The Importance of Predicting Bowel Necrosis in Acute Mesenteric Ischemia: Narrative Review
Abstract
1. Introduction
- Acute Venous Mesenteric Ischemia (AVMI): the least frequent due to venous thrombosis of the SMV or in one of its confluent branches [11].
- Non-occlusive Mesenteric Ischemia (NOMI): 5–15%, due to non-occlusive causes, essentially resulting in hypovolemia, absolute (e.g., hemorrhagic shock) or relative (septic shock, cardiogenic shock, major surgery, certain vasoactive substances, etc.) [11].
2. CT Scan Protocol
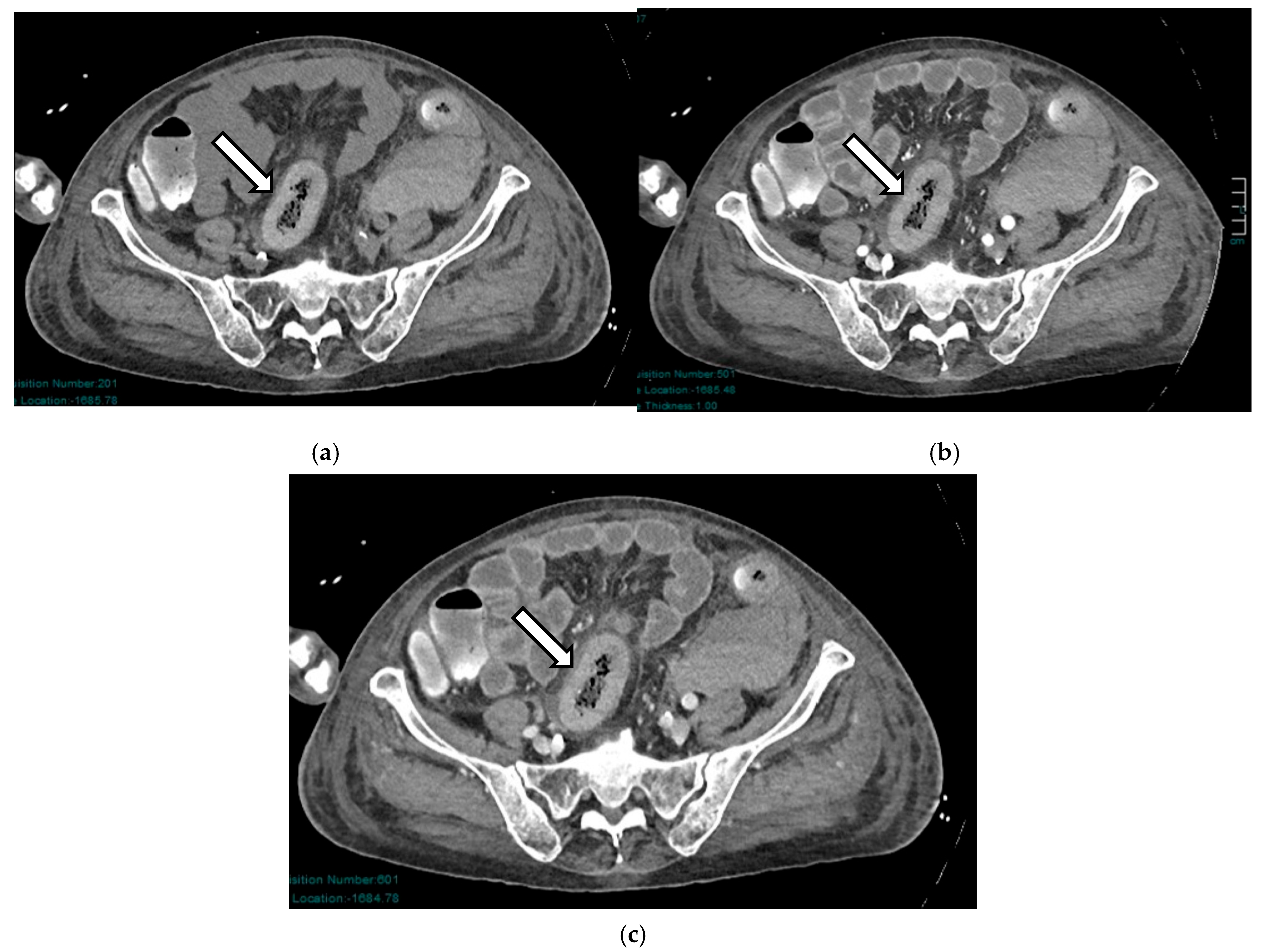
3. Arterial Occlusive or Stenotic Mesenteric Ischemia (AAMI)
3.1. Bowel Involvement in AAMI
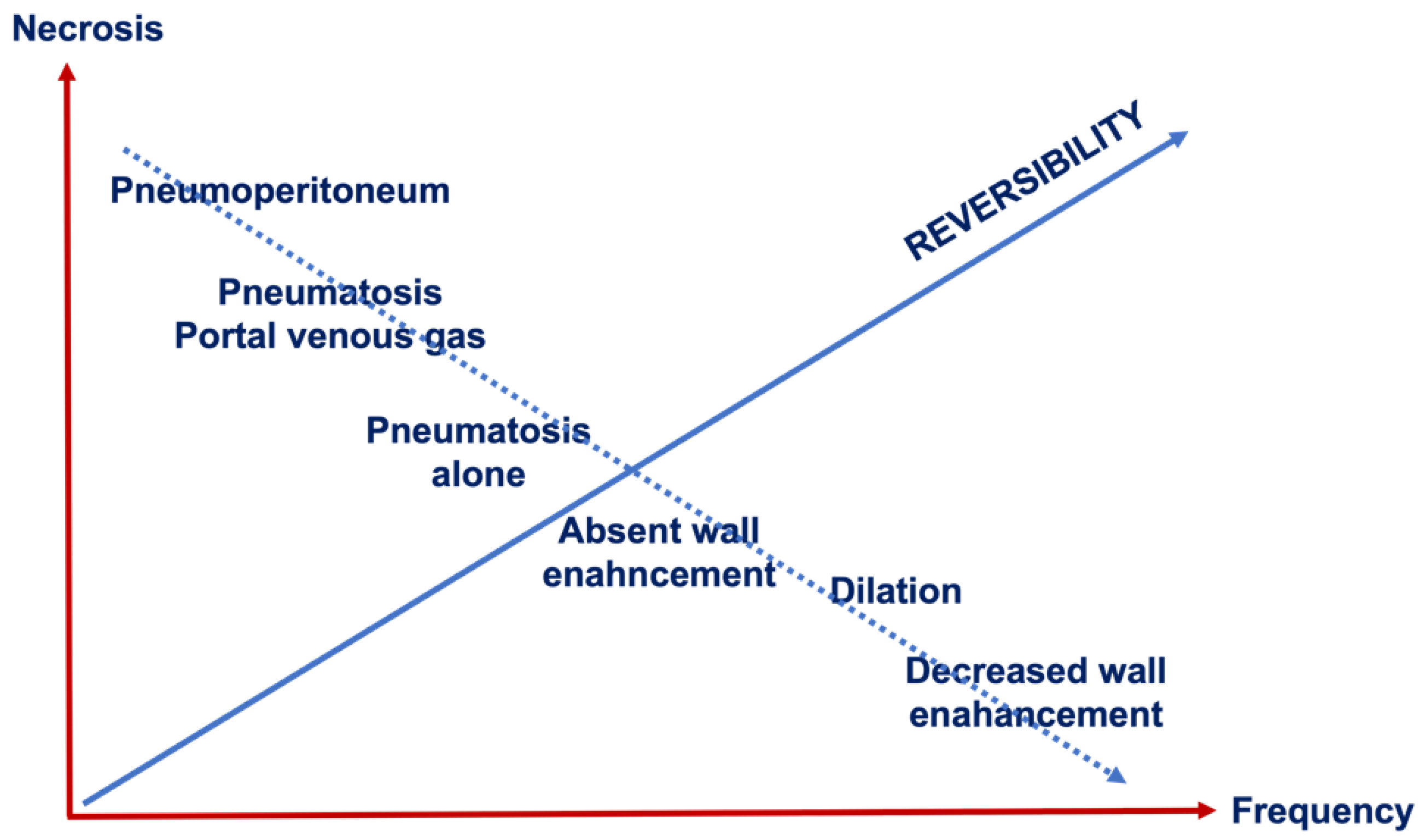
3.1.1. Decreased/Absent Wall Enhancement
3.1.2. Bowel Dilatation
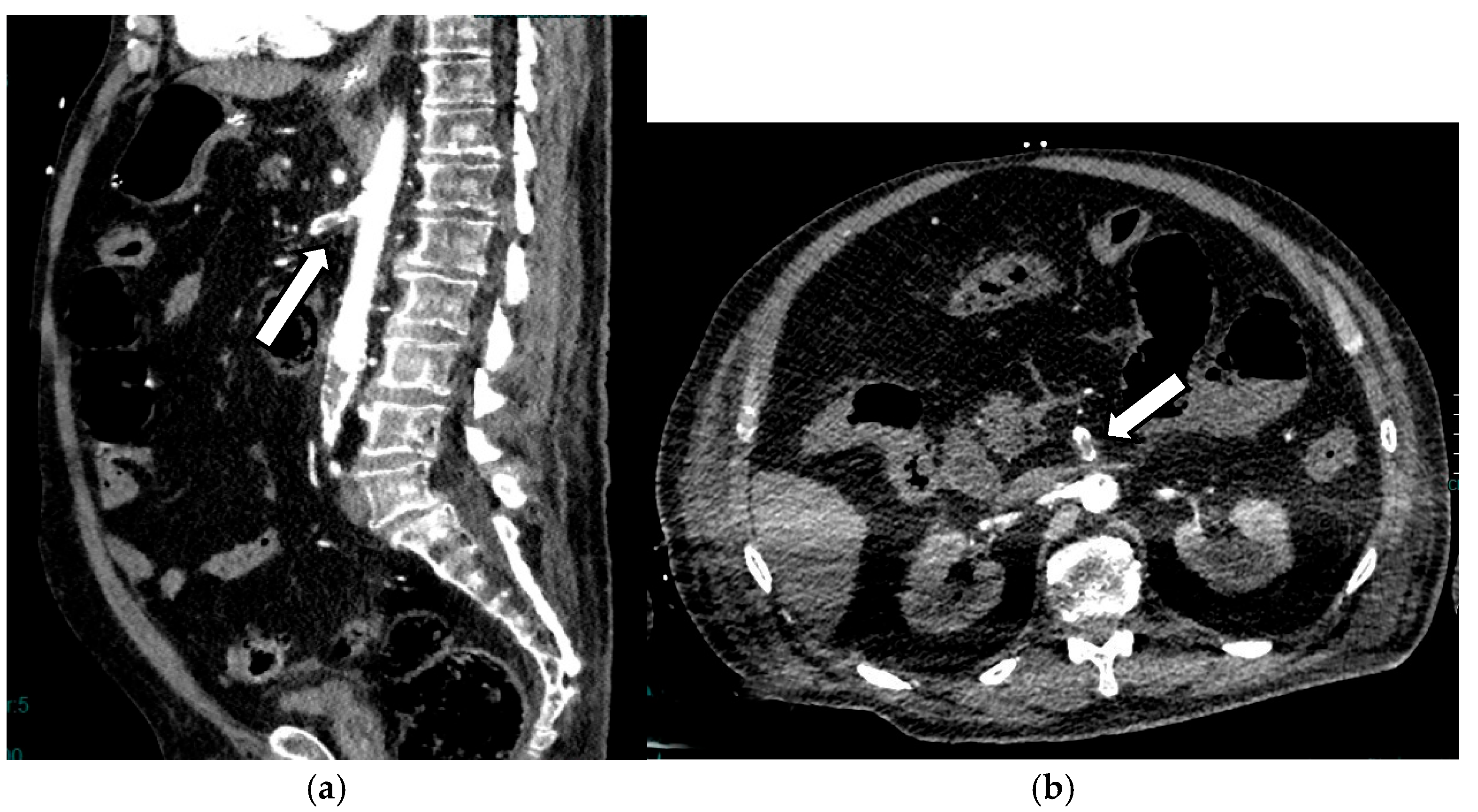
3.1.3. Pneumatosis
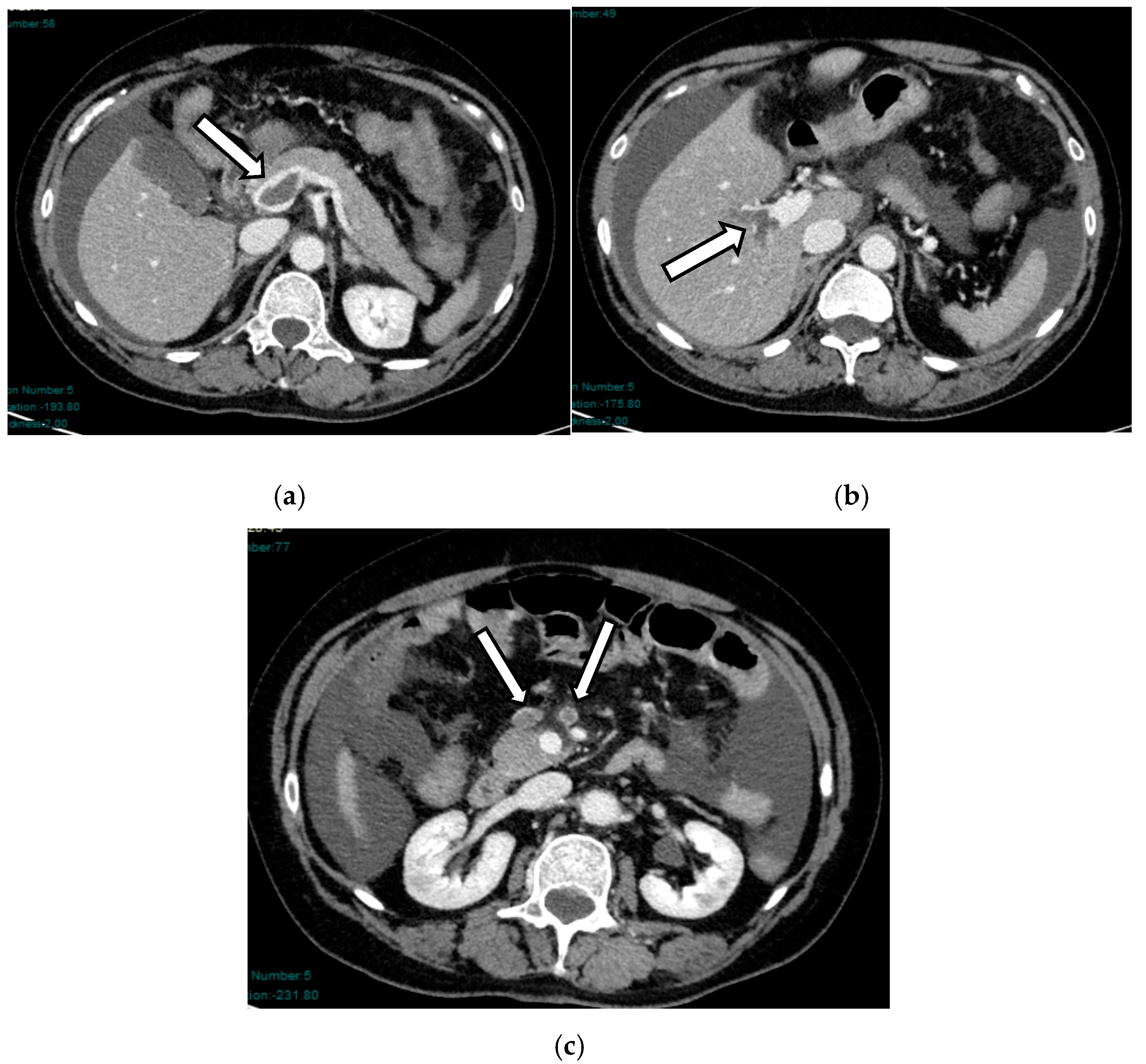
4. Acute Venous Mesenteric Infarction (AVMI)
4.1. Risk Factor for AMVI
4.2. Clinical Presentation and Laboratory Findings
4.3. Diagnostic Imaging
- Pre-contrast phase: Recent thrombi may appear hyperdense, aiding early diagnosis.
- Arterial and venous phases: These highlight bowel wall thickening and layered enhancement, a hallmark of impaired venous outflow. This results in vascular congestion of the bowel wall and mesentery. Prolonged obstruction may lead to free abdominal fluid.
4.4. Pathophysiological Progression
4.5. Management and Treatment of AVMI
5. Non-Occlusive Mesenteric Ischemia (NOMI)
5.1. Vascular Involvement in NOMI
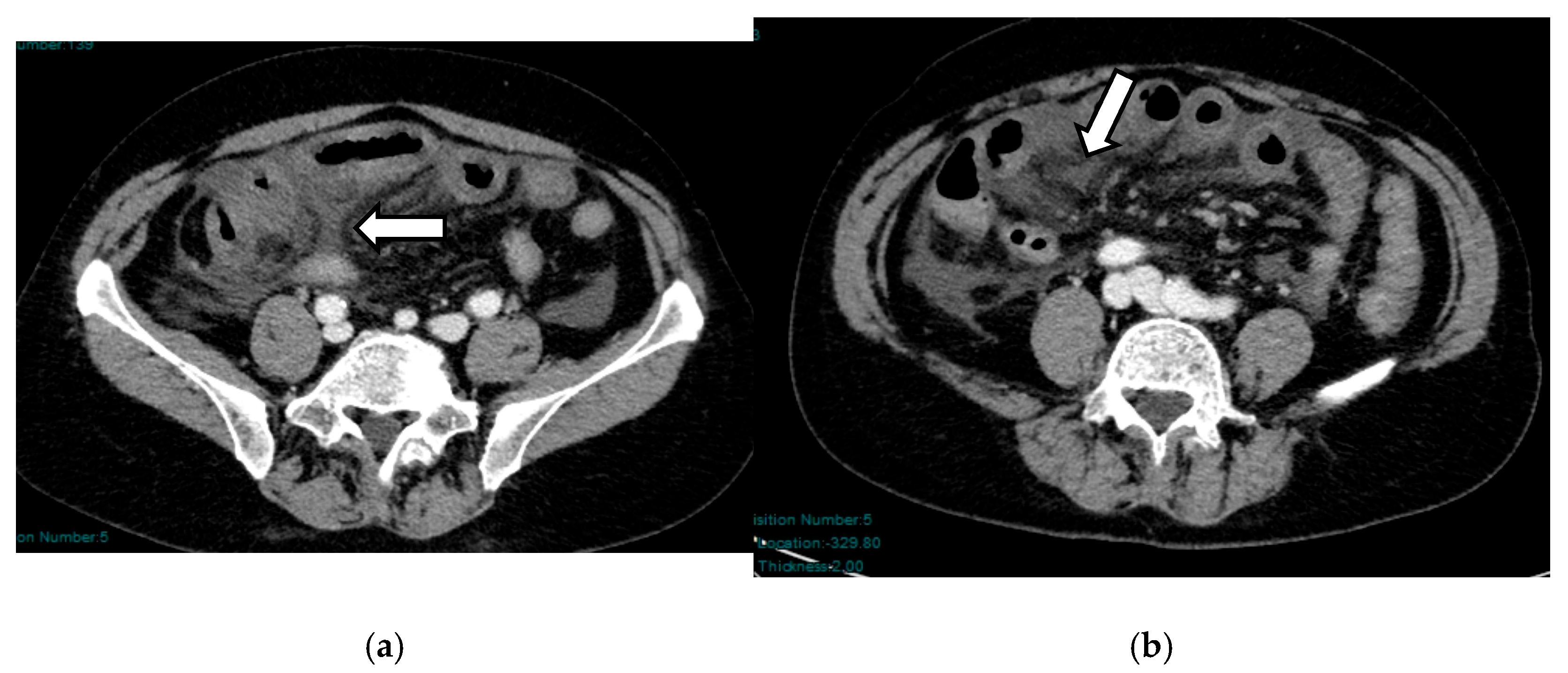
5.2. Bowel Involvement in NOMI
5.2.1. Ischemic Injury—Early Stage
5.2.2. Necrotic Injury—Late Stage
6. Chronic Mesenteric Ischemia (CMI)
6.1. Risk Factors for CMI
6.2. Diagnosis
6.3. Management and Treatment of CMI
7. Predictor Signs of Transmural Necrosis
8. Imaging Limitations
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AAMI | Acute Arterial Mesenteric Ischemia |
| AMI | Acute Mesenteric Ischemia |
| AMVI | Acute Venous Mesenteric Ischemia |
| CECT | Contrast-Enhanced CT |
| CMI | Chronic Mesenteric Ischemia |
| CTA | Computed Tomography Angiography |
| DECT | Dual-Energy CT |
| I-FABP | Intestinal Fatty Acid Binding Protein |
| IMA | Inferior Mesenteric Artery |
| IMV | Inferior Mesenteric Vein |
| ITN | Irreversible Transmural Necrosis |
| IVC | Inferior Vena Cava |
| MPR | Multiplanar Reformation |
| NOMI | Non-Occlusive Mesenteric Ischemia |
| SMA | Superior Mesenteric Artery |
| SMV | Superior Mesenteric Vein |
References
- Kärkkäinen, J.M. Acute Mesenteric Ischemia: A Challenge for the Acute Care Surgeon. Scand. J. Surg. 2021, 110, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Björck, M.; Koelemay, M.; Acosta, S.; Bastos Goncalves, F.; Kölbel, T.; Kolkman, J.J.; Lees, T.; Lefevre, J.H.; Menyhei, G.; Oderich, G.; et al. Editor’s Choice—Management of the Diseases of Mesenteric Arteries and Veins: Clinical Practice Guidelines of the European Society of Vascular Surgery (ESVS). Eur. J. Vasc. Endovasc. Surg. 2017, 53, 460–510. [Google Scholar] [CrossRef]
- Kanasaki, S.; Furukawa, A.; Fumoto, K.; Hamanaka, Y.; Ota, S.; Hirose, T.; Inoue, A.; Shirakawa, T.; Nguyen, L.D.H.; Tulyeubai, S. Acute Mesenteric Ischemia: Multidetector CT Findings and Endovascular Management. Radiographics 2018, 38, 945–961. [Google Scholar] [CrossRef]
