A Comparative Study of Rapid Fresh Pathology Imaging and Standard FFPE H&E Histopathology: A High Concordance in the Evaluation of Lung and Breast Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Tissue Preparation
2.2. Optical Imaging via PATHOscope
2.3. Validation of RFP Imaging Against Standard FFPE H&E-Stained Slides for Pathological Evaluation
3. Results
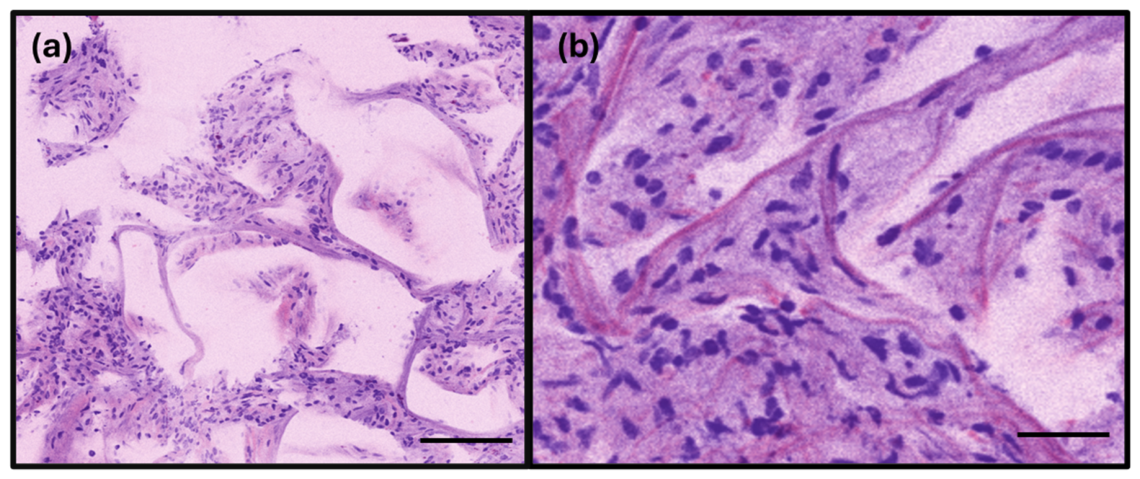
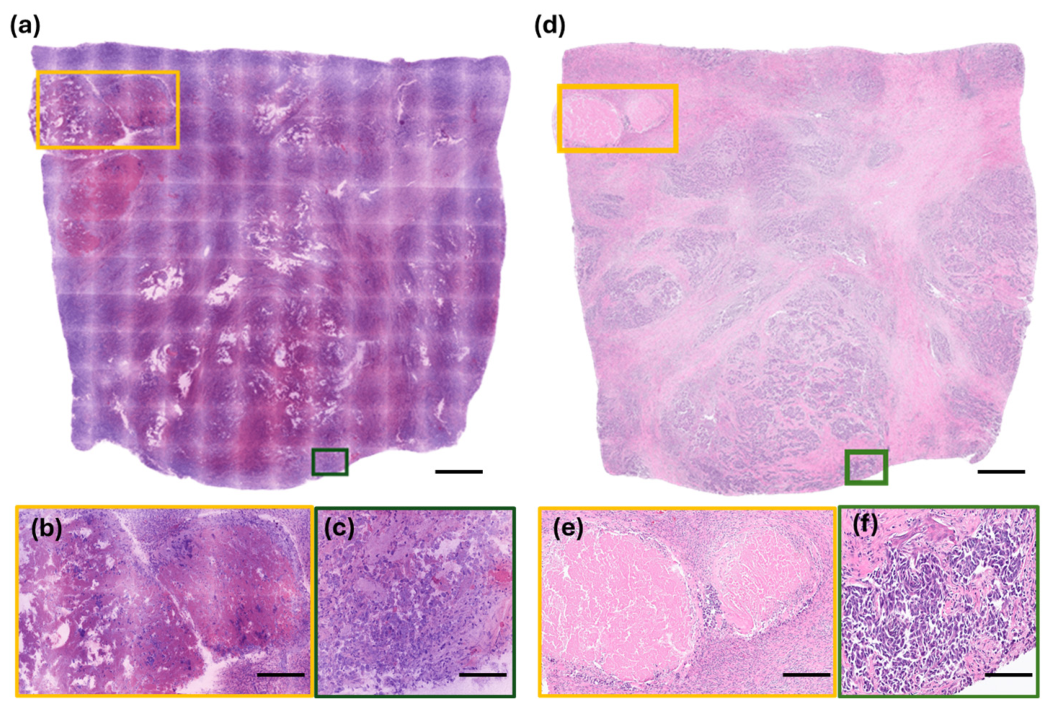
3.1. RFP Results of Lung Specimens
3.2. RFP Results of Breast Specimens
3.3. Rapid Fresh Pathology (RFP) as a High-Concordance Alternative to FFPE H&E Histopathology for Lung and Breast Cancer Diagnosis
4. Discussion
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Raab, S.S.; Grzybicki, D.M. Quality in cancer diagnosis. CA Cancer J. Clin. 2010, 60, 139–165. [Google Scholar] [CrossRef] [PubMed]
- Azari, F.; Kennedy, G.; Singhal, S. Intraoperative detection and assessment of lung nodules. Surg. Oncol. Clin. N. Am. 2020, 29, 525–541. [Google Scholar] [CrossRef] [PubMed]
- Brenner, A.T.; Malo, T.L.; Margolis, M.; Elston Lafata, J.; James, S.; Vu, M.B.; Reuland, D.S. Evaluating shared decision making for lung cancer screening. JAMA Netw. Open. 2018, 1, e180032. [Google Scholar] [CrossRef] [PubMed]
- Christofidis, K.; Pergaris, A.; Kalfa, M.; Klapsinou, E.; Kyriakidou, V.; Chodrodimou, Z.; Savvidou, K.; Terzi, M.; Stamou, C.; Mallios, D.; et al. Intraoperative rapid on-site evaluation of lung tumors: A diagnostic accuracy study. Acta Cytol. 2025, 69, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Li, X. Development of intraoperative assessment of margins in breast conserving surgery: A narrative review. Gland Surg. 2022, 11, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, V.; Khalid, U.; Dimov, R. Margin matters: Advances in intraoperative margin assessment for breast-conserving surgery. Diagnostics 2025, 15, 2804. [Google Scholar] [CrossRef] [PubMed]
- Rana, M.K.; Rana, A.P.; Sharma, U.; Barwal, T.S.; Jain, A. Evolution of frozen section in carcinoma breast: Systematic review. J. Oncol. 2022, 2022, 4958580. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, B.A.; Engrav, S.K.; Roden, A.C.; Boland, J.M.; Aubry, M.C.; Abdallah, F.A.; Yi, E.S.; Saddoughi, S.A.; Cassivi, S.D.; Wigle, D.A.; et al. Impact of frozen section pathology examination of surgical margins in sublobar pulmonary resections for clinical stage IA non-small-cell lung cancer. Ann. Thorac. Surg. 2025, 120, 1044–1051. [Google Scholar] [CrossRef] [PubMed]
