A Hypothesis on the Progression of Insulin-Induced Lipohypertrophy: An Integrated Result of High-Frequency Ultrasound Imaging and Blood Glucose Control of Patients
Abstract
1. Introduction
2. Research Design and Methods
2.1. Study Participants
2.2. Data Collection
2.3. Statistical Analysis
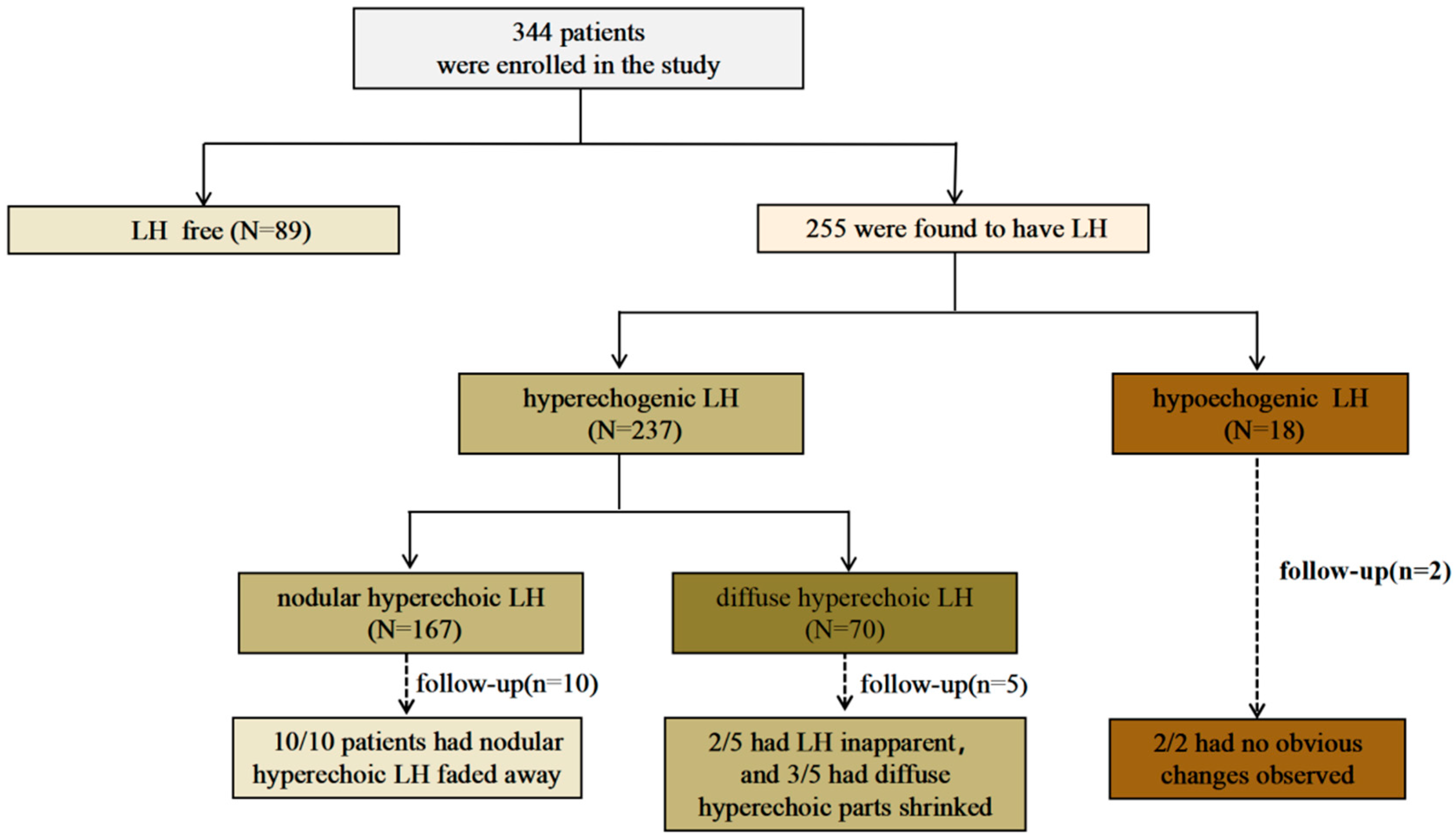
3. Results
3.1. Sample Characteristics
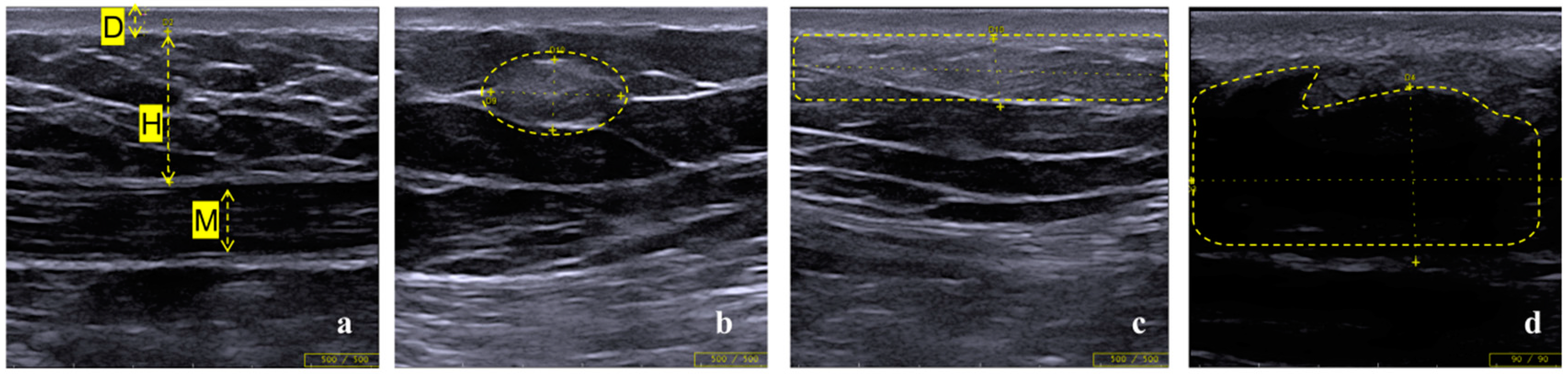
