Evaluation of Nailfold Capillaroscopic Findings in Pediatric Patients with Celiac Disease: A Cross-Sectional and Comparative Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Data Collection
2.3. Nailfold Capillaroscopy Assessment
- Capillary density: The number of capillaries per linear millimeter; a density of <7/mm was considered abnormal.
- Capillary morphology:
- Normal: Hairpin-shaped, crossing once or twice, or tortuous capillaries with a convex tip.
- Abnormal: Branching or bushy morphology, features suggestive of neoangiogenesis, non-convex tips, or capillaries exhibiting three or more crossings.
- Capillary dilations: Capillaries with an apical loop diameter of 20–50 μm (normal: <20 μm).
- Giant capillaries: Homogeneously dilated capillaries with an apical diameter of ≥50 μm.
- Microhemorrhages: Presence or absence of punctate or flame-shaped hemorrhagic spots in the pericapillary area.
- Scleroderma pattern: The presence of giant capillaries and/or multiple architectural irregularities, typically accompanied by a significant decrease in capillary density. (≤3/mm).
- Non-scleroderma patterns:
- Normal pattern: Homogeneous capillary size (<20 μm), normal morphology, and capillary density ≥7/mm.
- Nonspecific pattern: Features including abnormal morphology, presence of dilations, or a density <7/mm without fulfilling criteria for the scleroderma pattern.
2.4. Ethical Approval and Informed Consent
2.5. Statistical Analysis
3. Results
3.1. Demographic and Clinical Features of the Participants
3.2. Nailfold Videocapillaroscopy Findings
3.2.1. Capillaroscopic Differences Between CD Patients and Healthy Controls
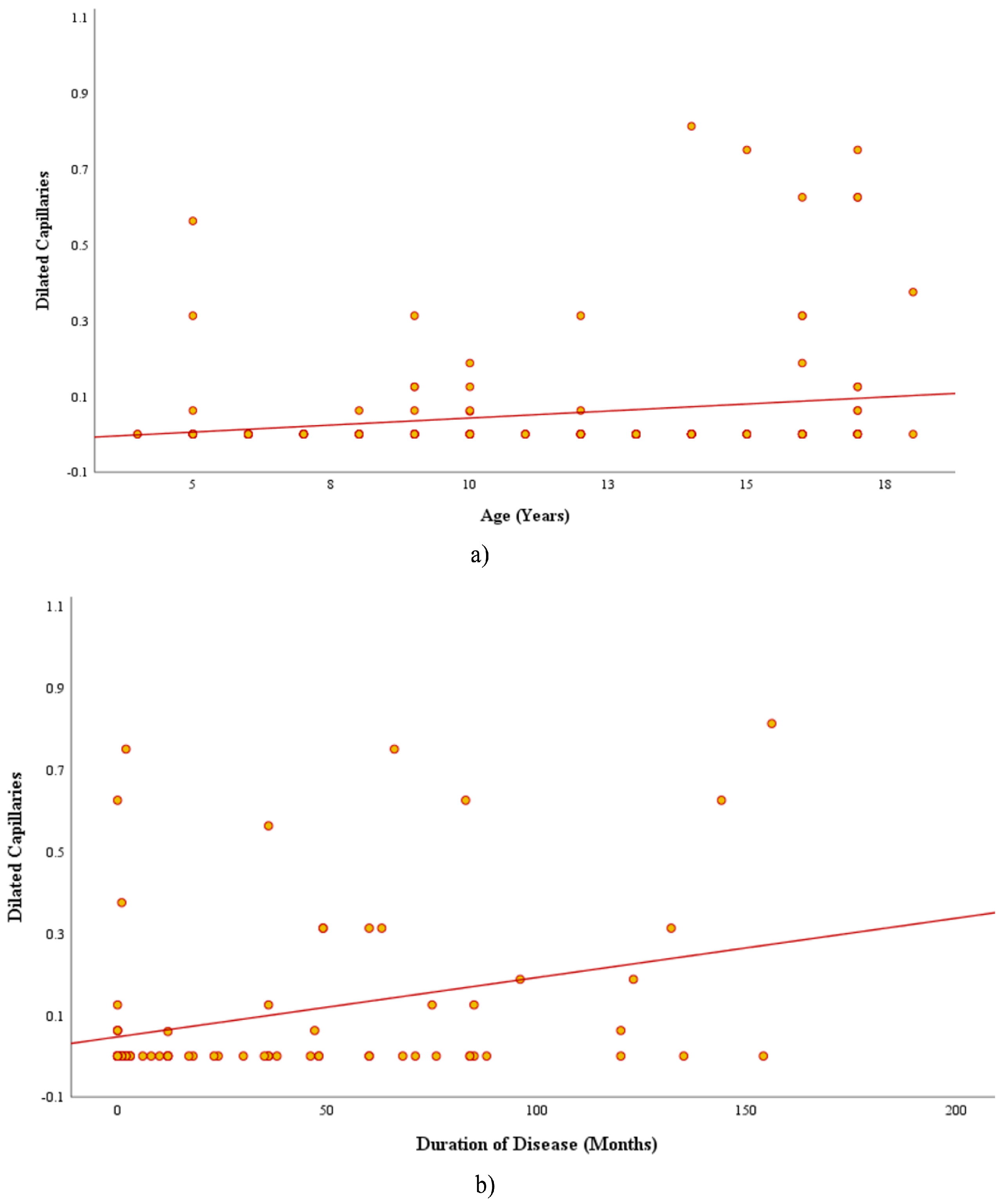
3.2.2. Correlations and Subgroup Analyses
3.2.3. The Effect of Adherence to a Gluten-Free Diet on Capillaroscopic Parameters
3.2.4. Capillaroscopic Findings According to Antinuclear Antibody Status
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Catassi, C.; Verdu, E.F.; Bai, J.C.; Lionetti, E. Coeliac disease. Lancet 2022, 399, 2413–2426. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, B.; Ciaccio, E.J.; Jneid, H.; Virani, S.S.; Lavie, C.J.; Lebovits, J.; Green, P.H.R.; Krittanawong, C. Celiac Disease and the Risk of Cardiovascular Diseases. Int. J. Mol. Sci. 2023, 24, 9974. [Google Scholar] [CrossRef]
