Severity of Atelectasis during Bronchoscopy: Descriptions of a New Grading System (Atelectasis Severity Scoring System—“ASSESS”) and At-Risk-Lung Zones
Abstract
1. Introduction
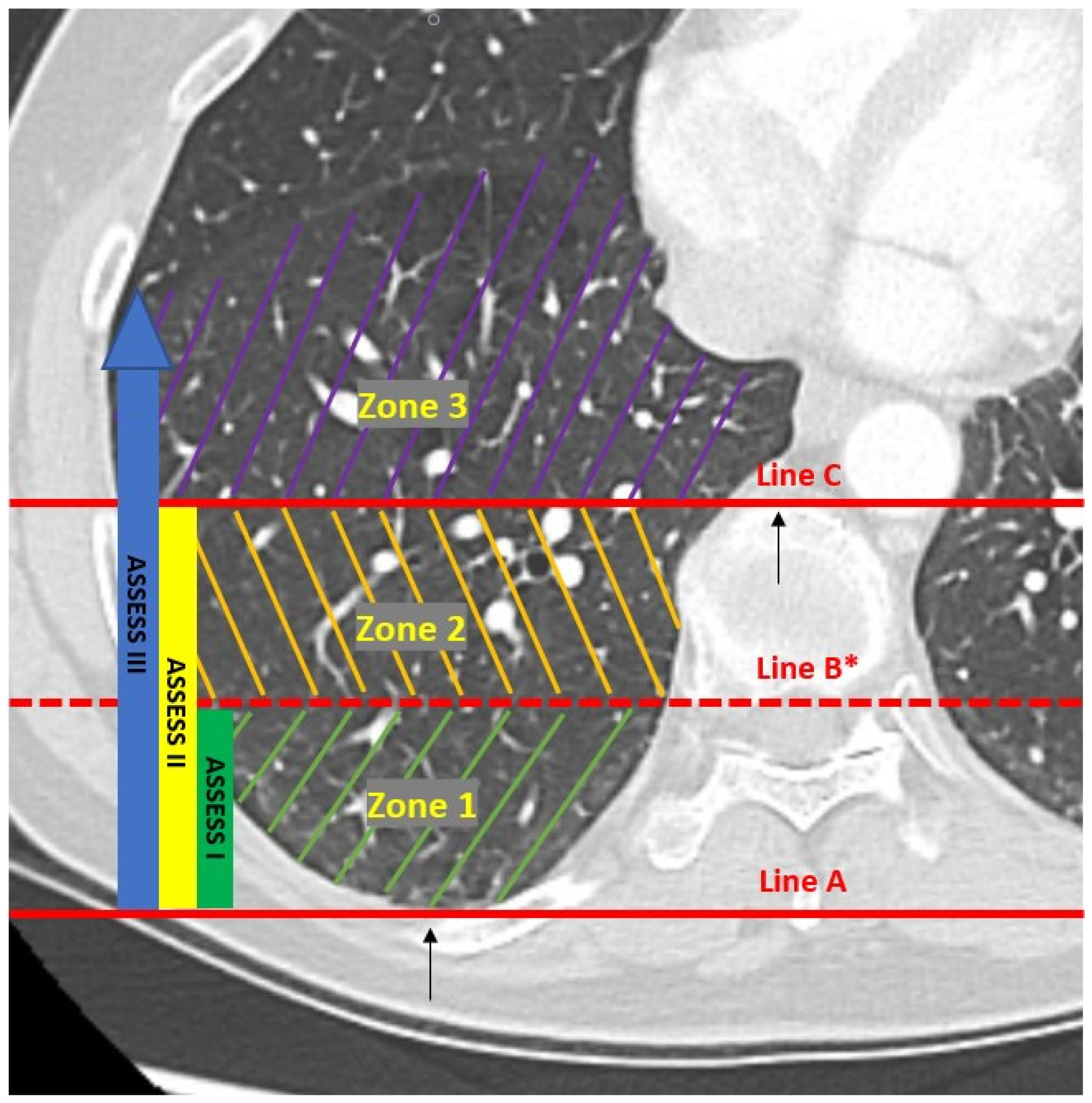
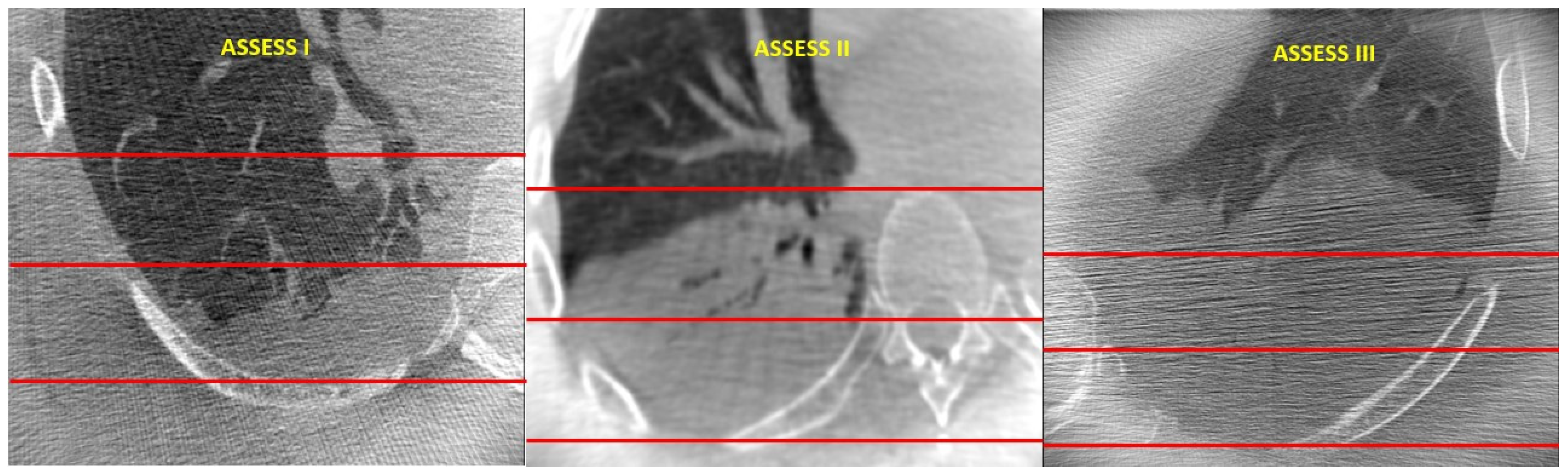
2. Materials and Methods
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Finley, T.N. Anesthesia and Atelectasis. Anesthesiology 1968, 29, 863–864. [Google Scholar] [CrossRef] [PubMed]
- Zeng, C.; Lagier, D.; Lee, J.W.; Vidal Melo, M.F. Perioperative Pulmonary Atelectasis: Part I. Biology and Mechanisms. Anesthesiology 2022, 136, 181–205. [Google Scholar] [CrossRef] [PubMed]
- Lagier, D.; Zeng, C.; Fernandez-Bustamante, A.; Vidal Melo, M.F. Perioperative Pulmonary Atelectasis: Part II. Clinical Implications. Anesthesiology 2022, 136, 206–236. [Google Scholar] [CrossRef] [PubMed]
- Hartland, B.L.; Newell, T.J.; Damico, N. Alveolar recruitment maneuvers under general anesthesia: A systematic review of the literature. Respir. Care 2015, 60, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Reinius, H.; Jonsson, L.; Gustafsson, S.; Sundbom, M.; Duvernoy, O.; Pelosi, P.; Hedenstierna, G.; Fredén, F. Prevention of atelectasis in morbidly obese patients during general anesthesia and paralysis: A computerized tomography study. Anesthesiology 2009, 111, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Ren, Q.; Li, X.; Han, H.; Peng, M.; Xie, K.; Wang, Z.; Wang, G. Effect of sigh in lateral position on postoperative atelectasis in adults assessed by lung ultrasound: A randomized, controlled trial. BMC Anesthesiol. 2022, 22, 215. [Google Scholar] [CrossRef]
- Casal, R.F.; Sarkiss, M.; Jones, A.K.; Stewart, J.; Tam, A.; Grosu, H.B.; Ost, D.E.; Jimenez, C.A.; Eapen, G.A. Cone beam computed tomography-guided thin/ultrathin bronchoscopy for diagnosis of peripheral lung nodules: A prospective pilot study. J. Thorac. Dis. 2018, 10, 6950–6959. [Google Scholar] [CrossRef]
