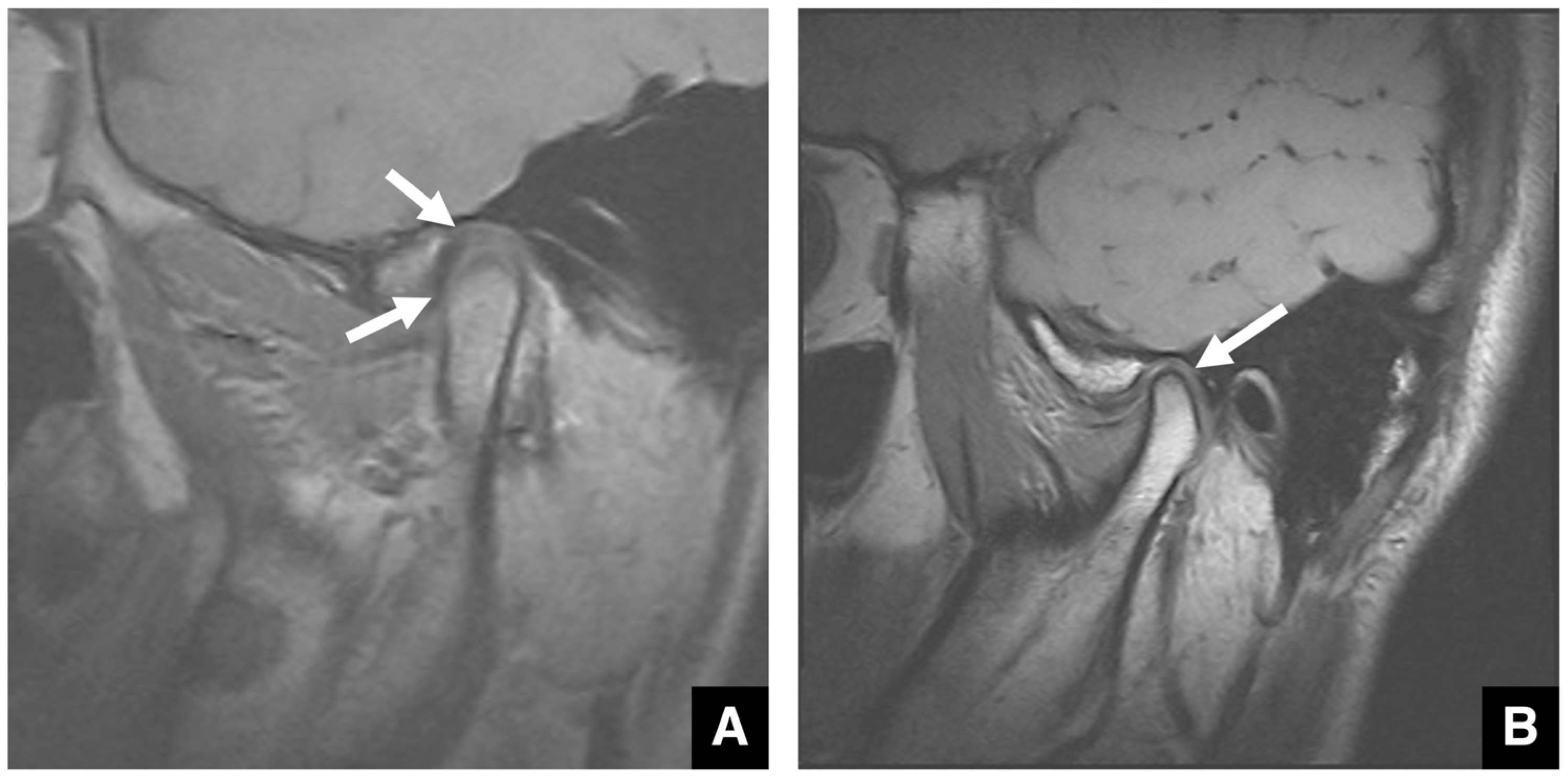
2.6. Imaging Evaluation of the TMJ in Jaw Deformities
2.7. Imaging Evaluation to Prevent Complications During Jaw Deformity Surgery
2.8. Imaging Evaluation for Genioplasty
2.9. Imaging Evaluation for Le Fort I Osteotomy
2.10. Intraoperative and Immediate Postoperative Imaging Evaluation in Jaw Deformity Surgery
2.11. Studies Related to Pre- and Post-Treatment Evaluation of Jaw Deformities
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- The Japan Society for Jaw Deformities. Basic Knowledge for Treatment of Jaw Deformities; Quintessence Publishing: Tokyo, Japan, 2022; pp. 8–9. (In Japanese) [Google Scholar]
- White, T.C.; Gardiner, J.H.; Leighton, B.C. Orthodontics for Dental Students; Macmillan Press: London, UK, 1976; pp. 58–85. [Google Scholar]
- Japanese Society of Oral and Maxillofacial Surgeons. Guidelines for the Diagnosis and Treatment of Jaw Deformities; Japanese Society of Oral and Maxillofacial Surgeons: Tokyo, Japan, 2008. (In Japanese) [Google Scholar]
- Horner, K.; Oslam, M.; Flygare, L.; Tsiklalis, K.; Whaites, E. Basic principles for use of dental cone beam computed tomography: Consensus guidelines of the European Academy of Dental and Maxillofacial Radiology. Dentomaxillofac. Radiol. 2009, 38, 187–195. [Google Scholar] [CrossRef]
- Narita, M.; Yamamoto, M.; Takaki, T.; Iwamoto, M.; Wakoh, M.; Yakushiji, T.; Katakura, A.; Takano, M.; Kamio, T. Incidental findings on preoperative head and neck MRI for orthognathic surgery in jaw deformity patients. J. Oral Maxillofac. Surg. Med. Pathol. 2024, 36, 28–33. [Google Scholar]
- Daghighi, M.H.; Rezaei, V.; Zarrintan, S.; Pourfathi, H. Intracranial physiological calcifications in adults on computed tomography in Tabriz, Iran. Folia Morphol. 2007, 66, 115–119. [Google Scholar]
- Langlais, R.P.; Langland, O.E.; Nortje, C.J. Diagnostic Imaging of the Jaws; Williams & Wilkins: Baltimore, MD, USA, 1995; pp. 266–291. [Google Scholar]
- White, S.C.; Pharoah, M.J. Oral Radiology Principles and Interpretation, 5th ed.; Mosby: Philadelphia, PA, USA, 2004; pp. 388–392. [Google Scholar]
- Ueda, N.; Tanaka, T.; Oda, M.; Wakasugi-Sato, N.; Matsumoto-Takeda, S.; Miyamura, Y.; Jyoujima, T.; Kiyota, K.; Tsutsumi, K.; Morimoto, Y. Advocacy of diagnostic criteria for maxillary incisive canal cysts based on alteration of normal maxillary incisive canals according to aging in Japanese populations. Head Face Med. 2019, 15, 25. [Google Scholar] [CrossRef] [PubMed]
- Mraiwa, N.; Jacobs, R.; van Cleynenbreugel, J.; Sanderink, G.; Schutyser, F.; Suetens, P.; van Steenberghe, D.; Quirynen, M. The nasopalatine canal revisited using 2D and 3D CT imaging. Dentomaxillofac. Radiol. 2004, 33, 396–402. [Google Scholar] [CrossRef]
- Thakur, A.R.; Burde, K.; Guttal, K.; Naikmasur, V.G. Anatomy and morphology of the nasopalatine canal using cone-beam computed tomography. Imaging Sci. Dent. 2013, 43, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.H.; Liang, H.; Benson, B.W.; Flint, D.J.; Cho, B.H. The assessment of impacted maxillary canine position with panoramic radiography and cone beam CT. Dentomaxillofac. Radiol. 2012, 41, 356–360. [Google Scholar] [CrossRef]
