Association of Obesity with SARS-CoV-2 and Its Relationship with the Humoral Response Prior to Vaccination in the State of Mexico: A Cross-Sectional Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Demographic Variables
2.3. Anthropometric Measurements
2.4. Detection of IgG Antibodies against S1/S2 Antigens of SARS-CoV-2
2.5. Neutralizing Activity of Antibodies against the RBD Antigen of SARS-CoV-2
2.6. Statistical Analysis
3. Results
3.1. BMI Association with Prior SARS-CoV-2 Infection
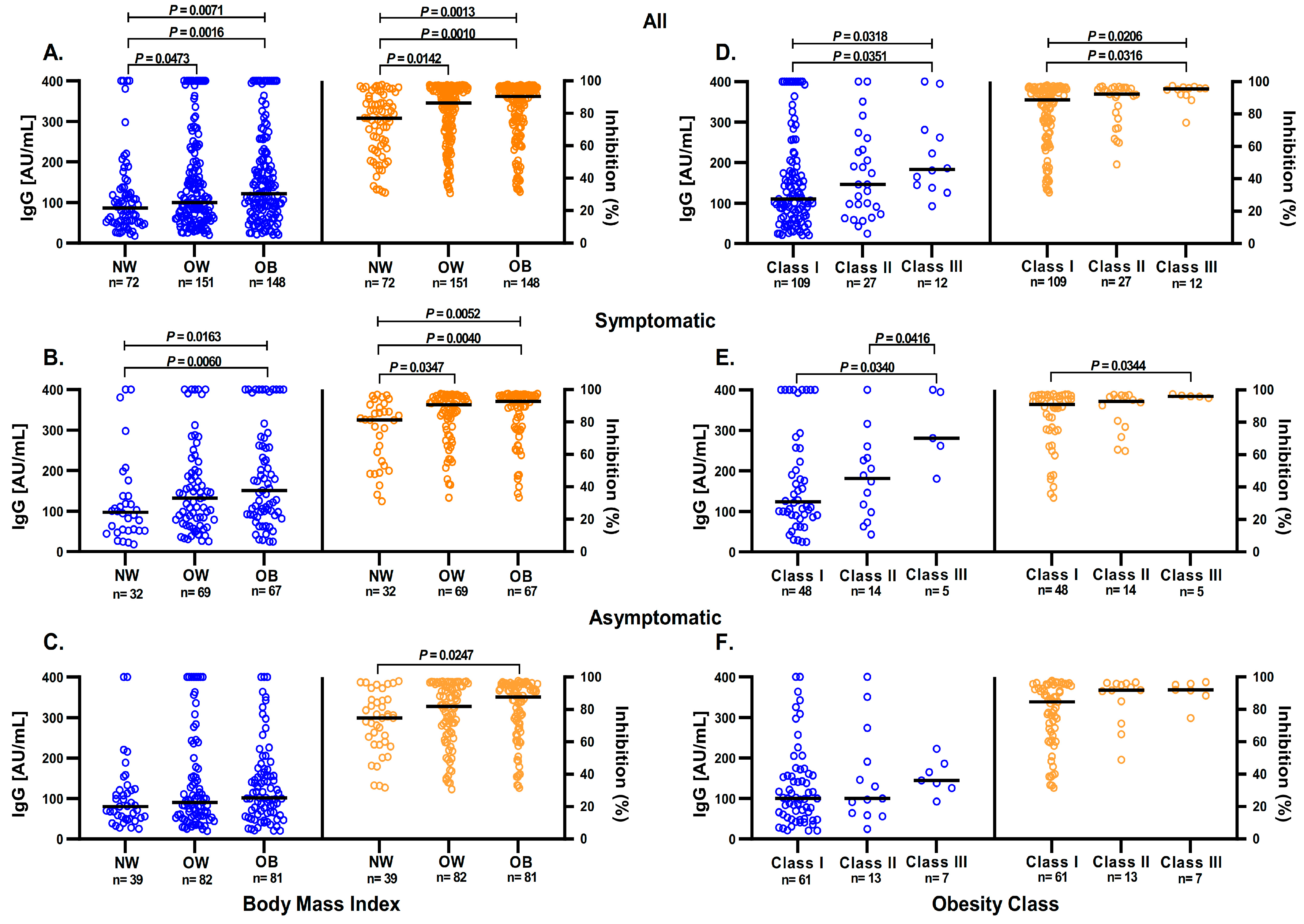
3.2. BMI Influence over Anti-S1/S2 and Anti-RBD IgG Antibodies Response
3.3. Obesity and Other Comorbidities Influence the Anti-S1/S2 and Anti-RBD IgG Antibodies Response
3.4. Anti-S1/S2 IgG Antibodies and Anti-RBD Antibodies in Symptomatic Participants
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- COVID-19 México. Información General Nacional (Confirmados). Available online: https://datos.covid-19.conacyt.mx/ (accessed on 2 January 2023).
