The Impacts of Enlarged Subarachnoid Space on Brain Growth and Cortex Maturation in Very Preterm Infants
Abstract
1. Introduction
2. Methods
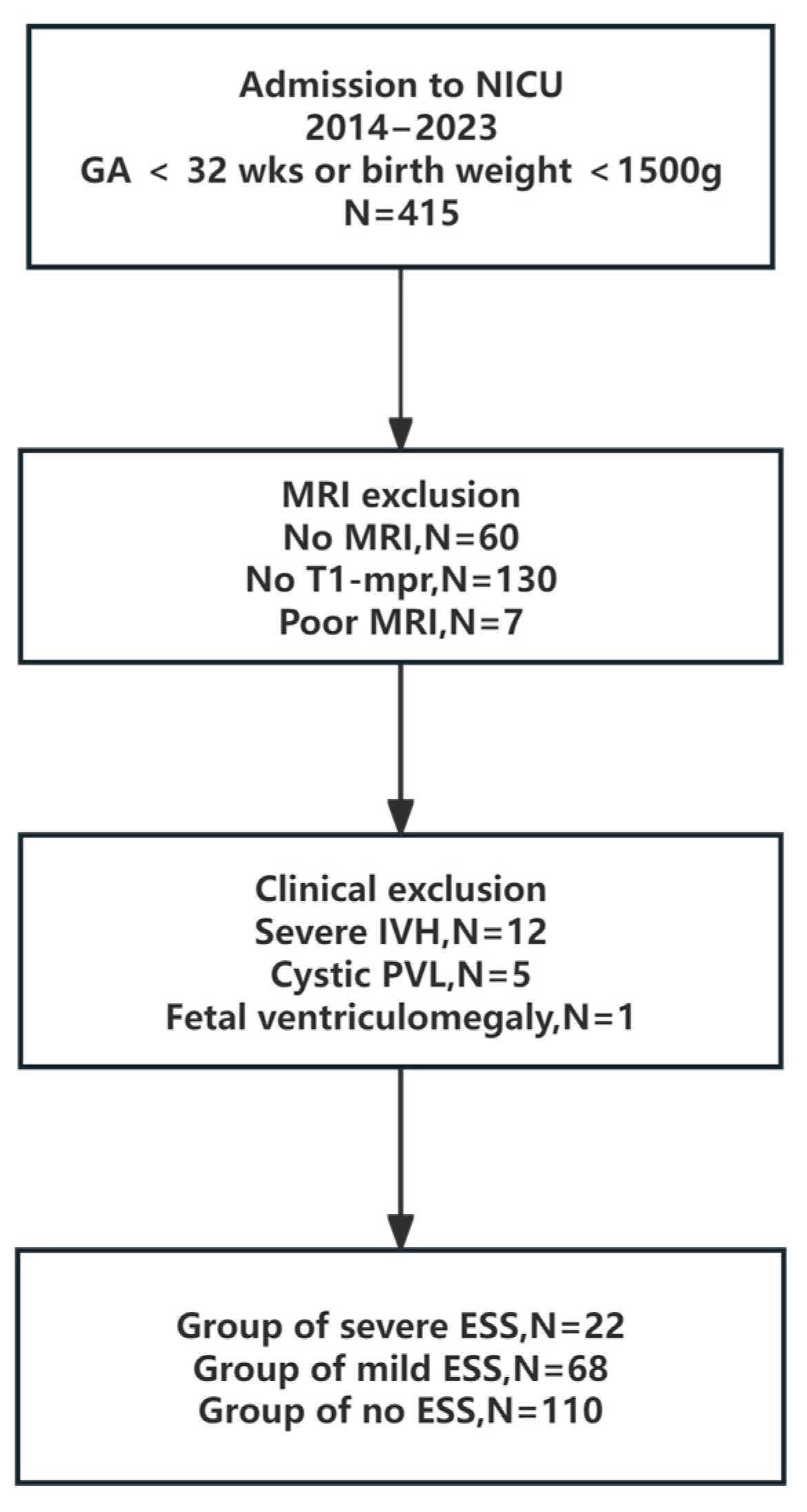
2.1. Patient Selection and Data Collection
2.2. Study Design and Rationale
2.3. MRI Data Acquisition
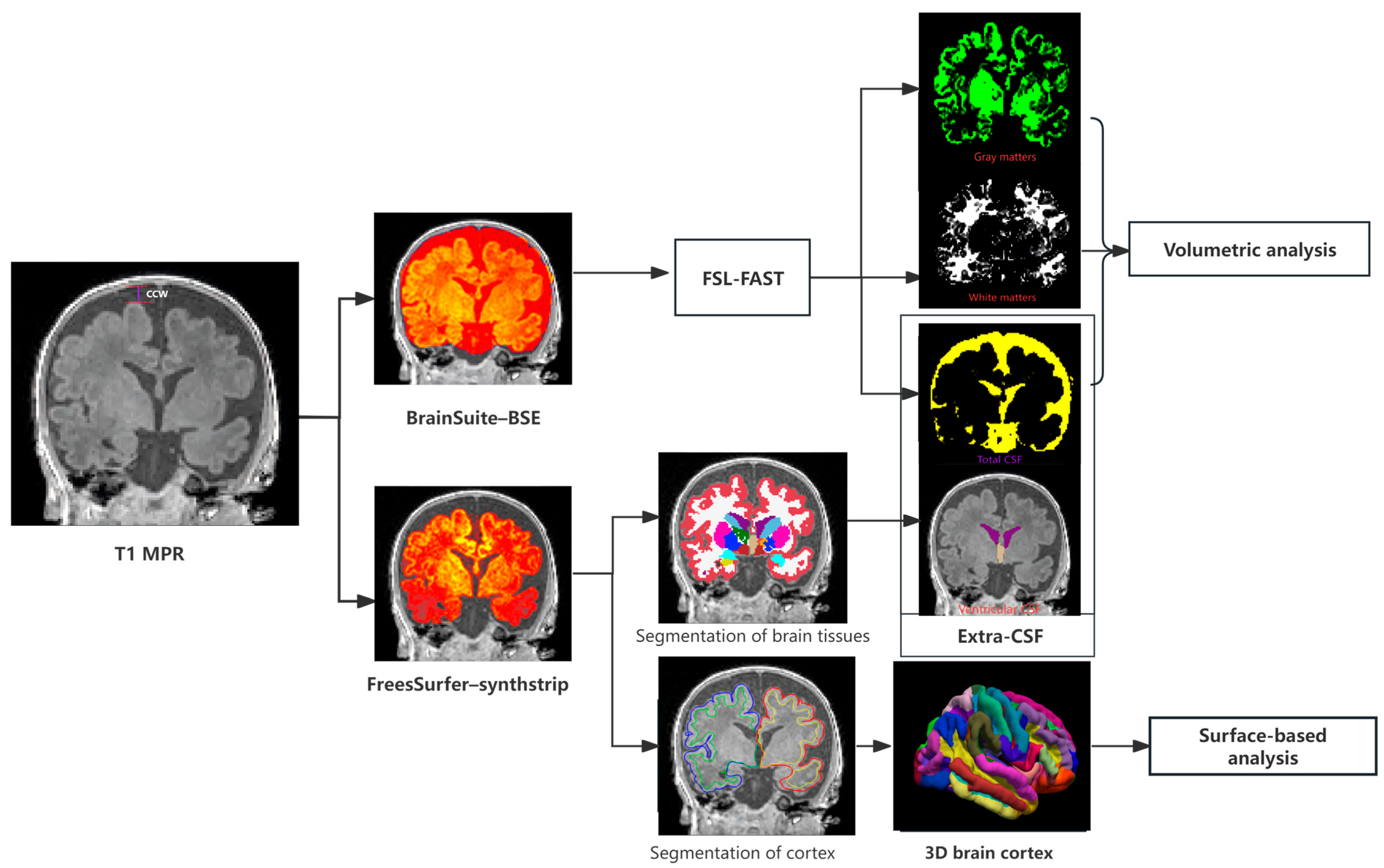
2.4. MR Image Analysis
2.4.1. Measurement of the Craniocortical Width (CCW)
2.4.2. Brain Tissue Segmentation
2.4.3. Brain Surface Segmentation
2.5. Statistical Analysis
3. Results
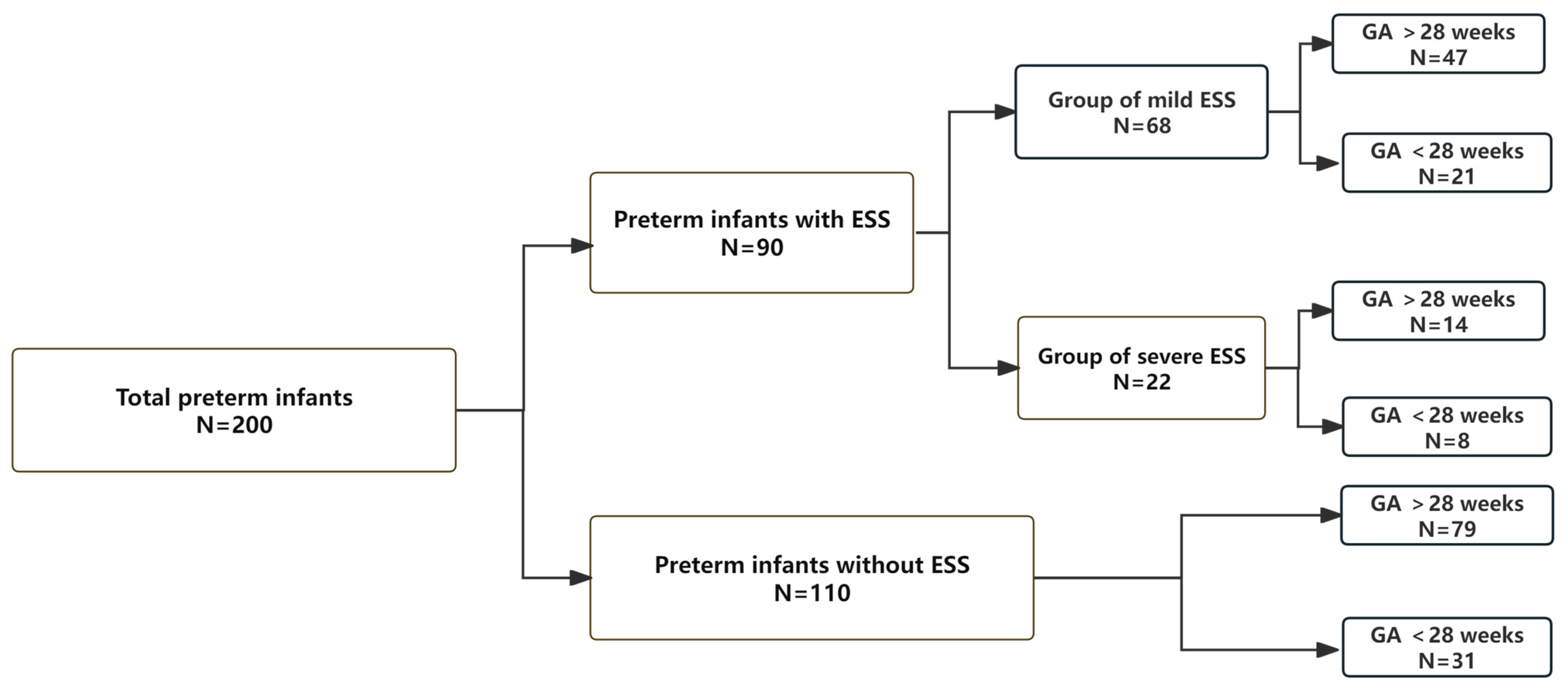
3.1. Participant Characteristics
3.2. Comparison of Basic Clinical Characteristics Between the Three Groups
3.3. Comparison of Brain Volume Between Three Groups
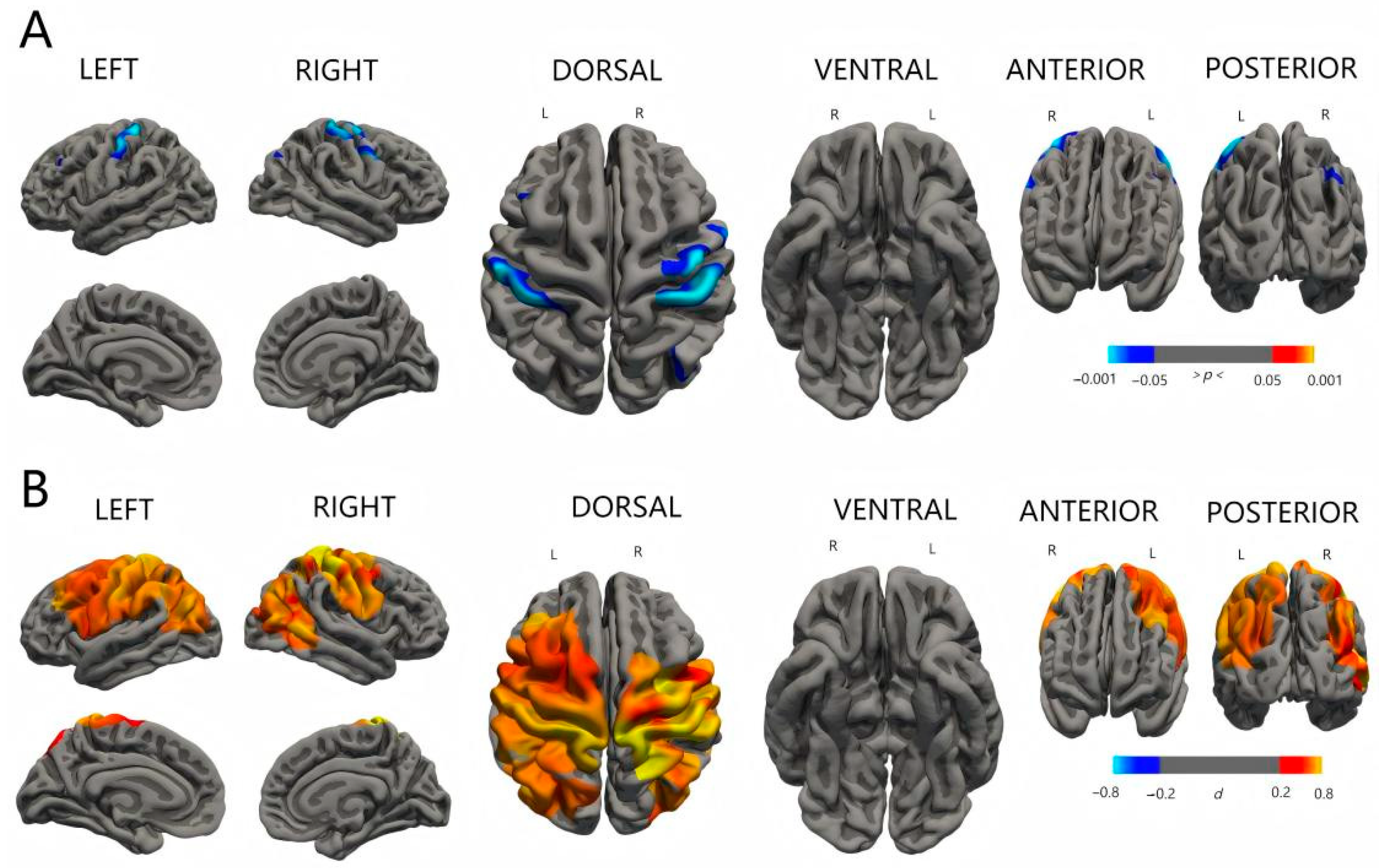
3.4. Comparison of Indices of Cortex Maturation Among Three Groups
4. Discussion
4.1. ESS and Brain Volume
4.2. ESS and Cortex Development
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marino, M.A.; Morabito, R.; Vinci, S.; Germanò, A.; Briguglio, M.; Alafaci, C.; Mormina, E.; Longo, M.; Granata, F. Benign External Hydrocephalus in Infants. Neuroradiol. J. 2014, 27, 245–250. [Google Scholar] [CrossRef]
- Zahl, S.M.; Egge, A.; Helseth, E.; Wester, K. Benign external hydrocephalus: A review, with emphasis on management. Neurosurg. Rev. 2011, 34, 417–432. [Google Scholar] [CrossRef] [PubMed]
- Yum, S.K.; Im, S.A.; Seo, Y.M.; Sung, I.K. Enlarged subarachnoid space on cranial ultrasound in preterm infants: Neurodevelopmental implication. Sci. Rep. 2019, 9, 19072. [Google Scholar] [CrossRef] [PubMed]
- Hammerl, M.; Zagler, M.; Zimmermann, M.; Griesmaier, E.; Janjic, T.; Gizewski, E.R.; Kiechl-Kohlendorfer, U.; Neubauer, V. Supratentorial Brain Metrics Predict Neurodevelopmental Outcome in Very Preterm Infants without Brain Injury at Age 2 Years. Neonatology 2020, 117, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, R.; Rødevand, L.N.; Wiig, U.S.; Zahl, S.M.; Berntsen, T.; Skarbø, A.