- Sagar, A.E.S.; Sabath, B.F.; Eapen, G.A.; Song, J.; Marcoux, M.; Sarkiss, M.; Arain, M.H.; Grosu, H.B.; Ost, D.E.; Jimenez, C.A.; et al. Incidence and Location of Atelectasis Developed During Bronchoscopy Under General Anesthesia: The I-LOCATE Trial. Chest 2020, 158, 2658–2666. [Google Scholar] [CrossRef] [PubMed]
- Avasarala, S.K.; Machuzak, M.S.; Gildea, T.R. Multidimensional precision: Hybrid mobile 2D/3D C-arm assisted biopsy of peripheral lung nodules. J. Bronchol. Interv. Pulmonol. 2020, 27, 153–155. [Google Scholar] [CrossRef]
- Pritchett, M.A.; Lau, K.; Skibo, S.; Phillips, K.A.; Bhadra, K. Anesthesia considerations to reduce motion and atelectasis during advanced guided bronchoscopy. BMC Pulm. Med. 2021, 21, 240. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Pastis, N.; Furukawa, B.; Silvestri, G.A. The effect of respiratory motion on pulmonary nodule location during electromagnetic navigation bronchoscopy. Chest 2015, 147, 1275–1281. [Google Scholar] [CrossRef]
- Reisenauer, J.; Duke, J.D.; Kern, R.; Fernandez-Bussy, S.; Edell, E. Combining Shape-Sensing Robotic Bronchoscopy with Mobile Three-Dimensional Imaging to Verify Tool-in-Lesion and Overcome Divergence: A Pilot Study. Mayo Clin. Proc. Innov. Qual. Outcomes 2022, 6, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Tanner, N.T.; Yarmus, L.; Chen, A.; Memoli, J.W.; Mehta, H.J.; Pastis, N.J.; Silvestri, G.A. Standard Bronchoscopy with Fluoroscopy vs Thin Bronchoscopy and Radial Endobronchial Ultrasound for Biopsy of Pulmonary Lesions: A Multicenter, Prospective, Randomized Trial. Chest 2018, 154, 1035–1043. [Google Scholar] [CrossRef]
- Salahuddin, M.; Sarkiss, M.; Sagar, A.-E.S.; Vlahos, I.; Chang, C.H.; Shah, A.; Sabath, B.F.; Lin, J.; Song, J.; Moon, T.; et al. Ventilatory Strategy to Prevent Atelectasis During Bronchoscopy Under General Anesthesia. Chest 2022, 162, 1393–1401. [Google Scholar] [CrossRef] [PubMed]
