Functioning and Safety of the Non-Invasive Corneal Esthesiometer Brill: A Multicenter Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Subjects
2.2. Primary and Secondary Endpoints
2.3. Variables Assessed
2.4. Study Visits and Procedures
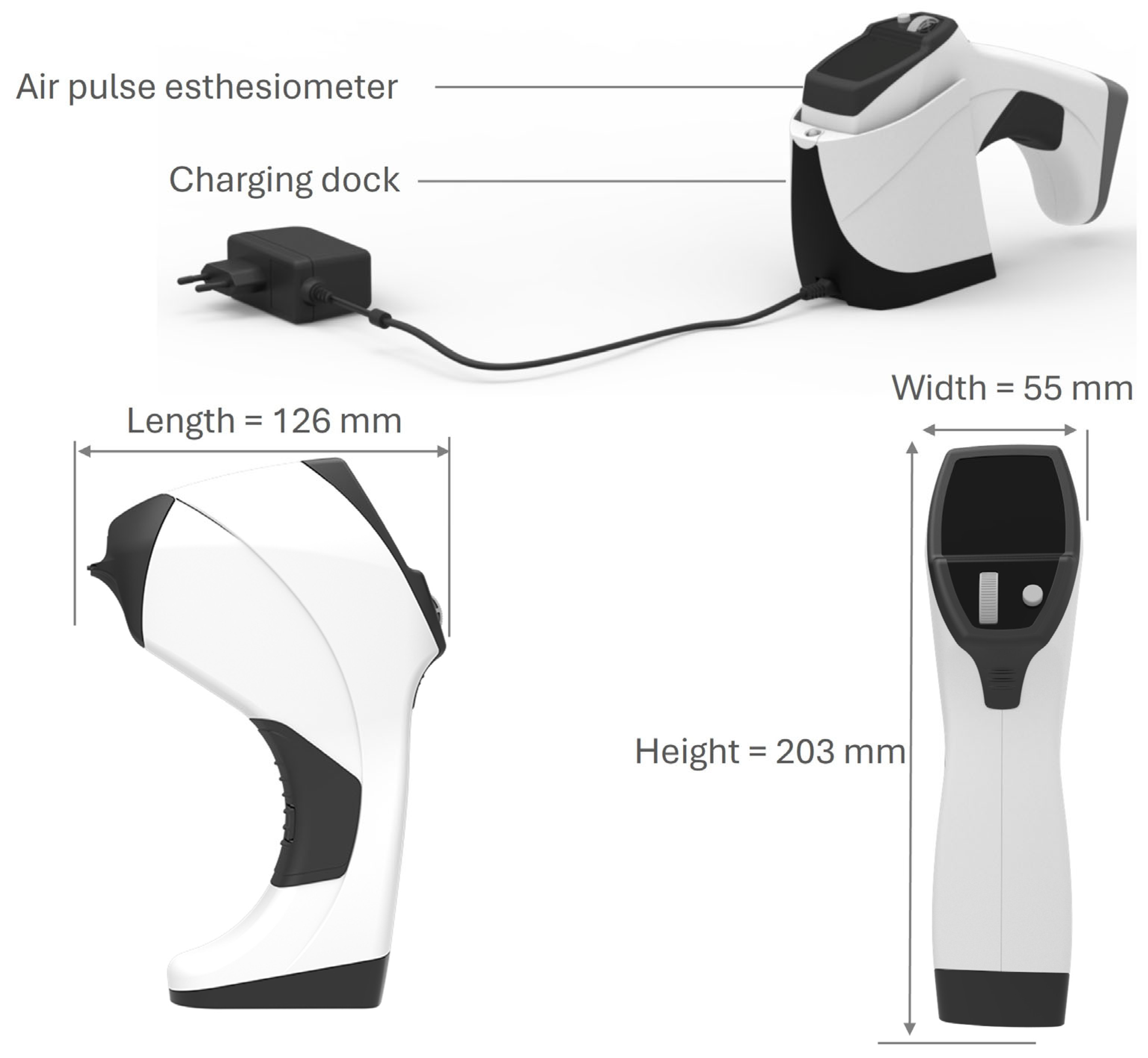
2.5. Corneal Sensitivity Assessment with the CEB
2.6. Safety Assessments
2.7. Sample Size
2.8. Data Processing and Statistical Analysis
3. Results
3.1. Participant Characteristics
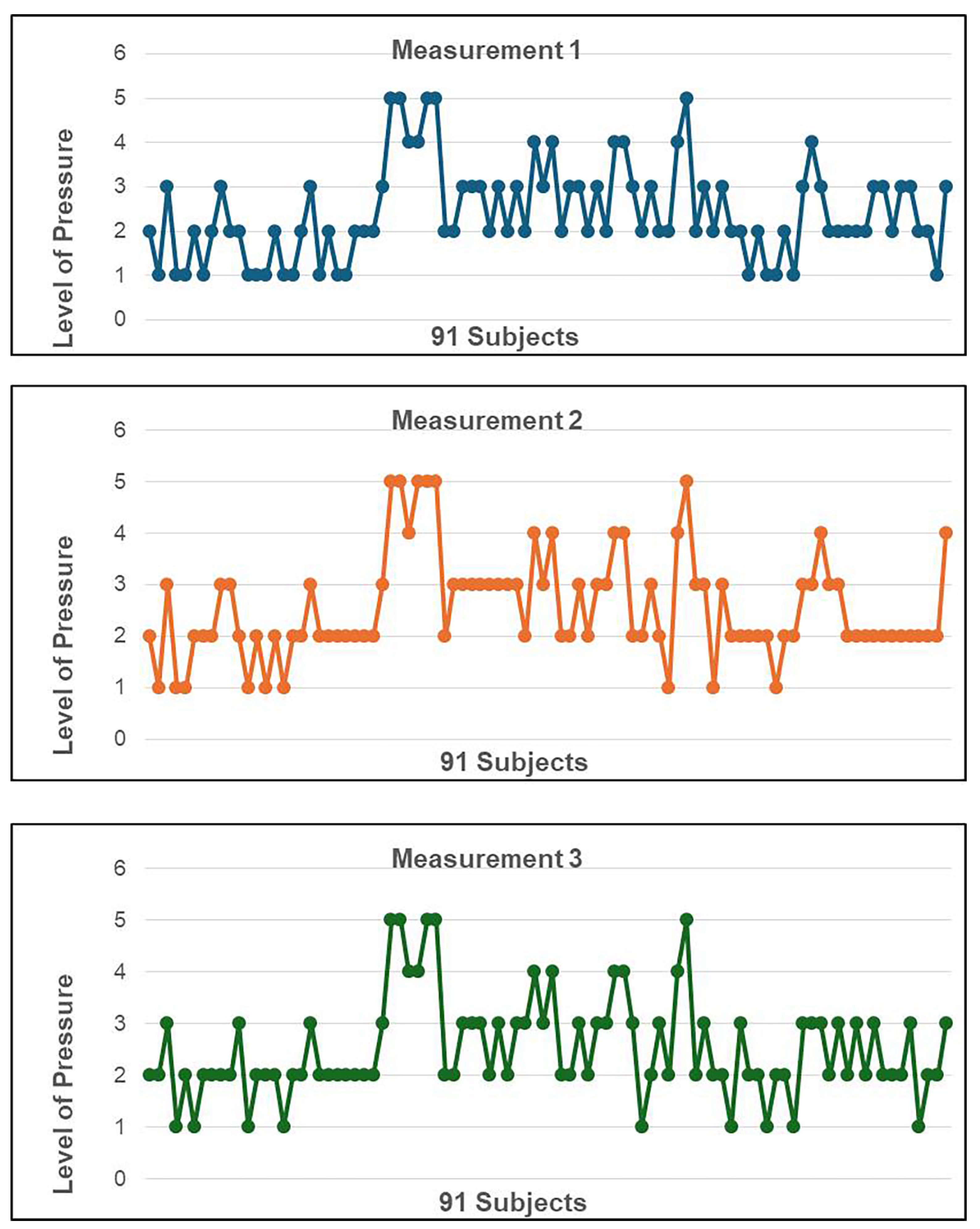
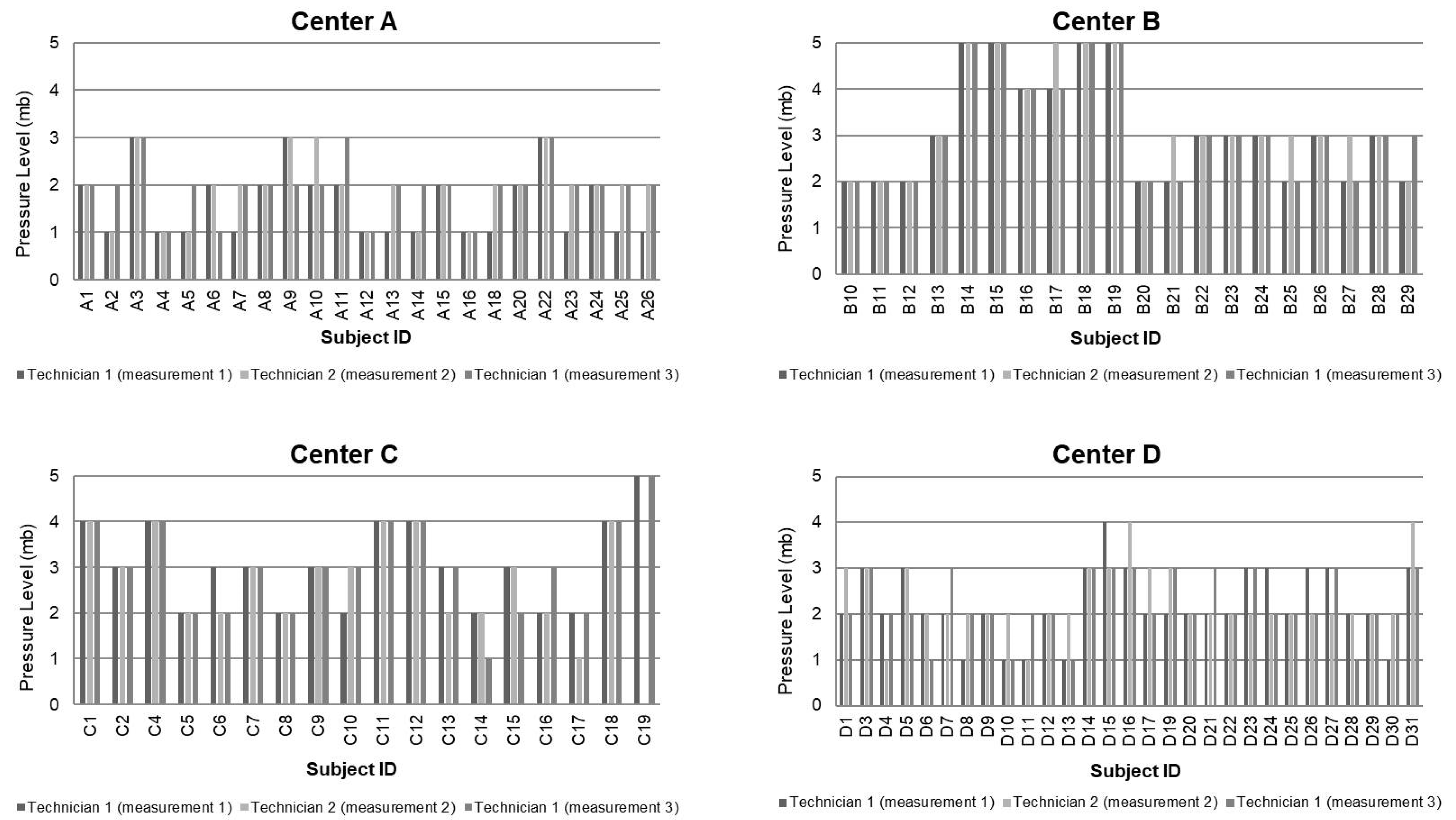
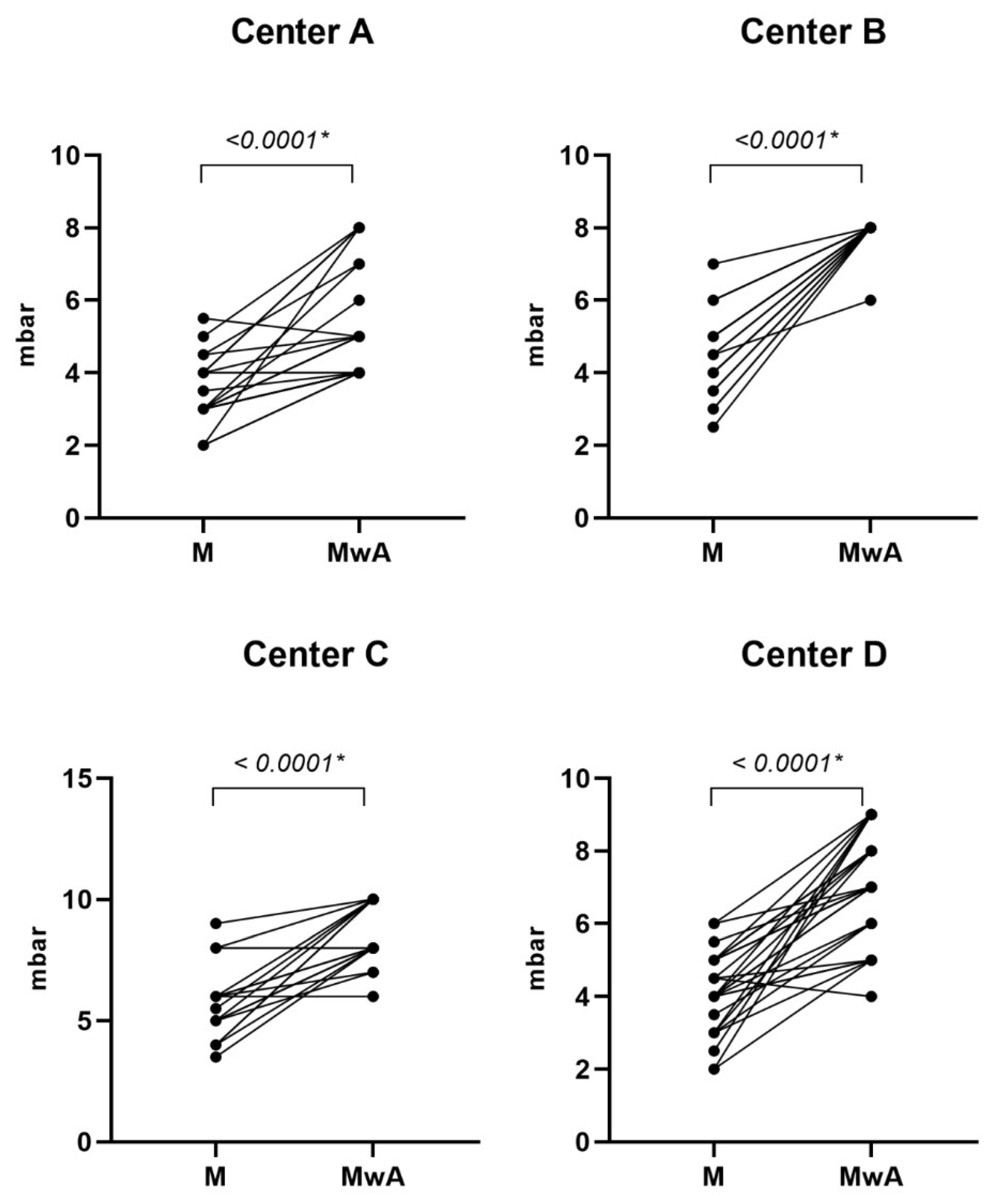
3.2. Primary Efficacy Endpoint
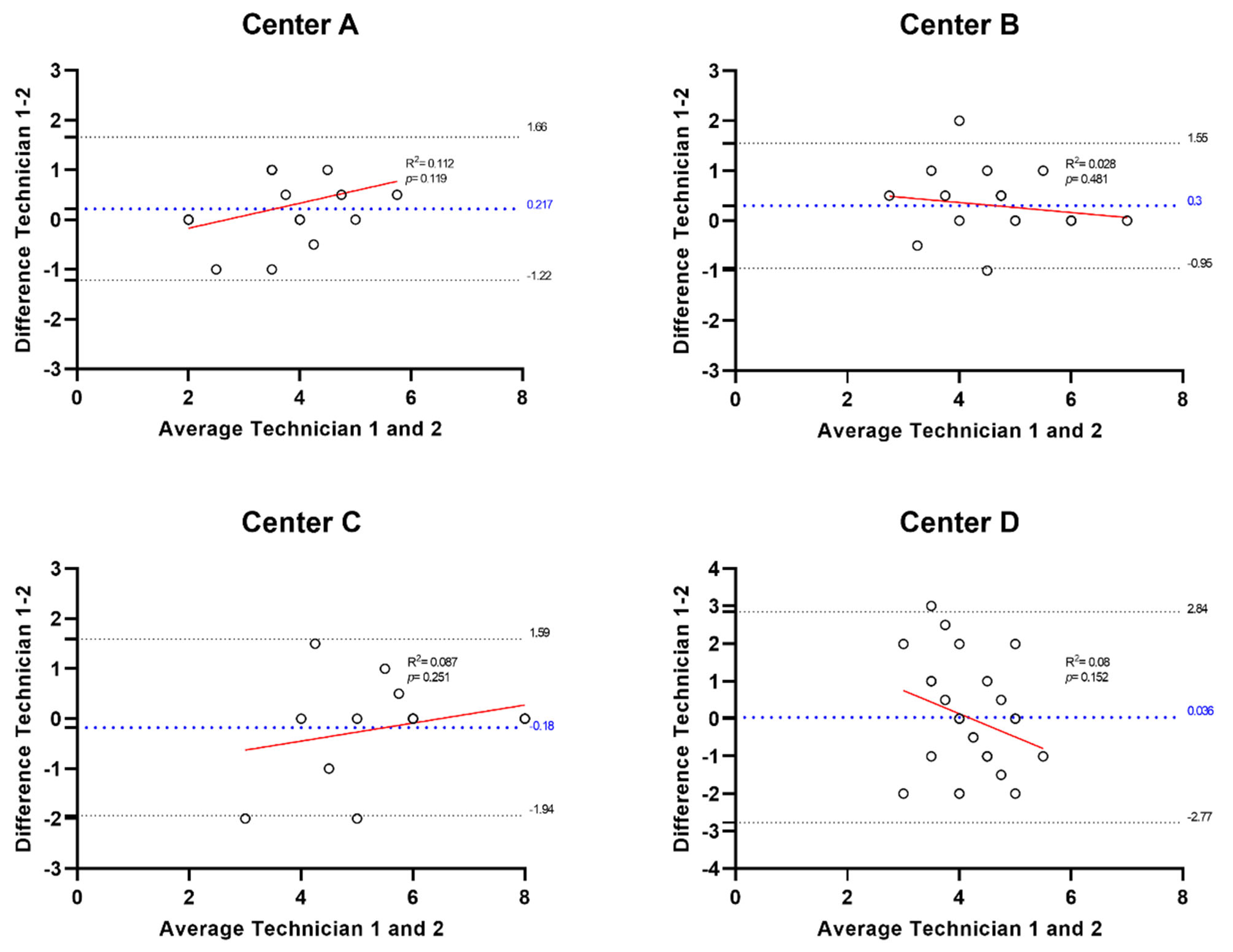
3.3. Secondary Efficacy Endpoints
3.4. Safety Data
4. Discussion
Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CBE | Cochet-Bonnet esthesiometer |
| CEB | Corneal Esthesiometer Brill |
| FDA | Food and Drug Administration |
| LEDs | Light-emitting diodes |
| OSDI | Ocular surface disease index |
| SD | Standard deviation |
References
- Guerrero-Moreno, A.; Baudouin, C.; Parsadaniantz, S.M.; Goazigo, A.R.-L. Morphological and Functional Changes of Corneal Nerves and Their Contribution to Peripheral and Central Sensory Abnormalities. Front. Cell. Neurosci. 2020, 14, 610342. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, M.F.; Soto, L.F.; Requena, D.; Ruiz-Alejos, A.O.; Huaylinos, Y.; Velasquez, R.; Bernabe-Ortiz, A.; Gilman, R.H. Tear biomarkers and corneal sensitivity as an indicator of neuropathy in type 2 diabetes. Diabetes Res. Clin. Pract. 2020, 163, 108143. [Google Scholar] [CrossRef]
