Occipital Pial AVM Rupture in a Young Adult: Dual Intranidal Aneurysms, Solitary Parasagittal SSS Drainage, and Hematoma-Corridor Microsurgical Cure
Abstract
1. Introduction
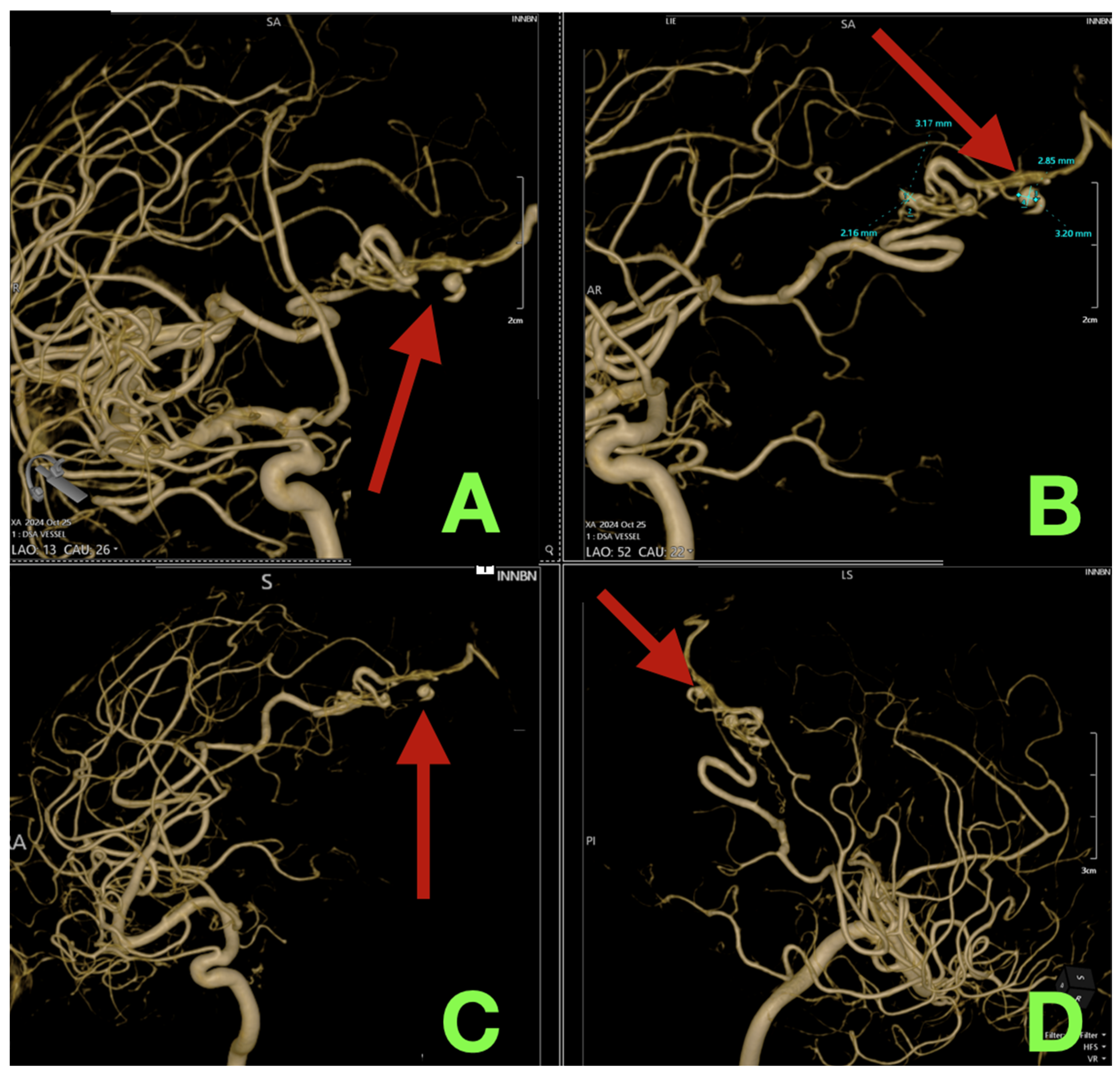
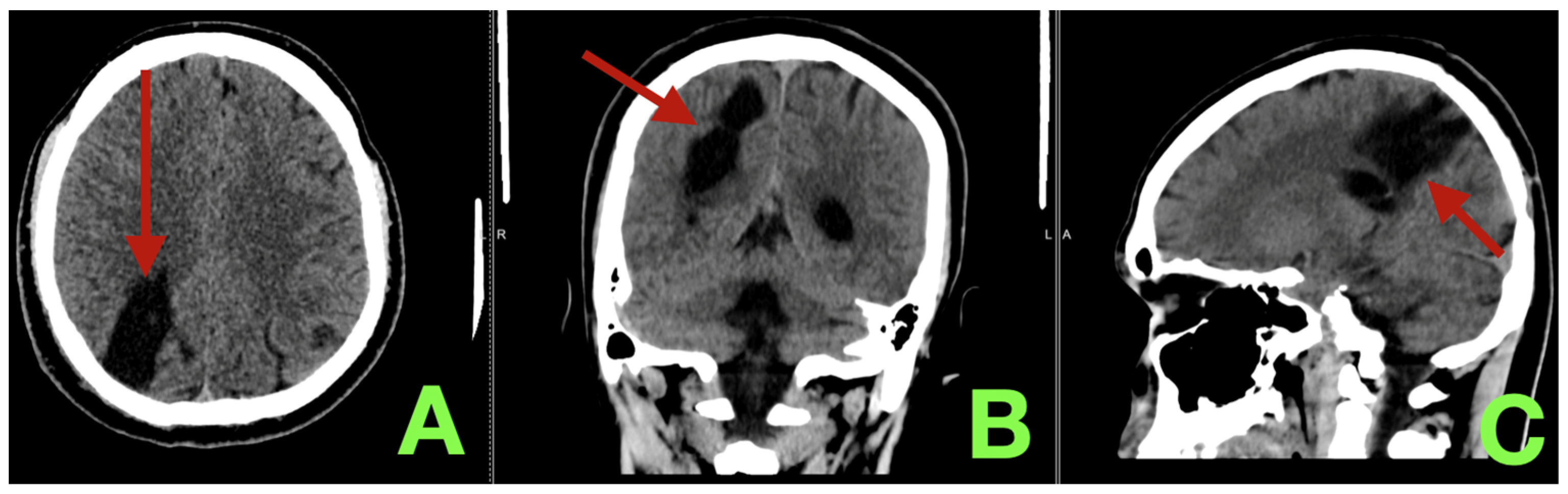
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jabarkheel, R.; Li, L.; Frankfurter, M.; Zhang, D.Y.; Gajjar, A.; Muhammad, N.; Srinivasan, V.M.; Burkhardt, J.-K.; Kahn, M. Untangling Sporadic Brain Arteriovenous Malformations: Towards Targeting the KRAS/MAPK Pathway. Front. Surg. 2024, 11, 1504612. [Google Scholar] [CrossRef] [PubMed]
- Mansour, M.A.; Malaeb, R.W.; Mostafa, H.N.; Kamal, M.I.; Ayoub, B. Diagnostic and Therapeutic Dilemmas of Asymptomatic Intracranial AVMs: A Case Report and Evidence-Based Review. Radiol. Case Rep. 2024, 19, 6452–6459. [Google Scholar] [CrossRef]
- Nakisli, S.; Lagares, A.; Nielsen, C.M.; Cuervo, H. Pericytes and Vascular Smooth Muscle Cells in Central Nervous System Arteriovenous Malformations. Front. Physiol. 2023, 14, 1210563. [Google Scholar] [CrossRef]
- Zhu, H.; Liu, L.; Chang, Y.; Song, Y.; Liang, S.; Ma, C.; Zhang, L.; Liang, F.; Jiang, C.; Zhang, Y. Quantitative Evaluation of the Subsequent Hemorrhage with Arteriography-Derived Hemodynamic Features in Patients with Untreated Cerebral Arteriovenous Malformation. Front. Neurol. 2023, 14, 1174245. [Google Scholar] [CrossRef]
- Suhartono, R.; Suhendro; Rahardjo, H.E.; Harahap, A.R.; Mochtar, C.A.; Muradi, A.; Alwi, I.; Lydia, A.; Kekalih, A.; Soetikno, V.; et al. Effect of Primary Balloon Angioplasty on Draining Vein Diameter and Volume Flow in Patients with Arteriovenous Fistula: A Cohort Study. Ann. Med. Surg. 2022, 81, 104426. [Google Scholar] [CrossRef]
- D’Aliberti, G.; Talamonti, G.; Cenzato, M.; La Camera, A.; Debernardi, A.; Valvassori, L.; Mariangela, P.; Nichelatti, M. Arterial and Venous Aneurysms Associated with Arteriovenous Malformations. World Neurosurg. 2015, 83, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Gokce, G.; Oren, N.C.; Ceylan, O.M.; Mumcuoglu, T.; Hurmeric, V. Scattered Depressions with Temporal Preponderance in Visual Field Test Coexisting with Optic Disc Temporal Atrophy in Cerebral Arteriovenous Malformation. Clin. Ophthalmol. 2013, 7, 2031–2035. [Google Scholar] [CrossRef] [PubMed]
- Lawton, M.T.; Lang, M.J. The Future of Open Vascular Neurosurgery: Perspectives on Cavernous Malformations, AVMs, and Bypasses for Complex Aneurysms. J. Neurosurg. 2019, 130, 1409–1425. [Google Scholar] [CrossRef]