- Ghai, S.; Choudhury, S. Role of panoramic imaging and cone beam CT for assessment of inferior alveolar nerve exposure and subsequent paresthesia following removal of impacted mandibular third molar. J. Oral Maxillofac. Surg. 2018, 17, 242–247. [Google Scholar] [CrossRef]
- Georgalis, K.; Woods, M.G. A study of Class III treatment: Orthodontic camouflage vs orthognathic surgery. Australas. Orthod. J. 2015, 31, 138–148. [Google Scholar] [CrossRef]
- Rabie, A.-B.M.; Wong, R.W.K.; Min, G.U. Treatment in Borderline Class III Malocclusion: Orthodontic Camouflage (Extraction) Versus Orthognathic Surgery. Open Dent. J. 2008, 2, 38–48. [Google Scholar] [CrossRef]
- Profit, W.R.; Phillips, C.; Tulloch, J.F.; Medland, P.H. Surgical versus orthodontic correction of skeletal Class II malocclusion in adolescents: Effects and indications. Int. J. Adult Orthod. Orthognath. Surg. 1992, 7, 209–220. [Google Scholar]
- Lannetti, G.; Fadda, M.T.; Marianetti, T.M.; Terenzi, V.; Cassoni, A. Long-term skeletal stability after surgical correction in Class III open-bite patients: A retrospective study on 40 patients treated with mono- or bimaxillary surgery. J. Craniofac. Surg. 2007, 18, 350–354. [Google Scholar] [CrossRef] [PubMed]
- Epker, B.N.; Fish, L. Surgical–orthodontic correction of open-bite deformity. Am. J. Orthod. 1977, 71, 278–299. [Google Scholar] [PubMed]
- Burford, D.; Noar, J.H. The causes, diagnosis and treatment of anterior open bite. Dent Update 2003, 30, 235–241. [Google Scholar] [CrossRef]
- Yoshioka, I.; Tanaka, T.; Habu, M.; Oda, M.; Kodama, M.; Kokuryo, S.; Kito, S.; Wakasugi-Sato, N.; Matsumoto-Takeda, S.; Seta, Y.; et al. Criteria and limitations for selecting a sagittal split ramus osteotomy for patients with skeletal mandibular prognathism and open bite. J. Oral Med. Oral Surg. Oral Pathol. Oral Radiol. 2013, 115, 455–465. [Google Scholar] [CrossRef]
- Wolford, M.; Chemello, P.D.; Hilliard, F. Occlusal plane alteration in orthognathic surgery—Part I: Effects on function and esthetics. Am. J. Orthod. Dentofac. Orthop. 1994, 106, 304–316. [Google Scholar] [CrossRef]
- Reyneke, J.P. Surgical manipulation of the occlusal plane: New concepts in geometry. Int. J. Adult Orthod. Orthognath. Surg. 1998, 13, 307–316. [Google Scholar]
- Hajeer, M.Y.; Millett, D.T.; Ayoub, A.F.; Siebert, J.P. Applications of 3D imaging in orthodontics: Part I. J. Orthod. 2004, 31, 62–70. [Google Scholar] [CrossRef]
- Lopes, P.M.L.; Moreira, C.R.; Perrella, A.; Antunes, J.L.; Cavalcanti, M.G.P. 3-D volume rendering maxillofacial analysis of angular measurements by multislice CT. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontology 2008, 105, 224–230. [Google Scholar] [CrossRef]
- Yitschaky, O.; Redlich, M.; Abed, Y.; Faerman, M.; Casap, N.; Hiller, N. Comparison of common hard tissue cephalometric measurements between computed tomography 3D reconstruction and conventional 2D cephalometric images. Angle Orthod. 2011, 81, 11–16. [Google Scholar] [CrossRef]
- Muramatsu, A.; Nawa, H.; Kimura, M.; Yoshida, K.; Maeda, M.; Katsumata, A.; Ariji, E.; Goto, S. Reproducibility of Maxillofacial Anatomic Landmarks on 3-Dimensional Computed Tomographic Images Determined with the 95% Confidence Ellipse Method. Angle Orthod. 2008, 78, 396–402. [Google Scholar] [CrossRef]