3.2. Lipohypertrophy Findings and Characteristics
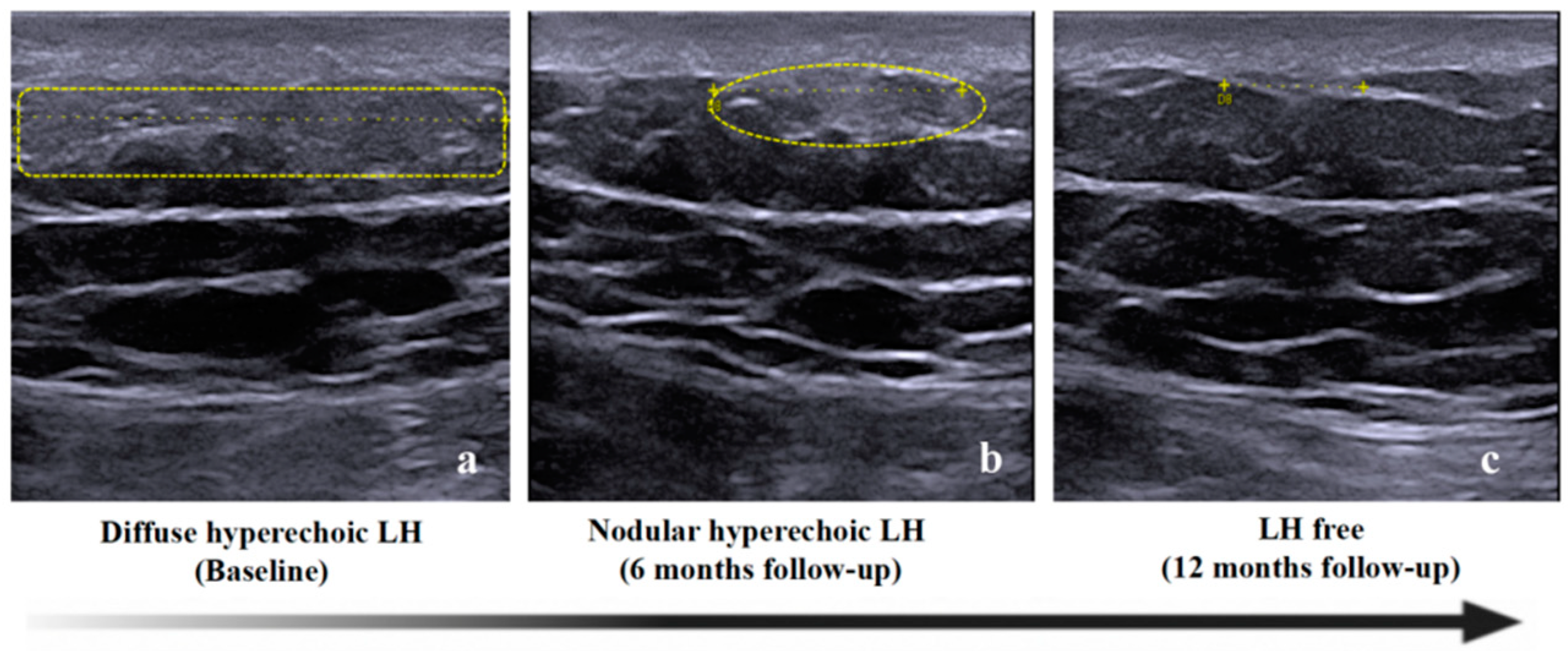
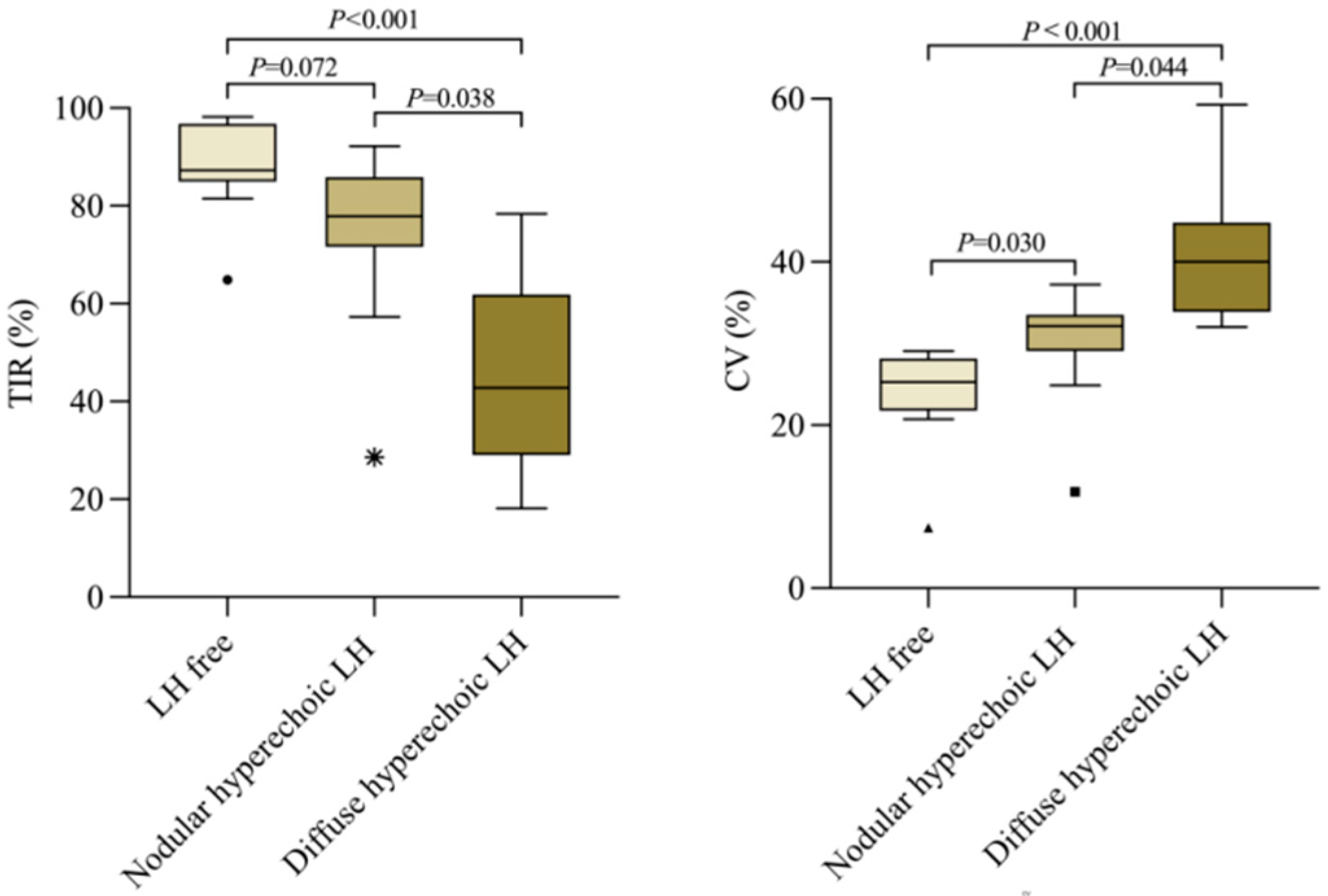
3.3. LH Follow-Up by Ultrasound Assessment
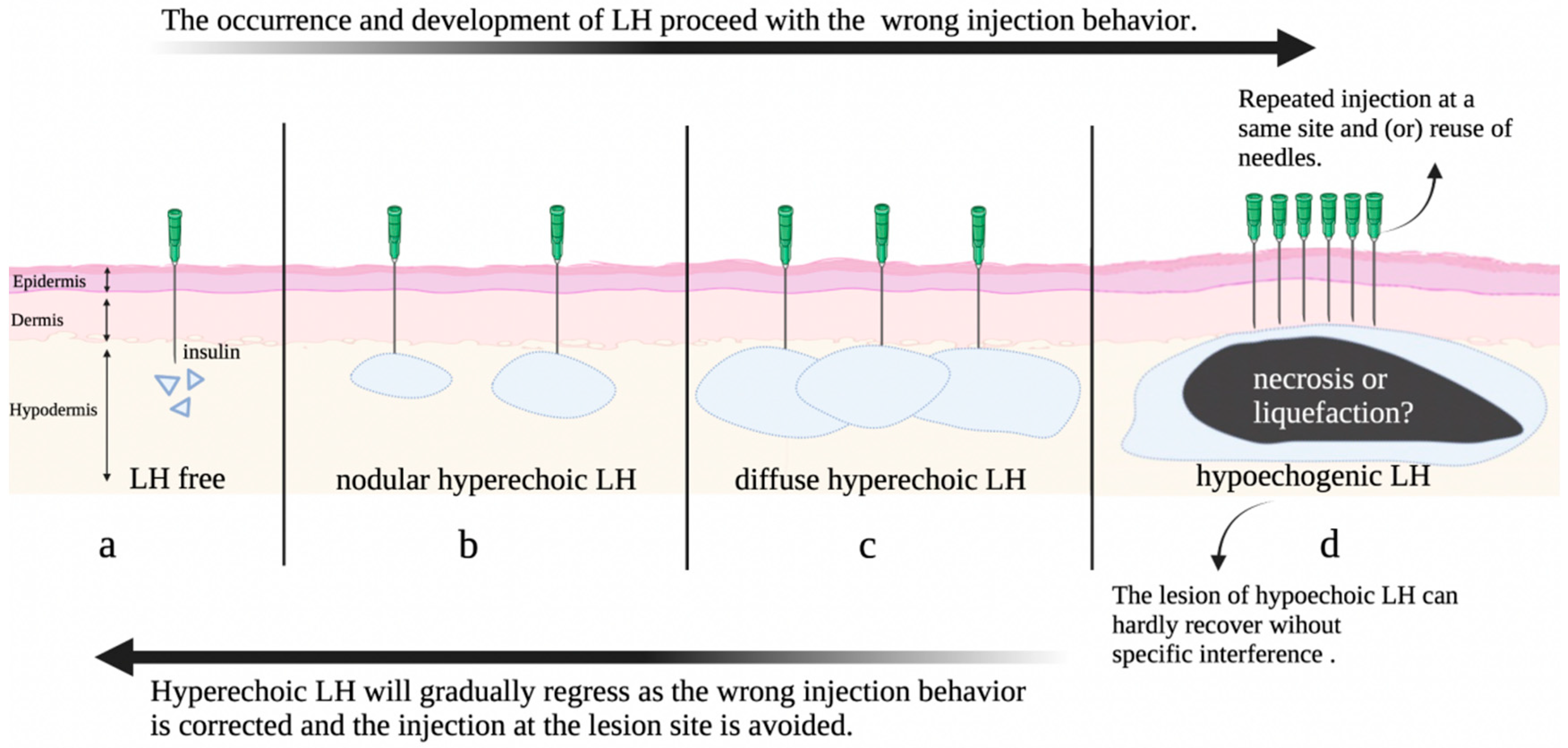