- Mansoor, H.; Tan, H.C.; Lin, M.T.-Y.; Mehta, J.S.; Liu, Y.-C. Diabetic Corneal Neuropathy. J. Clin. Med. 2020, 9, 3956. [Google Scholar] [CrossRef]
- Nepp, J.; Wirth, M. Fluctuations of Corneal Sensitivity in Dry Eye Syndromes—A Longitudinal Pilot Study. Cornea 2015, 34, 1221–1226. [Google Scholar] [CrossRef] [PubMed]
- Guzel, B.; Oshitari, T.; Baba, T.; Mukharram, B.; Yamamoto, S. Diabetic corneal neuropathy: Clinical perspectives. Clin. Ophthalmol. 2018, 12, 981–987. [Google Scholar] [CrossRef]
- Marcos-Fernández, M.Á.; Tabernero, S.S.; Herreras, J.M.; Galarreta, D.J. Impact of herpetic stromal immune keratitis in corneal biomechanics and innervation. Graefe’s Arch. Clin. Exp. Ophthalmol. 2018, 256, 155–161. [Google Scholar] [CrossRef]
- Lin, X.; Xu, B.; Sun, Y.; Zhong, J.; Huang, W.; Yuan, J. Comparison of deep anterior lamellar keratoplasty and penetrating keratoplasty with respect to postoperative corneal sensitivity and tear film function. Graefe’s Arch. Clin. Exp. Ophthalmol. 2014, 252, 1779–1787. [Google Scholar] [CrossRef] [PubMed]
- Dell’Omo, R.; Cifariello, F.; De Turris, S.; Romano, V.; Di Renzo, F.; Di Taranto, D.; Coclite, G.; Agnifili, L.; Mastropasqua, L.; Costagliola, C. Confocal microscopy of corneal nerve plexus as an early marker of eye involvement in patients with type 2 diabetes. Diabetes Res. Clin. Pract. 2018, 142, 393–400. [Google Scholar] [CrossRef]