- Fuyamada, M.; Shibata, M.; Nawa, H.; Yoshida, K.; Kise, Y.; Katsumata, A.; Ariji, E.; Goto, S. Reproducibility of maxillofacial landmark identification on three-dimensional cone-beam computed tomography images of patients with mandibular prognathism: Comparative study of a tentative method and traditional cephalometric analysis. Angle Orthod. 2014, 84, 966–973. [Google Scholar] [CrossRef] [PubMed]
- Lisboa, C.d.O.; Masterson, D.; da Motta, A.F.; Motta, A.T. Reliability and reproducibility of three-dimensional cephalometric landmarks using CBCT: A systematic review. J. Appl. Oral Sci. 2015, 23, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Li, J.T.W.; Leung, Y.Y. Digital Diagnostics and Treatment Planning for Orthognathic Surgery. Aust. Dent. J. 2025, 70, S82–S92. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, D.W.; Ryu, J.; Jung, S.; Park, H.J.; Kook, M.S. Correlation analysis of eye and lip canting correction after bimaxillary orthognathic surgery and orthodontic treatment. Maxillofac. Plast. Reconstr. Surg. 2025, 47, 39. [Google Scholar] [CrossRef]
- Hwang, J.Y.; Hwang, D.S.; Choi, B. Management of the pterygoid plate in orthognathic surgery: A narrative review. Maxillofac. Plast. Reconstr. Surg. 2025, 47, 30. [Google Scholar] [CrossRef]
- Kobravi, S.; Jafari, A.; Lotfalizadeh, M.; Azimi, A. Digital Innovations in Orthognathic Surgery: A Systematic Review of Virtual Surgical Planning, Digital Transfer, and Conventional Model Surgery. Orthod. Craniofac. Res. 2025, 28, 783–798. [Google Scholar] [CrossRef]
- Hajeer, M.Y.; Ayoub, A.F.; Millett, D.T.; Bock, M.; Siebert, J.P. Three-dimensional imaging in orthognathic surgery: The clinical application of a new method. Int. J. Adult Orthod. Orthognath. Surg. 2002, 17, 318–330. [Google Scholar]
- Hajeer, M.Y.; Mao, Z.; Millett, D.T.; Ayoub, A.F.; Siebert, J.P. A new three-dimensional method of assessing facial volumetric changes after orthognathic treatment. Cleft Palate Craniofac. J. 2005, 42, 113–120. [Google Scholar] [CrossRef]
- Jaber, S.T.; Hajeer, M.Y.; Alkhouli, K.W.; Al-Shamak, R.M.; Darwich, K.M.; Aljabban, O.; Alam, M.K.; Kara-Boulad, J.M.; Hajeer, M.Y.; Darwich, K.; et al. Evaluation of Three-Dimensional Digital Models Formulated From Direct Intra-oral Scanning of Dental Arches in Comparison With Extra-oral Scanning of Poured Dental Models in Terms of Dimensional Accuracy and Reliability. Cureus 2024, 16, e54869. [Google Scholar]
- Sinnott, P.M.; Hodges, S. An incidental dense bone island: A review of potential medical and orthodontic implications of dense bone islands and case report. J. Orthod. 2020, 47, 251–256. [Google Scholar] [CrossRef]
- Ünsal, G.; Orhan, K.; Tamimi, D. Imaging of the Temporomandibular Joint Interventions. Neuroimaging Clin. N. Am. 2025, 35, 539–552. [Google Scholar] [CrossRef]
- Lübbers, H.T.; Bornstein, M.M.; Dagassan-Berndt, D.; Filippi, A.; Suter, V.G.A.; Dula, K. Revised consensus guidelines for the use of cone-beam computed tomography/digital volume tomography. Swiss Dent. J. 2024, 134, 86–115. [Google Scholar] [CrossRef]