- Yang, H.S.; Costa, V.; Racine-Brzostek, S.E.; Acker, K.P.; Yee, J.; Chen, Z.; Karbaschi, M.; Zuk, R.; Rand, S.; Sukhu, A.; et al. Association of Age with SARS-CoV-2 Antibody Response. JAMA Netw. Open 2021, 4, e214302. [Google Scholar] [CrossRef]
- Eftekhari, A.; Alipour, M.; Chodari, L.; Maleki Dizaj, S.; Ardalan, M.; Samiei, M.; Sharifi, S.; Zununi Vahed, S.; Huseynova, I.; Khalilov, R.; et al. A Comprehensive Review of Detection Methods for SARS-CoV-2. Microorganisms 2021, 9, 232. [Google Scholar] [CrossRef]
- Carter, L.J.; Garner, L.V.; Smoot, J.W.; Li, Y.; Zhou, Q.; Saveson, C.J.; Sasso, J.M.; Gregg, A.C.; Soares, D.J.; Beskid, T.R.; et al. Assay Techniques and Test Development for COVID-19 Diagnosis. ACS Cent. Sci. 2020, 6, 591–605. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Medina, J.E.; Grajales-Muñiz, C.; Salas-Lais, A.G.; Fernandes-Matano, L.; López-Macías, C.; Monroy-Muñoz, I.E.; Santos Coy-Arechavaleta, A.; Palomec-Nava, I.D.; Duque-Molina, C.; Madera-Sandoval, R.L.; et al. SARS-CoV-2 IgG Antibodies Seroprevalence and Sera Neutralizing Activity in MEXICO: A National Cross-Sectional Study during 2020. Microorganisms 2021, 9, 850. [Google Scholar] [CrossRef] [PubMed]
- Winter, A.K.; Hegde, S.T. The important role of serology for COVID-19 control. Lancet Infect. Dis. 2020, 20, 758–759. [Google Scholar] [CrossRef] [PubMed]
- Krammer, F.; Simon, V. Serology assays to manage COVID-19. Science 2020, 368, 1060–1061. [Google Scholar] [CrossRef] [PubMed]
- Oved, K.; Olmer, L.; Shemer-Avni, Y.; Wolf, T.; Supino-Rosin, L.; Prajgrod, G.; Shenhar, Y.; Payorsky, I.; Cohen, Y.; Kohn, Y.; et al. Multi-center nationwide comparison of seven serology assays reveals a SARS-CoV-2 non-responding seronegative subpopulation. EClinicalMedicine 2020, 29, 100651. [Google Scholar] [CrossRef]
- Horn, M.P.; Jonsdottir, H.R.; Brigger, D.; Damonti, L.; Suter-Riniker, F.; Endrich, O.; Froehlich, T.K.; Fiedler, M.; Largiadèr, C.R.; Marschall, J.; et al. Serological testing for SARS-CoV-2 antibodies in clinical practice: A comparative diagnostic accuracy study. Allergy 2022, 77, 2090–2103. [Google Scholar] [CrossRef]