- Stahl, J.; McGuire, L.S.; Abou-Mrad, T.; Saalfeld, S.; Behme, D.; Alaraj, A.; Berg, P. Feasibility Study for Multimodal Image-Based Assessment of Patient-Specific Intracranial Arteriovenous Malformation Hemodynamics. J. Clin. Med. 2025, 14, 2638. [Google Scholar] [CrossRef]
- Zhao, Y.C.; Vatankhah, P.; Goh, T.; Michelis, R.; Kyanian, K.; Zhang, Y.; Li, Z.; Ju, L.A. Hemodynamic Analysis for Stenosis Microfluidic Model of Thrombosis with Refined Computational Fluid Dynamics Simulation. Sci. Rep. 2021, 11, 6875. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, Y.; Si, J.; Xiao, C.; Liu, X.; Wang, C.; Chen, F.; Guo, Y. An Innovative Model for Diagnosing Lesions in Coronary Angiography Imagery Using an Improved YOLOv4 Model. Bioengineering 2025, 12, 1241. [Google Scholar] [CrossRef]
- Miron, I.; Pruna, V.M.; Visarion, D.M.; Petrescu, G.E.D.; Gorgan, R.M. Surgical Outcomes and Risk Factors for Overall Mortality in Brain Arteriovenous Malformations Patients: A Retrospective Analysis. Front. Neurol. 2024, 15, 1428718. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez Pineda, F.; Quiceno, E.; Suarez Marin, M.M.; Londoño Ocampo, F.J. Early versus Delayed Surgical Management of Ruptured Arteriovenous Brain Malformations in a Tertiary Referral Center in Colombia, South America. World Neurosurg. 2024, 191, e107–e115. [Google Scholar] [CrossRef]
- Musmar, B.; Abdalrazeq, H.; Adeeb, N.; Roy, J.M.; Aslan, A.; Tjoumakaris, S.I.; Salim, H.A.; Ogilvy, C.S.; Baskaya, M.K.; Kondziolka, D.; et al. Outcomes of Arteriovenous Malformations with Single versus Multiple Draining Veins: A Multicenter Study. J. Neurol. Sci. 2025, 473, 123503. [Google Scholar] [CrossRef]
- Ma, L.; Chen, Y.; Chen, P.; Ma, L.; Yan, D.; Li, R.; Li, Z.; Zhang, H.; Han, H.; Yuan, K.; et al. Quantitative Hemodynamics of Draining Veins in Brain Arteriovenous Malformation: A Preliminary Study Based on Computational Fluid Dynamics. Front. Neurol. 2024, 15, 1474857. [Google Scholar] [CrossRef]
- Li, Z.; Han, H.; Ma, L.; Li, R.; Li, A.; Zhang, H.; Zhu, Q.; Yuan, K.; Wang, K.; Wang, C.; et al. Venous Aneurysms in Unruptured Supratentorial Brain Arteriovenous Malformations: A Protective Factor against Hemorrhagic Stroke and Insights into Hemodynamic Mechanisms. Eur. Radiol. 2025, 35, 2660–2669. [Google Scholar] [CrossRef]
- Jacobs, R.C.; Chilukuri, A.; Abou-Al-Shaar, H.; Garcia, J.H.; Agarwal, P.; McDowell, M.M.; Al-Bayati, A.R.; Greene, S. Arteriovenous Malformation-Associated Aneurysms in the Pediatric Population: The University of Pittsburgh Medical Center Experience. J. Neurosurg. Pediatr. 2025, 35, 79–84. [Google Scholar] [CrossRef]