-B.; Egge, A.; Helseth, E.; Andersson, S.; Wester, K. Neurocognitive and psychosocial function in children with benign external hydrocephalus (BEH)-a long-term follow-up study. Child’s Nerv. Syst. 2017, 33, 91–99. [Google Scholar] [CrossRef]
- Maruccia, F.; Gomáriz, L.; Rosas, K.; Durduran, T.; Paredes-Carmona, F.; Sahuquillo, J.; Poca, M.A. Neurodevelopmental profile in children with benign external hydrocephalus syndrome. A pilot cohort study. Child’s Nerv. Syst. 2021, 37, 2799–2806. [Google Scholar] [CrossRef]
- Zahl, S.M.; Egge, A.; Helseth, E.; Skarbø, A.B.; Wester, K. Quality of life and physician-reported developmental, cognitive, and social problems in children with benign external hydrocephalus-long-term follow-up. Child’s Nerv. Syst. 2019, 35, 245–250. [Google Scholar] [CrossRef]
- Padilla, N.; Alexandrou, G.; Blennow, M.; Lagercrantz, H.; Ådén, U. Brain Growth Gains and Losses in Extremely Preterm Infants at Term. Cereb. Cortex 2015, 25, 1897–1905. [Google Scholar] [CrossRef]
- Bouyssi-Kobar, M.; du Plessis, A.J.; McCarter, R.; Brossard-Racine, M.; Murnick, J.; Tinkleman, L.; Robertson, R.L.; Limperopoulos, C. Third Trimester Brain Growth in Preterm Infants Compared With In Utero Healthy Fetuses. Pediatrics 2016, 138, e20161640. [Google Scholar] [CrossRef]
- Wandel, A.; Weissbach, T.; Katorza, E.; Ziv-Baran, T. Subarachnoid Space Measurements in Apparently Healthy Fetuses Using MR Imaging. Am. J. Neuroradiol. 2023, 44, 716–721. [Google Scholar] [CrossRef]
- Maunu, J.; Parkkola, R.; Rikalainen, H.; Lehtonen, L.; Haataja, L.; Lapinleimu, H. Brain and ventricles in very low birth weight infants at term: A comparison among head circumference, ultrasound, and magnetic resonance imaging. Pediatrics 2009, 123, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Beare, R.J.; Chen, J.; Kelly, C.E.; Alexopoulos, D.; Smyser, C.D.; Rogers, C.E.; Loh, W.Y.; Matthews, L.G.; Cheong, J.L.Y.; Spittle, A.J.; et al. Neonatal Brain Tissue Classification with Morphological Adaptation and Unified Segmentation. Front. Neuroinform. 