3.4. Hypothesis on the Progression of Insulin Induced Lipohypertrophy
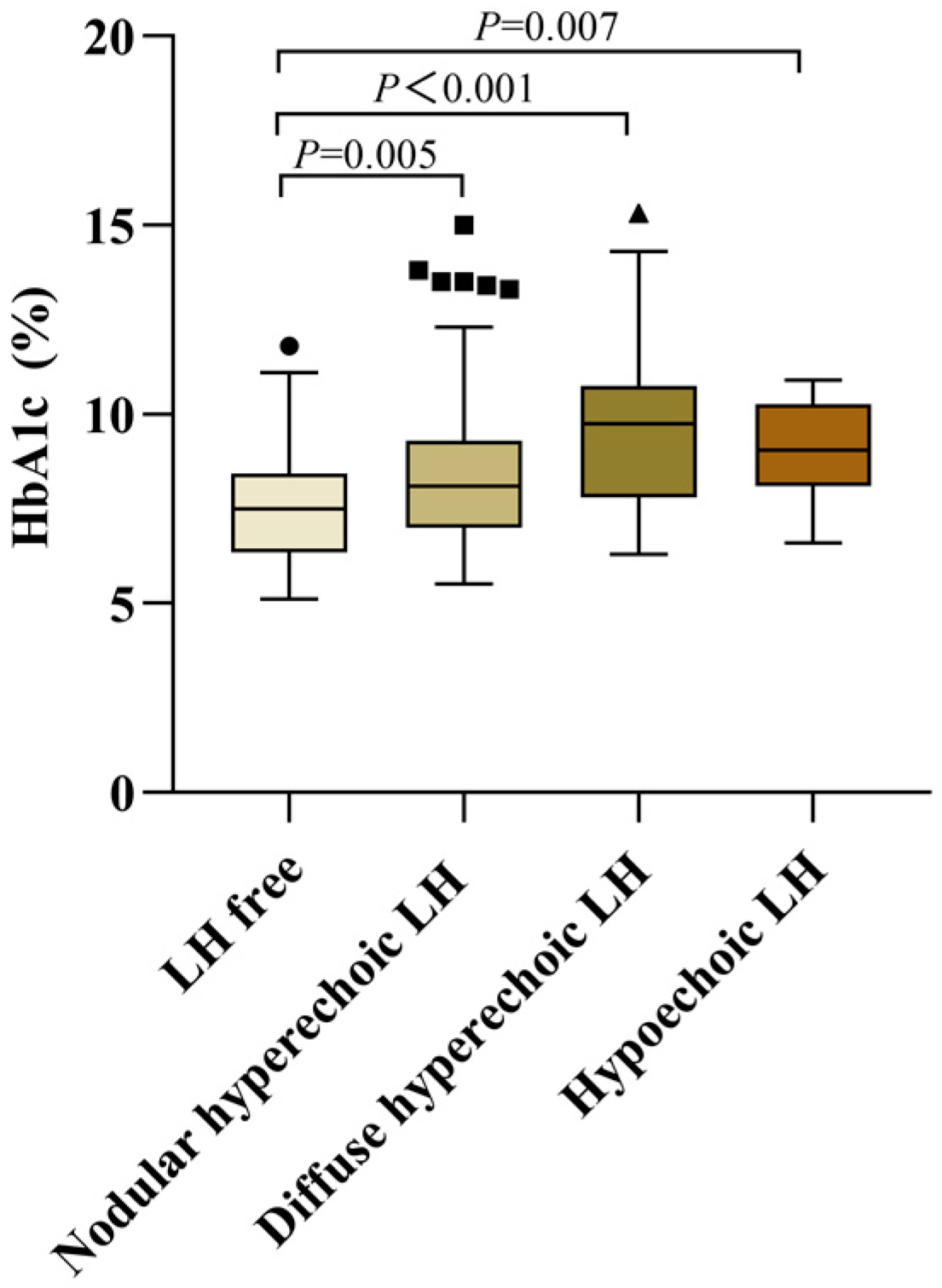
3.5. Differences of Blood Glucose Control in Patients with Different Types of LH
4. Discussion
5. Limitation
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- International Diabetes Federation. Diabetes Atlas 2021-10th Edition. Available online: www.diabetesatlas.org (accessed on 1 February 2022).