- Verónica Velasco González. Umbrales de Sensibilidad en Pacientes con Sintomatología de Superficie ocular Después de Cirugía Corneal; Universidad de Valladolid, Instituto Universitario de Oftalmología Aplicada: Valladolid, Spain, 2016. [Google Scholar]
- Yang, A.Y.; Chow, J.; Liu, J. Corneal Innervation and Sensation: The Eye and Beyond. Yale J. Biol. Med. 2018, 91, 13–21. [Google Scholar]
- Wu, K.Y.; Kulbay, M.; Tanasescu, C.; Jiao, B.; Nguyen, B.H.; Tran, S.D. An Overview of the Dry Eye Disease in Sjögren’s Syndrome Using Our Current Molecular Understanding. Int. J. Mol. Sci. 2023, 24, 1580. [Google Scholar] [CrossRef]
- Zhou, T.; Lee, A.; Lo, A.C.Y.; Kwok, J.S.W.J. Diabetic Corneal Neuropathy: Pathogenic Mechanisms and Therapeutic Strategies. Front. Pharmacol. 2022, 13, 816062. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-C.; So, W.; Wong, N.Q.; Tan, H.; Lin, M.Y.; Lee, I.Y.; Mehta, J. Diabetic corneal neuropathy as a surrogate marker for diabetic peripheral neuropathy. Neural Regen. Res. 2022, 17, 2172–2178. [Google Scholar] [CrossRef]
- Zhao, H.; He, Y.; Ren, Y.-R.; Chen, B.-H. Corneal alteration and pathogenesis in diabetes mellitus. Int. J. Ophthalmol. 2019, 12, 1939–1950. [Google Scholar] [CrossRef]
- NaPier, E.; Camacho, M.; McDevitt, T.F.; Sweeney, A.R. Neurotrophic keratopathy: Current challenges and future prospects. Ann. Med. 2022, 54, 666–673. [Google Scholar] [CrossRef] [PubMed]