- Troude, L.; Sahan, S.; Fischer, I.; Koeskemeier, P., Jr.; Faust, K.; Muhammad, S. Clinical and Radiological Outcome after Microsurgical Management of Ruptured and Unruptured Brain Arteriovenous Malformations. Neurosurg. Rev. 2025, 48, 632. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Cajiao, F.; Quispe-Alcócer, J.; Flores-Enderica, G.; Rodríguez-Cajiao, M.; González-Andrade, F. Arterial Afferent Count Is Associated with Arteriovenous Malformation Grade but Not with Rupture Risk in a South American Cohort. Discov. Neurosci. 2025, 20, 19. [Google Scholar] [CrossRef]
- Janicijevic, A.; Kostic, J.; Jovicevic, N.; Milosavljevic, A.; Vidovic, D.; Cancarevic-Janicijevic, M.; Repac, N.; Tasic, G. Predictors of Functional Outcome After Microsurgical Resection of Brain Arteriovenous Malformations: A Retrospective Single-Center Study. J. Clin. Med. 2025, 14, 8680. [Google Scholar] [CrossRef]
- Dobrocky, T.; Matzinger, M.; Piechowiak, E.I.; Kaesmacher, J.; Pilgram-Pastor, S.; Goldberg, J.; Bervini, D.; Klail, T.; Pereira, V.M.; Z’Graggen, W.; et al. Benefit of Advanced 3D DSA and MRI/CT Fusion in Neurovascular Pathology. Clin. Neuroradiol. 2023, 33, 669–676. [Google Scholar] [CrossRef]
- Ricciardelli, A.R.; Robledo, A.; Fish, J.E.; Kan, P.T.; Harris, T.H.; Wythe, J.D. The Role and Therapeutic Implications of Inflammation in the Pathogenesis of Brain Arteriovenous Malformations. Biomedicines 2023, 11, 2876. [Google Scholar] [CrossRef]
- Guo, H.; Liu, J.-F.; Li, C.-H.; Wang, J.-W.; Li, H.; Gao, B.-L. Greater Hemodynamic Stresses Initiate Aneurysms on Major Cerebral Arterial Bifurcations. Front. Neurol. 2023, 14, 1265484. [Google Scholar] [CrossRef]
- Bateman, G.A.; Bateman, A.R. A Lumped Parameter Modelling Study of Idiopathic Intracranial Hypertension Suggests the CSF Formation Rate Varies with the Capillary Transmural Pressure. Brain Sci. 2025, 15, 527. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Y.; Han, H.; Ma, L.; Li, R.; Li, Z.; Yan, D.; Zhang, H.; Yuan, K.; Wang, K.; et al. Timing of Microsurgical Resection for Ruptured Brain Arteriovenous Malformations: A Propensity Score-Matched Analysis Using Prospective Single-Center Registry Data. J. Neurosurg. 2024, 140, 164–171. [Google Scholar] [CrossRef]
- Zhang, C.; Ge, H.; Zhang, S.; Liu, D.; Jiang, Z.; Lan, C.; Li, L.; Feng, H.; Hu, R. Hematoma Evacuation via Image-Guided Para-Corticospinal Tract Approach in Patients with Spontaneous Intracerebral Hemorrhage. Neurol. Ther. 2021, 10, 1001–1013. [Google Scholar] [CrossRef] [PubMed]
- Miron, I.; Prună, V.M.; Visarion, D.; Giovani, A.; Sandu, A.M.; Brehar, F.M.; Petrescu, G.E.D.; Gorgan, R.M. Clinical Outcomes of Posterior Fossa Arteriovenous Malformations: A Single Center Experience. Acta Neurochir. 2024, 166, 215. [Google Scholar] [CrossRef]