- Laurikka, P.; Kivelä, L.; Kurppa, K.; Kaukinen, K. Review article: Systemic consequences of coeliac disease. Aliment. Pharmacol. Ther. 2022, 56, 64–72. [Google Scholar] [CrossRef]
- Keppeler, K.; Pesi, A.; Lange, S.; Helmstädter, J.; Strohm, L.; Ubbens, H.; Kuntić, M.; Kuntić, I.; Mihaliková, D.; Vujačić-Mirski, K.; et al. Vascular Dysfunction and Arterial Hypertension in Experimental Celiac Disease Are Mediated by Gut-Derived Inflammation and Oxidative Stress. Redox Biol. 2024, 70, 103071. [Google Scholar] [CrossRef]
- Rohrer, T.R.; Wolf, J.; Liptay, S.; Zimmer, K.P.; Fröhlich-Reiterer, E.; Scheuing, N.; Marg, W.; Stern, M.; Kapellen, T.M.; Hauffa, B.P.; et al. Microvascular Complications in Childhood-Onset Type 1 Diabetes and Celiac Disease: A Multicenter Longitudinal Analysis of 56,514 Patients From the German-Austrian DPV Database. Diabetes Care 2015, 38, 801–807. [Google Scholar] [CrossRef]
- Kıran Taşcı, E.; Taner, S.; Doğan, E.; Karakoyun, M.; Kaplan Bulut, İ.; Kabasakal, C. Evaluation of Vascular Involvement in Children with Celiac Disease. J. Pediatr. Res. 2023, 10, 167–172. [Google Scholar] [CrossRef]
- Cutolo, M.; Sulli, A.; Secchi, M.E.; Olivieri, M.; Pizzorni, C. The Contribution of Capillaroscopy to the Differential Diagnosis of Connective Autoimmune Diseases. Best. Pract. Res. Clin. Rheumatol. 2007, 21, 1093–1108. [Google Scholar] [CrossRef]
- Smith, V.; Ickinger, C.; Hysa, E.; Snow, M.; Frech, T.; Sulli, A.; Cutolo, M. Nailfold Capillaroscopy. Best. Pract. Res. Clin. Rheumatol. 2023, 37, 101849. [Google Scholar] [CrossRef] [PubMed]
- Schonenberg-Meinema, D.; Cutolo, M.; Smith, V. Capillaroscopy in the Daily Clinic of the Pediatric Rheumatologist. Best. Pract. Res. Clin. Rheumatol. 2024, 38, 101978. [Google Scholar] [CrossRef]
- Komai, M.; Takeno, D.; Fujii, C.; Nakano, J.; Ohsaki, Y.; Shirakawa, H. Nailfold Capillaroscopy: A Comprehensive Review on Its Usefulness in Both Clinical Diagnosis and Improving Unhealthy Dietary Lifestyles. Nutrients 2024, 16, 1914. [Google Scholar] [CrossRef]