- Bonelli, F.; Sarasini, A.; Zierold, C.; Calleri, M.; Bonetti, A.; Vismara, C.; Blocki, F.A.; Pallavicini, L.; Chinali, A.; Campisi, D.; et al. Clinical and Analytical Performance of an Automated Serological Test That Identifies S1/S2-Neutralizing IgG in COVID-19 Patients Semiquantitatively. J. Clin. Microbiol. 2020, 58, e01224-20. [Google Scholar] [CrossRef]
- Meyer, B.; Reimerink, J.; Torriani, G.; Brouwer, F.; Godeke, G.J.; Yerly, S.; Hoogerwerf, M.; Vuilleumier, N.; Kaiser, L.; Eckerle, I.; et al. Validation and clinical evaluation of a SARS-CoV-2 surrogate virus neutralisation test (sVNT). Emerg. Microbes Infect. 2020, 9, 2394–2403. [Google Scholar] [CrossRef]
- Paidi, R.K.; Jana, M.; Raha, S.; Mishra, R.K.; Jeong, B.; Sheinin, M.; Pahan, K. Prenol, but Not Vitamin C, of Fruit Binds to SARS-CoV-2 Spike S1 to Inhibit Viral Entry: Implications for COVID-19. J. Immunol. 2023, 210, 1938–1949. [Google Scholar] [CrossRef] [PubMed]
- Paidi, R.K.; Jana, M.; Mishra, R.K.; Dutta, D.; Pahan, K. Selective Inhibition of the Interaction between SARS-CoV-2 Spike S1 and ACE2 by SPIDAR Peptide Induces Anti-Inflammatory Therapeutic Responses. J. Immunol. 2021, 207, 2521–2533. [Google Scholar] [CrossRef] [PubMed]
- Bayram, A.; Demirbakan, H.; Günel Karadeniz, P.; Erdoğan, M.; Koçer, I. Quantitation of antibodies against SARS-CoV-2 spike protein after two doses of CoronaVac in healthcare workers. J. Med. Virol. 2021, 93, 5560–5567. [Google Scholar] [CrossRef] [PubMed]
- Hulme, K.D.; Noye, E.C.; Short, K.R.; Labzin, L.I. Dysregulated Inflammation During Obesity: Driving Disease Severity in Influenza Virus and SARS-CoV-2 Infections. Front. Immunol. 2021, 12, 770066. [Google Scholar] [CrossRef]
- Ritter, A.; Kreis, N.N.; Louwen, F.; Yuan, J. Obesity and COVID-19: Molecular Mechanisms Linking Both Pandemics. Int. J. Mol. Sci. 2020, 21, 5793. [Google Scholar] [CrossRef]
- World Health Organization. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 26 October 2022).
- Encuesta Nacional de Salud y Nutrición 2018. Available online: https://ensanut.insp.mx/encuestas/ensanut2018/doctos/informes/ensanut_2018_presentacion_resultados.pdf (accessed on 26 October 2022).
- WHO Guidance on Regulations for the Transport of Infectious Substances 2021–2022. Available online: https://www.who.int/publications/i/item/9789240019720 (accessed on 26 October 2022).