- Tamimi, A.; Sheikhzadeh, F.; Ezabadi, S.G.; Islampanah, M.; Parhiz, P.; Fathabadi, A.; Poudineh, M.; Khanjani, Z.; Pourmontaseri, H.; Orandi, S.; et al. Post-LASIK dry eye disease: A comprehensive review of management and current treatment options. Front. Med. 2023, 10, 57685. [Google Scholar] [CrossRef] [PubMed]
- Cochet, P.; Bonnet, R. L’Esthesie Corneenne. Sa mesure clinique. Ses variations physiologiques et pathologiques. La Clin. Ophtalomologique 1960, 4, 3–27. [Google Scholar]
- Belmonte, C.; Acosta, M.C.; Schmelz, M.; Gallar, J. Measurement of corneal sensitivity to mechanical and chemical stimulation with a CO2 esthesiometer. Investig. Ophthalmol. Vis. Sci. 1999, 40, 513–519. [Google Scholar]
- Nosch, D.S.; Oscity, M.; Steigmeier, P.; Käser, E.; Loepfe, M.; E Joos, R. Working principle and relevant physical properties of the Swiss Liquid Jet Aesthesiometer for Corneal Sensitivity (SLACS) evaluation. Ophthalmic Physiol. Opt. 2022, 42, 609–618. [Google Scholar] [CrossRef]
- Lozano, J.; Barros, A.; del Castilo, J.M.B.; Montero, J.; Merayo-Lloves, J. Corneal sensitivity repeatability with a new non-contact air jet corneal esthesiometer and comparison with Cochet-Bonnet esthesiometer. Investig. Ophthalmol. Vis. Sci. 2021, 62, 746. [Google Scholar]
- Merayo-Lloves, J.; Martín, C.G.; Lozano-Sanroma, J.; Laguna, C.R. Assessment and safety of the new esthesiometer BRILL: Comparison with the Cochet-Bonnet Esthesiometer. Eur. J. Ophthalmol. 2023, 34, 1036–1045. [Google Scholar] [CrossRef]