2016, 10, 12. [Google Scholar] [CrossRef]
- Smith, S.M. Fast robust automated brain extraction. Hum. Brain Mapp. 2002, 17, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Brady, M.; Smith, S. Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans. Med. Imaging 2001, 20, 45–57. [Google Scholar] [CrossRef]
- Zöllei, L.; Iglesias, J.E.; Ou, Y.; Grant, P.E.; Fischl, B. Infant FreeSurfer: An automated segmentation and surface extraction pipeline for T1-weighted neuroimaging data of infants 0–2 years. NeuroImage 2020, 218, 116946. [Google Scholar] [CrossRef]
- Kelley, W.; Ngo, N.; Dalca, A.V.; Fischl, B.; Zöllei, L.; Hoffmann, M. Boosting skull-stripping performance for pediatric brain images. In Proceedings of the IEEE International Symposium on Biomedical Imaging, Athens, Greece, 27–30 May 2024. [Google Scholar]
- Yushkevich, P.A.; Piven, J.; Hazlett, H.C.; Smith, R.G.; Ho, S.; Gee, J.C.; Gerig, G. User-guided 3D active contour segmentation of anatomical structures: Significantly improved efficiency and reliability. NeuroImage 2006, 31, 1116–1128. [Google Scholar] [CrossRef] [PubMed]
- Alexander, B.; Kelly, C.E.; Adamson, C.; Beare, R.; Zannino, D.; Chen, J.; Murray, A.L.; Loh, W.Y.; Matthews, L.G.; Warfield, S.K.; et al. Changes in neonatal regional brain volume associated with preterm birth and perinatal factors. NeuroImage 2019, 185, 654–663. [Google Scholar] [CrossRef]
- Boardman, J.P.; Counsell, S.J.; Rueckert, D.; Hajnal, J.V.; Bhatia, K.K.; Srinivasan, L.; Kapellou, O.; Aljabar, P.; Dyet, L.E.; Rutherford, M.A.; et al. Early growth in brain volume is preserved in the majority of preterm infants. Ann. Neurol. 2007, 62, 185–192. [Google Scholar] [CrossRef]