- Kurowski, J.A.; Patel, S.R.; Wechsler, J.B.; Izaguirre, M.R.; Morgan, G.A.; Pachman, L.M.; Brown, J.B. Nailfold Capillaroscopy as a Biomarker in the Evaluation of Pediatric Inflammatory Bowel Disease. Crohns Colitis 360 2021, 3, otab069. [Google Scholar] [CrossRef]
- Okyar, B.; Yıldırım, A.E.; Barutçu, S. The Relationship Between Gluten Enteropathy and Nail Capillaroscopy Findings and Disease Activation. Clin. Exp. Health Sci. 2022, 12, 760–764. [Google Scholar] [CrossRef]
- Husby, S.; Koletzko, S.; Korponay-Szabó, I.; Kurppa, K.; Mearin, M.L.; Ribes-Koninckx, C.; Shamir, R.; Troncone, R.; Auricchio, R.; Castillejo, G.; et al. European Society for Paediatric Gastroenterology, Hepatology and Nutrition Guidelines for Diagnosing Coeliac Disease 2020. J. Pediatr. Gastroenterol. Nutr. 2020, 70, 141–156. [Google Scholar] [CrossRef]
- Marsh, M.N. Gluten, Major Histocompatibility Complex, and the Small Intestine: A Molecular and Immunobiologic Approach to the Spectrum of Gluten Sensitivity (“Celiac Sprue”). Gastroenterology 1992, 102, 330–354. [Google Scholar] [CrossRef]
- Mearin, M.L.; Agardh, D.; Antunes, H.; Al-Toma, A.; Auricchio, R.; Castillejo, G.; Catassi, C.; Ciacci, C.; Discepolo, V.; Dolinsek, J.; et al. ESPGHAN Special Interest Group on Celiac Disease. ESPGHAN Position Paper on Management and Follow-up of Children and Adolescents With Celiac Disease. J. Pediatr. Gastroenterol. Nutr. 2022, 75, 369–386. [Google Scholar] [CrossRef]
- Smith, V.; Herrick, A.L.; Ingegnoli, F.; Damjanov, N.; De Angelis, R.; Denton, C.P.; Distler, O.; Espejo, K.; Foeldvari, I.; Frech, T.; et al. Standardisation of Nailfold Capillaroscopy for the Assessment of Patients with Raynaud’s Phenomenon and Systemic Sclerosis. Autoimmun. Rev. 2020, 19, 102458. [Google Scholar] [CrossRef]
- Melsens, K.; Cutolo, M.; Schonenberg-Meinema, D.; Foeldvari, I.; Leone, M.C.; Mostmans, Y.; Badot, V.; Cimaz, R.; Dehoorne, J.; Deschepper, E.; et al. Standardized Nailfold Capillaroscopy in Children with Rheumatic Diseases: A Worldwide Study. Rheumatology 2023, 62, 1605–1615. [Google Scholar] [CrossRef]