- Reintam Blaser, A.; Mändul, M.; Björck, M.; Acosta, S.; Bala, M.; Bodnar, Z.; Casian, D.; Demetrashvili, Z.; D’Oria, M.; Durán Muñoz-Cruzado, V.; et al. Incidence, Diagnosis, Management and Outcome of Acute Mesenteric Ischaemia: A Prospective, Multicentre Observational Study (AMESI Study). Crit. Care 2024, 28, 32. [Google Scholar] [CrossRef]
- Kärkkäinen, J.M.; Lehtimäki, T.T.; Manninen, H.; Paajanen, H. Acute Mesenteric Ischemia Is a More Common Cause than Expected of Acute Abdomen in the Elderly. J. Gastrointest. Surg. 2015, 19, 1407–1414. [Google Scholar] [CrossRef]
- Schoots, I.G.; Koffeman, G.I.; Legemate, D.A.; Levi, M.; van Gulik, T.M. Systematic Review of Survival after Acute Mesenteric Ischaemia According to Disease Aetiology. J. Br. Surg. 2004, 91, 17–27. [Google Scholar] [CrossRef]
- Adaba, F.; Askari, A.; Dastur, J.; Patel, A.; Gabe, S.M.; Vaizey, C.J.; Faiz, O.; Nightingale, J.M.D.; Warusavitarne, J. Mortality after Acute Primary Mesenteric Infarction: A Systematic Review and Meta-Analysis of Observational Studies. Color. Dis. 2015, 17, 566–577. [Google Scholar] [CrossRef]
- Magnus, L.; Lejay, A.; Philouze, G.; Chakfé, N.; Collange, O.; Thaveau, F.; Georg, Y. Mortality and Delays of Management of Acute Mesenteric Ischemia: The Need of a Dedicated Program. Ann. Vasc. Surg. 2023, 91, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Lemma, A.; Tolonen, M.; Vikatmaa, P.; Mentula, P.; Kantonen, I.; But, A.; Leppäniemi, A.; Sallinen, V. Editor’s Choice—Epidemiology, Diagnostics, and Outcomes of Acute Occlusive Arterial Mesenteric Ischaemia: A Population Based Study. Eur. J. Vasc. Endovasc. Surg. 2022, 64, 646–653. [Google Scholar] [CrossRef] [PubMed]
- Sinha, D.; Kale, S.; Kundaragi, N.G.; Sharma, S. Mesenteric Ischemia: A Radiologic Perspective. Abdom. Radiol. 2022, 47, 1514–1528. [Google Scholar] [CrossRef]
- Clair, D.G.; Beach, J.M. Mesenteric Ischemia. N. Engl. J. Med. 2016, 374, 959–968. [Google Scholar] [CrossRef]
- Garzelli, L.; Ben Abdallah, I.; Nuzzo, A.; Zappa, M.; Corcos, O.; Burgio, M.D.; Cazals-Hatem, D.; Rautou, P.E.; Vilgrain, V.; Calame, P.; et al. Insights into Acute Mesenteric Ischaemia: An up-to-Date, Evidence-Based Review from a Mesenteric Stroke Centre Unit. Br. J. Radiol. 2023, 96, 20230232. [Google Scholar] [CrossRef]
- Nuzzo, A.; Guedj, K.; Curac, S.; Hercend, C.; Bendavid, C.; Gault, N.; Tran-Dinh, A.; Ronot, M.; Nicoletti, A.; Bouhnik, Y.; et al. Accuracy of Citrulline, I-FABP and d-Lactate in the Diagnosis of Acute Mesenteric Ischemia. Sci. Rep. 2021, 11, 18929. [Google Scholar] [CrossRef]
- Nuzzo, A.; Joly, F.; Ronot, M.; Castier, Y.; Huguet, A.; Paugam-Burtz, C.; Cazals-Hatem, D.; Tran-Dinh, A.; Becq, A.; Panis, Y.; et al. Normal Lactate and Unenhanced CT-Scan Result in Delayed Diagnosis of Acute Mesenteric Ischemia. Am. J. Gastroenterol. 2020, 115, 1902–1905. [Google Scholar] [CrossRef]
- Nuzzo, A.; Maggiori, L.; Ronot, M.; Becq, A.; Plessier, A.; Gault, N.; Joly, F.; Castier, Y.; Vilgrain, V.; Paugam, C.; et al. Predictive Factors of Intestinal Necrosis in Acute Mesenteric Ischemia: Prospective Study from an Intestinal Stroke Center. Am. J. Gastroenterol. 2017, 112, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Mihaileanu, F.V.; Popa, S.L.; Grad, S.; Dumitrascu, D.I.; Ismaiel, A.; Rus, E.; Brata, V.D.; Padureanu, A.M.; Dita, M.O.; Turtoi, D.C.; et al. The Efficiency of Serum Biomarkers in Predicting the Clinical Outcome of Patients with Mesenteric Ischemia during Follow-Up: A Systematic Review. Diagnostics 2024, 14, 670. [Google Scholar] [CrossRef]