- Mahe, E.; Ara, S.; Bishara, M.; Kurian, A.; Tauqir, S.; Ursani, N.; Vasudev, P.; Aziz, T.; Ross, C.; Lytwyn, A. Intraoperative pathology consultation error, cause and impact. Can. J. Surg. 2013, 56, E13. [Google Scholar] [CrossRef] [PubMed]
- Voskuil, F.J.; Vonk, J.; van der Vegt, B.; Kruijff, S.; Ntziachristos, V.; van der Zaag, P.J.; Witjes, M.J.; van Dam, G.M. Intraoperative imaging in pathology-assisted surgery. Nat. Biomed. Eng. 2022, 6, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huang, B.; Wu, J.; Wong, T.T.W. Advances in optical microscopy revolutionize the practice of surgical pathology with rapid and non-destructive tissue assessment. Eur. Phys. J. Spec. Top. 2022, 231, 763–779. [Google Scholar] [CrossRef]
- Lopez, D.R.; Sgroi, D.; Krishnamourthy, S.; Tearney, G. Is real-time microscopy on the horizon? A brief review of the potential future directions in clinical breast tumor microscopy implementation. Virchows Arch. 2022, 480, 211–227. [Google Scholar] [CrossRef] [PubMed]
- Borah, B.J.; Tseng, Y.C.; Wang, K.C.; Wang, H.C.; Huang, H.Y.; Chang, K.; Lin, J.R.; Liao, Y.H.; Sun, C.K. Rapid digital pathology of H&E-stained fresh human brain specimens as an alternative to frozen biopsy. Commun. Med. 2023, 3, 77. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.-K.; Lee, Y.-H.; Lien, H.-C.; Wang, M.-Y. A comparative study on H&E-based rapid fresh digital pathology and frozen section analysis for intraoperative margin assessment in breast-conserving surgery. Lab. Investig. 2025, 105, 102454. [Google Scholar] [CrossRef]
- Tseng, Y.C.; Hsieh, M.S.; Lu, Y.T.; Huang, C.Y.; Lee, Y.H.; Chen, P.H.; Chuang, J.H.; Tsai, T.M.; Liao, Y.H.; Hsu, H.H.; et al. Accelerating precision diagnosis in CT-guided lung biopsies using rapid fresh pathology. Lab. Investig. 2026, 106, 104807. [Google Scholar] [CrossRef]
- Hsieh, M.S.; Tseng, Y.C.; Lu, Y.T.; Huang, C.Y.; Lee, Y.H.; Chen, P.H.; Chuang, J.H.; Tsai, T.M.; Hsu, H.H.; Liao, Y.H.; et al. Rapid fresh pathology enables subtype-specific intraoperative diagnosis of lung cancer within 10 minutes. Lab. Investig. 2026, 106, 104762. [Google Scholar] [CrossRef]
- Giacomelli, M.G.; Husvogt, L.; Vardeh, H.; Faulkner-Jones, B.E.; Hornegger, J.; Connolly, J.L.; Fujimoto, J.G. Virtual Hematoxylin and Eosin Transillumination Microscopy Using Epi-Fluorescence Imaging. PLoS ONE 2016, 11, e0159337. [Google Scholar] [CrossRef] [PubMed]





| Specimen Type | Lung | Breast | Total |
|---|---|---|---|
| Patients (n) | 10 | 10 | 20 |
| Tumor part (n) | 53 | 34 | 87 |
| Non-tumor part (n) | 24 | 16 | 40 |