- Riechmann, M.; Schmidt, C.; Ahlers, M.O.; Feurer, I.; Kleinheinz, J.; Kolk, A.; Pautke, C.; Schön, A.; Teschke, M.; Toferer, A.; et al. Controversial Aspects of Diagnostics and Therapy of Idiopathic Condylar Resorption: An Analysis of Evidence- and Consensus-Based Recommendations Based on an Interdisciplinary Guideline Project. J. Clin. Med. 2023, 12, 4946. [Google Scholar] [CrossRef]
- Hamel, C.; Avard, B.; Campbell, R.; Kontolemos, M.; Murphy, A. Canadian Association of Radiologists Head and Neck Imaging Referral Guideline. Can. Assoc. Radiol. J. 2024, 75, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Kvaratskhelia, S.; Nemsadze, T. The influence of the orthodontic treatment on the development of the temporomandibular joint disorder—Literature review. Georgian Med. News 2022, 331, 22–26. [Google Scholar]
- Nunes de Lima, V.; Faverani, L.P.; Santiago, J.F., Jr.; Palmieri, C., Jr.; Magro Filho, O.; Pellizzer, E.P. Evaluation of condylar resorption rates after orthognathic surgery in class II and III dentofacial deformities: A systematic review. J. Craniomaxillofac. Surg. 2018, 46, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Hwang, K.; Nam, Y.S.; Han, S.H. Vulnerable structures during intraoral sagittal split ramus osteotomy. J. Craniofac. Surg. 2009, 20, 229–232. [Google Scholar] [CrossRef]
- Jo, H.W.; Kim, Y.S.; Kang, D.H.; Lee, S.H.; Kwon, T.G. Pseudoaneurysm of the facial artery occurred after mandibular sagittal split ramus osteotomy. J. Oral Maxillofac. Surg. 2013, 17, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Tsurushima, H.; Yoshioka, I. Imaging diagnosis for safe mandibular orthodontic surgery. Jpn. J. Oral Diag. 2023, 36, 99–105. (In Japanese) [Google Scholar] [CrossRef]
- Yoshioka, I.; Tanaka, T.; Oda, M.; Kodama, M.; Habu, M.; Kito, S.; Okabe, S.; Wakasugi-Sato, N.; Kuroiwa, H.; Tominaga, K.; et al. Usefulness of CT scan images for planning the bone cut for intra-oral vertical ramus osteotomy. J. Oral Maxillofac. Surg. 2008, 7, 245–250. [Google Scholar]
- Yoshioka, I.; Tanaka, T.; Khanal, A.; Habu, M.; Kito, S.; Kodama, M.; Oda, M.; Wakasugi-Sato, N.; Matsumoto-Takeda, S.; Fukai, Y.; et al. Relationship between inferior alveolar nerve canal positions at the mandibular second molar in patients with prognathism and the possible occurrence of neurosensory disturbance after sagittal split ramus osteotomy. J. Oral Maxillofac. Surg. 2010, 68, 3022–3027. [Google Scholar] [CrossRef]
- Yoshioka, I.; Tanaka, T.; Habu, M.; Oda, M.; Kodama, M.; Kito, S.; Seta, Y.; Tominaga, K.; Sakoda, S.; Morimoto, Y. Effect of bone quality and position of the inferior alveolar nerve canal in continuous, long-term, neurosensory disturbance after sagittal split ramus osteotomy. J. Craniomaxillofac. Surg. 2012, 40, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.P.; Murtadha, L.; Brown, J. Bifurcation of the inferior dental nerve canal: An anatomical study. Br. J. Oral Maxillofac. Surg. 2018, 56, 267–271. [Google Scholar] [CrossRef] [PubMed]
- von Arx, T.; Bornstein, M.M. The bifid mandibular canal in three-dimensional radiography: Morphologic and quantitative characteristics. Swiss Dent. J. 2021, 131, 10–28. [Google Scholar] [CrossRef]
- Aung, N.M.; Myint, K.K. Bifid mandibular canal: A proportional meta-analysis of computed tomography studies. Int. J. Dent. 2023, 2023, 9939076. [Google Scholar] [CrossRef]
