Bridging the Diagnostic Gap in Peripheral Arterial Disease (PAD): Leveraging Fatty Acid Binding Protein 3 (FABP3) for Biomarker-Guided Detection
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Quantification of FABP3 Levels and Analytical Methodology
2.3. Previously Identified Cutoff Points in PAD Diagnosis
2.4. Statistical Analysis
3. Results
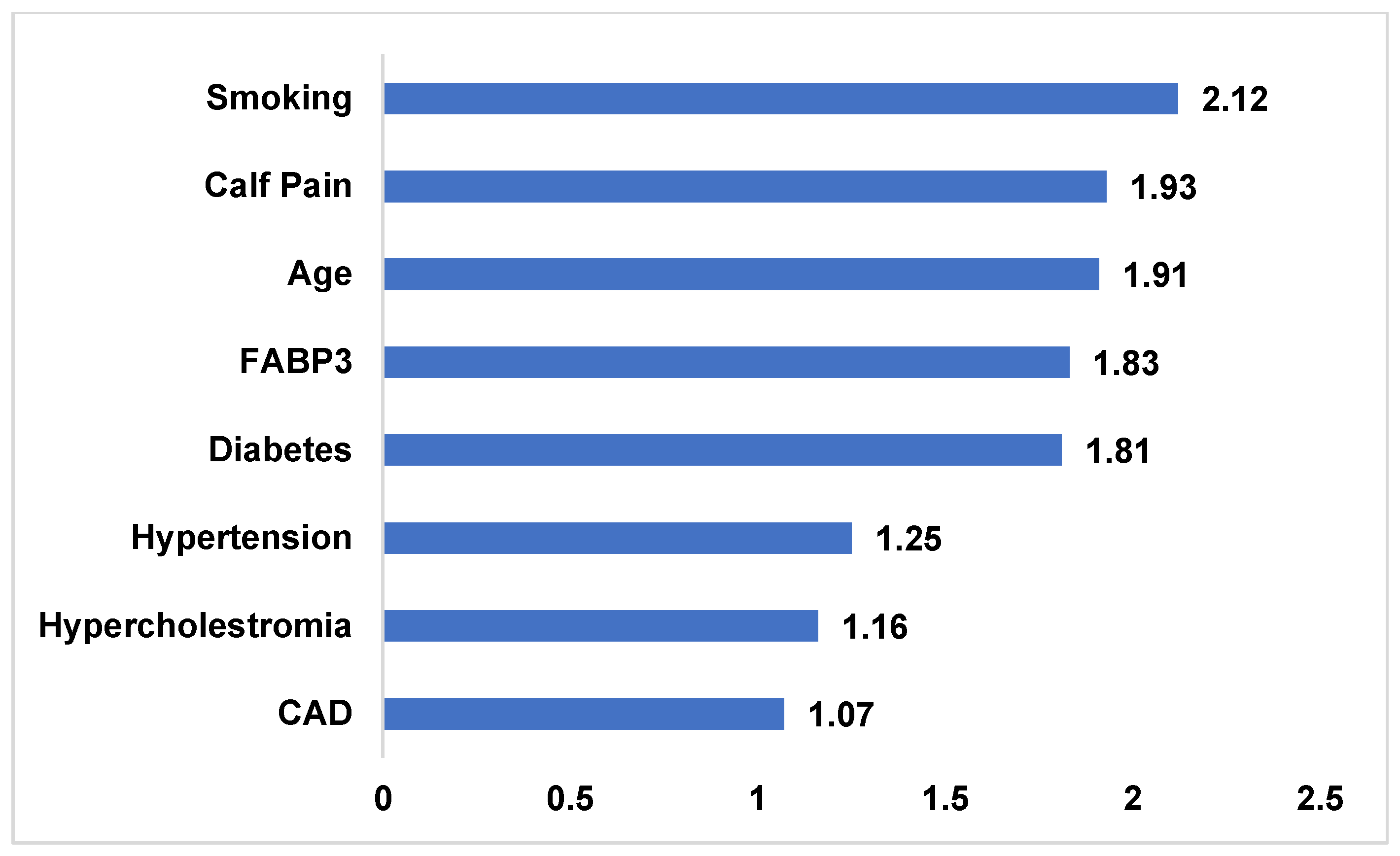
3.1. Model Performance: Clinical Risk Factors vs. FABP3-Integrated Model
3.2. Subgroup Analyses: Asymptomatic vs. Symptomatic PAD
3.3. Grey Zone Analysis (FABP3 1.55–3.55 ng/mL)
4. Discussion
4.1. FABP3 as a Biomarker for PAD
4.2. Clinical Implications
4.3. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fowkes, F.G.; Rudan, D.; Rudan, I.; Aboyans, V.; Denenberg, J.O.; McDermott, M.M.; Norman, P.E.; Sampson, U.K.; Williams, L.J.; Mensah, G.A.; et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: A systematic review and analysis. Lancet 2013, 382, 1329–1340. [Google Scholar] [CrossRef] [PubMed]
- Criqui, M.H.; Aboyans, V. Epidemiology of Peripheral Artery Disease. Circ. Res. 2015, 116, 1509–1526. [Google Scholar] [CrossRef] [PubMed]
- McDermott, M.M.; Mehta, S.; Ahn, H.; Greenland, P. Atherosclerotic risk factors are less intensively treated in patients with peripheral arterial disease than in patients with coronary artery disease. J. Gen. Intern. Med. 1997, 12, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Aboyans, V.; Criqui, M.H.; Abraham, P.; Allison, M.A.; Creager, M.A.; Diehm, C.; Fowkes, F.G.; Hiatt, W.R.; Jönsson, B.; Lacroix, P.; et al. Measurement and interpretation of the ankle-brachial index: A scientific statement from the American Heart Association. Circulation 2012, 126, 2890–2909. [Google Scholar] [CrossRef]
- American Diabetes Association. Peripheral arterial disease in people with diabetes. Diabetes Care 2003, 26, 3333–3341. [Google Scholar] [CrossRef]
- Collins, R.; Cranny, G.; Burch, J.; Aguiar-Ibáñez, R.; Craig, D.; Wright, K.; Berry, E.; Gough, M.; Kleijnen, J.; Westwood, M. A systematic review of duplex ultrasound, magnetic resonance angiography and computed tomography angiography for the diagnosis and assessment of symptomatic, lower limb peripheral arterial disease. Health Technol. Assess. (Winch. Engl.) 2007, 11, 3–184. [Google Scholar] [CrossRef]
- Chiu, L.Y.; Syed, M.H.; Zamzam, A.; Rotstein, O.D.; Abdin, R.; Laraya, N.; Qadura, M. Perceived challenges to routine uptake of the ankle brachial index within primary care practice. J. Clin. Med. 2021, 10, 4371. [Google Scholar] [CrossRef]
- Thygesen, K.; Mair, J.; Giannitsis, E.; Mueller, C.; Lindahl, B.; Blankenberg, S.; Huber, K.; Plebani, M.; Biasucci, L.M.; Tubaro, M.; et al. How to use high-sensitivity cardiac troponins in acute cardiac care. Eur. Heart J. 2012, 33, 2252–2257. [Google Scholar] [CrossRef]
