Comparative Study of the Myocardium of Patients from Four COVID-19 Waves
Abstract
1. Introduction
2. Methods
2.1. Patients
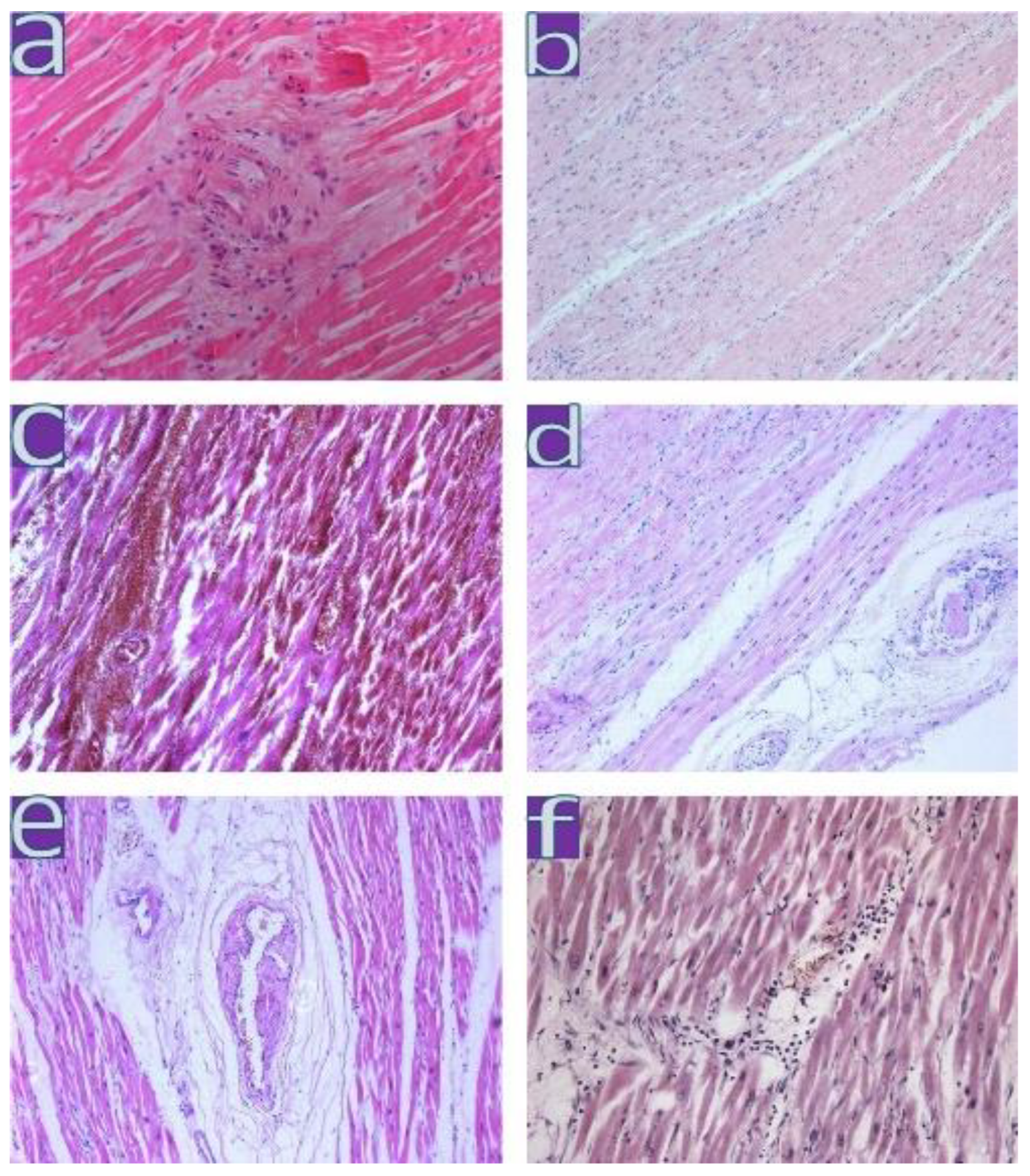
2.2. Histological Examination
2.3. Immunohistochemical Study
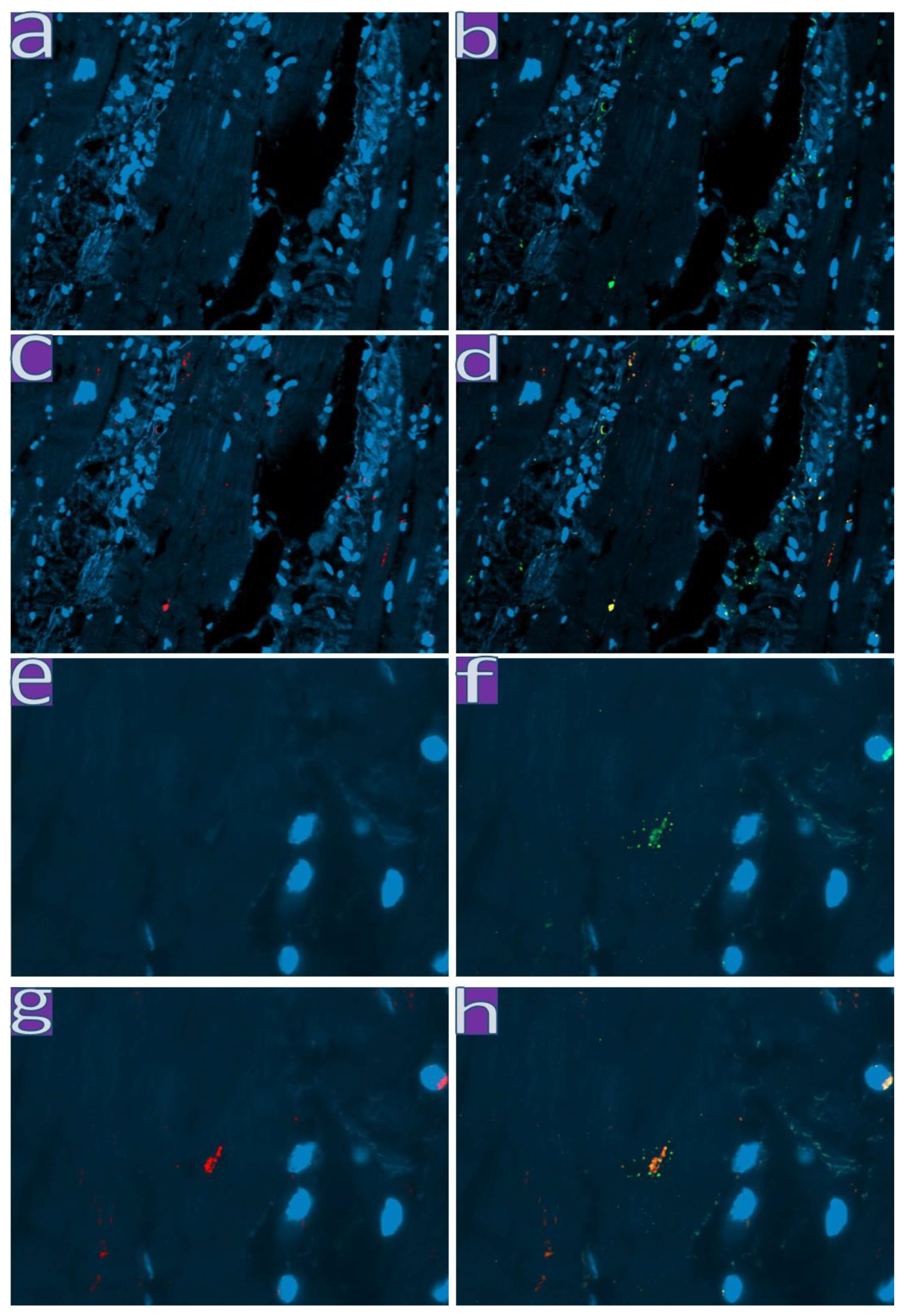
2.4. Immunofluorescence Study
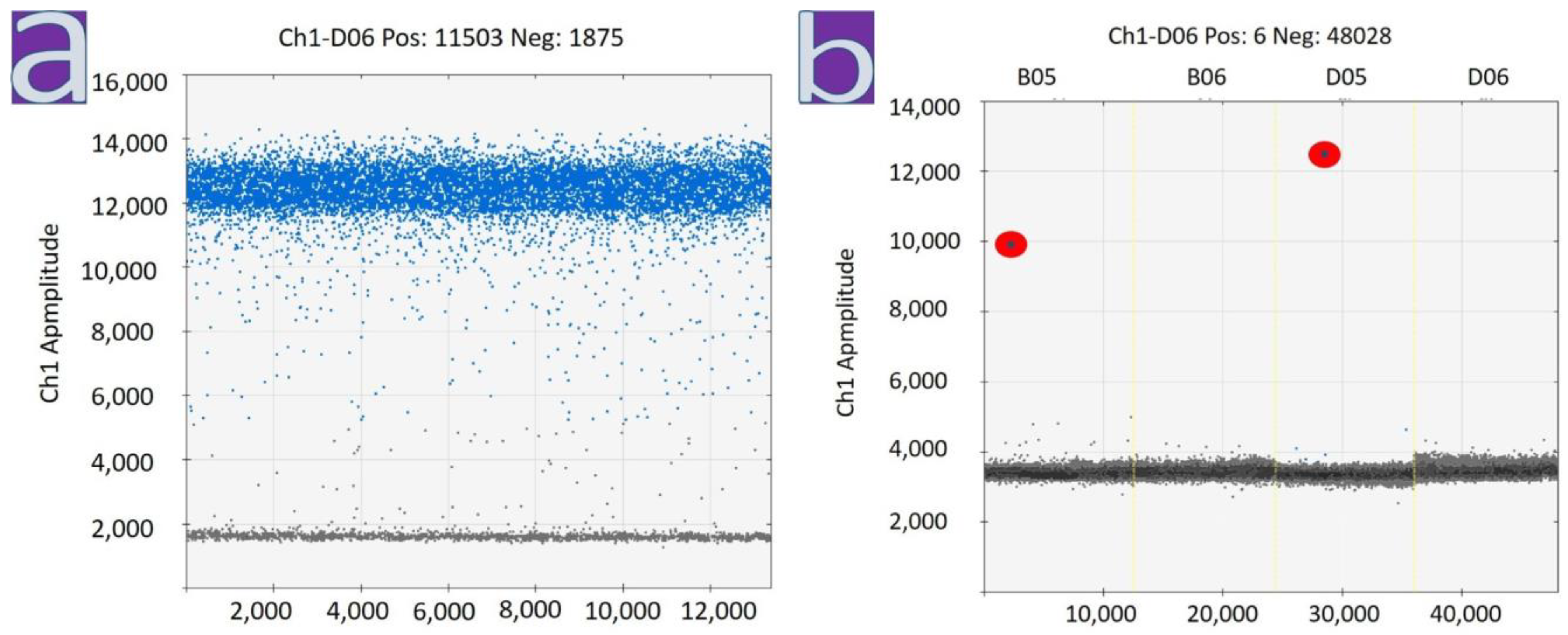
2.5. SARS-CoV-2 RT-ddPCR
3. Morphometry and Statistics
Electron Microscopy
4. Results
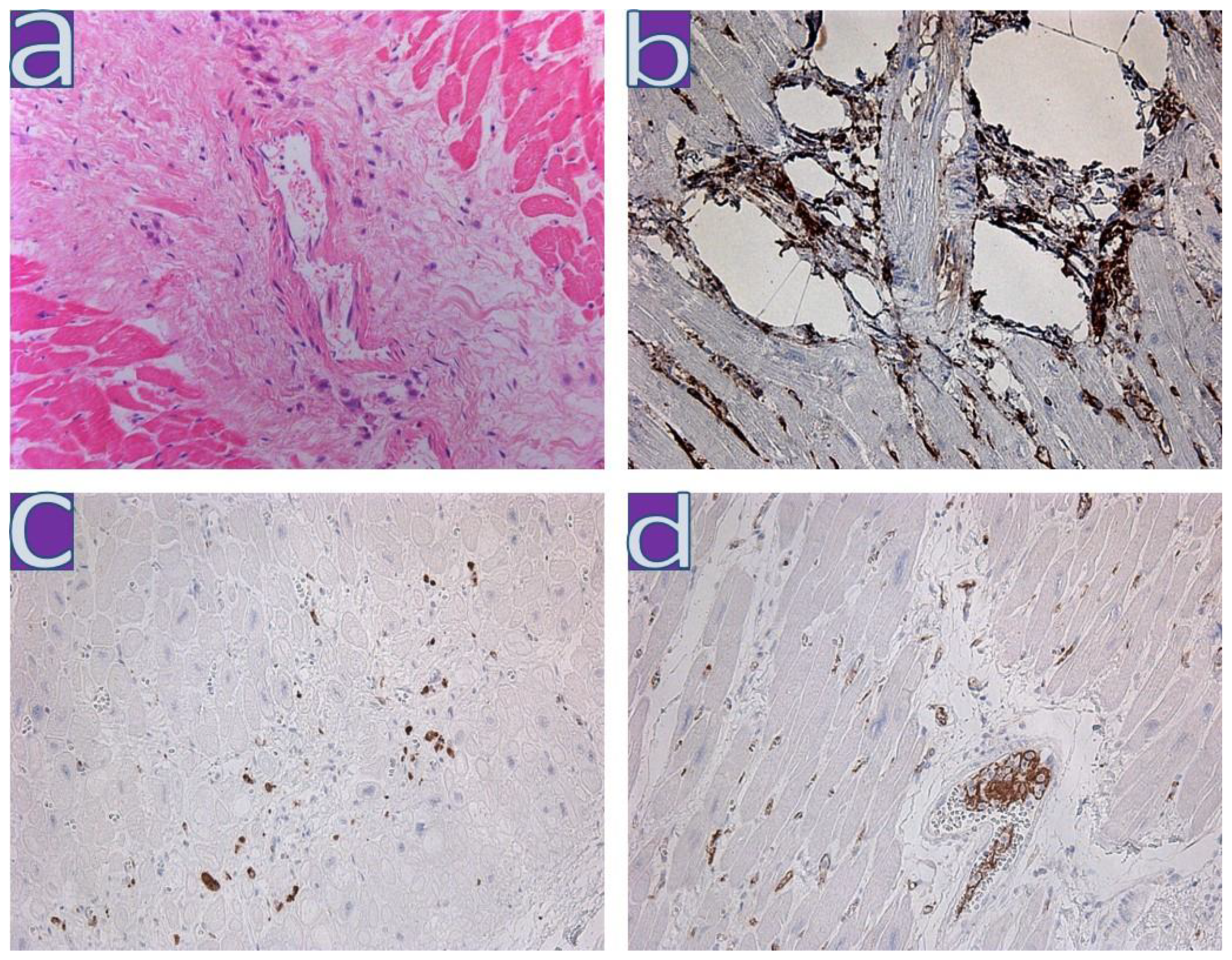
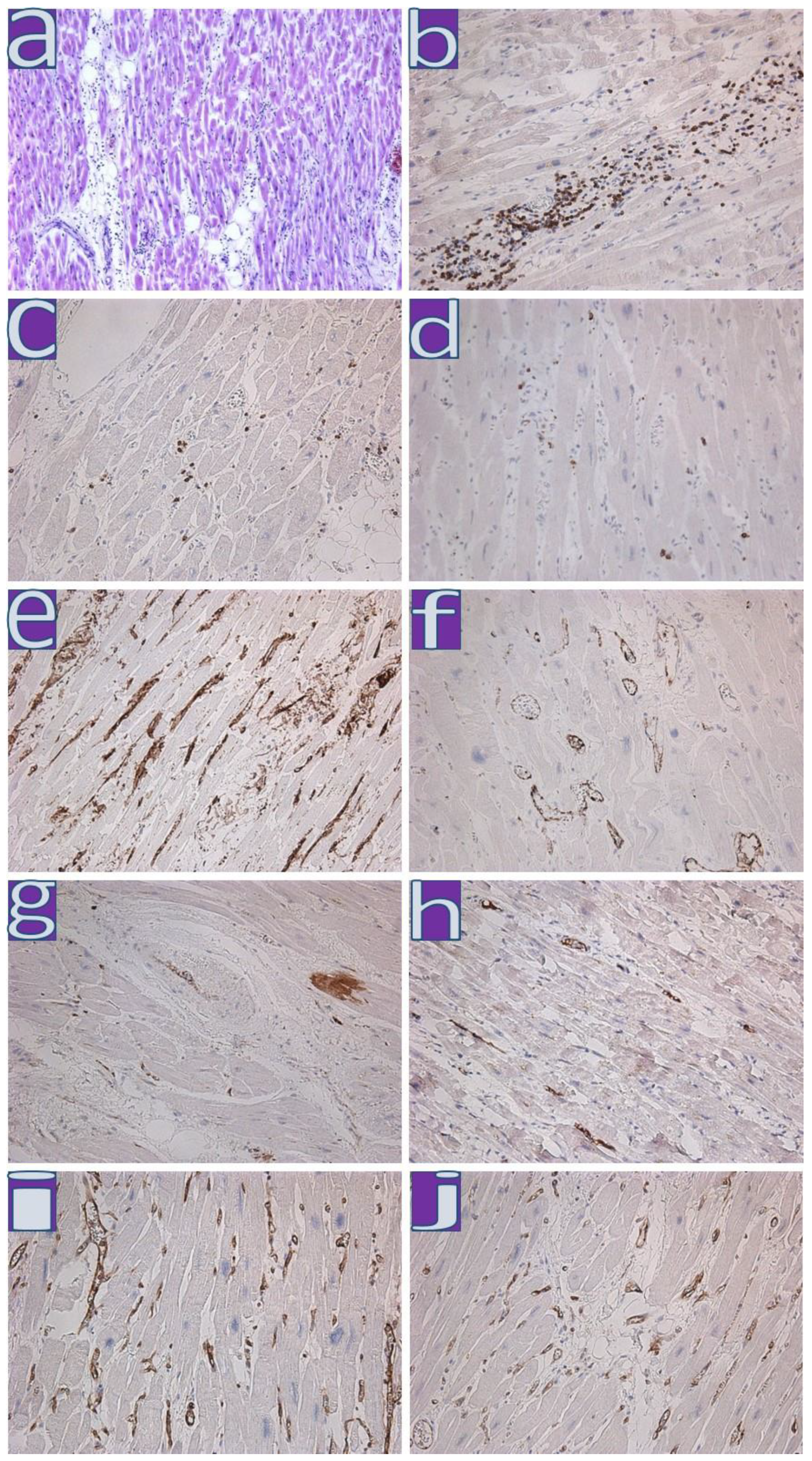
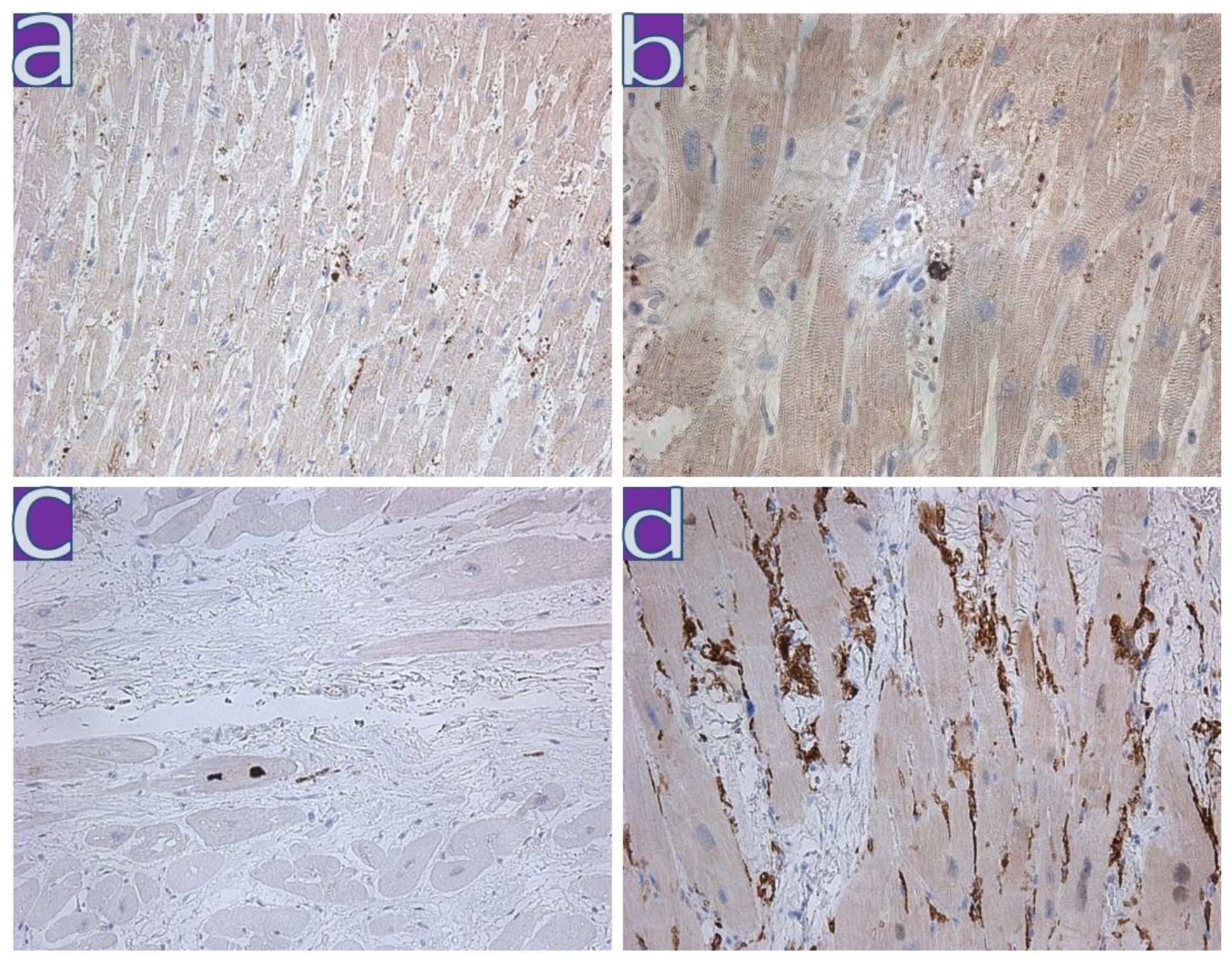
4.1. Clinical and Morphological Characteristics of the Myocardium of Patients from the First Wave
4.2. Clinical and Morphological Characteristics of the Myocardium of Patients from the Second Wave
4.3. Clinical and Morphological Characteristics of the Myocardium of Patients from the Third Wave
4.4. Clinical and Morphological Characteristics of the Myocardium of Patients from the Fourth Wave
| Myocardial Sample | Cq ORF1 | Cq ORF8 | Cq N |
|---|---|---|---|
| № 1 | 31, 10 | 32, 85 | 34, 27 |
| № 2 | NEGATIVE | NEGATIVE | NEGATIVE |
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Study Limitation
Abbreviations
| ACE 2 | angiotensin-converting enzyme 2 |
| AF | atrial fibrillation |
| AH | arterial hypertension |
| AMI | acute myocardial infarction |
| Ang1 | angiopoietin 1 |
| CCVD | chronic cerebrovascular disease |
| COPD | chronic obstructive pulmonary disease |
| CT | computed tomography |
| DM | diabetes mellitus |
| ECG | electrocardiography |
| EM | electron microscopy |
| HIV | human immunodeficiency virus |
| IF | immunofluorescence |
| IHC | immunohistochemistry |
| IHD | ischemic heart disease |
| RNA | ribonucleic acid |
| RT-ddPCR | droplet digital polymerase chain reaction after reverse transcription |
| RT-qPCR | quantitative polymerase chain reaction after reverse transcription |
| VEGF | vascular endothelial growth factor |
| VWF | von Willebrand factor |
References
- Saito, S.; Asai, Y.; Matsunaga, N.; Hayakawa, K.; Terada, M.; Ohtsu, H.; Tsuzuki, S.; Ohmagari, N. First and second COVID-19 waves in Japan: A comparison of disease severity and characteristics. J. Infect. 2021, 82, 84–123. [Google Scholar] [CrossRef]
- Iftimie, S.; López-Azcona, A.F.; Vallverdú, I.; Hernández-Flix, S.; de Febrer, G.; Parra, S.; Hernández-Aguilera, A.; Riu, F.; Joven, J.; Andreychuk, N.; et al. First and second waves of coronavirus disease-19: A comparative study in hospitalized patients in Reus, Spain. PLoS ONE 2021, 16, e0248029. [Google Scholar] [CrossRef]