- Aretha, D.; Dimitriou, E.; Antoniou, S. Ventilatory Strategy to Prevent Atelectasis During Bronchoscopy with General Anesthesia: The Role of Laryngeal Mask Airway. Chest 2022, 162, e281. [Google Scholar] [CrossRef]
- Lentz, R.; Shojaee, S. The Intersection of Ventilatory Strategy to Prevent Atelectasis and Teslas in Navigational Bronchoscopy. Chest 2022, 162, 1236–1237. [Google Scholar] [CrossRef] [PubMed]
- Bhadra, K.; Setser, R.M.; Condra, W.R.; Pritchett, M.A.D. Lung Navigation Ventilation Protocol to Optimize Biopsy of Peripheral Lung Lesions. J. Bronc. Interv. Pulmonol. 2022, 29, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Bhadra, K.; Baleeiro, C.; Patel, S.; Condra, W.R.; Bader, B.A.M.; Setser, R.M.; Youngblood, S. High Tidal Volume, High Positive End Expiratory Pressure and Apneic Breath Hold Strategies (Lung Navigation Ventilation Protocol) with Cone Beam Computed Tomography Bronchoscopic Biopsy of Peripheral Lung Lesions. J. Bronc. Interv. Pulmonol. 2023; ahead of print. [Google Scholar] [CrossRef]
- Bhadra, K.; Condra, W.; Setser, R.M. Out of the Box Thinking: Prone Bronchoscopy to Reduce Atelectasis. Bronchol. Interv. Pulmonol. 2022, 29, e57–e60. [Google Scholar] [CrossRef] [PubMed]
- Ho, E.; Hedstrom, G.; Murgu, S. Robotic bronchoscopy in diagnosing lung cancer-the evidence, tips and tricks: A clinical practice review. Ann. Transl. Med. 2023, 11, 359. [Google Scholar] [CrossRef] [PubMed]
| Covariate | Level | OR (95% CI) | p-Value |
|---|---|---|---|
| Age | In 1 Unit Change | 0.98 (0.93–1.03) | 0.3956 |
| BMI | In 1 Unit Change | 1.22 (1.00–1.48) | 0.0527 |
| Time from Induction to Intubation | In 1 Unit Change | 0.85 (0.66–1.10) | 0.2205 |
| Time from Induction to Time 2 Atelectasis Survey | In 1 Unit Change | 0.89 (0.78–1.02) | 0.1054 |