- Gentile, S.; Strollo, F.; Satta, E.; Della Corte, T.; Romano, C.; Guarino, G. Insulin-Related Lipohypertrophy in Hemodialyzed Diabetic People: A Multicenter Observational Study and a Methodological Approach. Diabetes Ther. 2019, 10, 1423–1433. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; Sun, Z.; Li, Q.; Qin, G.; Wei, Z.; Liu, J.; Chandran, A.B.A.B.; Hirsch, L.J.L.J. Lipohypertrophy in China: Prevalence, Risk Factors, Insulin Consumption, and Clinical Impact. Diabetes Technol. Ther. 2017, 19, 61–67. [Google Scholar] [CrossRef]
- Volkova, N.I.; Davidenko, I.Y. Clinical significance of lipohypertrophy without visual and palpable changes detected by ultrasonography of subcutaneous fat. Ter. arkhiv 2019, 91, 62–66. [Google Scholar] [CrossRef]
- Luo, D.; Shi, Y.; Zhu, M.; Wang, H.; Yan, D.; Yu, J.; Ji, J.; Liu, X.; Fan, B.; Xu, Y.; et al. Subclinical lipohypertrophy—Easily ignored complications of insulin therapy. J. Diabetes Complicat. 2020, 35, 107806. [Google Scholar] [CrossRef] [PubMed]
- Kapeluto, J.E.; Paty, B.W.; Chang, S.D.; Meneilly, G.S. Ultrasound detection of insulin-induced lipohypertrophy in Type 1 and Type 2 diabetes. Diabet. Med. 2018, 35, 1383–1390. [Google Scholar] [CrossRef]
- Bertuzzi, F.; Meneghini, E.; Bruschi, E.; Luzi, L.; Nichelatti, M.; Epis, O. Ultrasound characterization of insulin induced lipohypertrophy in type 1 diabetes mellitus. J. Endocrinol. Investig. 2017, 40, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Lin, L.; Wang, W.; Hong, J.; Zeng, H. Insulin-related lipohypertrophy: Ultrasound characteristics, risk factors, and impact of glucose fluctuations. Endocrine 2021, 75, 768–775. [Google Scholar] [CrossRef]
- Yu, J.; Wang, H.; Zhu, M.; Yan, D.; Fan, B.; Shi, Y.; Shen, M.; Liu, X.; He, W.; Luo, D.; et al. Detection sensitivity of ultrasound scanning vs. clinical examination for insulin injection-related lipohypertrophy. Chin. Med. J. 2021, 135, 353–355. [Google Scholar] [CrossRef]
- Tsadik, A.G.; Atey, T.M.; Nedi, T.; Fantahun, B.; Feyissa, M. Effect of Insulin-Induced Lipodystrophy on Glycemic Control among Children and Adolescents with Diabetes in Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia. J. Diabetes Res. 2018, 2018, 4910962. [Google Scholar] [CrossRef]
- Famulla, S.; Hövelmann, U.; Fischer, A.; Coester, H.-V.; Hermanski, L.; Kaltheuner, M.; Kaltheuner, L.; Heinemann, L.; Heise, T.; Hirsch, L. Insulin Injection Into Lipohypertrophic Tissue: Blunted and More Variable Insulin Absorption and Action and Impaired Postprandial Glucose Control. Diabetes Care 2016, 39, 1486–1492. [Google Scholar] [CrossRef]
- Gupta, S.S.; Gupta, K.S.; Gathe, S.S.; Bamrah, P.; Gupta, S.S. Clinical Implications of Lipohypertrophy Among People with Type 1 Diabetes in India. Diabetes Technol. Ther. 2018, 20, 483–491. [Google Scholar] [CrossRef]
- Rochester, D.; Bowie, J.D.; Kunzmann, A.; Lester, E. Ultrasound in the Staging of Lymphoma. Radiology 1977, 124, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Schüller, J.; Walther, V.; Schmiedt, E.; Staehler, G.; Bauer, H.; Schilling, A. Intravesical Ultrasound Tomography in Staging Bladder Carcinoma. J. Urol. 1982, 128, 264–266. [Google Scholar] [CrossRef]