- Wolfisberg, S.; Gregoriano, C.; Struja, T.; Kutz, A.; Koch, D.; Bernasconi, L.; Hammerer-Lercher, A.; Mohr, C.; Haubitz, S.; Conen, A.; et al. Comparison of characteristics, predictors and outcomes between the first and second COVID-19 waves in a tertiary care centre in Switzerland: An observational analysis. Swiss Med. Wkly. 2021, 151, w20569. [Google Scholar] [CrossRef]
- Diebold, M.; Martinez, A.E.; Adam, K.-M.; Bassetti, S.; Osthoff, M.; Kassi, E.; Steiger, J.; Pargger, H.; Siegemund, M.; Battegay, M.; et al. Temporal trends of COVID-19 related in-hospital mortality and demographics in Switzerland—A retrospective single centre cohort study. Swiss Med. Wkly. 2021, 151, w20572. [Google Scholar] [CrossRef]
- Vardavas, C.I.; Mathioudakis, A.G.; Nikitara, K.; Stamatelopoulos, K.; Georgiopoulos, G.; Phalkey, R.; Leonardi-Bee, J.; Fernandez, E.; Carnicer-Pont, D.; Vestbo, J.; et al. Prognostic factors for mortality, intensive care unit and hospital admission due to SARS-CoV-2: A systematic review and meta-analysis of cohort studies in Europe. Eur. Respir. Rev. 2022, 31, 220098. [Google Scholar] [CrossRef]
- Friedrich, M.G.; Cooper, L.T. What we (don’t) know about myocardial injury after COVID-19. Eur. Heart J. 2021, 42, 1879–1882. [Google Scholar] [CrossRef]
- Salabei, J.K.; Asnake, Z.T.; Ismail, Z.H.; Charles, K.; Stanger, G.-T.; Abdullahi, A.H.; Abraham, A.T.; Okonoboh, P. COVID-19 and the cardiovascular system: An update. Am. J. Med. Sci. 2022, 364, 139–147. [Google Scholar] [CrossRef]
- Buckley, B.J.; Harrison, S.L.; Fazio-Eynullayeva, E.; Underhill, P.; Lane, D.A.; Lip, G.Y. Prevalence and clinical outcomes of myocarditis and pericarditis in 718,365 COVID-19 patients. Eur. J. Clin. Investig. 2021, 51, e13679. [Google Scholar] [CrossRef]
- Merugu, G.P.; Nesheiwat, Z.; Balla, M.; Patel, M.; Fatima, R.; Sheikh, T.; Kotturi, V.; Bommana, V.; Pulagam, G.; Do, B.K. Predictors of mortality in 217 COVID-19 patients in Northwest Ohio, United States: A retrospective study. J. Med. Virol. 2021, 93, 2875–2882. [Google Scholar] [CrossRef]
- Linschoten, M.; Peters, S.; van Smeden, M.; Jewbali, L.S.; Schaap, J.; Siebelink, H.-M.; Smits, P.C.; Tieleman, R.G.; van der Harst, P.; van Gilst, W.H.; et al. Cardiac complications in patients hospitalised with COVID-19. Eur. Heart J. Acute Cardiovasc. Care 2020, 9, 817–823. [Google Scholar] [CrossRef]
- Giustino, G.; Pinney, S.P.; Lala, A.; Reddy, V.Y.; Johnston-Cox, H.A.; Mechanick, J.I.; Halperin, J.L.; Fuster, V. Coronavirus and Cardiovascular Disease, Myocardial Injury, and Arrhythmia. J. Am. Coll. Cardiol. 2020, 76, 2011–2023. [Google Scholar] [CrossRef]
- Zhao, S.; Lin, Q.; Ran, J.; Musa, S.S.; Yang, G.; Wang, W.; Lou, Y.; Gao, D.; Yang, L.; He, D.; et al. Preliminary estimation of the basic reproduction number of novel coronavirus (2019-nCoV) in China, from 2019 to 2020: A data-driven analysis in the early phase of the outbreak. Int. J. Infect. Dis. 2020, 92, 214–217. [Google Scholar] [CrossRef]