- Esler, S.R.; Ment, L.R.; Vohr, B.; Pajot, S.K.; Schneider, K.C.; Katz, K.H.; Ebbitt, T.B.; Duncan, C.C.; Makuch, R.W.; Reiss, A.L. Volumetric analysis of regional cerebral development in preterm children. Pediatr. Neurol. 2004, 31, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.Y.; Zhu, M.; Wang, G.; Gui, Y.; Jiang, F.; Dong, S.Z. Quantification of Intracranial Structures Volume in Fetuses Using 3-D Volumetric MRI: Normal Values at 19 to 37 Weeks’ Gestation. Front. Neurosci. 2022, 16, 886083. [Google Scholar] [CrossRef]
- Zhou, M.; Li, X.; Huang, T.; Wang, M.; Giorgio, A. Analysis of automatic fetal intracranial volume (ICV) measurement based on the optimized ultrasound Smart ICV method at 16–34 weeks of gestation. Quant. Imaging Med. Surg. 2024, 14, 9361–9373. [Google Scholar] [CrossRef]
- Onn-Margalit, L.; Weissbach, T.; Gafner, M.; Fried, S.; Wandel, A.; Ziv-Baran, T.; Katorza, E. An Association Between Fetal Subarachnoid Space and Various Pathologies Using MR Imaging. Diagnostics 2024, 14, 2535. [Google Scholar] [CrossRef]
- Rekate, H.L.; Nadkarni, T.D.; Wallace, D. The importance of the cortical subarachnoid space in understanding hydrocephalus. J. Neurosurg. Pediatr. 2008, 2, 1–11. [Google Scholar] [CrossRef]
- Hladky, S.B.; Barrand, M.A. Regulation of brain fluid volumes and pressures: Basic principles, intracranial hypertension, ventriculomegaly and hydrocephalus. Fluids Barriers CNS 2024, 21, 57. [Google Scholar] [CrossRef]
- Garcia, K.E.; Robinson, E.C.; Alexopoulos, D.; Dierker, D.L.; Glasser, M.F.; Coalson, T.S.; Ortinau, C.M.; Rueckert, D.; Taber, L.A.; Van Essen, D.C.; et al. Dynamic patterns of cortical expansion during folding of the preterm human brain. Proc. Natl. Acad. Sci. USA 2018, 115, 3156–3161. [Google Scholar] [CrossRef]