- Schonenberg-Meinema, D.; Bergkamp, S.C.; Nassar-Sheikh Rashid, A.; van der Aa, L.B.; de Bree, G.J.; Ten Cate, R.; Cutolo, M.; Hak, A.E.; Hissink Muller, P.C.; van Onna, M.; et al. Nailfold Capillary Abnormalities in Childhood-Onset Systemic Lupus Erythematosus: A Cross-Sectional Study Compared with Healthy Controls. Lupus 2021, 30, 818–827. [Google Scholar] [CrossRef]
- Chanprapaph, K.; Fakprapai, W.; Limtong, P.; Suchonwanit, P. Nailfold Capillaroscopy with USB Digital Microscopy in Connective Tissue Diseases: A Comparative Study of 245 Patients and Healthy Controls. Front. Med. 2021, 8, 683900. [Google Scholar] [CrossRef]
- Dundar, H.A.; Adrovic, A.; Demir, S.; Demir, F.; Cakmak, F.; Ayaz, N.A.; Sözeri, B.; Bilginer, Y.; Kasapçopur, O.; Unsal, E. Description of the characteristics of the nailfold capillary structure in healthy children: A multi-centric study. Rheumatology 2024, 63, SI152–SI159. [Google Scholar] [CrossRef]
- Gidrewicz, D.; Trevenen, C.L.; Lyon, M.; Butzner, J.D. Normalization Time of Celiac Serology in Children on a Gluten-free Diet. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 362–367. [Google Scholar] [CrossRef]
- Comba, A.; Çaltepe, G.; Yank, K.; Gör, U.; Yüce, Ö.; Kalayc, A.G. Assessment of Endothelial Dysfunction With Adhesion Molecules in Patients With Celiac Disease. J. Pediatr. Gastroenterol. Nutr. 2016, 63, 247–252. [Google Scholar] [CrossRef]
- Lupu, V.V.; Sasaran, M.O.; Jechel, E.; Starcea, I.M.; Ioniuc, I.; Mocanu, A.; Rosu, S.T.; Munteanu, V.; Nedelcu, A.H.; Danielescu, C.; et al. Celiac Disease—A Pluripathological Model in Pediatric Practice. Front. Immunol. 2024, 15, 1390755. [Google Scholar] [CrossRef]
- Lupu, V.V.; Jechel, E.; Mihai, C.M.; Mitrofan, E.C.; Lupu, A.; Starcea, I.M.; Fotea, S.; Mocanu, A.; Ghica, D.C.; Mitrofan, C.; et al. Connection between Celiac Disease and Systemic Lupus Erythematosus in Children—A Development Model of Autoimmune Diseases Starting from What We Inherit to What We Eat. Nutrients 2023, 15, 2535. [Google Scholar] [CrossRef]