- Daniels, L.B.; Maisel, A.S. Natriuretic peptides. J. Am. Coll. Cardiol. 2007, 50, 2357–2368. [Google Scholar] [CrossRef]
- Melnikov, I.; Kozlov, S.; Saburova, O.; Avtaeva, Y.; Guria, K.; Gabbasov, Z. Monomeric C-Reactive Protein in Atherosclerotic Cardiovascular Disease: Advances and Perspectives. Int. J. Mol. Sci. 2023, 24, 2079. [Google Scholar] [CrossRef]
- Veerkamp, J.; Paulussen, R.; Peeters, R.; Maatman, R.; Van Moerkerk, H.; Van Kuppevelt, T. Detection, tissue distribution and (sub) cellular localization of fatty acid-binding protein types. Mol. Cell. Biochem. 1990, 98, 11–18. [Google Scholar] [CrossRef]
- Besnard, P.; Niot, I.; Poirier, H.; Clément, L.; Bernard, A. New insights into the fatty acid-binding protein (FABP) family in the small intestine. Mol. Cell. Biochem. 2002, 239, 139–147. [Google Scholar] [CrossRef]
- Glatz, J.F.; van der Vusse, G.J. Cellular fatty acid-binding proteins: Their function and physiological significance. Prog. Lipid Res. 1996, 35, 243–282. [Google Scholar] [CrossRef]
- Otaki, Y.; Watanabe, T.; Kubota, I. Heart-type fatty acid-binding protein in cardiovascular disease: A systemic review. Clin. Chim. Acta Int. J. Clin. Chem. 2017, 474, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Goel, H.; Melot, J.; Krinock, M.D.; Kumar, A.; Nadar, S.K.; Lip, G.Y.H. Heart-type fatty acid-binding protein: An overlooked cardiac biomarker. Ann. Med. 2020, 52, 444–461. [Google Scholar] [CrossRef] [PubMed]
- Warren, W.; Osborn, M.; Yates, A.; O’Sullivan, S. The emerging role of fatty acid-binding protein 3 (FABP3) in cancer. Drug Discov. Today 2025, 30, 104504. [Google Scholar] [CrossRef] [PubMed]
- Ishii, J.; Ozaki, Y.; Lu, J.; Kitagawa, F.; Kuno, T.; Nakano, T.; Nakamura, Y.; Naruse, H.; Mori, Y.; Matsui, S. Prognostic value of serum concentration of heart-type fatty acid–binding protein relative to cardiac troponin T on admission in the early hours of acute coronary syndrome. Clin. Chem. 2005, 51, 1397–1404. [Google Scholar] [CrossRef]
- Ye, X.-D.; He, Y.; Wang, S.; Wong, G.T.; Irwin, M.G.; Xia, Z. Heart-type fatty acid binding protein (H-FABP) as a biomarker for acute myocardial injury and long-term post-ischemic prognosis. Acta Pharmacol. Sin. 2018, 39, 1155–1163. [Google Scholar] [CrossRef]
- Thumser, A.E.; Moore, J.B.; Plant, N.J. Fatty acid binding proteins: Tissue-specific functions in health and disease. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 124–129. [Google Scholar] [CrossRef]
- Pritt, M.L.; Hall, D.G.; Recknor, J.; Credille, K.M.; Brown, D.D.; Yumibe, N.P.; Schultze, A.E.; Watson, D.E. Fabp3 as a biomarker of skeletal muscle toxicity in the rat: Comparison with conventional biomarkers. Toxicol. Sci. 2008, 103, 382–396. [Google Scholar] [CrossRef]