- Long, Q.X.; Tang, X.J.; Shi, Q.L.; Li, Q.; Deng, H.J.; Yuan, J.; Hu, J.L.; Xu, W.; Zhang, Y.; Lv, F.J.; et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat. Med. 2020, 26, 1200–1204. [Google Scholar] [CrossRef]
- Shields, A.M.; Faustini, S.E.; Perez-Toledo, M.; Jossi, S.; Allen, J.D.; Al-Taei, S.; Backhouse, C.; Dunbar, L.A.; Ebanks, D.; Emmanuel, B.; et al. Serological responses to SARS-CoV-2 following non-hospitalised infection: Clinical and ethnodemographic features associated with the magnitude of the antibody response. BMJ Open Respir. Res. 2021, 8, e000872. [Google Scholar] [CrossRef]
- Sancilio, A.E.; D’Aquila, R.T.; McNally, E.M.; Velez, M.P.; Ison, M.G.; Demonbreun, A.R.; McDade, T.W. A surrogate virus neutralization test to quantify antibody-mediated inhibition of SARS-CoV-2 in finger stick dried blood spot samples. Sci. Rep. 2021, 11, 15321. [Google Scholar] [CrossRef]
- Favresse, J.; Gillot, C.; Di Chiaro, L.; Eucher, C.; Elsen, M.; Van Eeckhoudt, S.; David, C.; Morimont, L.; Dogné, J.M.; Douxfils, J. Neutralizing Antibodies in COVID-19 Patients and Vaccine Recipients after Two Doses of BNT162b2. Viruses 2021, 13, 1364. [Google Scholar] [CrossRef]
- Lau, E.H.Y.; Tsang, O.T.Y.; Hui, D.S.C.; Kwan, M.Y.W.; Chan, W.H.; Chiu, S.S.; Ko, R.L.W.; Chan, K.H.; Cheng, S.M.S.; Perera, R.A.P.M.; et al. Neutralizing antibody titres in SARS-CoV-2 infections. Nat. Commun. 2021, 12, 63. [Google Scholar] [CrossRef]
- Lee, N.; Chan, P.K.; Ip, M.; Wong, E.; Ho, J.; Ho, C.; Cockram, C.S.; Hui, D.S. Anti-SARS-CoV IgG response in relation to disease severity of severe acute respiratory syndrome. J. Clin. Virol. 2006, 35, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Frasca, D.; Reidy, L.; Cray, C.; Diaz, A.; Romero, M.; Kahl, K.; Blomberg, B.B. Influence of obesity on serum levels of SARS-CoV-2-specific antibodies in COVID-19 patients. PLoS ONE 2021, 16, e0245424. [Google Scholar] [CrossRef] [PubMed]
- Popkin, B.M.; Du, S.; Green, W.D.; Beck, M.A.; Algaith, T.; Herbst, C.H.; Alsukait, R.F.; Alluhidan, M.; Alazemi, N.; Shekar, M. Individuals with obesity and COVID-19: A global perspective on the epidemiology and biological relationships. Obes. Rev. 2020, 21, e13128. [Google Scholar] [CrossRef]
- Jung, C.Y.; Park, H.; Kim, D.W.; Lim, H.; Chang, J.H.; Choi, Y.J.; Kim, S.W.; Chang, T.I. Association between Body Mass Index and Risk of Coronavirus Disease 2019 (COVID-19): A Nationwide Case-control Study in South Korea. Clin. Infect. Dis. 2021, 73, e1855–e1862. [Google Scholar] [CrossRef] [PubMed]
- Ghoneim, S.; Butt, M.U.; Hamid, O.; Shah, A.; Asaad, I. The incidence of COVID-19 in patients with metabolic syndrome and non-alcoholic steatohepatitis: A population-based study. Metabol. Open 2020, 8, 100057. [Google Scholar] [CrossRef]