- Lehtimäki, T.T.; Kärkkäinen, J.M.; Saari, P.; Manninen, H.; Paajanen, H.; Vanninen, R. Detecting Acute Mesenteric Ischemia in CT of the Acute Abdomen Is Dependent on Clinical Suspicion: Review of 95 Consecutive Patients. Eur. J. Radiol. 2015, 84, 2444–2453. [Google Scholar] [CrossRef]
- Anglaret, S.; Dallongeville, A.; Beaussier, H.; Touloupas, C.; Boulay, I.; Tardivel, A.M.; Béranger, S.; Silvera, S.; Chatelier, G.; Ronot, M.; et al. Influence of Clinical Suspicion on CT Accuracy of Acute Mesenteric Ischemia: Retrospective Study of 362 Patients. Eur. J. Radiol. 2021, 138, 109652. [Google Scholar] [CrossRef] [PubMed]
- Nuzzo, A.; Peoc’h, K.; Vaittinada Ayar, P.; Tran-Dinh, A.; Weiss, E.; Panis, Y.; Ronot, M.; Garzelli, L.; Eloy, P.; Ben Abdallah, I.; et al. Improving Clinical Suspicion of Acute Mesenteric Ischemia among Patients with Acute Abdomen: A Cross-Sectional Study from an Intestinal Stroke Center. World J. Emerg. Surg. 2023, 18, 37. [Google Scholar] [CrossRef]
- Garzelli, L.; Nuzzo, A.; Copin, P.; Calame, P.; Corcos, O.; Vilgrain, V.; Ronot, M. Contrast-Enhanced CT for the Diagnosis of Acute Mesenteric Ischemia. Am. J. Roentgenol. 2020, 215, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Bala, M.; Catena, F.; Kashuk, J.; De Simone, B.; Gomes, C.A.; Weber, D.; Sartelli, M.; Coccolini, F.; Kluger, Y.; Abu-Zidan, F.M.; et al. Acute Mesenteric Ischemia: Updated Guidelines of the World Society of Emergency Surgery. World J. Emerg. Surg. 2022, 17, 54. [Google Scholar] [CrossRef]
- Pinto, A.; Lanzetta, M.M.; Addeo, G.; Bonini, M.C.; Grazzini, G.; Miele, V. Errors in MDCT diagnosis of acute mesenteric ischemia. Abdom. Radiol. 2022, 47, 1699–1713. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, L.A.; Rivers-Bowerman, M.D.; Thipphavong, S.; Clarke, S.E.; Rowe, J.A.; Costa, A.F. Pearls, Pitfalls, and Conditions That Mimic Mesenteric Ischemia at CT. Radiographics 2020, 40, 545–561. [Google Scholar] [CrossRef]
- Chuong, A.M.; Corno, L.; Beaussier, H.; Isabelle, B.C.; Millet, I.; Hodel, J.; Taourel, P.; Chatellier, G.; Zins, M. Assessment of Bowel Wall Enhancement for the Diagnosis of Intestinal Ischemia in Patients with Small Bowel Obstruction: Value of Adding Unenhanced CT to Contrast-Enhanced CT. Radiology 2016, 280, 98–107. [Google Scholar] [CrossRef]
- Wadman, M.; Block, T.; Ekberg, O.; Syk, I.; Elmståhl, S.; Acosta, S. Impact of MDCT with Intravenous Contrast on the Survival in Patients with Acute Superior Mesenteric Artery Occlusion. Emerg. Radiol. 2010, 17, 171–178. [Google Scholar] [CrossRef]
- Horton, K.M.; Fishman, E.K. Multi-Detector Row CT of Mesenteric Ischemia: Can It Be Done? Radiographics 2001, 21, 1463–1473. [Google Scholar] [CrossRef]
- Mazzei, M.A.; Volterrani, L. Nonocclusive Mesenteric Ischaemia: Think about It. Radiol. Medica 2015, 120, 85–95. [Google Scholar] [CrossRef]
- Copin, P.; Ronot, M.; Nuzzo, A.; Maggiori, L.; Bouhnik, Y.; Corcos, O.; Vilgrain, V. Inter-Reader Agreement of CT Features of Acute Mesenteric Ischemia. Eur. J. Radiol. 2018, 105, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Nakhaei, M.; Mojtahedi, A.; Brook, O.R. Split-Bolus CTA for Mesenteric Ischemia with a Single Scan Opacifying Arterial and Mesenteric Venous Systems. Eur. Radiol. 2020, 30, 3987–3995. [Google Scholar] [CrossRef]