- Kelly, C.E.; Thompson, D.K.; Adamson, C.L.; Ball, G.; Dhollander, T.; Beare, R.; Matthews, L.G.; Alexander, B.; Cheong, J.L.Y.; Doyle, L.W.; et al. Cortical growth from infancy to adolescence in preterm and term-born children. Brain 2024, 147, 1526–1538. [Google Scholar] [CrossRef]
- Makropoulos, A.; Aljabar, P.; Wright, R.; Hüning, B.; Merchant, N.; Arichi, T.; Tusor, N.; Hajnal, J.V.; Edwards, A.D.; Counsell, S.J.; et al. Regional growth and atlasing of the developing human brain. NeuroImage 2016, 125, 456–478. [Google Scholar] [CrossRef]
- Kline, J.E.; Sita Priyanka Illapani, V.; He, L.; Parikh, N.A. Automated brain morphometric biomarkers from MRI at term predict motor development in very preterm infants. NeuroImage Clin. 2020, 28, 102475. [Google Scholar] [CrossRef] [PubMed]
- Shimony, J.S.; Smyser, C.D.; Wideman, G.; Alexopoulos, D.; Hill, J.; Harwell, J.; Dierker, D.; Van Essen, D.C.; Inder, T.E.; Neil, J.J. Comparison of cortical folding measures for evaluation of developing human brain. NeuroImage 2016, 125, 780–790. [Google Scholar] [CrossRef] [PubMed]
- de Vareilles, H.; Rivière, D.; Mangin, J.F.; Dubois, J. Development of cortical folds in the human brain: An attempt to review biological hypotheses, early neuroimaging investigations and functional correlates. Dev. Cogn. Neurosci. 2023, 61, 101249. [Google Scholar] [CrossRef] [PubMed]
- Pagnozzi, A.M.; van Eijk, L.; Pannek, K.; Boyd, R.N.; Saha, S.; George, J.; Bora, S.; Bradford, D.; Fahey, M.; Ditchfield, M.; et al. Early brain morphometrics from neonatal MRI predict motor and cognitive outcomes at 2-years corrected age in very preterm infants. NeuroImage 2023, 267, 119815. [Google Scholar] [CrossRef]
- Pagnozzi, A.M.; Pannek, K.; Boyd, R.N.; van Eijk, L.; George, J.M.; Bora, S.; Bradford, D.; Fahey, M.; Ditchfield, M.; Malhotra, A.; et al. Brain MRI before and at term equivalent age predicts motor and cognitive outcomes in very preterm infants. Neuroimage Rep. 2025, 5, 100262. [Google Scholar] [CrossRef] [PubMed]