- Chen, K.-W.; Su, I.-C.; Lee, J.-E.; Liu, H.-M. Treatment of Arteriovenous Malformation with High-Flow Fistula and Bilateral Transverse-Sigmoid Sinuses Stenosis Resulting Diffuse Cortical Vein Engorgement and Symptoms Resembling Carotid-Cavernous Fistula. Asian J. Neurosurg. 2015, 10, 310–312. [Google Scholar] [CrossRef]
- Shi, J.; Xu, S.; Feng, Y.; Wei, W.; Zou, Y.; Xiong, W.; Zhao, W.; Zhang, T.; Peng, H.; Chen, J. Predicting Intraoperative Major Blood Loss in Microsurgery for Brain Arteriovenous Malformations. Front. Med. 2024, 11, 1446088. [Google Scholar] [CrossRef] [PubMed]
- Le, N.; Li, Y.; Walker, G.; Nguyen, B.-N.; Bornak, A.; Deo, S.K.; Velazquez, O.C.; Liu, Z.-J. Arteriovenous Malformations (AVMs): Molecular Pathogenesis, Clinical Features, and Emerging Therapeutic Strategies. Biomolecules 2025, 15, 1661. [Google Scholar] [CrossRef] [PubMed]
- Salam, R.A.; Dunlop, K.L.A.; Gide, T.N.; Wilmott, J.; Smith, A.; Cust, A.E. Factors Associated with Implementation of Biomarker Testing and Strategies to Improve Its Clinical Uptake in Cancer Care: Systematic Review Using Theoretical Domains Framework. JCO Precis. Oncol. 2025, 9, e2500063. [Google Scholar] [CrossRef]
- Maalim, A.A.; Zhu, M.; Shu, K.; Wu, Y.; Zhang, S.; Ye, F.; Zeng, Y.; Huang, Y.; Lei, T. Microsurgical Treatment of Arteriovenous Malformations: A Single-Center Study Experience. Brain Sci. 2023, 13, 1183. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Hasegawa, H.; Shibuya, K.; Fujiwara, H.; Oishi, M. Clinical and Hemodynamic Features of Aneurysm Rupture in Coil Embolization of Intracranial Aneurysms. Diagnostics 2024, 14, 1203. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Lin, F.; Qiu, H.; Cao, Y.; Wang, S.; Zhao, J. Comparison of Endovascular Embolization Plus Simultaneous Microsurgical Resection vs. Primary Microsurgical Resection for High-Grade Brain Arteriovenous Malformations. Front. Neurol. 2021, 12, 756307. [Google Scholar] [CrossRef] [PubMed]
- Guest, W.; Krings, T. Brain Arteriovenous Malformations: The Role of Imaging in Treatment Planning and Monitoring Response. Neuroimaging Clin. 2021, 31, 205–222. [Google Scholar] [CrossRef]
- Pagola, J.; Rubiera, M.; Flores, A.; Rodríguez-Luna, D.; Piñeiro, S.; Muchada, M.A.; Quintana, M.; Alvarez-Sabin, J.; Molina, C.A.; Ribo, M. Selecting Endovascular Treatment Strategy According to the Location of Intracranial Occlusion in Acute Stroke. Cerebrovasc. Dis. 2013, 35, 502–506. [Google Scholar] [CrossRef]


| References | Design/Cohort | Key Population | Therapy | Outcomes | Practice-Relevant Notes |
|---|---|---|---|---|---|