- D’Angelo, T.; Bucolo, G.M.; Yel, I.; Koch, V.; Gruenewald, L.D.; Martin, S.S.; Alizadeh, L.S.; Vogl, T.J.; Ascenti, G.; Lanzafame, L.R.M.; et al. Dual-Energy CT Late Arterial Phase Iodine Maps for the Diagnosis of Acute Non-Occlusive Mesenteric Ischemia. Radiol. Medica 2024, 129, 1611–1621. [Google Scholar] [CrossRef] [PubMed]
- Ota, T.; Onishi, H.; Itoh, T.; Fukui, H.; Tsuboyama, T.; Nakamoto, A.; Enchi, Y.; Tatsumi, M.; Tomiyama, N. Investigation of Abdominal Artery Delineation by Photon-Counting Detector CT. Radiol. Medica 2024, 129, 1265–1274. [Google Scholar] [CrossRef] [PubMed]
- van den Berg, J.C. A New Computed Tomography Classification for Acute Mesenteric Ischaemia: More Than a “Gut Feeling”? Eur. J. Vasc. Endovasc. Surg. 2023, 65, 809–810. [Google Scholar] [CrossRef] [PubMed]
- Najdawi, M.I.; Garzelli, L.; Nuzzo, A.; Huguet, A.; Raynaud, L.; Paulatto, L.; Panis, Y.; Ben Abdallah, I.; Castier, Y.; Sibert, A.; et al. Endovascular Revascularization of Acute Arterial Mesenteric Ischemia: Report of a 3-Year Experience from an Intestinal Stroke Center Unit. Eur. Radiol. 2022, 32, 5606–5615. [Google Scholar] [CrossRef]
- Tual, A.; Garzelli, L.; Nuzzo, A.; Corcos, O.; Castier, Y.; Ben Abdallah, I.; Ronot, M. Strengthening the Description of Superior Mesenteric Artery Occlusions in Acute Mesenteric Ischaemia: Proposition for an Anatomical Classification. Eur. J. Vasc. Endovasc. Surg. 2023, 65, 802–808. [Google Scholar] [CrossRef] [PubMed]
- Acosta, S.; Ögren, M.; Sternby, N.H.; Bergqvist, D.; Björck, M. Clinical Implications for the Management of Acute Thromboembolic Occlusion of the Superior Mesenteric Artery: Autopsy Findings in 213 Patients. Ann. Surg. 2005, 241, 516–522. [Google Scholar] [CrossRef]
- Bordet, M.; Tresson, P.; Huvelle, U.; Long, A.; Passot, G.; Bergoin, C.; Lermusiaux, P.; Millon, A.; Della Schiava, N. Natural History of Asymptomatic Superior Mesenteric Arterial Stenosis Depends on Coeliac and Inferior Mesenteric Artery Status. Eur. J. Vasc. Endovasc. Surg. 2021, 61, 810–818. [Google Scholar] [CrossRef]
- Park, W.M.; Gloviczki, P.; Cherry, K.J.; Hallett, J.W.; Bower, T.C.; Panneton, J.M.; Schleck, C.; Ilstrup, D.; Harmsen, W.S.; Noel, A.A. Contemporary Management of Acute Mesenteric Ischemia: Factors Associated with Survival. J. Vasc. Surg. 2002, 35, 445–452. [Google Scholar] [CrossRef]
- Tang, W.; Zhang, J.; Kuang, L.Q.; Yi, K.M.; Li, C.X.; Wang, Y. Relationship of Superior Mesenteric Artery Thrombus Density with Transmural Intestinal Necrosis on Multidetector Computed Tomography in Acute Mesenteric Ischemia. Quant. Imaging Med. Surg. 2021, 11, 3120–3132. [Google Scholar] [CrossRef]
- Emile, S.H.; Khan, S.M.; Barsoum, S.H. Predictors of Bowel Necrosis in Patients with Acute Mesenteric Ischemia: Systematic Review and Meta-Analysis. Updates Surg. 2021, 73, 47–57. [Google Scholar] [CrossRef]
- Zeng, Y.; Yang, F.; Hu, X.; Zhu, F.; Chen, W.; Lin, W. Radiological Predictive Factors of Transmural Intestinal Necrosis in Acute Mesenteric Ischemia: Systematic Review and Meta-Analysis. Eur. Radiol. 2023, 33, 2792–2799. [Google Scholar] [CrossRef]
- Sinz, S.; Schneider, M.A.; Graber, S.; Alkadhi, H.; Rickenbacher, A.; Turina, M. Prognostic Factors in Patients with Acute Mesenteric Ischemia—Novel Tools for Determining Patient Outcomes. Surg. Endosc. 2022, 36, 8607–8618. [Google Scholar] [CrossRef]