- Yoshioka, I.; Tanaka, T.; Khanal, A.; Habu, M.; Kito, S.; Kodama, M.; Oda, M.; Wakasugi-Sato, N.; Matsumoto-Takeda, S.; Seta, Y.; et al. Correlation of mandibular bone quality with neurosensory disturbance after sagittal split ramus osteotomy. Br. J. Oral Maxillofac. Surg. 2011, 49, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, A.; Narita, M.; Muramatsu, K.; Shibui, T.; Takano, M.; Takagi, T.; Ohata, H.; Takano, N. Our team’s management of the sagittal splitting method of the mandibular ramus. Jpn. J. Jaw Deform. 2016, 26, 1–8. (In Japanese) [Google Scholar]
- Balaban, E.; Köse, T.E.; Günaçar, D.N.; Naralan, M.E.; Gonca, M. Comparison of methods for detecting mandibular lingula and can antilingula be used in lingula mandibula detection? BMC Oral Health 2025, 25, 430. [Google Scholar] [CrossRef]
- Chen, H.S.; Hsu, K.J.; Chen, P.J.; Lee, H.N.; Chen, J.H.; Chen, C.M. Investigating the relation among the needle insertion plane, occlusal plane, mandibular foramen, and mandibular lingula for inferior alveolar nerve block. J. Dent. Sci. 2025, 20, 1792–1801. [Google Scholar] [CrossRef]
- Kuroyanagi, N.; Miyachi, H.; Kanazawa, T.; Kamiya, N.; Nagao, T.; Shimozato, K. Morphologic features of the mandibular ramus associated with increased surgical time and blood loss in sagittal split-ramus osteotomy. J. Oral Maxillofac. Surg. 2013, 7, e31–e41. [Google Scholar] [CrossRef]
- Choi, B.K.; Lee, S.S.; Yun, L.S.; Yang, E.J. Vascular anatomy for the prevention of sublingual hematomas: Life-threatening complication of genioplasty. J. Craniofac. Surg. 2023, 34, 1308–1311. [Google Scholar] [CrossRef] [PubMed]
- Oth, O.; Orellana, M.F.; Glineur, R. The minimally invasive guided genioplasty technique using piezosurgery and 3D printed surgical guide: An innovative technique. Ann. Maxillofac. Surg. 2020, 10, 178–181. [Google Scholar] [CrossRef]
- Katsumi, Y.; Takagi, R.; Ohshima, H. Variations in the venous supply of the floor of the oral cavity: Assessment of relative hemorrhage risk during surgery. Clin. Anat. 2021, 34, 1087–1094. [Google Scholar] [CrossRef]
- Ritter, E.F.; Moelleken, B.R.; Mathes, S.J.; Ousterhout, D.K. The course of the inferior alveolar neurovascular canal in relation to sliding genioplasty. J. Craniofac. Surg. 1992, 3, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Hwang, K.; Lee, W.J.; Song, Y.B.; Chung, I.H. Vulnerability of the inferior alveolar nerve and mental nerve during genioplasty: An anatomic study. J. Craniofac. Surg. 2005, 16, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Suo, N.; Tian, X.; Li, F.; Zhong, G.; Liu, X.; Bao, Y.; Song, T.; Tian, H. Anatomic study on mental canal and incisive nerve canal in interforaminal region in Chinese population. Surg. Radiol. Anat. 2015, 37, 585–589. [Google Scholar] [CrossRef]
- Avelar, R.L.; Sá, C.D.; Esses, D.F.; Becker, O.E.; Soares, E.C.; de Oliveira, R.B. Unusual complication after genioplasty. J. Craniofacial Surg. 2014, 25, e180–e182. [Google Scholar] [CrossRef]
- Ito, Y.; Kojima, T.; Maeda, T.; Fujii, M. Iatrogenic arteriovenous fistula between the middle meningeal artery and pterygoid venous plexus secondary to a Le Fort I osteotomy: A case report. NMC Case Rep. J. 2023, 10, 299–302. [Google Scholar] [CrossRef]