- Maiese, A.; Frati, P.; Del Duca, F.; Santoro, P.; Manetti, A.C.; La Russa, R.; Di Paolo, M.; Turillazzi, E.; Fineschi, V. Myocardial Pathology in COVID-19-Associated Cardiac Injury: A Systematic Review. Diagnostics 2021, 11, 1647. [Google Scholar] [CrossRef]
- Ferro, M.D.; Bussani, R.; Paldino, A.; Nuzzi, V.; Collesi, C.; Zentilin, L.; Schneider, E.; Correa, R.; Silvestri, F.; Zacchigna, S.; et al. SARS-CoV-2, myocardial injury and inflammation: Insights from a large clinical and autopsy study. Clin. Res. Cardiol. 2021, 110, 1822–1831. [Google Scholar] [CrossRef]
- Lindner, D.; Fitzek, A.; Bräuninger, H.; Aleshcheva, G.; Edler, C.; Meissner, K.; Scherschel, K.; Kirchhof, P.; Escher, F.; Schultheiss, H.-P.; et al. Association of Cardiac Infection With SARS-CoV-2 in Confirmed COVID-19 Autopsy Cases. JAMA Cardiol. 2020, 5, 1281–1285. [Google Scholar] [CrossRef]
- Roshdy, A.; Zaher, S.; Fayed, H.; Coghlan, J.G. COVID-19 and the Heart: A Systematic Review of Cardiac Autopsies. Front. Cardiovasc. Med. 2021, 7, 626975. [Google Scholar] [CrossRef]
- Tavazzi, G.; Pellegrini, C.; Maurelli, M.; Belliato, M.; Sciutti, F.; Bottazzi, A.; Sepe, P.A.; Resasco, T.; Camporotondo, R.; Bruno, R.; et al. Myocardial localization of coronavirus in COVID-19 cardiogenic shock. Eur. J. Heart Fail. 2020, 22, 911–915. [Google Scholar] [CrossRef]
- Fox, S.E.; Li, G.; Akmatbekov, A.; Harbert, J.L.; Lameira, F.S.; Brown, J.Q.; Heide, R.S.V. Unexpected Features of Cardiac Pathology in COVID-19 Infection. Circulation 2020, 142, 1123–1125. [Google Scholar] [CrossRef]
- Bulfamante, G.P.; Perrucci, G.L.; Falleni, M.; Sommariva, E.; Tosi, D.; Martinelli, C.; Songia, P.; Poggio, P.; Carugo, S.; Pompilio, G. Evidence of SARS-CoV-2 Transcriptional Activity in Cardiomyocytes of COVID-19 Patients without Clinical Signs of Cardiac Involvement. Biomedicines 2020, 8, 626. [Google Scholar] [CrossRef]
- Mukerji, S.S.; Solomon, I.H. What can we learn from brain autopsies in COVID-19? Neurosci. Lett. 2021, 742, 135528. [Google Scholar] [CrossRef]
- Romanova, E.S.; Vasilyev, V.V.; Startseva, G.; Karev, V.; Rybakova, M.G.; Platonov, P.G. Cause of death based on systematic post-mortem studies in patients with positive SARS-CoV-2 tissue PCR during the COVID-19 pandemic. J. Intern. Med. 2021, 290, 655–665. [Google Scholar] [CrossRef]
- Barchuk, A.; Skougarevskiy, D.; Kouprianov, A.; Shirokov, D.; Dudkina, O.; Tursun-Zade, R.; Sergeeva, M.; Tychkova, V.; Komissarov, A.; Zheltukhina, A.; et al. COVID-19 pandemic in Saint Petersburg, Russia: Combining population-based serological study and surveillance data. PLoS ONE 2022, 17, e0266945. [Google Scholar] [CrossRef]