- Racine-Brzostek, S.E.; Yang, H.S.; Jack, G.A.; Chen, Z.; Chadburn, A.; Ketas, T.J.; Francomano, E.; Klasse, P.J.; Moore, J.P.; McDonough, K.A.; et al. Postconvalescent SARS-CoV-2 IgG and Neutralizing Antibodies are Elevated in Individuals with Poor Metabolic Health. J. Clin. Endocrinol. Metab. 2021, 106, e2025–e2034. [Google Scholar] [CrossRef]
- Gudbjartsson, D.F.; Norddahl, G.L.; Melsted, P.; Gunnarsdottir, K.; Holm, H.; Eythorsson, E.; Arnthorsson, A.O.; Helgason, D.; Bjarnadottir, K.; Ingvarsson, R.F.; et al. Humoral Immune Response to SARS-CoV-2 in Iceland. N. Engl. J. Med. 2020, 383, 1724–1734. [Google Scholar] [CrossRef]
- Nilles, E.J.; Siddiqui, S.M.; Fischinger, S.; Bartsch, Y.C.; de St Aubin, M.; Zhou, G.; Gluck, M.J.; Berger, S.; Rhee, J.; Petersen, E.; et al. Epidemiological and Immunological Features of Obesity and SARS-CoV-2. Viruses 2021, 13, 2235. [Google Scholar] [CrossRef]
- Frasca, D.; Reidy, L.; Romero, M.; Diaz, A.; Cray, C.; Kahl, K.; Blomberg, B.B. The majority of SARS-CoV-2-specific antibodies in COVID-19 patients with obesity are autoimmune and not neutralizing. Int. J. Obes. 2022, 46, 427–432. [Google Scholar] [CrossRef]
- Zhang, B.; Zhou, X.; Zhu, C.; Song, Y.; Feng, F.; Qiu, Y.; Feng, J.; Jia, Q.; Song, Q.; Zhu, B.; et al. Immune Phenotyping Based on the Neutrophil-to-Lymphocyte Ratio and IgG Level Predicts Disease Severity and Outcome for Patients with COVID-19. Front. Mol. Biosci. 2020, 7, 157. [Google Scholar] [CrossRef]
- Ko, J.H.; Joo, E.J.; Park, S.J.; Baek, J.Y.; Kim, W.D.; Jee, J.; Kim, C.J.; Jeong, C.; Kim, Y.J.; Shon, H.J.; et al. Neutralizing Antibody Production in Asymptomatic and Mild COVID-19 Patients, in Comparison with Pneumonic COVID-19 Patients. J. Clin. Med. 2020, 9, 2268. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.L.; Pekosz, A.; Park, H.S.; Ursin, R.L.; Shapiro, J.R.; Benner, S.E.; Littlefield, K.; Kumar, S.; Naik, H.M.; Betenbaugh, M.J.; et al. Sex, age, and hospitalization drive antibody responses in a COVID-19 convalescent plasma donor population. J. Clin. Investig. 2020, 130, 6141–6150. [Google Scholar] [CrossRef] [PubMed]
- Karbiener, M.; Farcet, M.R.; Ilk, R.; Schreiner, J.; Lenart, J.; Powers, N.; Stewart, J.M.; Tallman, H.; Kreil, T.R. Longitudinal analysis of SARS-CoV-2 antibodies in 8000 U.S. first-time convalescent plasma donations. Transfusion 2021, 61, 1141–1147. [Google Scholar] [CrossRef] [PubMed]
- Skorek, A.; Jaźwińska-Curyłło, A.; Romanowicz, A.; Kwaśniewski, K.; Narożny, W.; Tretiakow, D. Assessment of anti-SARS-CoV-2 antibodies level in convalescents plasma. J. Med. Virol. 2022, 94, 1130–1137. [Google Scholar] [CrossRef]
- Terpos, E.; Stellas, D.; Rosati, M.; Sergentanis, T.N.; Hu, X.; Politou, M.; Pappa, V.; Ntanasis-Stathopoulos, I.; Karaliota, S.; Bear, J.; et al. SARS-CoV-2 antibody kinetics eight months from COVID-19 onset: Persistence of spike antibodies but loss of neutralizing antibodies in 24% of convalescent plasma donors. Eur. J. Intern. Med. 2021, 89, 87–96. [Google Scholar] [CrossRef]