- Nakajima, M. Standard surgical technique for jaw deformity-Le Fort Ⅰ osteotomy-. Jpn. J. Oral Maxillofac. Surg. 2019, 58, 473–479. (In Japanese) [Google Scholar] [CrossRef]
- Chung, S.W.; Park, K.R.; Jung, Y.S.; Park, H.S. Fracture of the clivus as an unusual complication of LeFort I osteotomy: Case report. Br. J. Oral Maxillofac. Surg. 2014, 52, 467–469. [Google Scholar] [CrossRef]
- Gaspar, C.; Santos, R.B.; Rodrigues, M.; Zenha, H.; Costa, H. A new pattern of cranial nerve injuries caused by unfavorable fractures during Le Fort I osteotomy. J. Stomatol. Oral Maxillofac. Surg. 2022, 123, e140–e144. [Google Scholar] [CrossRef]
- Eshghpour, M.; Mianbandi, V.; Samieirad, S. Intra- and postoperative complications of Le Fort I maxillary osteotomy. J. Craniofac. Surg. 2018, 29, e797–e803. [Google Scholar] [CrossRef]
- Niazi, M.H.; El-Ghanem, M.; Al-Mufti, F.; Wajswol, E.; Dodson, V.; Abdulrazzaq, A.; Sami, T.; Nuoman, R.; Aziz, S.; Gandhi, C.D. Endovascular management of epistaxis secondary to dissecting pseudoaneurysm of the descending palatine artery following orthognathic surgery. J. Vasc. Interv. Neurol. 2018, 10, 41–46. [Google Scholar] [PubMed]
- Ulker, O.; Demirbas, A.E.; Kutuk, N.; Kilic, E.; Alkan, A. Vascular complications in Le Fort I osteotomy: Incidence, reasons, and management of the intraoperative hemorrhage. J. Craniofac. Surg. 2021, 32, 325–328. [Google Scholar] [CrossRef] [PubMed]
- Yun, Y.; Kurasawa, S.; Morita, M.; Kawachi, R.; Murata, H.; Sawada, S.; Kojima, Y.; Yagi, M.; Asako, M.; Iwai, H. Postoperative hemorrhage after Le Fort I osteotomy hemostasis with angiographic embolization: Report of two cases. J. Surg. Case Rep. 2023, 12, rjad663. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Filho, V.A.; Gabrielli, M.F.; Gabrielli, M.A.; Pinto, F.A.; Rodrigues-Junior, A.L.; Klüppel, L.E.; Passeri, L.A. Incidence of maxillary sinusitis following Le Fort I osteotomy: Clinical, radiographic, and endoscopic study. J. Oral Maxillofac. Surg. 2011, 69, 346–351. [Google Scholar] [CrossRef]
- Joujima, T.; Oda, M.; Sasaguri, M.; Habu, M.; Kataoka, S.; Miyamura, Y.; Wakasui-Sato, N.; Matsumoto-Takeda, S.; Takahashi, O.; Kokuryo, S.; et al. Evaluations of velopharyngeal function using high-speed cine-MRI based on T2-weighted sequences: A preliminary study. Int. J. Oral Maxillofac. Surg. 2020, 49, 432–441. [Google Scholar] [CrossRef]
- Morimoto, Y.; Tominaga, K.; Konoo, T.; Tanaka, T.; Ohba, T. Detection and significance of the characteristic magnetic resonance signals of mandibular condyles in children. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2004, 97, 269–275. [Google Scholar] [CrossRef]
- Morimoto, Y.; Tominaga, K.; Konoo, T.; Tanaka, T.; Yamaguchi, K.; Fukuda, J.; Ohba, T. Alternation of the magnetic resonance signals characteristic of mandibular condyles during growth. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2004, 98, 348–354. [Google Scholar]
- Morimoto, Y.; Konoo, T.; Tominaga, K.; Tanaka, T.; Yamaguchi, K.; Fukuda, J.; Ohba, T. Relationship between cortical bone formation on mandibular condyles and alternation of the magnetic resonance signals characteristic during growth. Am. J. Orthod. Dentofac. Orthop. 2007, 131, 473–480. [Google Scholar] [CrossRef] [PubMed]