- Moeskops, P.; Benders, M.J.N.L.; Kersbergen, K.J.; Groenendaal, F.; de Vries, L.S.; Viergever, M.A.; Išgum, I.; He, H. Development of Cortical Morphology Evaluated with Longitudinal MR Brain Images of Preterm Infants. PLoS ONE 2015, 10, e0131552. [Google Scholar] [CrossRef] [PubMed]
- Luders, E.; Thompson, P.M.; Narr, K.L.; Toga, A.W.; Jancke, L.; Gaser, C. A curvature-based approach to estimate local gyrification on the cortical surface. NeuroImage 2006, 29, 1224–1230. [Google Scholar] [CrossRef] [PubMed]
- Kline, J.E.; Illapani, V.S.P.; He, L.; Altaye, M.; Logan, J.W.; Parikh, N.A. Early cortical maturation predicts neurodevelopment in very preterm infants. Arch. Dis. Child. Fetal Neonatal Ed. 2020, 105, 460–465. [Google Scholar] [CrossRef]
- Hedderich, D.M.; Bäuml, J.G.; Berndt, M.T.; Menegaux, A.; Scheef, L.; Daamen, M.; Zimmer, C.; Bartmann, P.; Boecker, H.; Wolke, D.; et al. Aberrant gyrification contributes to the link between gestational age and adult IQ after premature birth. Brain 2019, 142, 1255–1269. [Google Scholar] [CrossRef]
- Kim, H.; Ahn, J.-H.; Lee, J.Y.; Jang, Y.H.; Kim, Y.-E.; Kim, J.I.; Kim, B.-N.; Lee, H.J. Altered Cerebral Curvature in Preterm Infants Is Associated with the Common Genetic Variation Related to Autism Spectrum Disorder and Lipid Metabolism. J. Clin. Med. 2022, 11, 3135. [Google Scholar] [CrossRef]
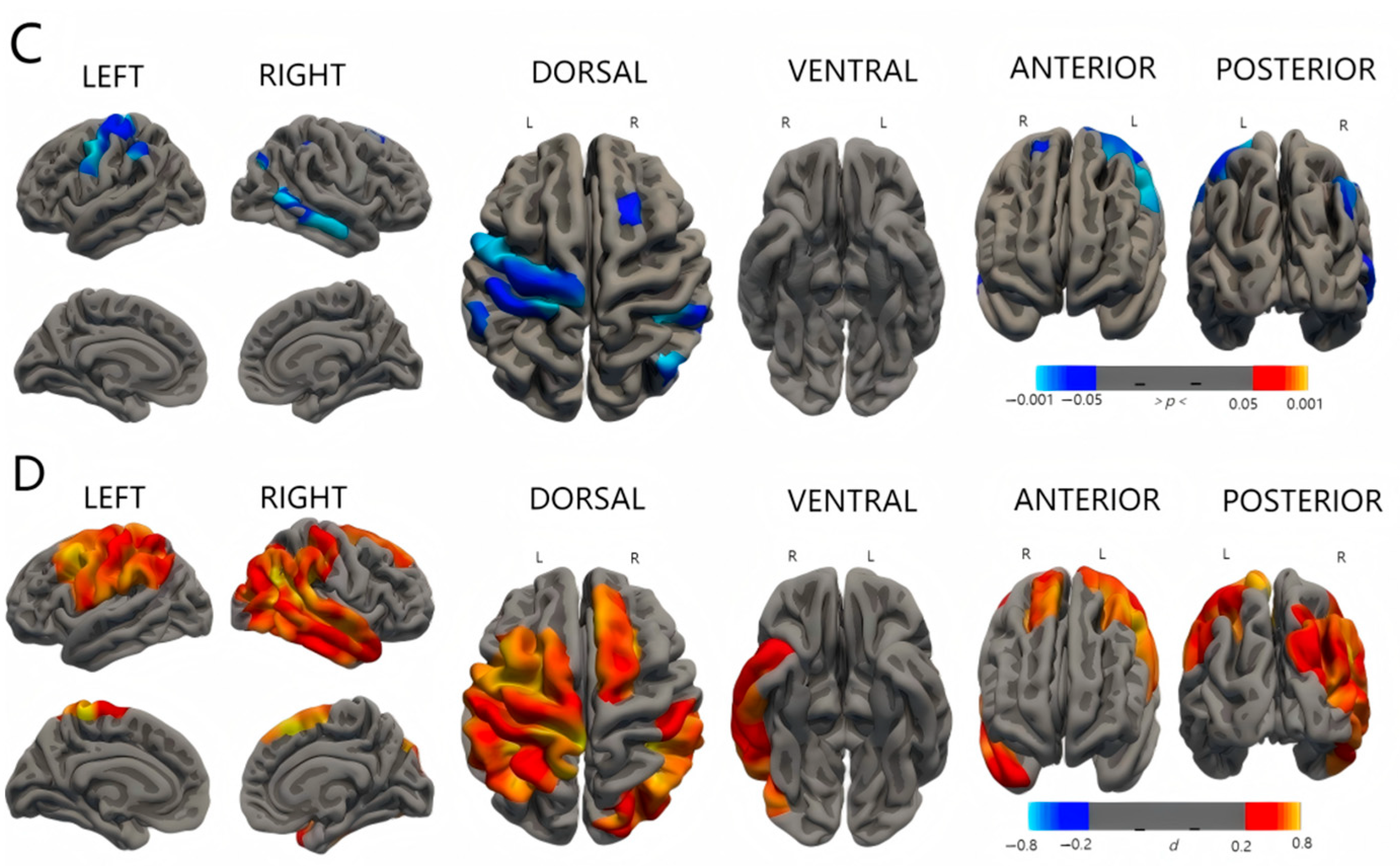
| No ESS (n = 110) | Mild ESS (n = 68) | Severe ESS (n = 22) | p Value | |
|---|---|---|---|---|
| Birth weight (g) | 1201 ± 276 | 1125 ± 279 | 1078 ± 245 | 0.798 |
| Gestational age (weeks) | 29.3 ± 2.5 | 28.5 ± 2.6 | 28.1 ± 2.4 | 0.522 |
| Head circumference at birth (cm) | 26.1 ± 2.2 | 26.3 ± 2.4 | 25.6 ± 1.8 | 0.854 |