- Zhu, M.; Whang, H.; Shen, M.; Yu, J.; Yan, D.; Luo, D.; Shi, Y.; Hang, J.; Xu, J.; Yang, T. Clinical application of high-frequency ultrasound in the screening of lipohypertrophy in diabetic patients. Chin. J. Diabetes Melltus 2021, 13, 848–853. [Google Scholar] [CrossRef]
- Kapeluto, J.; Paty, B.W.; Chang, S.D.; Eddy, C.; Meneilly, G. Criteria for the Detection of Insulin-induced Lipohypertrophy Using Ultrasonography. Can. J. Diabetes 2015, 39, 534. [Google Scholar] [CrossRef]
- Greenstein, Y.Y.; Guevarra, K. Point-of-Care Ultrasound in the Intensive Care Unit: Applications, Limitations, and the Evolution of Clinical Practice. Clin. Chest Med. 2022, 43, 373–384. [Google Scholar] [CrossRef]
- Izzetti, R.; Nisi, M.; Aringhieri, G.; Vitali, S.; Oranges, T.; Romanelli, M.; Caramella, D.; Graziani, F.; Gabriele, M. Ultra-high frequency ultrasound in the differential diagnosis of oral pemphigus and pemphigoid: An explorative study. Skin Res. Technol. 2021, 27, 682–691. [Google Scholar] [CrossRef] [PubMed]
- Abu Ghazaleh, H.; Hashem, R.; Forbes, A.; Dilwayo, T.R.; Duaso, M.; Sturt, J.; Halson-Brown, S.; Mulnier, H. A Systematic Review of Ultrasound-Detected Lipohypertrophy in Insulin-Exposed People with Diabetes. Diabetes Ther. 2018, 9, 1741–1756. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Standards of Medical Care in Diabetes—2010. Diabetes Care 2010, 33 (Suppl. 1), S11–S61. [Google Scholar] [CrossRef]
- Gentile, S.; Strollo, F.; Della Corte, T.; Marino, G.; Guarino, G. Skin complications of insulin injections: A case presentation and a possible explanation of hypoglycaemia. Diabetes Res. Clin. Pract. 2018, 138, 284–287. [Google Scholar] [CrossRef]
- Demir, G.; Er, E.; Altınok, Y.A.; Özen, S.; Darcan, Ş.; Gökşen, D. Local complications of insulin administration sites and effect on diabetes management. J. Clin. Nurs. 2021, 31, 2530–2538. [Google Scholar] [CrossRef]
- Ucieklak, D.; Mrozinska, S.; Wojnarska, A.; Malecki, M.T.; Klupa, T.; Matejko, B. Insulin-induced Lipohypertrophy in Patients with Type 1 Diabetes Mellitus Treated with an Insulin Pump. Int. J. Endocrinol. 2022, 2022, 9169296. [Google Scholar] [CrossRef] [PubMed]
- Hashem, R.; Mulnier, H.; Abu Ghazaleh, H.; Halson-Brown, S.; Duaso, M.; Rogers, R.; Karalliedde, J.; Forbes, A. Characteristics and morphology of lipohypertrophic lesions in adults with type 1 diabetes with ultrasound screening: An exploratory observational study. BMJ Open Diabetes Res. Care 2021, 9, e002553. [Google Scholar] [CrossRef]
- Xu, X.-H.; Carvalho, V.; Wang, X.-H.; Qiu, S.-H.; Sun, Z.-L. Lipohypertrophy: Prevalence, clinical consequence, and pathogenesis. Chin. Med. J. 2020, 134, 47–49. [Google Scholar] [CrossRef] [PubMed]
- Gentile, S.; Guarino, G.; Della Corte, T.; Marino, G.; Fusco, A.; Corigliano, G.; Colarusso, S.; Piscopo, M.; Improta, M.R.; Corigliano, M.; et al. Insulin-Induced Skin Lipohypertrophy in Type 2 Diabetes: A Multicenter Regional Survey in Southern Italy. Diabetes Ther. 2020, 11, 2001–2017. [Google Scholar] [CrossRef] [PubMed]