- Mitrofanova, L.B.; Konovalov, P.V.; Krylova, J.S.; Polyakova, V.O.; Kvetnoy, I.M. Plurihormonal cells of normal anterior pituitary: Facts and conclusions. Oncotarget 2017, 8, 29282–29299. [Google Scholar] [CrossRef]
- Suo, T.; Liu, X.; Feng, J.; Guo, M.; Hu, W.; Guo, D.; Ullah, H.; Yang, Y.; Zhang, Q.; Wang, X.; et al. ddPCR: A more accurate tool for SARS-CoV-2 detection in low viral load specimens. Emerg. Microbes Infect. 2020, 9, 1259–1268. [Google Scholar] [CrossRef]
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.W.; Bleicker, T.; Brünink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Eurosurveillance 2020, 25, 2000045. [Google Scholar] [CrossRef]
- Caforio, A.L.P.; Pankuweit, S.; Arbustini, E.; Basso, C.; Gimeno-Blanes, J.; Felix, S.B.; Fu, M.; Heliö, T.; Heymans, S.; Jahns, R.; et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: A position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 2013, 34, 2636–2648. [Google Scholar] [CrossRef]
- Verduri, A.; Short, R.; Carter, B.; Braude, P.; Vilches-Moraga, A.; Quinn, T.J.; Collins, J.; Lumsden, J.; McCarthy, K.; Evans, L.; et al. Comparison between first and second wave of COVID-19 outbreak in older people: The COPE multicentre European observational cohort study. Eur. J. Public Health 2022, 32, 807–812. [Google Scholar] [CrossRef]
- Contou, D.; Fraissé, M.; Pajot, O.; Tirolien, J.-A.; Mentec, H.; Plantefève, G. Comparison between first and second wave among critically ill COVID-19 patients admitted to a French ICU: No prognostic improvement during the second wave? Crit. Care 2021, 25, 3. [Google Scholar] [CrossRef]
- Buttenschøn, H.N.; Lynggaard, V.; Sandbøl, S.G.; Glassou, E.N.; Haagerup, A. Comparison of the clinical presentation across two waves of COVID-19: A retrospective cohort study. BMC Infect. Dis. 2022, 22, 423. [Google Scholar] [CrossRef]
- Mollinedo-Gajate, I.; Villar-Álvarez, F.M.; Zambrano-Chacón, M.D.L.; Núñez-García, L.; de la Dueña-Muñoz, L.I.; López-Chang, C.; Górgolas, M.M.; Cabello, A.M.; Sánchez-Pernaute, O.M.; Romero-Bueno, F.M.; et al. First and Second Waves of Coronavirus Disease 2019 in Madrid, Spain: Clinical Characteristics and Hematological Risk Factors Associated With Critical/Fatal Illness. Crit. Care Explor. 2021, 3, e0346. [Google Scholar] [CrossRef]
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [CrossRef]
- Escher, F.; Pietsch, H.; Aleshcheva, G.; Bock, T.; Baumeier, C.; Elsaesser, A.; Wenzel, P.; Hamm, C.; Westenfeld, R.; Schultheiss, M.; et al. Detection of viral SARS-CoV-2 genomes and histopathological changes in endomyocardial biopsies. ESC Heart Fail. 2020, 7, 2440–2447. [Google Scholar] [CrossRef]