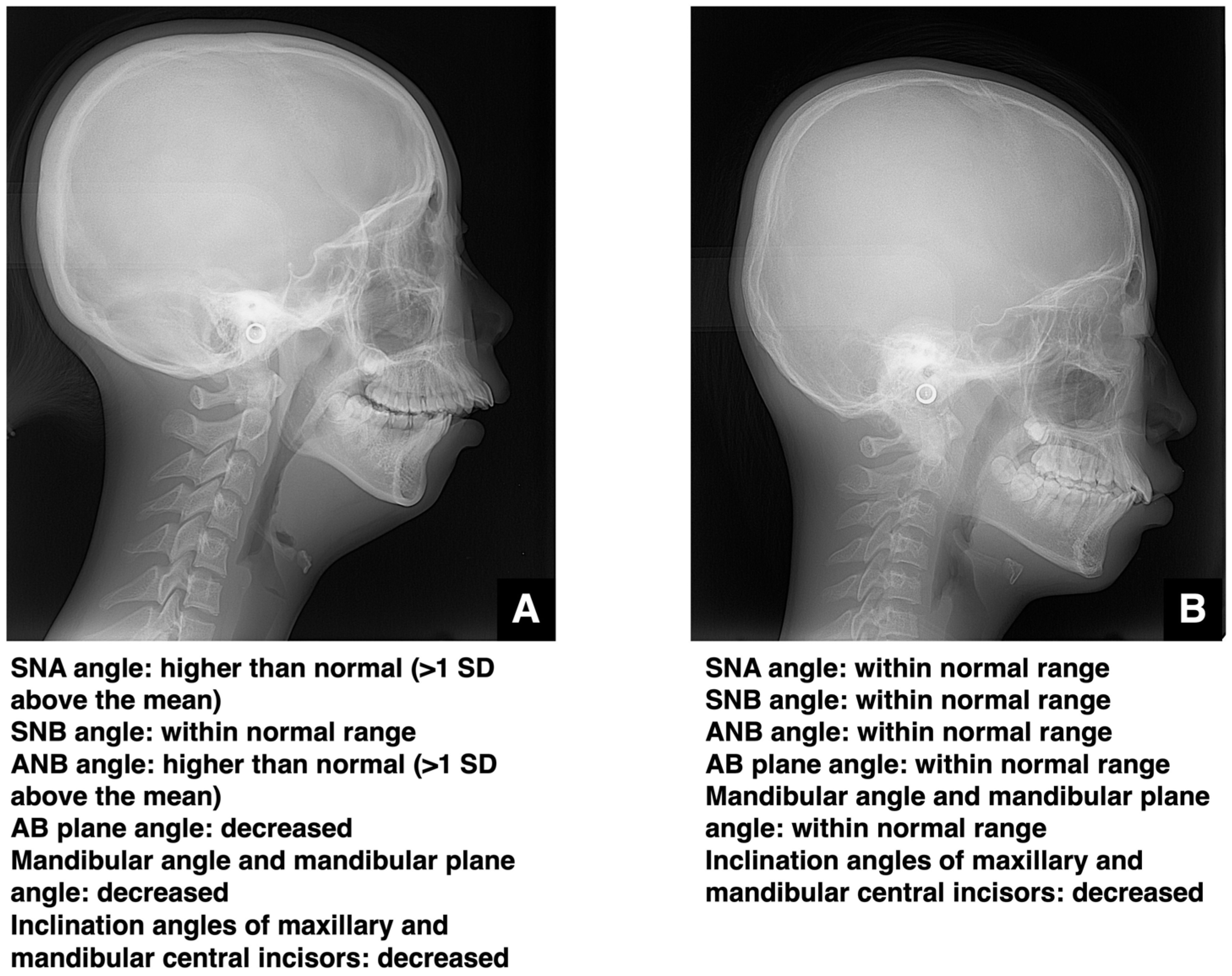
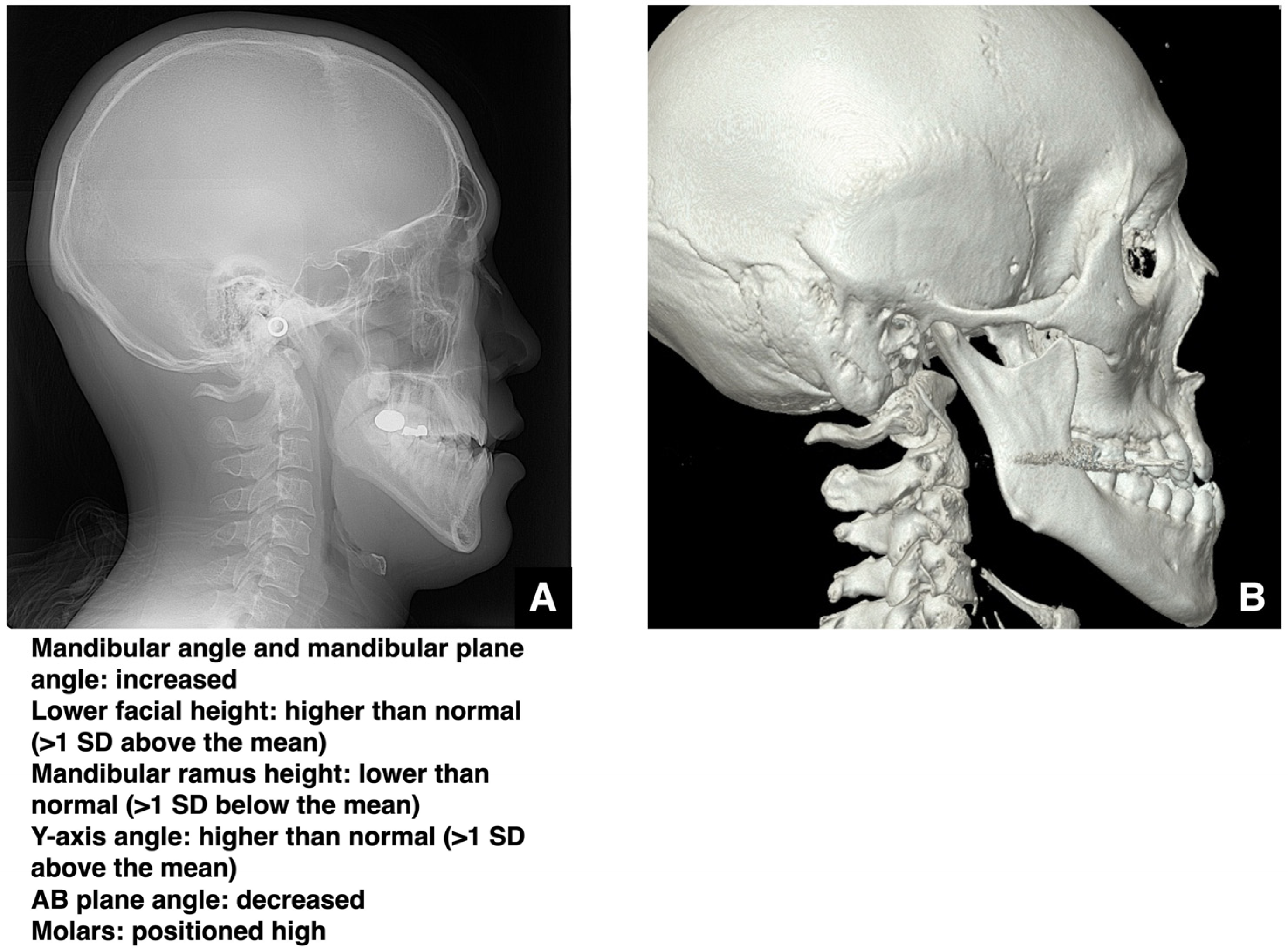


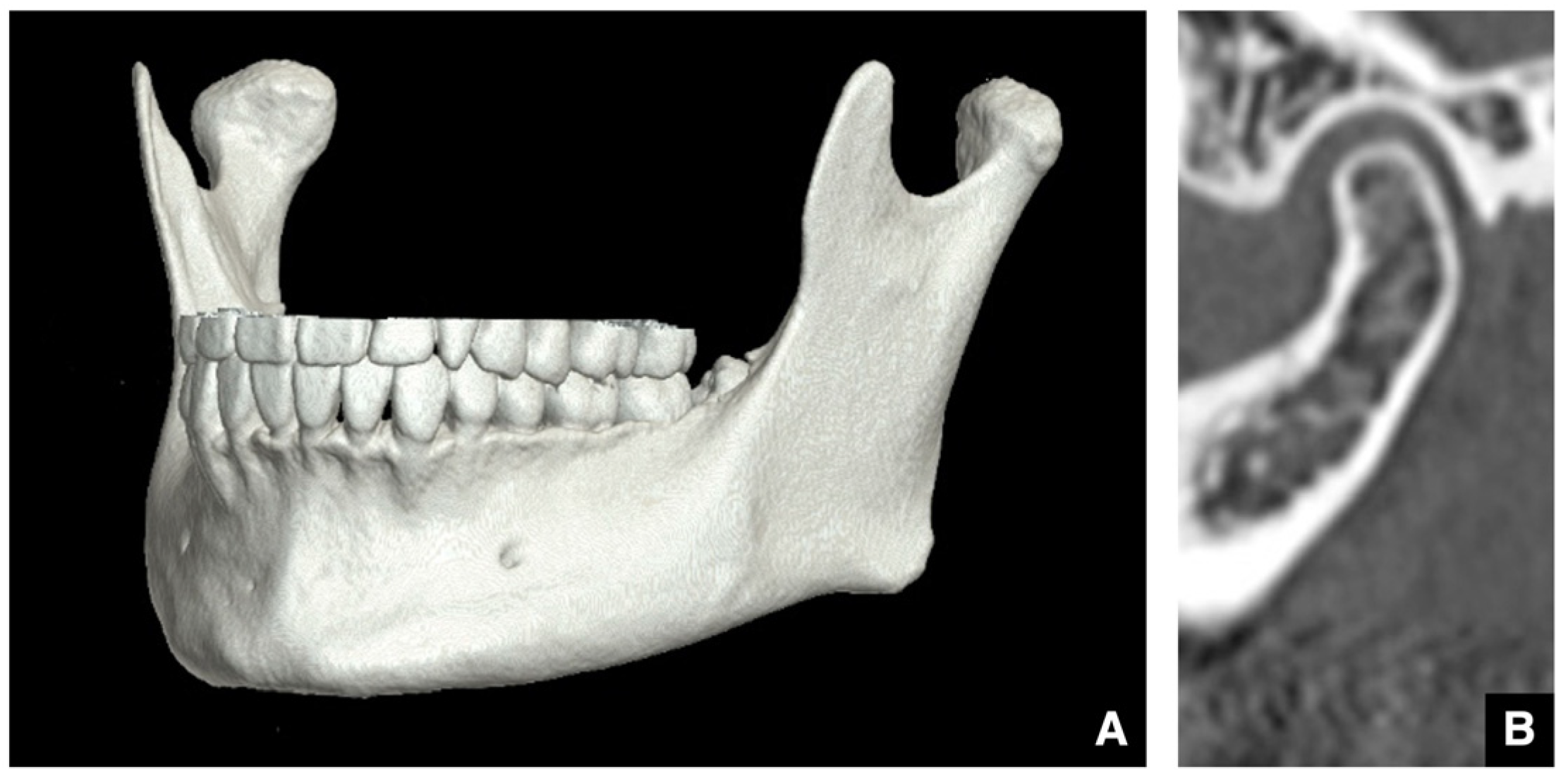





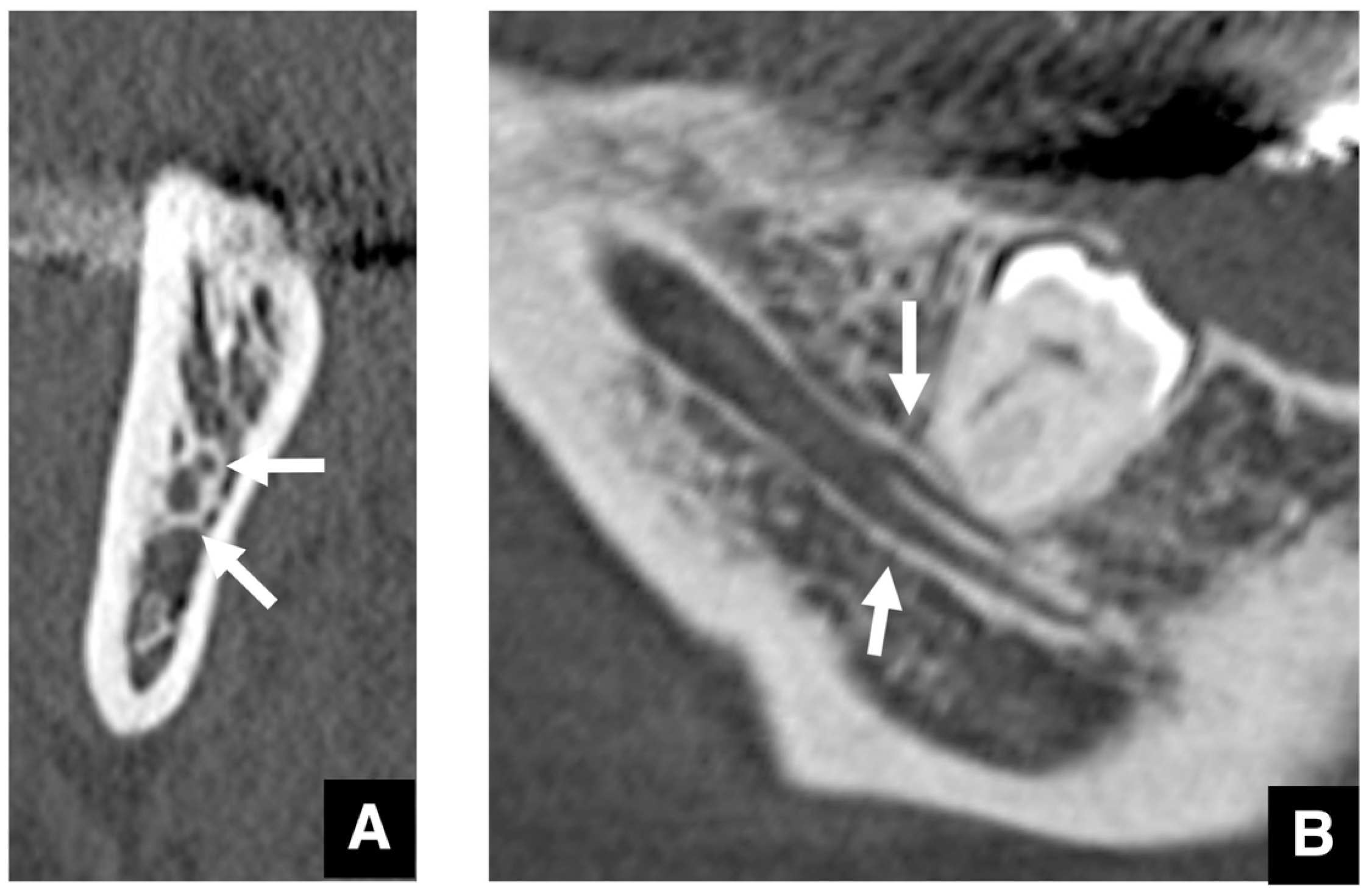

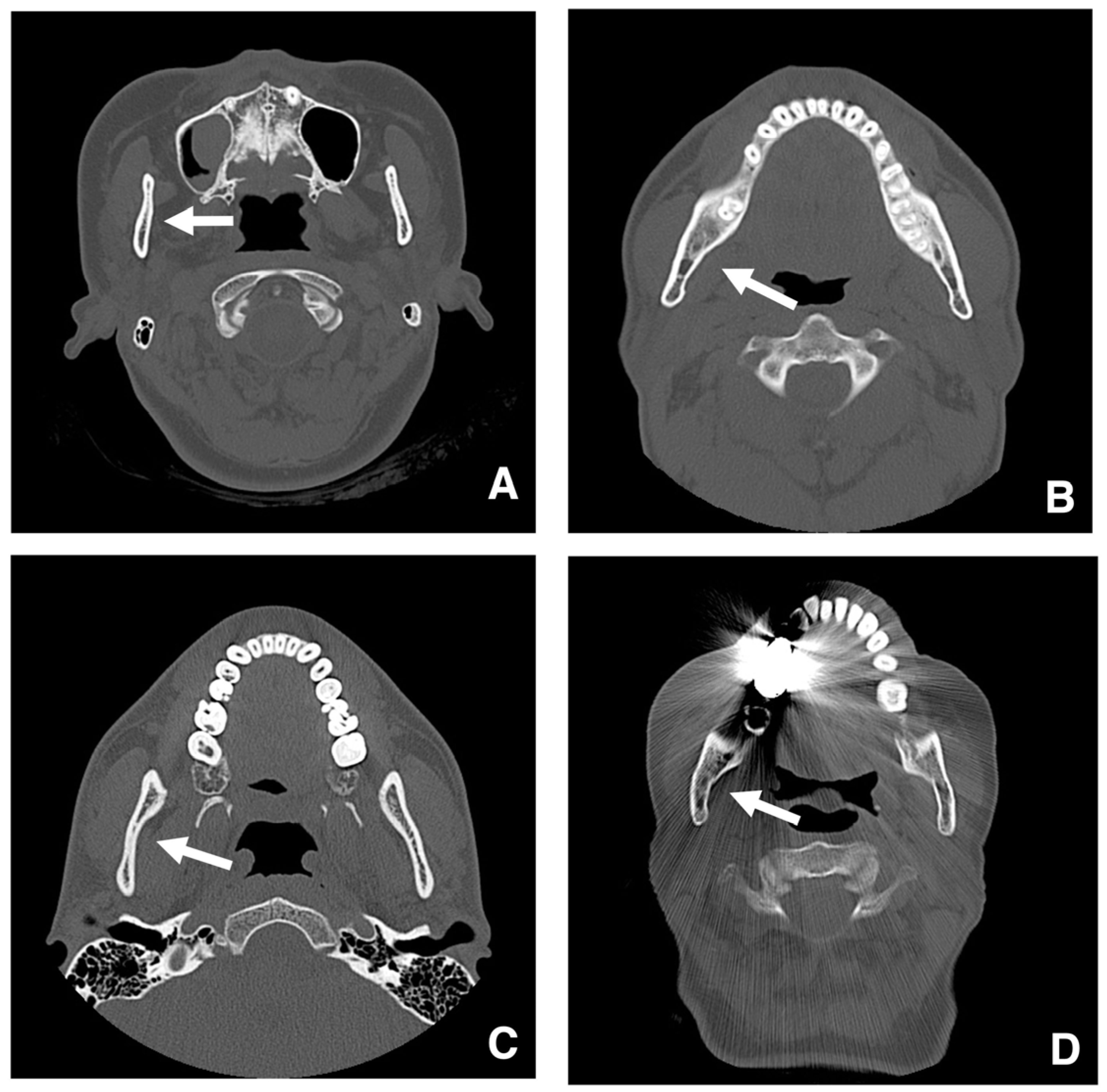

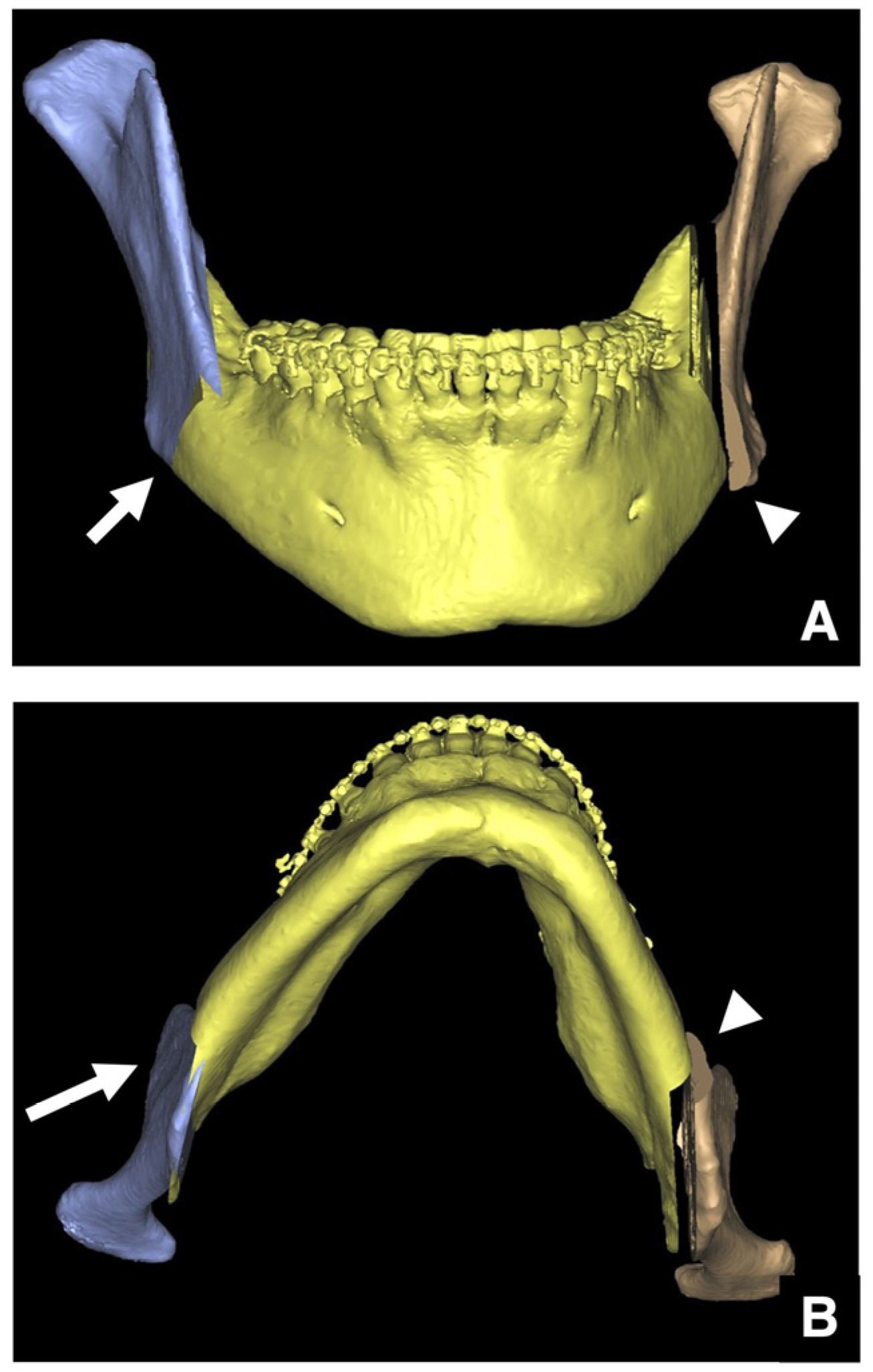



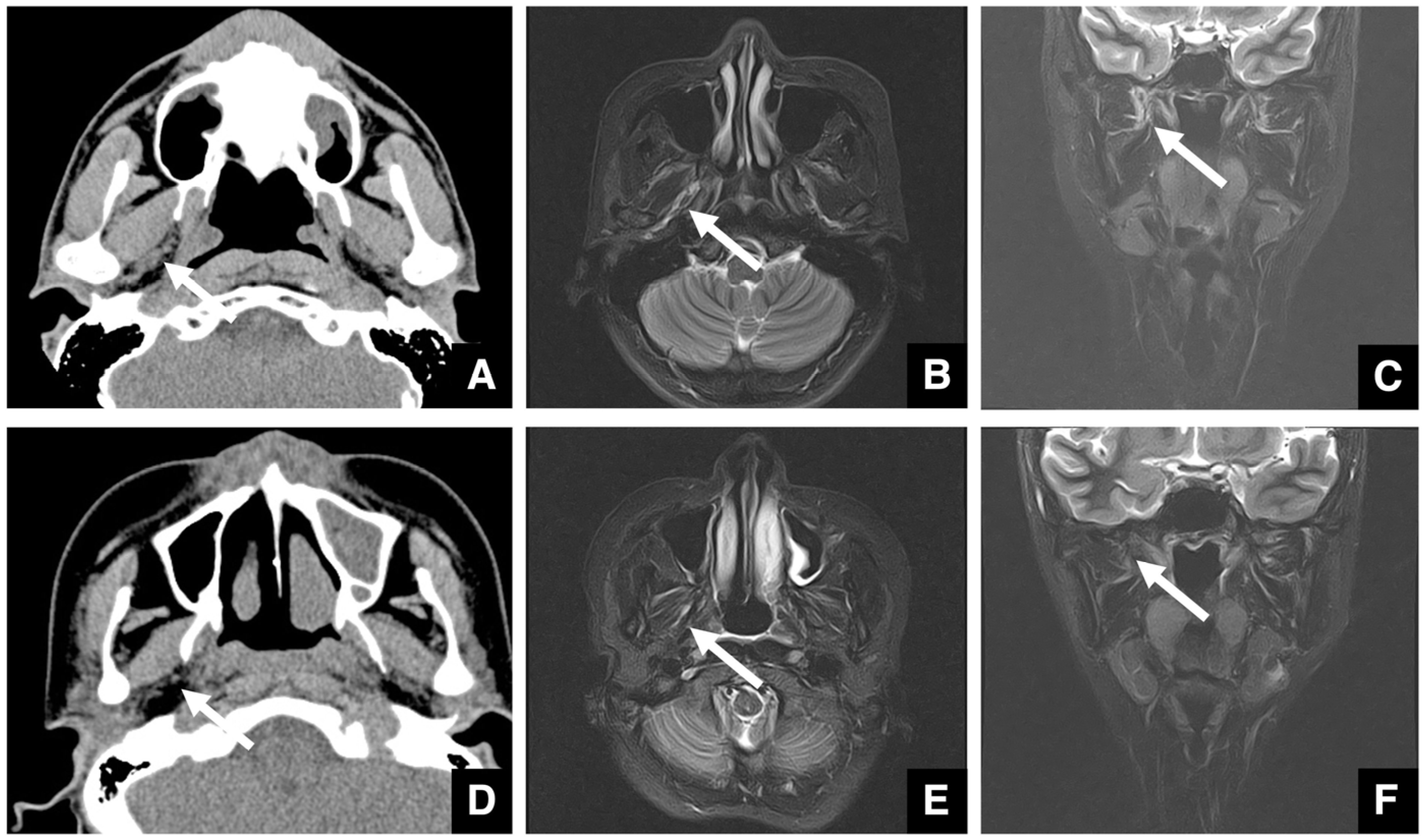

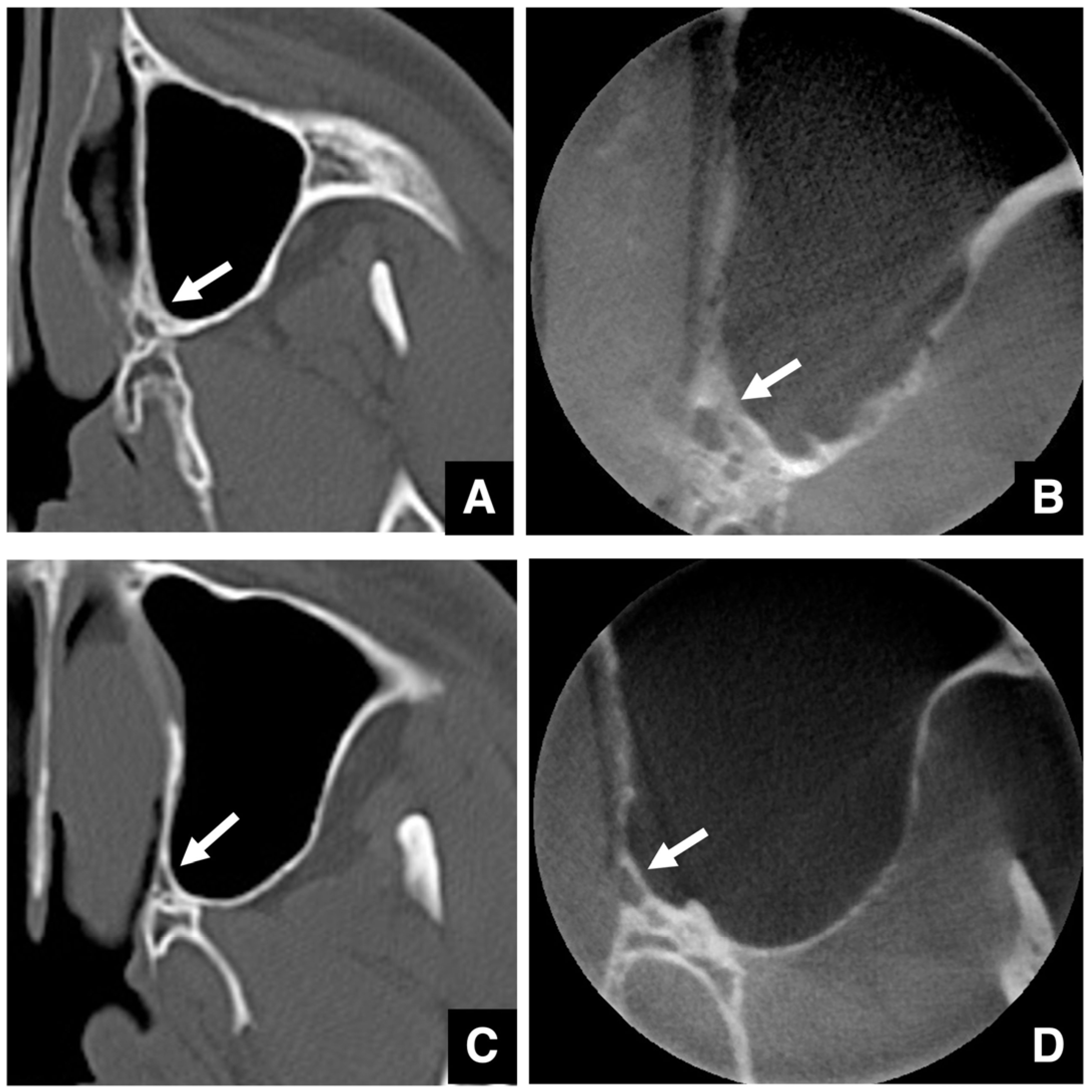


Types of Malocclusion
Causes of Malocclusion
|
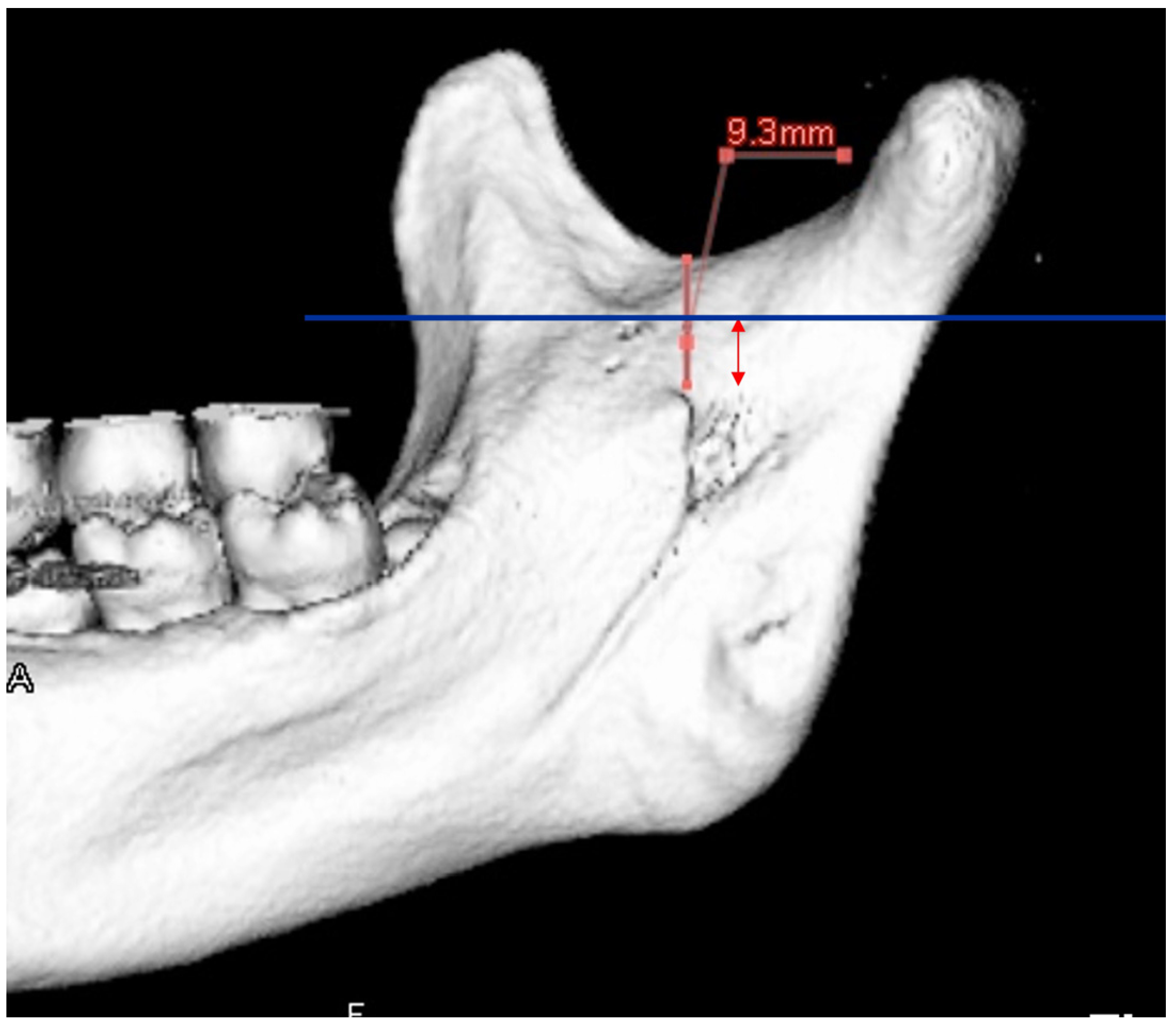
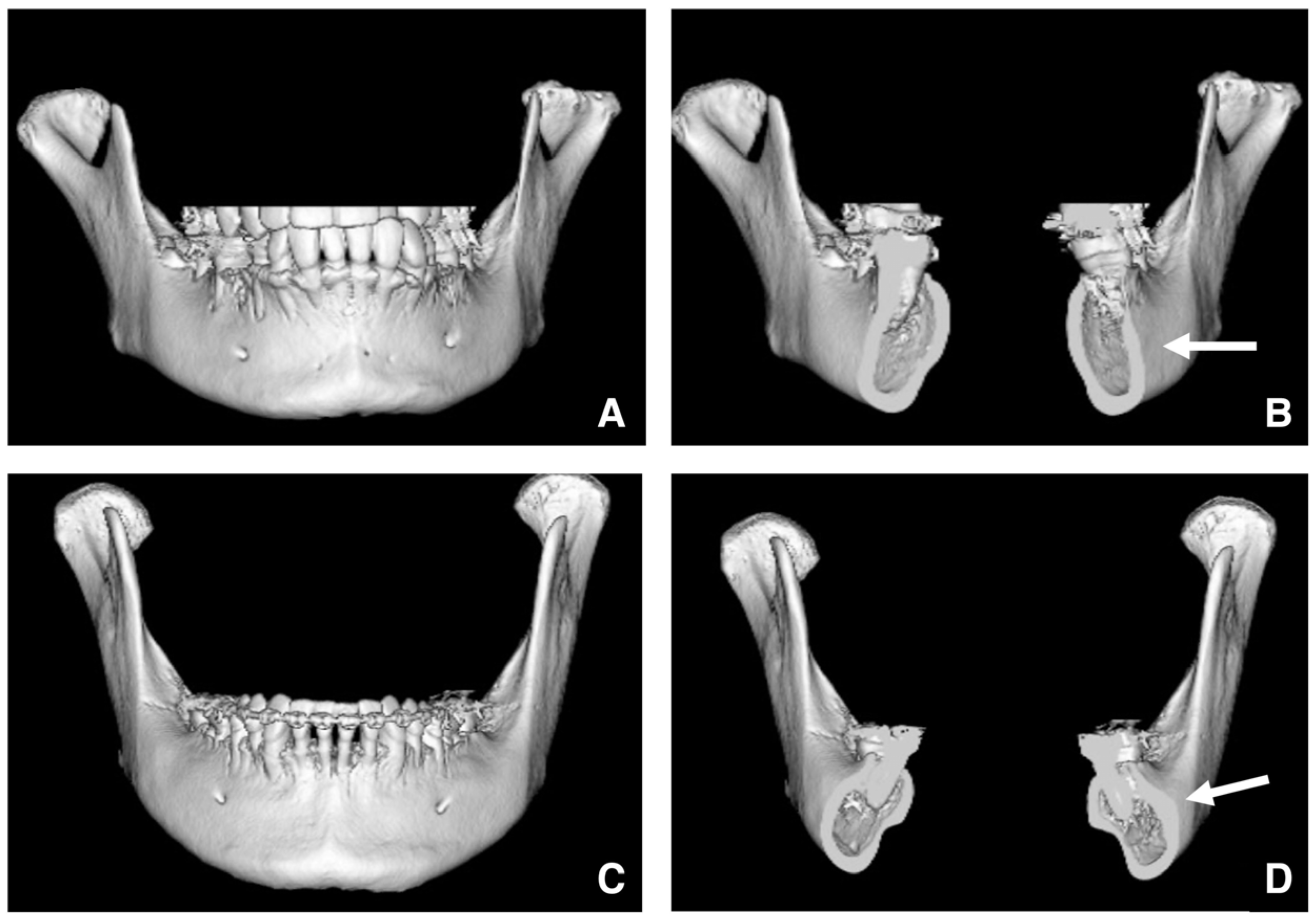
| Setting the osteotomy line 1. Evaluation of the position of the mandibular canal and surrounding bone quality Presence or absence of mandibular canal bifurcation and course of resection site Distance from the buccal wall of the mandibular canal to the buccal cortical bone margin Bone quality around the mandibular canal (CT number) 2. Distance from the mandibular notch to the lingula Whether it is 14 mm or more 3. Morphology of the mandible and variation in cortical bone thickness Cortical bone thickness in the medial osteotomy area and lateral osteotomy area Enhanced safety through improved visibility 1. Medial curvature of the mandibular ramus Mandibular ramus is straight or strong curvature 2. Course of small blood vessels along the bone surface Depression of the buccal-lingual cortical bone from the mandibular fossa to the mandibular ramus region, trabecular bone defect Improved success rates through enhanced postoperative bone integration 1. Degree of interference between bone segments The presence or absence of interference between the proximal and distal bone segments formed during virtual mandibular deformity surgery |
| Enhanced safety through improved visibility 1. Course of the submental artery and its branches Identification of the submandibular gland and its medial aspect 2. Course of the sublingual artery and the mental nerve Identification of the mentalis muscle, hyoglossus muscle, and anterior belly of the digastric muscle and their medial aspects (Observed within the fatty tissue beneath the chin) 3. Course of small blood vessels along the bone surface Degree of looping at the mentum foramen during opening, course of the incisive branch Setting the osteotomy line 1. Position of the mental foramen and course of the incisive branch Record the position of each root for each lower tooth 2. Position of the mandibular anterior tooth roots Record the thickness of the cortical bone using the mandibular teeth as a reference point 3. Variation in cortical bone thickness in the anterior mandible Depression of the cortical bone in the mandibular anterior region, trabecular bone defect |
| Improving safety through prediction of surgical difficulty and complication 1. Circumstance of pterygoid venous plexus Carefully observe the fat tissue surrounding the medial and lateral pterygoid muscles to assess the development of the internal vascular structures (using CT and MR to evaluate the size of the fat tissue) 2. Fusion status of the maxillary tuberosity and pterygomaxillary suture When the fusion is narrow and the degree of calcification (CT number) is low or when the fusion is wide and high 3. Course of the descending palatine artery