- Kirkpatrick, I.D.C.; Kroeker, M.A.; Greenberg, H.M. Biphasic CT with Mesenteric CT Angiography in the Evaluation of Acute Mesenteric Ischemia: Initial Experience. Radiology 2003, 229, 91–98. [Google Scholar] [CrossRef]
- Schieda, N.; Fasih, N.; Shabana, W. Triphasic CT in the Diagnosis of Acute Mesenteric Ischaemia. Eur. Radiol. 2013, 23, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Taourel, P.G.; Deneuville, M.; Pradel, J.A.; Régent, D.; Bruel, J.M. Acute Mesenteric Ischemia: Diagnosis with Contrast-Enhanced CT. Radiology 1996, 199, 632–636. [Google Scholar] [CrossRef] [PubMed]
- Yikilmaz, A.; Karahan, O.I.; Senol, S.; Tuna, I.S.; Akyildiz, H.Y. Value of Multislice Computed Tomography in the Diagnosis of Acute Mesenteric Ischemia. Eur. J. Radiol. 2011, 80, 297–302. [Google Scholar] [CrossRef]
- Atre, I.D.; Eurboonyanun, K.; O’Shea, A.; Lahoud, R.M.; Shih, A.; Kalva, S.; Harisinghani, M.G.; Hedgire, S. Predictors of Transmural Intestinal Necrosis in Patients Presenting with Acute Mesenteric Ischemia on Computed Tomography. Abdom. Radiol. 2022, 47, 1636–1643. [Google Scholar] [CrossRef] [PubMed]
- Kernagis, L.Y.; Levine, M.S.; Jacobs, J.E. Pneumatosis Intestinalis in Patients with Ischemia: Correlation of CT Findings with Viability of the Bowel. Am. J. Roentgenol. 2003, 180, 733–736. [Google Scholar] [CrossRef]
- Milone, M.; Di Minno, M.N.D.; Musella, M.; Maietta, P.; Iaccarino, V.; Barone, G.; Milone, F. Computed Tomography Findings of Pneumatosis and Portomesenteric Venous Gas in Acute Bowel Ischemia. World J. Gastroenterol. 2013, 19, 6579–6584. [Google Scholar] [CrossRef]
- DuBose, J.J.; Lissauer, M.; Waldman, I.; Nooralian, J.; Johnson, S.; Maung, A.A.; Piper, G.L.; O’Callaghan, T.A.; Luo-Owen, X.; Turay, D.; et al. Pneumatosis Intestinalis Predictive Evaluation Study (PIPES): A Multicenter Epidemiologic Study of the Eastern Association for the Surgery of Trauma. J. Trauma Acute Care Surg. 2013, 75, 15–23. [Google Scholar] [CrossRef]
- Corcos, O.; Castier, Y.; Sibert, A.; Gaujoux, S.; Ronot, M.; Joly, F.; Paugam, C.; Bretagnol, F.; Abdel-Rehim, M.; Francis, F.; et al. Effects of a Multimodal Management Strategy for Acute Mesenteric Ischemia on Survival and Intestinal Failure. Clin. Gastroenterol. Hepatol. 2013, 11, 158–165.e2. [Google Scholar] [CrossRef]
- Acosta, S. Epidemiology of Mesenteric Vascular Disease: Clinical Implications. Semin. Vasc. Surg. 2010, 23, 4–8. [Google Scholar] [CrossRef]
- Hotoleanu, C.; Andercou, O.; Andercou, A. Mesenteric Venous Thrombosis: Clinical and Therapeutical Approach. Int. Angiol. 2008, 27, 462–465. [Google Scholar]
- Blom, R.M.; Bracke, P.; Brusselaers, H.; Degryse, H. Small Bowel Infarction Due to Mesenteric Venous Thrombosis. J. Belg. Soc. Radiol. 2012, 95, 130–131. [Google Scholar] [CrossRef][Green Version]
- Acosta, S.; Alhadad, A.; Svensson, P.; Ekberg, O. Epidemiology, Risk and Prognostic Factors in Mesenteric Venous Thrombosis. Br. J. Surg. 2008, 95, 1245–1251. [Google Scholar] [CrossRef]
- Dhatt, H.S.; Behr, S.C.; Miracle, A.; Wang, Z.J.; Yeh, B.M. Radiological Evaluation of Bowel Ischemia. Radiol. Clin. N. Am. 2015, 53, 1241–1254. [Google Scholar] [CrossRef]