- Caforio, A.L.; Baritussio, A.; Basso, C.; Marcolongo, R. Clinically Suspected and Biopsy-Proven Myocarditis Temporally Associated with SARS-CoV-2 Infection. Annu. Rev. Med. 2022, 73, 149–166. [Google Scholar] [CrossRef]
- Fraser, M.; Agdamag, A.C.C.; Maharaj, V.R.; Mutschler, M.; Charpentier, V.; Chowdhury, M.; Alexy, T. COVID-19-Associated Myocarditis: An Evolving Concern in Cardiology and Beyond. Biology 2022, 11, 520. [Google Scholar] [CrossRef]
- Andréoletti, L.; Bourlet, T.; Moukassa, D.; Rey, L.; Hot, D.; Li, Y.; Lambert, V.; Gosselin, B.; Mosnier, J.; Stankowiak, C.; et al. Enteroviruses Can Persist with or without Active Viral Replication in Cardiac Tissue of Patients with End-Stage Ischemic or Dilated Cardiomyopathy. J. Infect. Dis. 2000, 182, 1222–1227. [Google Scholar] [CrossRef]
- Marjomäki, V.; Kalander, K.; Hellman, M.; Permi, P. Enteroviruses and coronaviruses: Similarities and therapeutic targets. Expert Opin. Ther. Targets 2021, 25, 479–489. [Google Scholar] [CrossRef]
- Tao, Z.; Chen, B.; Tan, X.; Zhao, Y.; Wang, L.; Zhu, T.; Cao, K.; Yang, Z.; Kan, Y.W.; Su, H. Coexpression of VEGF and angiopoietin-1 promotes angiogenesis and cardiomyocyte proliferation reduces apoptosis in porcine myocardial infarction (MI) heart. Proc. Natl. Acad. Sci. USA 2011, 108, 2064–2069. [Google Scholar] [CrossRef]
- Dallabrida, S.M.; Ismail, N.; Oberle, J.R.; Himes, B.E.; Rupnick, M.A. Angiopoietin-1 Promotes Cardiac and Skeletal Myocyte Survival Through Integrins. Circ. Res. 2005, 96, e8–e24. [Google Scholar] [CrossRef]
- Ray, P.S.; Estrada-Hernandez, T.; Sasaki, H.; Zhu, L.; Maulik, N. Early effects of hypoxia/reoxygenation on VEGF, ang-1, ang-2 and their receptors in the rat myocardium: Implications for myocardial angiogenesis. Mol. Cell Biochem. 2000, 213, 145–153. [Google Scholar] [CrossRef]
- Mojiri, A.; Alavi, P.; Carrillo, M.A.L.; Nakhaei-Nejad, M.; Sergi, C.M.; Thebaud, B.; Aird, W.C.; Jahroudi, N. Endothelial cells of different organs exhibit heterogeneity in von Willebrand factor expression in response to hypoxia. Atherosclerosis 2019, 282, 1–10. [Google Scholar] [CrossRef]
- Spiel, A.; Gilbert, J.C.; Jilma, B. von Willebrand factor in cardiovascular disease: Focus on acute coronary syndromes. Circulation 2008, 117, 1449–1459. [Google Scholar] [CrossRef]
- Deng, H.; Tang, T.-X.; Chen, D.; Tang, L.-S.; Yang, X.-P.; Tang, Z.-H. Endothelial Dysfunction and SARS-CoV-2 Infection: Association and Therapeutic Strategies. Pathogens 2021, 10, 582. [Google Scholar] [CrossRef]
- Rauti, R.; Shahoha, M.; Leichtmann-Bardoogo, Y.; Nasser, R.; Paz, E.; Tamir, R.; Miller, V.; Babich, T.; Shaked, K.; Ehrlich, A.; et al. Effect of SARS-CoV-2 proteins on vascular permeability. Elife 2021, 10, e69314. [Google Scholar] [CrossRef]