| [12] | Retrospective surgical cohort | Adults with ruptured and unruptured bAVMs managed microsurgically (large single-center series) | Microsurgical resection (graded by SM/Ponce) | High cure rates for low-grade AVMs; functional outcome driven by grade, rupture status, venous anatomy | Confirms microsurgery as definitive modality for low-grade convexity AVMs, especially when a rupture corridor exists; supports your single-stage cure logic. |
| [13] | Comparative original study (early vs. delayed) | Ruptured bAVMs with ICH, stratified by timing | Early definitive surgery via hematoma corridor vs. delayed elective resection | Early resection not inferior in functional outcome when low-grade and surgically accessible; avoids rebleed window | Directly reinforces your corridor-first strategy: decompression converts pressure emergency into safe anatomical dissection without waiting for “cool-down.” |
| [14] | Multicenter adult cohort | AVMs with single vs. multiple draining veins | Multimodal management tracked by angioarchitecture | Single-drainage AVMs show higher hemorrhagic presentation and worse venous hemodynamics | Matches your case constraint: a solitary SSS vein is a pressure amplifier, not a benign simplification; mandates vein-last choreography. |
| [15] | Original CFD + angiographic hemodynamic study | Ruptured vs. unruptured AVMs with quantified venous flow parameters | Hemodynamic modeling of draining veins | Rupture associated with adverse venous profiles (higher shear heterogeneity/flow acceleration) | Supports the concept that rupture risk is venous-microphysics-linked, explaining catastrophic collapse despite small nidus size. |
| [16] | Prospective quantitative DSA registry | Supratentorial AVMs assessed with QDSA; focus on venous outflow lesions | Imaging-based risk stratification (no intervention assigned) | Venous aneurysms/outflow pathology correlate with hemorrhage and unstable transit patterns | Backs the idea that micro-compartment outflow behavior dominates rupture risk over nidus diameter; helpful when framing your dual-aneurysm/single-vein “high-risk-dense” topology. |
| [17] | Original cohort on AVM-associated aneurysms | AVMs with feeder/intranidal aneurysms (recent high-resolution series) | Natural history + treated subsets | Intranidal aneurysms strongly associate with hemorrhagic presentation and act as dominant weak points | Mechanistic foundation for your case’s dual intranidal aneurysms being the rupture substrate even in an 8 × 3 mm nidus. |