- Murphy, P.J.; Lawrenson, J.G.; Patel, S.; Marshall, J. Reliability of the non-contact corneal aesthesiometer and its comparison with the Cochet–Bonnet aesthesiometer. Ophthalmic Physiol. Opt. 1998, 18, 532–539. [Google Scholar] [CrossRef]
- Golebiowski, B.; Papas, E.; Stapleton, F. Assessing the sensory function of the ocular surface: Implications of use of a non-contact air jet aesthesiometer versus the Cochet–Bonnet aesthesiometer. Exp. Eye Res. 2011, 92, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Al-Aqaba, M.A.; Dhillon, V.K.; Mohammed, I.; Said, D.G.; Dua, H.S. Corneal nerves in health and disease. Prog. Retin. Eye Res. 2019, 73, 100762. [Google Scholar] [CrossRef] [PubMed]
- Kaur, K.; Gurnani, B. Slit-Lamp Biomicroscope. 2024. Available online: https://www.ncbi.nlm.nih.gov/pubmed/26020484 (accessed on 14 July 2025).
- Faul, F.; Erdfelder, E.; Lang, A.-G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Belmonte, C.; Nichols, J.J.; Cox, S.M.; Brock, J.A.; Begley, C.G.; Bereiter, D.A.; Dartt, D.A.; Galor, A.; Hamrah, P.; Ivanusic, J.J.; et al. TFOS DEWS II pain and sensation report. Ocul. Surf. 2017, 15, 404–437. [Google Scholar] [CrossRef]
- Rolando, M.; Zierhut, M.; Barabino, S. Should We Reconsider the Classification of Patients with Dry Eye Disease? Ocul. Immunol. Inflamm. 2021, 29, 521–523. [Google Scholar] [CrossRef]
- Versura, P.; Frigato, M.; Cellini, M.; Mulè, R.; Malavolta, N.; Campos, E.C. Diagnostic performance of tear function tests in Sjogren’s syndrome patients. Eye 2006, 21, 229–237. [Google Scholar] [CrossRef]