| Head circumference at MRI scan (cm) | 32.5 ± 0.9 1,2 | 33.6 ± 1.0 1,3 | 33.5 ± 1.2 2,3 | p = 0.001 1 p = 0.001 2 p = 0.921 3 |
| Gestational age at MRI scan (weeks) | 38.0 ± 2.3 | 38.2 ± 2.1 | 38.5 ± 2.5 | 0.588 |
| Male sex | 64 (58.2) | 38 (55.9) | 13 (59.0) | 0.885 |
| Grade 1–2 IVH | 14 (12.7) | 7 (10.3) | 4 (18.2) | 0.459 |
| PWML | 8 (7.3) | 4 (5.9) | 3 (13.6) | 0.631 |
| sBPD | 80 (72.7) | 38 (55.9) | 14 (63.6) | 0.954 |
| IUGR | 24 (21.8) | 15 (22.0) | 6 (27.3) | 0.559 |
| Postnatal steroid usage | 6 (5.5) | 4 (5.9) | 2 (9.0) | 0.452 |
| Pairwise Comparison p Value | ||||||
|---|---|---|---|---|---|---|
| No ESS (n = 110) | Mild ESS (n = 68) | Severe ESS (n = 22) | Mild vs. No ESS | Severe vs. No ESS | Severe vs. Mild ESS | |
| Volume of ICC (mL) | 356.78 ± 26.03 | 374.25 ± 26.45 | 384.66 ± 30.33 | p= 0.000 | p= 0.001 | p = 0.291 |
| Volume of brain parenchyma (mL) | 312.27 ± 20.75 | 310.37 ± 24.41 | 302.35 ± 26.43 | p = 0.426 | p= 0.003 | p= 0.001 |
| % ICC | 87.52 ± 3.1 | 82.93 ± 2.7 | 78.60 ± 4.6 | p= 0.013 | p= 0.000 | p= 0.010 |
| Total volume of CSF (mL) | 44.51 ± 8.9 | 63.88 ± 7.4 | 82.31 ± 8.2 | p= 0.000 | p= 0.000 | p= 0.000 |
| % ICC | 12.47 ± 2.2 | 17.06 ± 2.9 | 21.39 ± 3.7 | p= 0.013 | p= 0.000 | p= 0.010 |
| Volume of CSF in ventricular system (mL) | 6.41 ± 1.8 | 8.52 ± 2.1 | 10.33 ± 2.5 | p = 0.298 | p = 0.336 | p = 0.181 |
| % ICC | 1.79 ± 0.7 | 2.27 ± 0.5 | 2.68 ± 0.4 | p = 0.743 | p = 0.063 | p = 0.077 |
| Volume of extra CSF (mL) | 40.54 ± 4.3 | 55.36 ± 3.8 | 74.20 ± 5.1 | p= 0.000 | p= 0.000 | p= 0.000 |
| % ICC | 11.36 ± 1.9 | 14.79 ± 1.4 | 19.29 ± 2.0 | p= 0.009 | p= 0.000 | p= 0.027 |
| Severe ESS/No ESS | ||||||
|---|---|---|---|---|---|---|
| Annotation | Cluster Size (mm2) | MNI | Sig. | |||
| x | y | z | ||||
| Mean Curvature | lh_precentral | 985.35 | −43.79 | −11.40 | 61.76 | 0.001 |