- Bartoloni, E.; Bistoni, O.; Alunno, A.; Cavagna, L.; Nalotto, L.; Baldini, C.; Priori, R.; Fischetti, C.; Fredi, M.; Quartuccio, L.; et al. Celiac Disease Prevalence Is Increased in Primary Sjögren’s Syndrome and Diffuse Systemic Sclerosis: Lessons from a Large Multi-Center Study. J. Clin. Med. 2019, 8, 540. [Google Scholar] [CrossRef] [PubMed]
- Flores Monar, G.V.; Islam, H.; Puttagunta, S.M.; Islam, R.; Kundu, S.; Jha, S.B.; Rivera, A.P.; Sange, I. Association Between Type 1 Diabetes Mellitus and Celiac Disease: Autoimmune Disorders With a Shared Genetic Background. Cureus 2022, 14, e22912. [Google Scholar] [CrossRef]
- Dai, X.; Fan, Y.; Zhao, X. Systemic lupus erythematosus: Updated insights on the pathogenesis, diagnosis, prevention and therapeutics. Signal Transduct. Target. Ther. 2025, 10, 102. [Google Scholar] [CrossRef]
- Satoh, M.; Vázquez-Del Mercado, M.; Chan, E.K. Clinical interpretation of antinuclear antibody tests in systemic rheumatic diseases. Mod. Rheumatol. 2009, 19, 219–228. [Google Scholar] [CrossRef]
- Almeida, R.M.; da Silva, Z.D.L.; Leite, F.B.; de Medeiros Nóbrega, Y.K. Antinuclear antibodies patterns in patients with celiac disease. Biomed. J. 2019, 1, 3. [Google Scholar] [CrossRef]
- Dahan, S.; Shor, D.B.; Comaneshter, D.; Tekes-Manova, D.; Shovman, O.; Amital, H.; Cohen, A.D. All disease begins in the gut: Celiac disease co-existence with SLE. Autoimmun. Rev. 2016, 15, 848–853. [Google Scholar] [CrossRef]
- Wananukul, S.; Voramethkul, W.; Kaewopas, Y.; Hanvivatvong, O. Prevalence of positive antinuclear antibodies in healthy children. Asian Pac. J. Allergy Immunol. 2005, 23, 153–157. [Google Scholar] [PubMed]
- Xue, J.; Zhang, Z.; Sun, Y.; Jin, D.; Guo, L.; Li, X.; Zhao, D.; Feng, X.; Qi, W.; Zhu, H. Research Progress and Molecular Mechanisms of Endothelial Cells Inflammation in Vascular-Related Diseases. J. Inflamm. Res. 2023, 16, 3593–3617. [Google Scholar] [CrossRef]
- Auricchio, R.; Calabrese, I.; Galatola, M.; Cielo, D.; Carbone, F.; Mancuso, M.; Matarese, G.; Troncone, R.; Auricchio, S.; Greco, L. Gluten consumption and inflammation affect the development of celiac disease in at-risk children. Sci. Rep. 2022, 12, 5396, Erratum in Sci. Rep. 2022, 12, 8157. https://doi.org/10.1038/s41598-022-12636-0. [Google Scholar] [CrossRef]