- Reginelli, A.; Genovese, E.A.; Cappabianca, S.; Iacobellis, F.; Berritto, D.; Fonio, P.; Coppolino, F.; Grassi, R. Intestinal Ischemia: US-CT Findings Correlations. Crit. Ultrasound J. 2013, 5, S7. [Google Scholar] [CrossRef] [PubMed]
- Klein, H.M.; Lensing, R.; Klosterhalfen, B.; Töns, C.; Günther, R.W. Diagnostic Imaging of Mesenteric Infarction. Radiology 1995, 197, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Wu, Z.; Wang, J. The Target Sign: A Significant CT Sign for Predicting Small-Bowel Ischemia and Necrosis. Radiol. Medica 2024, 129, 368–379. [Google Scholar] [CrossRef] [PubMed]
- Caruso, M.; Rinaldo, C.; Iacobellis, F.; Dell’Aversano Orabona, G.; Grimaldi, D.; Di Serafino, M.; Schillirò, M.L.; Verde, F.; Sabatino, V.; Camillo, C.; et al. Abdominal Compartment Syndrome: What Radiologist Needs to Know. Radiol. Medica 2023, 128, 1447–1459. [Google Scholar] [CrossRef]
- Yu, H.; Kirkpatrick, I.D.C. An Update on Acute Mesenteric Ischemia. Can. Assoc. Radiol. J. 2023, 74, 160–171. [Google Scholar] [CrossRef]
- Reginelli, A.; Iacobellis, F.; Berritto, D.; Gagliardi, G.; Di Grezia, G.; Rossi, M.; Fonio, P.; Grassi, R. Mesenteric Ischemia: The Importance of Differential Diagnosis for the Surgeon. BMC Surg. 2013, 13, S51. [Google Scholar] [CrossRef]
- Nuzzo, A.; Corcos, O. Management of Mesenteric Ischemia in the Era of Intestinal Stroke Centers: The Gut and Lifesaving Strategy. Rev. Med. Interne 2017, 38, 592–602. [Google Scholar] [CrossRef] [PubMed]
- Landreneau, R.J.; Fry, W.J. The Right Colon as a Target Organ of Nonocclusive Mesenteric Ischemia. Case Report and Review of the Literature. Arch. Surg. 1990, 125, 591–594. [Google Scholar] [CrossRef] [PubMed]
- Henes, F.O.; Pickhardt, P.J.; Herzyk, A.; Lee, S.J.; Motosugi, U.; Derlin, T.; Lubner, M.G.; Adam, G.; Schön, G.; Bannas, P. CT Angiography in the Setting of Suspected Acute Mesenteric Ischemia: Prevalence of Ischemic and Alternative Diagnoses. Abdom. Radiol. 2017, 42, 1152–1161. [Google Scholar] [CrossRef] [PubMed]
- Mazzei, M.A.; Guerrini, S.; Squitieri, N.C.; Vindigni, C.; Imbriaco, G.; Gentili, F.; Berritto, D.; Mazzei, F.G.; Grassi, R.; Volterrani, L. Emergency Radiology Special Feature: Full Paper: Reperfusion in Non-Occlusivemesenteric Ischaemia (NOMI): Effectiveness of CT in an Emergency Setting. Br. J. Radiol. 2016, 89, 20150956. [Google Scholar] [CrossRef]
- Wasnik, A.; Kaza, R.K.; Al-Hawary, M.M.; Liu, P.S.; Platt, J.F. Multidetector CT Imaging in Mesenteric Ischemia—Pearls and Pitfalls. Emerg. Radiol. 2011, 18, 145–156. [Google Scholar] [CrossRef]
- Nakamura, Y.; Urashima, M.; Toyota, N.; Ono, C.; Iida, M.; Fukumoto, W.; Kaichi, Y.; Tani, C.; Honda, Y.; Komoto, D.; et al. Non-Occlusive Mesenteric Ischemia (NOMI): Utility of Measuring the Diameters of the Superior Mesenteric Artery and Superior Mesenteric Vein at Multidetector CT. Jpn. J. Radiol. 2013, 31, 737–743. [Google Scholar] [CrossRef]
- Pérez-García, C.; De Miguel Campos, E.; Gonzalo, A.F.; Malfaz, C.; Pinacho, J.J.M.; Álvarez, C.F.; Pérez, R.H. Non-Occlusive Mesenteric Ischaemia: CT Findings, Clinical Outcomes and Assessment of the Diameter of the Superior Mesenteric Artery. Br. J. Radiol. 2018, 91, 20170492. [Google Scholar] [CrossRef] [PubMed]
- Woodhams, R.; Nishimaki, H.; Fujii, K.; Kakita, S.; Hayakawa, K. Usefulness of Multidetector-Row CT (MDCT) for the Diagnosis of Non-Occlusive Mesenteric Ischemia (NOMI): Assessment of Morphology and Diameter of the Superior Mesenteric Artery (SMA) on Multi-Planar Reconstructed (MPR) Images. Eur. J. Radiol. 2010, 76, 96–102. [Google Scholar] [CrossRef]