- Smadja, D.M.; Mentzer, S.J.; Fontenay, M.; Laffan, M.A.; Ackermann, M.; Helms, J.; Jonigk, D.; Chocron, R.; Pier, G.B.; Gendron, N.; et al. COVID-19 is a systemic vascular hemopathy: Insight for mechanistic and clinical aspects. Angiogenesis 2021, 24, 755–788. [Google Scholar] [CrossRef]
- Smeeth, L.; Thomas, S.L.; Hall, A.J.; Hubbard, R.; Farrington, P.; Vallance, P. Risk of Myocardial Infarction and Stroke after Acute Infection or Vaccination. N. Engl. J. Med. 2004, 351, 2611–2618. [Google Scholar] [CrossRef]
- Hirsch, J.S.; Ng, J.H.; Ross, D.W.; Sharma, P.; Shah, H.H.; Barnett, R.L.; Hazzan, A.D.; Fishbane, S.; Jhaveri, K.D.; on behalf of theNorthwell COVID-19 Research Consortium and theNorthwell Nephrology COVID-19 Research Consortium. Acute kidney injury in patients hospitalized with COVID-19. Kidney Int. 2020, 98, 209–218. [Google Scholar] [CrossRef]
- Cascella, M.; Rajnik, M.; Aleem, A.; Dulebohn, S.C.; Di Napoli, R. Features, Evaluation, and Treatment of Coronavirus (COVID-19). In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]


| Assay Designation | Genbank Accession | Name | Sequence (5′ to 3′) |
|---|---|---|---|
| E | NC_045512.2 | E-F1 | ACAGGTACGTTAATAGTTAATAGCGT |
| E-R2 | ATATTGCAGCAGTACGCACACA | ||
| E-P1 | FAM-ACACTAGCCATCCTTACTGCGCTTCG-RTQ1 | ||
| N | NC_045512.2 | N1 | GGGGAACTTCTCCTGCTAGAAT |
| N2 | CAGACATTTTGCTCTCAAGCTG | ||
| N3 | FAM-TTGCTGCTGCTTGACAGATT-RTQ1 | ||
| Act | NM_001101.5 | Act-F | ACCGAGCGCGGCTACAG |
| Act-R | GGCCATCTCTTGCTCGAAGT | ||
| Act-Pr | FAM-AGCGGGAAATCGTGCGTGACATTA-RTQ1 |
| Patients without Myocarditis | Patients with Myocarditis | |||||
|---|---|---|---|---|---|---|
| I Wave (n = 77) | II Wave (n = 119) | III Wave (n = 78) | I Wave (n = 8) | II Wave (n = 1) | III Wave (n = 5) | |
| Age (year) | 74 (IQR 66–79) | 74 (IQR 71–80) | 72 (IQR 68–75) | 62 (IQR 61–68) | 58 | 82 (IQR 73–84) |
| Time for onset of symptoms (day) | 5 (IQR 3–6) | 7 (IQR 6–7) | 7 (IQR 8–9) | 4 (IQR 4–10) | 7 | 8 (IQR 7–11) |
| Time from onset of symptoms to death (day) | 18 (IQR 14–20) | 19 (IQR 17–22) | 20 (IQR 16–24) | 23 (IQR 20–23) | 29 | 22 (IQR 16–23) |
| C-reactive protein (mg/L) | 112.9 (IQR 60.6–114.1) | 88.3 (IQR 64.3–105.1) | 87.3 (IQR 54.1–116.9) | 124.5 (IQR 161.0–250.0) | 113.1 | 34.7 (IQR 27.8–50.5) |
| D-dimer (mcg/mL) | 2.1 (IQR 1.5–2.7) | 0.7 (IQR 0.6–0.8) | 0.6 (IQR 0.5–0.7) | 1.5 (IQR 1.3–4.9) | 1.0 | 0.4 (IQR 0.2–0.4) |
| Troponin (ng/mL) | 200 (IQR 130–230) | 100 (IQR 100–100) | 100 (IQR 100–100) | 570 (IQR 120–390) | 100 | 100 (IQR 100–670) |
| Ferritin (mcg/L) | 1159 (IQR 846–1880) | 621 (IQR 461–735) | 869 (IQR 638–1121) | 1246 (IQR 420–3175) | 1124 | 341 (IQR 420–1233) |