- Fontana, G.; Ziberna, F.; Barbi, E.; Di Leo, G.; De Leo, L. Intestinal celiac disease-related autoantibodies. Front. Immunol. 2025, 16, 1567416. [Google Scholar] [CrossRef]
- Lindeman, I.; Høydahl, L.S.; Christophersen, A.; Risnes, L.F.; Jahnsen, J.; Lundin, K.E.A.; Sollid, L.M.; Iversen, R. Generation of circulating autoreactive pre-plasma cells fueled by naive B cells in celiac disease. Cell Rep. 2024, 43, 114045. [Google Scholar] [CrossRef]
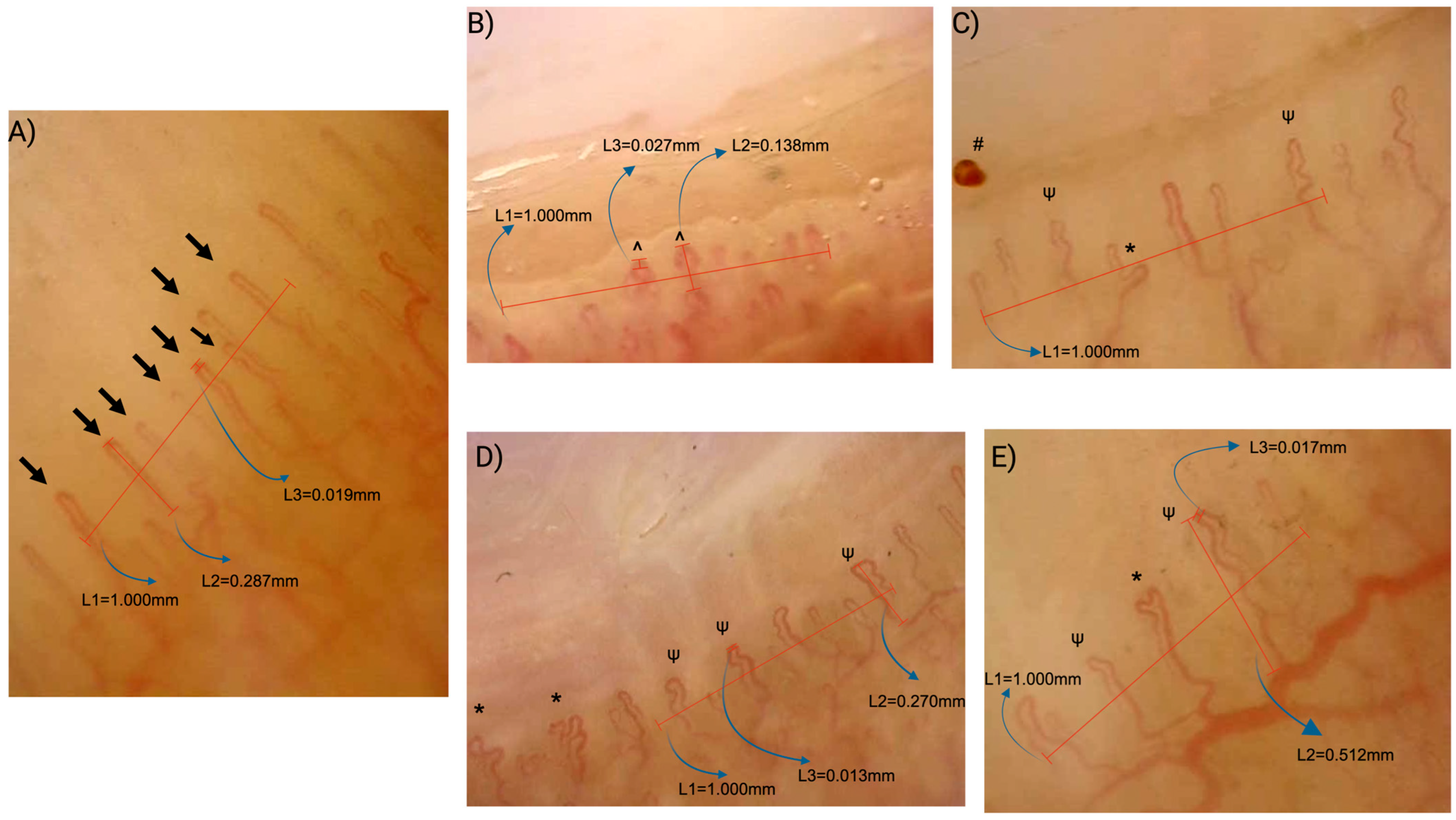
| Variables | Celiac Disease (n:76) | HCs (n:76) | p Value |
|---|---|---|---|
| Age years, median (min–max) | 13 (4–18) | 11 (5–17) | 0.08 µ |
| Gender (male/female) | 32/44 | 39/37 | 0.25 * |
| BMI-SDS median (min–max) | 0.2 (−3.2–2.1) | 0.45 (−2.1–2.2) | 0.005 µ |
| Disease duration months, median (min–max) | 23.5 (0–156) | - | - |
| tTG-IgA value U/mL, median (min–max) | 52 (10–200) | - | - |
| Patients without tTG-IgA normalization, n (%) | 44 (57.9) | - | - |
| ANA: Negative n (%) | 66 (86.8) | - | - |
| Variables | Celiac Disease (n:76) | HCs (n:76) | p Value |
|---|---|---|---|
| Capillaroscopic Findings (per mm) | |||
| Capillary density, median (IQR) | 7(6.813–7.063) | 7.063 (7–7.125) | <0.001 µ |
| Dilated capillaries, median (IQR) | 0 (0–0.094) | 0 (0–0) | <0.001 µ |
| Abnormal shapes, median (IQR) | 0.13 (0–0.19) | 0 (0–0) | <0.001 µ |
| Crossing capillaries, median (min–max) | 0.184 (0–0.938) | 0 (0–0.375) | <0.001 µ |
| Tortuous capillaries, median (min–max) | 0.125 (0–0.93) | 0 (0–0.313) | <0.001 µ |
| Capillary length (µm), median (IQR) | 275 (246.5–306.5) | 255 (230–290) | 0.026 µ |
| Reduced capillary density (<7), n (%) | 33 (43.4) | 5 (6.6) | <0.001 * |
| Presence of dilated capillaries, n (%) | 25 (32.9) | 3 (3.9) | <0.001 * |
| Presence of giant capillaries, n (%) | 0 | 0 | >0.05 |
| Presence of abnormal shapes, n (%) | 45 (59.2) | 0 (0) | <0.001 * |
| Presence of microhemorrhages, n (%) | 8 (10.5) | 0 (0) | 0.006 * |
| Presence of crossing capillaries, n (%) | 62 (81.6) | 15 (19.7) | <0.001 * |