- Moschetta, M.; Telegrafo, M.; Rella, L.; Stabile Ianora, A.A.; Angelelli, G. Multi-detector CT features of acute intestinal ischemia and their prognostic correlations. World J. Radiol. 2014, 6, 130. [Google Scholar] [CrossRef]
- Klotz, S.; Vestring, T.; Rötker, J.; Schmidt, C.; Scheld, H.H.; Schmid, C. Diagnosis and Treatment of Nonocclusive Mesenteric Ischemia after Open Heart Surgery. Ann. Thorac. Surg. 2001, 72, 1583–1586. [Google Scholar] [CrossRef]
- Turba, U.C.; Kapucu, I.; Matsumoto, A.H. Chronic Mesenteric Ischemia. In Image-Guided Interventions: Expert Radiology Series, 3rd ed.; Elsevier Health Sciences: Amsterdam, The Netherlands, 2025; pp. 246–254.e2. [Google Scholar] [CrossRef]
- Anand, U.; Maheshwari, N.; Khandelwal, V.; Sreevani, P.; Gorsi, U.; Savlania, A. Current Management of Chronic Mesenteric Ischemia: A Comprehensive Review. Indian J. Gastroenterol. 2025, 1–13. [Google Scholar] [CrossRef]
- van Petersen, A.S.; Vriens, B.H.; Huisman, A.B.; Kolkman, J.J.; Geelkerken, R.H. Retroperitoneal Endoscopic Release in the Management of Celiac Artery Compression Syndrome. J. Vasc. Surg. 2009, 50, 140–147. [Google Scholar] [CrossRef]
- Blauw, J.T.M.; Pastoors, H.A.M.; Brusse-Keizer, M.; Beuk, R.J.; Kolkman, J.J.; Geelkerken, R.H. The Impact of Revascularisation on Quality of Life in Chronic Mesenteric Ischemia. Can. J. Gastroenterol. Hepatol. 2019, 2019, 7346013. [Google Scholar] [CrossRef]
- Huber, T.S.; Björck, M.; Chandra, A.; Clouse, W.D.; Dalsing, M.C.; Oderich, G.S.; Smeds, M.R.; Murad, M.H. Chronic Mesenteric Ischemia: Clinical Practice Guidelines from the Society for Vascular Surgery. J. Vasc. Surg. 2021, 73, 87S–115S. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, P.J.; Pfarr, J.; Trentmann, J.; Wulff, A.M.; Langer, C.; Siggelkow, M.; Groß, J.; Knabe, H.; Schaefer, F.K.W. Comparison of Noninvasive Imaging Modalities for Stenosis Grading in Mesenteric Arteries. Rofo 2013, 185, 628–634. [Google Scholar] [CrossRef]
- Kärkkäinen, J.M.; Acosta, S. Acute Mesenteric Ischemia (Part I)—Incidence, Etiologies, and How to Improve Early Diagnosis. Best Pract. Res. Clin. Gastroenterol. 2017, 31, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Goodall, R.; Langridge, B.; Onida, S.; Ellis, M.; Lane, T.; Davies, A.H. Median Arcuate Ligament Syndrome. J. Vasc. Surg. 2020, 71, 2170–2176. [Google Scholar] [CrossRef]
- Cognet, F.; Ben Salem, D.; Dranssart, M.; Cercueil, J.P.; Weiller, M.; Tatou, E.; Boyer, L.; Krausé, D. Chronic Mesenteric Ischemia: Imaging and Percutaneous Treatment. Radiographics 2002, 22, 863–879. [Google Scholar] [CrossRef] [PubMed]
- Calame, P.; Winiszewski, H.; Doussot, A.; Malakhia, A.; Grillet, F.; Verdot, P.; Vuitton, L.; Ronot, M.; Pili-Floury, S.; Heyd, B.; et al. Evaluating the Risk of Irreversible Intestinal Necrosis Among Critically Ill Patients With Nonocclusive Mesenteric Ischemia. Am. J. Gastroenterol. 2021, 116, 1506–1513. [Google Scholar] [CrossRef]
- Peoc’H, K.; Nuzzo, A.; Guedj, K.; Paugam, C.; Corcos, O. Diagnosis Biomarkers in Acute Intestinal Ischemic Injury: So Close, yet so Far. Clin. Chem. Lab. Med. 2018, 56, 373–385. [Google Scholar] [CrossRef]