- Blanco, M.; Hernández, M.; Strauss, K.; Amaya, M. Prevalence and risk factors of lipohypertrophy in insulin-injecting patients with diabetes. Diabetes Metab. 2013, 39, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.; Clapham, L.; Strauss, K. UK lipohypertrophy interventional study. Diabetes Res. Clin. Pract. 2017, 126, 248–253. [Google Scholar] [CrossRef] [PubMed]
- Campinos, C.; Le Floch, J.-P.; Petit, C.; Penfornis, A.; Winiszewski, P.; Bordier, L.; Lepage, M.; Fermon, C.; Louis, J.; Almain, C.; et al. An Effective Intervention for Diabetic Lipohypertrophy: Results of a Randomized, Controlled, Prospective Multicenter Study in France. Diabetes Technol. Ther. 2017, 19, 623–632. [Google Scholar] [CrossRef]
- Wang, W.; Huang, R.; Chen, Y.; Tu, M. Values of ultrasound for diagnosis and management of insulin-induced lipohypertrophy. Medicine 2021, 100, e26743. [Google Scholar] [CrossRef]
- Perciun, R. Ultrasonographic aspect of subcutaneous tissue dystrophies as a result of insulin injections. Med. Ultrason. 2010, 12, 104–109. [Google Scholar]
- Guidelines and Consensus Compilation Committee of Chinese Journal of Diabetes. Technical Guide for Diabetes Drug Injection in China (2016 edition). Chin. J. Diabetes Mellit. 2017, 9, 79–105. [Google Scholar]
- Mantovani, A.; Grani, G.; Chioma, L.; Vancieri, G.; Giordani, I.; Rendina, R.; Rinaldi, M.E.; Andreadi, A.; Coccaro, C.; Boccardo, C.; et al. Severe hypoglycemia in patients with known diabetes requiring emergency department care: A report from an Italian multicenter study. J. Clin. Transl. Endocrinol. 2016, 5, 46–52. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, J.; Wang, H.; Zhou, M.; Zhu, M.; Hang, J.; Shen, M.; Jin, X.; Shi, Y.; Xu, J.; Yang, T. A Hypothesis on the Progression of Insulin-Induced Lipohypertrophy: An Integrated Result of High-Frequency Ultrasound Imaging and Blood Glucose Control of Patients. Diagnostics 2023, 13, 1515. https://doi.org/10.3390/diagnostics13091515
Yu J, Wang H, Zhou M, Zhu M, Hang J, Shen M, Jin X, Shi Y, Xu J, Yang T. A Hypothesis on the Progression of Insulin-Induced Lipohypertrophy: An Integrated Result of High-Frequency Ultrasound Imaging and Blood Glucose Control of Patients. Diagnostics. 2023; 13(9):1515. https://doi.org/10.3390/diagnostics13091515
Chicago/Turabian StyleYu, Jian, Hong Wang, Meijing Zhou, Min Zhu, Jing Hang, Min Shen, Xin Jin, Yun Shi, Jingjing Xu, and Tao Yang. 2023. "A Hypothesis on the Progression of Insulin-Induced Lipohypertrophy: An Integrated Result of High-Frequency Ultrasound Imaging and Blood Glucose Control of Patients" Diagnostics 13, no. 9: 1515. https://doi.org/10.3390/diagnostics13091515
APA StyleYu, J., Wang, H., Zhou, M., Zhu, M., Hang, J., Shen, M., Jin, X., Shi, Y., Xu, J., & Yang, T. (2023). A Hypothesis on the Progression of Insulin-Induced Lipohypertrophy: An Integrated Result of High-Frequency Ultrasound Imaging and Blood Glucose Control of Patients. Diagnostics, 13(9), 1515. https://doi.org/10.3390/diagnostics13091515