and thickness of surrounding bone When the palatal canal wall is thick, the probability of damage during transection is low. When it is thin, the possibility of damage cannot be ruled out. Setting the osteotomy line 1. Distance from the piriform rim to the greater palatine canal Long, short, cortical bone thickness, presence or absence of bone irregularities 2. Evaluation of the size, morphology, and internal features (mucosa, septa, etc.) of the maxillary sinus Size, shape, presence or absence of septa, presence or absence of masses, presence or absence of mucosal thickening 3. Relationship between the floor of the maxillary sinus and the apices of the molars Describe the maxillary sinus floor and the apex of each molar tooth 4. Presence of nasal septal deviation, size of the nasal passages, and thickness of the lateral nasal wall bone Presence or absence of nasal septal deviation, Size of the nasal passage, Thickness of the cortical bone of the lateral wall 5. Course of the mandibular artery The course from the pterygoid cleft to the pterygopalatine fossa in the posterior maxilla |
Vascular injury and massive bleeding
Abnormal position of the mandibular condyle and temporomandibular joint dislocation Nerve injury Accidental fractures Airway obstruction Pulmonary aeration status |
| Inflammation Status of bone healing Abnormal fractures Loosening or fracture of bone fixation plates Nerve injury Temporomandibular joint disorders and progressive condylar resorption Displacement or dislocation of the temporomandibular joint Condition of the muscles during occlusion Velopharyngeal insufficiency |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tsurushima, H.; Oda, M.; Kometani-Gunjikake, K.; Shirakawa, T.; Matsumoto-Takeda, S.; Wakasugi-Sato, N.; Nishimura, S.; Haraguchi, K.; Nishina, S.; Kawamoto, T.; et al. Imaging Evaluation for Jaw Deformities: Diagnostic Workup and Pre-Treatment Imaging Checklist for Orthognathic Surgery. Diagnostics 2026, 16, 367. https://doi.org/10.3390/diagnostics16020367
Tsurushima H, Oda M, Kometani-Gunjikake K, Shirakawa T, Matsumoto-Takeda S, Wakasugi-Sato N, Nishimura S, Haraguchi K, Nishina S, Kawamoto T, et al. Imaging Evaluation for Jaw Deformities: Diagnostic Workup and Pre-Treatment Imaging Checklist for Orthognathic Surgery. Diagnostics. 2026; 16(2):367. https://doi.org/10.3390/diagnostics16020367
Chicago/Turabian StyleTsurushima, Hiroki, Masafumi Oda, Kaori Kometani-Gunjikake, Tomohiko Shirakawa, Shinobu Matsumoto-Takeda, Nao Wakasugi-Sato, Shun Nishimura, Kazuya Haraguchi, Susumu Nishina, Tatsuo Kawamoto, and et al. 2026. "Imaging Evaluation for Jaw Deformities: Diagnostic Workup and Pre-Treatment Imaging Checklist for Orthognathic Surgery" Diagnostics 16, no. 2: 367. https://doi.org/10.3390/diagnostics16020367
APA StyleTsurushima, H., Oda, M., Kometani-Gunjikake, K., Shirakawa, T., Matsumoto-Takeda, S., Wakasugi-Sato, N., Nishimura, S., Haraguchi, K., Nishina, S., Kawamoto, T., Habu, M., Yoshioka, I., Arimatsu, T., & Morimoto, Y. (2026). Imaging Evaluation for Jaw Deformities: Diagnostic Workup and Pre-Treatment Imaging Checklist for Orthognathic Surgery. Diagnostics, 16(2), 367. https://doi.org/10.3390/diagnostics16020367