| Allocation Group | Control | 1.00 | |
| VESPA | 0.23 (0.05–1.02) | 0.0525 | |
| Gender | Female | 1.00 | |
| Male | 1.06 (0.32–3.56) | 0.9202 | |
| Time 1 Atelectasis Survey Score | 0 | 1.00 | |
| 1–2 | 2.14 (0.61–7.53) | 0.2364 |
| Covariate | Level | OR (95% CI) | p-Value |
|---|---|---|---|
| BMI | In 1 Unit Change | 1.5 (1.10–2.04) | 0.0098 |
| Allocation Group | Control | 1.00 | |
| VESPA | 0.05 (0.01–0.47) | 0.0080 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, A.; Bashour, S.; Sabath, B.; Lin, J.; Sarkiss, M.; Song, J.; Sagar, A.-E.S.; Shah, A.; Casal, R.F. Severity of Atelectasis during Bronchoscopy: Descriptions of a New Grading System (Atelectasis Severity Scoring System—“ASSESS”) and At-Risk-Lung Zones. Diagnostics 2024, 14, 197. https://doi.org/10.3390/diagnostics14020197
Khan A, Bashour S, Sabath B, Lin J, Sarkiss M, Song J, Sagar A-ES, Shah A, Casal RF. Severity of Atelectasis during Bronchoscopy: Descriptions of a New Grading System (Atelectasis Severity Scoring System—“ASSESS”) and At-Risk-Lung Zones. Diagnostics. 2024; 14(2):197. https://doi.org/10.3390/diagnostics14020197
Chicago/Turabian StyleKhan, Asad, Sami Bashour, Bruce Sabath, Julie Lin, Mona Sarkiss, Juhee Song, Ala-Eddin S. Sagar, Archan Shah, and Roberto F. Casal. 2024. "Severity of Atelectasis during Bronchoscopy: Descriptions of a New Grading System (Atelectasis Severity Scoring System—“ASSESS”) and At-Risk-Lung Zones" Diagnostics 14, no. 2: 197. https://doi.org/10.3390/diagnostics14020197
APA StyleKhan, A., Bashour, S., Sabath, B., Lin, J., Sarkiss, M., Song, J., Sagar, A.-E. S., Shah, A., & Casal, R. F. (2024). Severity of Atelectasis during Bronchoscopy: Descriptions of a New Grading System (Atelectasis Severity Scoring System—“ASSESS”) and At-Risk-Lung Zones. Diagnostics, 14(2), 197. https://doi.org/10.3390/diagnostics14020197