| Procalcitonin (ng/mL) | 0.49 (IQR 0.45–1.84) | 0.15 (IQR 0.11–0.18) | 0.14 (IQR 0.12–0.20) | 0.44 (IQR 0.35–0.94) | 0 | 0.15 (IQR 0.03–0.62) |
| Lymphocytes (×109/L) | 0.63 (IQR 0.55–0.80) | 0.91 (IQR 0.77–1.04) | 0.70 (IQR 0.58–0.76) | 0.59 (IQR 0.56–1.58) | 0.48 | 0.72 (IQR 0.61–0.83) |
| Lung lesions on CT till the moment of hospitalization (%) | 75 (IQR 60–80) | 45 (IQR 40–56) | 40 (IQR 35–50) | 83 (IQR 70–85) | 75 | 56 (IQR 20–56) |
| SpO2 (%) | 92 (IQR 88–94) | 90 (IQR 89–92) | 90 (IQR 88–92) | 89 (IQR 85–91) | 80 | 87 (IQR 75–93) |
| Mean left ventricular ejection fraction till the moment of hospitalization (%) | 60 (IQR 55–64) | 57 (IQR 53–60) | 59 (IQR 47–60) | 47 (IQR 30–70) | 65 | 55 (IQR 48–70) |
| № | Wave | Changes in the Myocardium | CD3/1mm2 | CD8/1mm2 | CD68/1mm2 | MHC1 (in Points) | HLA-DR (in Points) | C1q (in Points) | VP1 Enterovirus (in Points) | SARS-CoV-2 (in Points) | VEGF (in Points) | Ang1 (in Points) | VWF (in Points) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | I | Lymphocytic myocarditis | 15 | 0 | 1 | 4 V 0 C | 2 V 0 C | 0 V 0 C | 0 V 0 C | 0 V 0 C | 0 V 0 C | 2 V 4 C | 3 V 0 C |
| 2 | I | Lymphocytic myocarditis | 20 | 0 | 8 | 4 V 0 C | 2 V 0 C | 2 V 0 C | 4 V 2 C | 0 V 0 C | 4 V 0 C | 4 V 4 C | 3 V 0 C |
| 3 | I | Lymphocytic myocarditis Non-coronary necrosis, thrombi in intramyocardial arteries | 11 | 0 | 6 | 3 V 0 C | 0 V 0 C | 1 V 0 C | 0 V 0 C | 0 V 0 C | 4 V 2 C | 4 V 4 C | 1 V 0 C |
| 4 | I | Lymphocytic myocarditis | 8 | 0 | 14 | 4 V 0 C | 1 V 30 C | 2 V 0 C | 4 V 0 C | 0 V 0 C | 4 V 0 C | 4 V 2 C | 4 V 0 C |
| 5 | I | Lymphocytic myocarditis Acute myocardial infarction | 15 | 0 | 32 | 3 V 0 C | 0 V 0 C | 2 V 0 C | 4 V 0 C | 0 V 0 C | 2 V 2 C | 4 V 3 C | 4 V 0 C |
| 6 | I | Lymphocytic myocarditis Non-coronary necrosis, thrombi in intramyocardial arteries | 37 | 0 | 2 | 4 V 0 C | 3 V 0 C | 3 V 0 C | 4 V 3 C | 0 V 0 C | 4 V 2 C | 2 V 4 C | 1 V 0 C |
| 7 | I | Lymphocytic myocarditis | 21 | 0 | 0 | 4 V 0 C | 4 V 0 C | 0 V 0 C | 4 V 4 C | 1 V 0 C | 3 V 1 C | 4 V 4 C | 1 V 0 C |
| 8 | I | Lymphocytic myocarditis | 16 | 0 | 20 | 4 V 0 C | 0 V 0 C | 3 V 0 C | 4 V 0 C | 0 V 30 C | 3 V 0 C | 4 V 4 C | 4 V 0 C |
| 9 | II | Focal lymphocytic myocarditis | 123 | 0 | 38 | 4 V 0 C | 0 V 0 C | 3 V 0 C | 4 V 4 C | 0 V 0 C | 3 V 0 C | 4 V 4 C | 3 V 0 C |
| 10 | III | Focal lymphocytic myocarditis Cardiosclerosis | 19 | 0 | 56 | 4 V 0 C | 4 V 0 C | 2 V 0 C | 0 V 4 C | 0 V 0 C | 1 V 0 C | 4 V 4 C | 3 V 0 C |
| 11 | III | Lymphocytic myocarditis | 29 | 0 | 33 | 4 V 0 C | 2 V 0 C | 0 V 0 C | 0 V 2 C | 2 V 0 C | 1 V 0 C | 4 V 4 C | 4 V 0 C |
| 12 | III | Lymphocytic myocarditis | 7 | 0 | 25 | 4 V 0 C | 2 V 0 C | 0 V 0 C | 4 V 0 C | 1 V 0 C | 4 V 4 C | 4 V 4 C | 3 V 0 C |