- Kärkkäinen, J.M.; Saari, P.; Kettunen, H.P.; Lehtimäki, T.T.; Vanninen, R.; Paajanen, H.; Manninen, H. Interpretation of Abdominal CT Findings in Patients Who Develop Acute on Chronic Mesenteric Ischemia. J. Gastrointest. Surg. 2016, 20, 791–802. [Google Scholar] [CrossRef]
- Bourcier, S.; Klug, J.; Nguyen, L.S. Non-Occlusive Mesenteric Ischemia: Diagnostic Challenges and Perspectives in the Era of Artificial Intelligence. World J. Gastroenterol. 2021, 27, 4088. [Google Scholar] [CrossRef] [PubMed]
- Bourcier, S.; Oudjit, A.; Goudard, G.; Charpentier, J.; Leblanc, S.; Coriat, R.; Gouya, H.; Dousset, B.; Mira, J.P.; Pène, F. Diagnosis of Non-Occlusive Acute Mesenteric Ischemia in the Intensive Care Unit. Ann. Intensive Care 2016, 6, 112. [Google Scholar] [CrossRef]
- Firetto, M.C.; Lemos, A.A.; Marini, A.; Avesani, E.C.; Biondetti, P.R. Acute Bowel Ischemia: Analysis of Diagnostic Error by Overlooked Findings at MDCT Angiography. Emerg. Radiol. 2012, 20, 139–147. [Google Scholar] [CrossRef] [PubMed]
- van den Heijkant, T.C.; Aerts, B.A.C.; Teijink, J.A.; Buurman, W.A.; Luyer, M.D.P. Challenges in Diagnosing Mesenteric Ischemia. World J. Gastroenterol. 2013, 19, 1338–1341. [Google Scholar] [CrossRef] [PubMed]





| Predictors of Ischemic Bowel Necrosis | |
|---|---|
| Wall features |
|
| |
| |
| |
| Intestinal lumen |
|
| |
| Mesentery |
|
| |
| Ischemic lesions of other organs |
| AAMI | AVMI | NOMI | CMI | |
|---|---|---|---|---|
| Vessels | Intraluminal filling defect in the SMA | Intraluminal filling defect in the SMV | Non intraluminal filling defects | Chronic stenosis or occlusion of the celiac trunk, the SMV or the IMA Prominent collateral vessels Vascular calcifications |
| Bowel wall | Early AAMI: Bowel wall thinning with low enhancement | Early AVMI: Bowel wall thickening and dilatation, mucosal hyperenhancement | Dilated bowel loops with segmental hypoenhancing | Compensated CMI: regular bowel wall AOCMI: Bowel wall thickening, bowel wall pneumatosis |
| Advanced AAMI: Bowel dilatation with no enhancement, bowel wall pneumatosis | Advanced AVMI: Bowel wall pneumatosis, intravenous pneumatosis | After reperfusion: wall thickening with mucosal hypoenhancement |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Giannessi, C.; Cozzi, D.; Scalzone, L.; Treballi, F.; Anichini, M.; Sessa, B.; Ferrarelli, A.; Danti, G.; Miele, V. The Importance of Predicting Bowel Necrosis in Acute Mesenteric Ischemia: Narrative Review. Diagnostics 2026, 16, 332. https://doi.org/10.3390/diagnostics16020332
Giannessi C, Cozzi D, Scalzone L, Treballi F, Anichini M, Sessa B, Ferrarelli A, Danti G, Miele V. The Importance of Predicting Bowel Necrosis in Acute Mesenteric Ischemia: Narrative Review. Diagnostics. 2026; 16(2):332. https://doi.org/10.3390/diagnostics16020332
Chicago/Turabian StyleGiannessi, Caterina, Diletta Cozzi, Ludovica Scalzone, Francesca Treballi, Matilde Anichini, Barbara Sessa, Anna Ferrarelli, Ginevra Danti, and Vittorio Miele. 2026. "The Importance of Predicting Bowel Necrosis in Acute Mesenteric Ischemia: Narrative Review" Diagnostics 16, no. 2: 332. https://doi.org/10.3390/diagnostics16020332
APA StyleGiannessi, C., Cozzi, D., Scalzone, L., Treballi, F., Anichini, M., Sessa, B., Ferrarelli, A., Danti, G., & Miele, V. (2026). The Importance of Predicting Bowel Necrosis in Acute Mesenteric Ischemia: Narrative Review. Diagnostics, 16(2), 332. https://doi.org/10.3390/diagnostics16020332