- Zamzam, A.; Syed, M.H.; Rotstein, O.D.; Eikelboom, J.; Klein, D.J.; Singh, K.K.; Abdin, R.; Qadura, M. Validating fatty acid binding protein 3 as a diagnostic and prognostic biomarker for peripheral arterial disease: A three-year prospective follow-up study. EClinicalMedicine 2023, 55, 101766. [Google Scholar] [CrossRef]
- Zamzam, A.; Syed, M.H.; Harlock, J.; Eikelboom, J.; Singh, K.K.; Abdin, R.; Qadura, M. Urinary fatty acid binding protein 3 (uFABP3) is a potential biomarker for peripheral arterial disease. Sci. Rep. 2021, 11, 11061. [Google Scholar] [CrossRef]
- Ridker, P.M. Clinical application of C-reactive protein for cardiovascular disease detection and prevention. Circulation 2003, 107, 363–369. [Google Scholar] [CrossRef]
- Li, B.; Syed, M.H.; Qadura, M. Increasing Awareness for Peripheral Artery Disease through the Identification of Novel Biomarkers. Biomolecules 2023, 13, 1189. [Google Scholar] [CrossRef]
- Glatz, J.F.; Schaap, F.G.; Binas, B.; Bonen, A.; van der Vusse, G.J.; Luiken, J.J. Cytoplasmic fatty acid-binding protein facilitates fatty acid utilization by skeletal muscle. Acta Physiol. Scand. 2003, 178, 367–371. [Google Scholar] [CrossRef]
- Pelsers, M.M.; Hermens, W.T.; Glatz, J.F. Fatty acid-binding proteins as plasma markers of tissue injury. Clin. Chim. Acta Int. J. Clin. Chem. 2005, 352, 15–35. [Google Scholar] [CrossRef]
- Li, B.; Syed, M.H.; Khan, H.; Singh, K.K.; Qadura, M. The Role of Fatty Acid Binding Protein 3 in Cardiovascular Diseases. Biomedicines 2022, 10, 2283. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, A.T.; Haskal, Z.J.; Hertzer, N.R.; Bakal, C.W.; Creager, M.A.; Halperin, J.L.; Hiratzka, L.F.; Murphy, W.R.; Olin, J.W.; Puschett, J.B.; et al. ACC/AHA 2005 Practice Guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): A collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients With Peripheral Arterial Disease): Endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; TransAtlantic Inter-Society Consensus; and Vascular Disease Foundation. Circulation 2006, 113, e463–e654. [Google Scholar] [PubMed]




| Category | Criteria |
|---|---|
| Inclusion Criteria—PAD group | ABI < 0.9 and evidence of atherosclerotic disease on duplex ultrasound, with reduced peripheral pulses in at least one leg, with or without claudication |
| Inclusion Criteria—Non-PAD group | ABI ≥ 0.9, palpable distal pulses, and no history of claudication |
| Special diagnostic criterion for non-compressible vessels | For patients with erroneous ABI due to non-compressible tibial vessels (ABI > 1.4), TBI was used instead |
| PAD definition using TBI | TBI < 0.7 was considered diagnostic of PAD |
| Exclusion Criteria | Chronic kidney disease stage 3–5 |