| Total specimens (n) | 77 | 50 | 127 |
| Specimen Type | Pathologist 1 | Pathologist 2 | ||
|---|---|---|---|---|
| Lung | FFPE Benign | FFPE Malignant | FFPE Benign | FFPE Malignant |
| RFP Benign | 24 | 0 | 24 | 0 |
| RFP Malignant | 0 | 53 | 0 | 53 |
| Breast | ||||
| RFP Benign | 16 | 0 | 16 | 0 |
| RFP Malignant | 0 | 34 | 0 | 34 |
| Total | 40 | 87 | 40 | 87 |
| Parameter | Rapid Fresh Pathology (RFP) | Formalin-Fixed Paraffin-Embedded Pathology (FFPE) |
|---|---|---|
| Sample preparation | No freezing or physical sectioning required | Requires fixation, embedding, sectioning |
| Image acquisition | Optical imaging (no physical sectioning) | Physical thin section |
| Tissue integrity | Preserved (no cutting artifacts) | High structural fidelity |
| Architectural preservation | Excellent | Excellent |
| Clinical application | Rapid assessment | Gold-standard diagnosis |
| Limitations | Reduced cytological clarity in some cases | Long processing time |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Hsieh, M.-S.; Tseng, Y.-C.; Huang, C.-Y.; Wang, H.; Lee, Y.-H.; Lien, H.-C.; Huang, H.-Y.; Chang, K.; Wang, H.-C.; Wang, K.-C.; et al. A Comparative Study of Rapid Fresh Pathology Imaging and Standard FFPE H&E Histopathology: A High Concordance in the Evaluation of Lung and Breast Cancer. Diagnostics 2026, 16, 1503. https://doi.org/10.3390/diagnostics16101503
Hsieh M-S, Tseng Y-C, Huang C-Y, Wang H, Lee Y-H, Lien H-C, Huang H-Y, Chang K, Wang H-C, Wang K-C, et al. A Comparative Study of Rapid Fresh Pathology Imaging and Standard FFPE H&E Histopathology: A High Concordance in the Evaluation of Lung and Breast Cancer. Diagnostics. 2026; 16(10):1503. https://doi.org/10.3390/diagnostics16101503
Chicago/Turabian StyleHsieh, Min-Shu, Yao-Chen Tseng, Chung-Yen Huang, Hsuan Wang, Yi-Hsuan Lee, Huang-Chun Lien, Hsin-Yi Huang, Koping Chang, Huan-Chih Wang, Kuo-Chuan Wang, and et al. 2026. "A Comparative Study of Rapid Fresh Pathology Imaging and Standard FFPE H&E Histopathology: A High Concordance in the Evaluation of Lung and Breast Cancer" Diagnostics 16, no. 10: 1503. https://doi.org/10.3390/diagnostics16101503
APA StyleHsieh, M.-S., Tseng, Y.-C., Huang, C.-Y., Wang, H., Lee, Y.-H., Lien, H.-C., Huang, H.-Y., Chang, K., Wang, H.-C., Wang, K.-C., Wang, M.-Y., Liao, Y.-H., Sun, C.-K., & Chen, J.-S. (2026). A Comparative Study of Rapid Fresh Pathology Imaging and Standard FFPE H&E Histopathology: A High Concordance in the Evaluation of Lung and Breast Cancer. Diagnostics, 16(10), 1503. https://doi.org/10.3390/diagnostics16101503