| 13 | III | Lymphocytic myocarditis | 18 | 8 | 30 | 3 V 0 C | 2 V 0 C | 2 V 0 C | 4 V 0 C | 0 V 0 C | 4 V 3 C | 4 V 4 C | 3 V 0 C |
| 14 | III | Lymphocytic myocarditis Severe fibrosis of the endocardium | 98 | 25 | 41 | 4 V 0 C | 4 V 0 C | 3 V 0 C | 1 V 1 C | 0 V 0 C | 4 V 4 C | 4 V 4 C | 4 V 0 C |
| Histological Features | I Wave n = 77 | II Wave n = 119 | III Wave n = 78 |
|---|---|---|---|
| Large polymorphic cells located in the vascular zone | 56% | 1% | 1% |
| Infarction/necrosis | 18% | 11% | 4% |
| Hemorrhagic infarction | 6% | 6% | 1% |
| Thrombi in the arteries | 12% | 7% | 0% |
| Myocarditis | 19% | 1% | 6% |
| Postinfarction cardiosclerosis | 19% | 26% | 9% |
| Thrombi in the endocardium | 0% | 0% | 0% |
| Pericarditis | 1% | 2% | 4% |
| Amyloidosis/glycogenosis | 0% | 0% | 1% |
| Fragmentation of muscle fibers | 29% | 23% | 36% |
| Pustular myocarditis | 4% | 3% | 1% |
| Vasculitis | 9% | 1% | 1% |
| Macrophages | 25% | 21% | 6% |
| Hemorrhage | 48% | 24% | 17% |
| Edema with fibrin | 8% | 24% | 17% |
| Myocardial hypertrophy | 47% | 69% | 68% |
| Cancer metastasis | 0% | 3% | 0% |
| Lipomatosis | 3% | 10% | 6% |
| Spasm | 18% | 24% | 32% |
| Microbial emboli | 0% | 3% | 1% |
| Focal plasma impregnation | 13% | 8% | 19% |
| Post myocardial cardiosclerosis | 0% | 3% | 3% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mitrofanova, L.B.; Makarov, I.A.; Gorshkov, A.N.; Runov, A.L.; Vonsky, M.S.; Pisareva, M.M.; Komissarov, A.B.; Makarova, T.A.; Li, Q.; Karonova, T.L.; et al. Comparative Study of the Myocardium of Patients from Four COVID-19 Waves. Diagnostics 2023, 13, 1645. https://doi.org/10.3390/diagnostics13091645
Mitrofanova LB, Makarov IA, Gorshkov AN, Runov AL, Vonsky MS, Pisareva MM, Komissarov AB, Makarova TA, Li Q, Karonova TL, et al. Comparative Study of the Myocardium of Patients from Four COVID-19 Waves. Diagnostics. 2023; 13(9):1645. https://doi.org/10.3390/diagnostics13091645
Chicago/Turabian StyleMitrofanova, Lubov Borisovna, Igor Aleksandrovich Makarov, Andrey Nikolaevich Gorshkov, Andrey Leonidovich Runov, Maxim Sergeevich Vonsky, Maria Mikhailovna Pisareva, Andrey Borisovich Komissarov, Taiana Alekseevna Makarova, Qingli Li, Tatiana Leonidovna Karonova, and et al. 2023. "Comparative Study of the Myocardium of Patients from Four COVID-19 Waves" Diagnostics 13, no. 9: 1645. https://doi.org/10.3390/diagnostics13091645
APA StyleMitrofanova, L. B., Makarov, I. A., Gorshkov, A. N., Runov, A. L., Vonsky, M. S., Pisareva, M. M., Komissarov, A. B., Makarova, T. A., Li, Q., Karonova, T. L., Konradi, A. O., & Shlaykhto, E. V. (2023). Comparative Study of the Myocardium of Patients from Four COVID-19 Waves. Diagnostics, 13(9), 1645. https://doi.org/10.3390/diagnostics13091645