- Ruiz-Lozano, R.E.; Quiroga-Garza, M.E.; Ramos-Dávila, E.M.; Pantaleón-García, J.; Khodor, A.; Komai, S.; Rodriguez-Gutierrez, L.A.; Ma, S.; Mousa, H.M.; Mattes, R.; et al. Comparative Evaluation of the Corneal Sensitivity Thresholds between the Novel Non-Contact and Cochet-Bonnet Esthesiometers. Arch. Ophthalmol. 2024, 271, 407–416. [Google Scholar] [CrossRef]
- Murphy, P.J.; Patel, S.; Marshall, J. A new non-contact corneal aesthesiometer (NCCA). Ophthalmic Physiol. Opt. 1996, 16, 101–107. [Google Scholar] [CrossRef]
- Tuisku, I.S.; Konttinen, Y.T.; Konttinen, L.M.; Tervo, T.M. Alterations in corneal sensitivity and nerve morphology in patients with primary Sjögren’s syndrome. Exp. Eye Res. 2008, 86, 879–885. [Google Scholar] [CrossRef]
- Beni’tEz-Del-Castillo, J.M.; Acosta, M.C.; Wassfi, M.A.; Di’aZ-Valle, D.; Gegu’nDez, J.A.; Fernandez, C.; Garci’a-Sa’nChez, J. Relation between corneal innervation with confocal microscopy and corneal sensitivity with noncontact esthesiometry in patients with dry eye. Investig. Ophthalmol. Vis. Sci. 2007, 48, 173–181. [Google Scholar] [CrossRef]
- Bourcier, T.; Acosta, M.C.; Borderie, V.; Borra’s, F.; Gallar, J.; Bury, T.; Laroche, L.; Belmonte, C. Decreased corneal sensitivity in patients with dry eye. Investig. Ophthalmol. Vis. Sci. 2005, 46, 2341–2345. [Google Scholar] [CrossRef] [PubMed]