| lh_postcentral | 401.60 | −47.91 | −31.23 | 53.79 | 0.012 | |
| lh_supramarginal | 105.11 | −57.83 | −44.76 | 44.87 | 0.025 | |
| rh_middletemporal | 770.56 | 64.88 | −53.12 | 4.10 | 0.003 | |
| rh_inferiorparietal | 201.43 | 51.32 | −55.63 | 47.12 | 0.010 | |
| rh_supramarginal | 267.14 | 60.31 | −33.63 | 45.10 | 0.005 | |
| rh_caudalmiddlefrontal | 355.88 | 39.54 | 27.08 | 44.73 | 0.032 | |
| Mild ESS/No ESS | ||||||
| Annotation | Cluster Size (mm2) | MNI | Sig. | |||
| x | y | z | ||||
| lh_postcentral | 675.44 | −35.00 | −33.03 | 69.25 | 0.001 | |
| lh_caudalmiddlefrontal | 140.98 | −42.69 | 13.34 | 52.10 | 0.002 | |
| rh_precentral | 765.33 | 43.01 | −7.84 | 60.92 | 0.036 | |
| rh_postcentral | 512.43 | 38.19 | −31.29 | 68.28 | 0.008 | |
| rh_inferiorparietal | 266.34 | 54.67 | −48.01 | 39.42 | 0.040 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Zhuo, Y.; Lin, F.; Wan, X.; Yang, G.; He, J. The Impacts of Enlarged Subarachnoid Space on Brain Growth and Cortex Maturation in Very Preterm Infants. Diagnostics 2025, 15, 2206. https://doi.org/10.3390/diagnostics15172206
Wang L, Zhuo Y, Lin F, Wan X, Yang G, He J. The Impacts of Enlarged Subarachnoid Space on Brain Growth and Cortex Maturation in Very Preterm Infants. Diagnostics. 2025; 15(17):2206. https://doi.org/10.3390/diagnostics15172206
Chicago/Turabian StyleWang, Liangbing, Yubo Zhuo, Fang Lin, Xueqing Wan, Guohui Yang, and Jianlong He. 2025. "The Impacts of Enlarged Subarachnoid Space on Brain Growth and Cortex Maturation in Very Preterm Infants" Diagnostics 15, no. 17: 2206. https://doi.org/10.3390/diagnostics15172206
APA StyleWang, L., Zhuo, Y., Lin, F., Wan, X., Yang, G., & He, J. (2025). The Impacts of Enlarged Subarachnoid Space on Brain Growth and Cortex Maturation in Very Preterm Infants. Diagnostics, 15(17), 2206. https://doi.org/10.3390/diagnostics15172206