| [18] | Single-center original surgical outcomes study | Consecutive intracranial AVM resections, majority ruptured | Microsurgical management with modern intraop verification | Favorable long-term mRS for low-grade ruptured AVMs; low permanent morbidity when venous preservation respected | Emphasizes the same controllable variables you executed: feeder-first devascularization, en-passage preservation, delayed venous division. |
| [19] | Original morphologic predictor cohort | Adult bAVMs stratified by arterial afferent count + venous drainage | Imaging-risk association | Higher afferent complexity and adverse venous patterns independently associate with rupture | Gives a modern quantitative lens to describe why even compact AVMs can harbor rupture-grade energy, complementing your bedside-to-angiography logic. |
| [20] | Original predictive microsurgical series | Low-grade bAVMs resected after hemorrhage | Microsurgical resection; predictors of outcome | Favorable mRS mainly determined by grade, compactness, superficial drainage, and corridor availability | Cleanly aligns with your risk-benefit framing for immediate cure in a superficial posterior nidus. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Breazu, A.; Oprea, S.; Dobrin, N.; Diaconescu, I.B.; Munteanu, O.; Șerban, M.; Covache-Busuioc, R.-A.; Toader, C.; Rădoi, M.P.; Pantu, C. Occipital Pial AVM Rupture in a Young Adult: Dual Intranidal Aneurysms, Solitary Parasagittal SSS Drainage, and Hematoma-Corridor Microsurgical Cure. Diagnostics 2026, 16, 265. https://doi.org/10.3390/diagnostics16020265
Breazu A, Oprea S, Dobrin N, Diaconescu IB, Munteanu O, Șerban M, Covache-Busuioc R-A, Toader C, Rădoi MP, Pantu C. Occipital Pial AVM Rupture in a Young Adult: Dual Intranidal Aneurysms, Solitary Parasagittal SSS Drainage, and Hematoma-Corridor Microsurgical Cure. Diagnostics. 2026; 16(2):265. https://doi.org/10.3390/diagnostics16020265
Chicago/Turabian StyleBreazu, Alexandru, Stefan Oprea, Nicolaie Dobrin, Ionut Bogdan Diaconescu, Octavian Munteanu, Matei Șerban, Răzvan-Adrian Covache-Busuioc, Corneliu Toader, Mugurel Petrinel Rădoi, and Cosmin Pantu. 2026. "Occipital Pial AVM Rupture in a Young Adult: Dual Intranidal Aneurysms, Solitary Parasagittal SSS Drainage, and Hematoma-Corridor Microsurgical Cure" Diagnostics 16, no. 2: 265. https://doi.org/10.3390/diagnostics16020265
APA StyleBreazu, A., Oprea, S., Dobrin, N., Diaconescu, I. B., Munteanu, O., Șerban, M., Covache-Busuioc, R.-A., Toader, C., Rădoi, M. P., & Pantu, C. (2026). Occipital Pial AVM Rupture in a Young Adult: Dual Intranidal Aneurysms, Solitary Parasagittal SSS Drainage, and Hematoma-Corridor Microsurgical Cure. Diagnostics, 16(2), 265. https://doi.org/10.3390/diagnostics16020265