- Golebiowski, B.; Lim, M.; Papas, E.; Stapleton, F. Understanding the stimulus of an air-jet aesthesiometer: Computerised modelling and subjective interpretation. Ophthalmic Physiol. Opt. 2013, 33, 104–113. [Google Scholar] [CrossRef]
- Surico, P.L.; Saricay, L.Y.; Singh, R.B.; Kahale, F.; Romano, F.M.; Dana, R.M. Corneal Sensitivity and Neuropathy in Patients With Ocular Graft-Versus-Host Disease. Cornea 2024, 44, 838–844. [Google Scholar] [CrossRef]
- Vera-Duarte, G.R.; Ramirez-Miranda, A.J.; Manuel-Garcia, A.; Padilla, L.A.G.; Morales, A.M.; Kahuam-López, N.; Navas, A.; Graue-Hernandez, E.O. Corneal Mechanical Sensitivity by Non-Contact Brill Engines Esthesiometer in a Healthy Mexican Population. Investig. Ophthalmol. Vis. Sci. 2024, 65, 2654. [Google Scholar]
- Jalavisto, E.; Orma, E.; Tawast, M. Ageing and Relation between Stimulus Intensity and Duration in Corneal Sensibility. Acta Physiol. Scand. 1951, 23, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Millodot, M. The influence of age onthe sensitivity of the cornea. Investig. Ophthalmol. Vis. Sci. 1977, 16, 240–242. [Google Scholar]
- Boberg-Ans, J. Experience in Clinical Examination of Corneal Sensitivity: Corneal Sensitivity and the Naso-Lacrimal Reflex after Retrobulbar Anaesthesia. Br. J. Ophthalmol. 1955, 39, 705–726. [Google Scholar] [CrossRef] [PubMed][Green Version]

| Test. Intra-Observer | Test. Inter-Observer | |||
|---|---|---|---|---|
| Normal Approximation | Normal Approximation with C.C. | Normal Approximation | Normal Approximation with C.C. | |