| Presence of tortuous capillaries, n (%) | 45 (59.2) | 19 (25) | <0.001 * |
| Overall NVC pattern | |||
| Normal pattern, n (%) | 22 (28.9) | 68 (89.5) | <0.001 * |
| Nonspecific pattern, n (%) | 54 (71.1) | 8 (10.5) |
| Capillaroscopic Parameters (per mm) | Group 1 (n:44) | Group 2 (n:32) | HCs (n:76) | p Ƙ Value | p1−2 | p1−3 | p2−3 |
|---|---|---|---|---|---|---|---|
| Capillary length (µm), median (IQR) | 279.5 (242.5–325.5) | 270 (249–302) | 255 (230–290) | 0.081 | - | - | - |
| Capillary density, median (IQR) | 6.93 (6.81–7.06) | 7 (6.87–7) | 7.06 (7–7.12) | <0.001 | >0.999 | <0.001 | 0.003 |
| Dilated capillaries, median (IQR) | 0 (0–0.12) | 0 (0–0.03) | 0 (0–0) | <0.001 | 0.33 | <0.001 | 0.029 |
| Capillaries with abnormal shape, median (IQR) | 0.12 (0–0.18) | 0 (0–0.18) | 0 (0–0) | <0.001 | 0.022 | <0.001 | <0.001 |
| Crossing capillaries, median (min–max) | 0.18 (0–0.93) | 0.12 (0–0.68) | 0 (0–0.37) | <0.001 | >0.999 | <0.001 | <0.001 |
| Tortuous capillaries, median (min–max) | 0.12 (0–0.93) | 0.12 (0–0.62) | 0 (0–0.03) | <0.001 | >0.999 | <0.001 | 0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Çirkin, G.; Taskin, R.B. Evaluation of Nailfold Capillaroscopic Findings in Pediatric Patients with Celiac Disease: A Cross-Sectional and Comparative Study. Diagnostics 2025, 15, 2102. https://doi.org/10.3390/diagnostics15162102
Çirkin G, Taskin RB. Evaluation of Nailfold Capillaroscopic Findings in Pediatric Patients with Celiac Disease: A Cross-Sectional and Comparative Study. Diagnostics. 2025; 15(16):2102. https://doi.org/10.3390/diagnostics15162102
Chicago/Turabian StyleÇirkin, Gül, and Raziye Burcu Taskin. 2025. "Evaluation of Nailfold Capillaroscopic Findings in Pediatric Patients with Celiac Disease: A Cross-Sectional and Comparative Study" Diagnostics 15, no. 16: 2102. https://doi.org/10.3390/diagnostics15162102
APA StyleÇirkin, G., & Taskin, R. B. (2025). Evaluation of Nailfold Capillaroscopic Findings in Pediatric Patients with Celiac Disease: A Cross-Sectional and Comparative Study. Diagnostics, 15(16), 2102. https://doi.org/10.3390/diagnostics15162102