| Exclusion Criteria | Acute coronary syndrome, stroke, or transient ischemic attack within the past 30 days |
| Exclusion Criteria | Elevated troponin levels |
| Characteristic | Overall (n = 657) | Non-PAD (n = 234) | PAD (n = 423) | p-Value |
|---|---|---|---|---|
| Age, mean (SD) | 66 (12) | 60 (13) | 69 (10) | <0.001 |
| Male | 417 (64) | 138 (59) | 279 (66) | 0.069 |
| Female | 239 (36) | 96 (41) | 143 (34) | |
| Hypertension | 442 (67) | 117 (50) | 325 (77) | <0.001 |
| Dyslipidemia | 456 (69) | 114 (49) | 342 (81) | <0.001 |
| Diabetes | 235 (36) | 38 (16) | 197 (47) | <0.001 |
| Past smoking | 313 (48) | 89 (38) | 224 (53) | <0.001 |
| Current smoking | 168 (26) | 39 (17) | 129 (31) | <0.001 |
| History of congestive heart failure | 15 (2) | 5 (2) | 10 (2) | 0.085 |
| History of coronary artery disease | 182 (28) | 42 (18) | 140 (33) | < 0.001 |
| History of stroke | 63 (10) | 10 (5) | 53 (13) | <0.001 |
| Chronic Limb Threatening Ischemia (CLTI) | 43 (7) | 0 (0) | 43 (10) | <0.001 |
| Leg pain | 484 (74) | 143 (61) | 341 (81) | <0.001 |
| N = 657 | TP | TN | FP | FN | Sensitivity | Specificity | LR+ | LR− | PPV | NPV | Diagnostic Accuracy |
|---|---|---|---|---|---|---|---|---|---|---|---|
| ModelA (Clinical Risk Factors) | 341 | 156 | 78 | 82 | 81% | 67% | 2.42 | 0.29 | 81% | 65% | 76% |
| Model B (Clinical Risk Factors + FABP3) | 410 | 197 | 37 | 13 | 96% | 84% | 6.16 | 0.04 | 92% | 94% | 93% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Syed, M.H.; Zamzam, A.; Shaikh, F.; Rotstein, O.D.; Klein, D.J.; Younes, H.; Abdin, R.; Qadura, M. Bridging the Diagnostic Gap in Peripheral Arterial Disease (PAD): Leveraging Fatty Acid Binding Protein 3 (FABP3) for Biomarker-Guided Detection. Diagnostics 2026, 16, 1457. https://doi.org/10.3390/diagnostics16101457
Syed MH, Zamzam A, Shaikh F, Rotstein OD, Klein DJ, Younes H, Abdin R, Qadura M. Bridging the Diagnostic Gap in Peripheral Arterial Disease (PAD): Leveraging Fatty Acid Binding Protein 3 (FABP3) for Biomarker-Guided Detection. Diagnostics. 2026; 16(10):1457. https://doi.org/10.3390/diagnostics16101457
Chicago/Turabian StyleSyed, Muzammil H., Abdelrahman Zamzam, Farah Shaikh, Ori D. Rotstein, David J. Klein, Houssam Younes, Rawand Abdin, and Mohammad Qadura. 2026. "Bridging the Diagnostic Gap in Peripheral Arterial Disease (PAD): Leveraging Fatty Acid Binding Protein 3 (FABP3) for Biomarker-Guided Detection" Diagnostics 16, no. 10: 1457. https://doi.org/10.3390/diagnostics16101457
APA StyleSyed, M. H., Zamzam, A., Shaikh, F., Rotstein, O. D., Klein, D. J., Younes, H., Abdin, R., & Qadura, M. (2026). Bridging the Diagnostic Gap in Peripheral Arterial Disease (PAD): Leveraging Fatty Acid Binding Protein 3 (FABP3) for Biomarker-Guided Detection. Diagnostics, 16(10), 1457. https://doi.org/10.3390/diagnostics16101457