| Z value | 1.4606 | 1.4582 | 0.7071 | 0.7047 |
| p-value | 0.14413 | 0.14479 | 0.4795 | 0.48098 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Renedo Laguna, C.; Gómez Martín, C.; Lozano-Sanroma, J.; Benítez del Castillo, J.M.; Montero Iruzubieta, J.; García Delpech, S.; Merayo-Lloves, J. Functioning and Safety of the Non-Invasive Corneal Esthesiometer Brill: A Multicenter Study. Diagnostics 2025, 15, 2208. https://doi.org/10.3390/diagnostics15172208
Renedo Laguna C, Gómez Martín C, Lozano-Sanroma J, Benítez del Castillo JM, Montero Iruzubieta J, García Delpech S, Merayo-Lloves J. Functioning and Safety of the Non-Invasive Corneal Esthesiometer Brill: A Multicenter Study. Diagnostics. 2025; 15(17):2208. https://doi.org/10.3390/diagnostics15172208
Chicago/Turabian StyleRenedo Laguna, Concepción, Carmen Gómez Martín, Javier Lozano-Sanroma, José Manuel Benítez del Castillo, Jesús Montero Iruzubieta, Salvador García Delpech, and Jesús Merayo-Lloves. 2025. "Functioning and Safety of the Non-Invasive Corneal Esthesiometer Brill: A Multicenter Study" Diagnostics 15, no. 17: 2208. https://doi.org/10.3390/diagnostics15172208
APA StyleRenedo Laguna, C., Gómez Martín, C., Lozano-Sanroma, J., Benítez del Castillo, J. M., Montero Iruzubieta, J., García Delpech, S., & Merayo-Lloves, J. (2025). Functioning and Safety of the Non-Invasive Corneal Esthesiometer Brill: A Multicenter Study. Diagnostics, 15(17), 2208. https://doi.org/10.3390/diagnostics15172208