- Gonzalez-Quintela, A.; Alende, R.; Gude, F.; Campos, J.; Rey, J.; Meijide, L.M.; Fernandez-Merino, C.; Vidal, C. Serum levels of immunoglobulins (IgG, IgA, IgM) in a general adult population and their relationship with alcohol consumption, smoking and common metabolic abnormalities. Clin. Exp. Immunol. 2008, 151, 42–50. [Google Scholar] [CrossRef]
- Tadic, M.; Cuspidi, C.; Mancia, G.; Dell’Oro, R.; Grassi, G. COVID-19, hypertension and cardiovascular diseases: Should we change the therapy? Pharmacol. Res. 2020, 158, 104906. [Google Scholar] [CrossRef]
- Sardu, C.; D’Onofrio, N.; Balestrieri, M.L.; Barbieri, M.; Rizzo, M.R.; Messina, V.; Maggi, P.; Coppola, N.; Paolisso, G.; Marfella, R. Outcomes in Patients with Hyperglycemia Affected by COVID-19: Can We Do More on Glycemic Control? Diabetes Care 2020, 43, 1408–1415. [Google Scholar] [CrossRef]
- De Greef, J.; Scohy, A.; Zech, F.; Aboubakar, F.; Pilette, C.; Gerard, L.; Pothen, L.; Yildiz, H.; Belkhir, L.; Yombi, J.C. Determinants of IgG antibodies kinetics after severe and critical COVID-19. J. Med. Virol. 2021, 93, 5416–5424. [Google Scholar] [CrossRef]
- Gadi, N.; Wu, S.C.; Spihlman, A.P.; Moulton, V.R. What’s Sex Got to Do with COVID-19? Gender-Based Differences in the Host Immune Response to Coronaviruses. Front. Immunol. 2020, 11, 2147. [Google Scholar] [CrossRef]
- Cruz-Arenas, E.; Cabrera-Ruiz, E.; Laguna-Barcenas, S.; Colin-Castro, C.A.; Chavez, T.; Franco-Cendejas, R.; Ibarra, C.; Perez-Orive, J. Serological prevalence of SARS-CoV-2 infection and associated factors in healthcare workers in a “non-COVID” hospital in Mexico City. PLoS ONE 2021, 16, e0255916. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.; Dai, C.; Cai, P.; Wang, J.; Xu, L.; Li, J.; Hu, G.; Wang, Z.; Zheng, F.; Wang, L. A comparison study of SARS-CoV-2 IgG antibody between male and female COVID-19 patients: A possible reason underlying different outcome between sex. J. Med. Virol. 2020, 92, 2050–2054. [Google Scholar] [CrossRef] [PubMed]
- Fernandes-Matano, L.; Salas-Lais, A.G.; Grajales-Muñiz, C.; Hernández-Ávila, M.; Garfias-Becerra, Y.O.; Rodríguez-Sepúlveda, M.C.; Segura-Sánchez, C.; Montes-Herrera, D.; Mendoza-Sánchez, D.; Angeles-Martínez, J.; et al. Longevity and Neutralizing Capacity of IgG Antibodies against SARS-CoV-2 Generated by the Application of BNT162b2, AZD1222, Convidecia, Sputnik V, and CoronaVac Vaccines: A Cohort Study in the Mexican Population. Microbiol. Spectr. 2023, 11, e0237622. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Acuña, N.; Avalos-Nolazco, D.M.; Rodriguez-Rodriguez, D.R.; Martinez-Liu, C.G.; Galan-Huerta, K.A.; Padilla-Rivas, G.R.; Ramos-Jimenez, J.; Ayala-de-la-Cruz, S.; Cienfuegos-Pecina, E.; Diaz-Chuc, E.A.; et al. Seroprevalence of Anti-SARS-CoV-2 Antibodies in Blood Donors from Nuevo Leon State, Mexico, during 2020: A Retrospective Cross-Sectional Evaluation. Viruses 2021, 13, 1225. [Google Scholar] [CrossRef] [PubMed]
- Gebhard, C.; Regitz-Zagrosek, V.; Neuhauser, H.K.; Morgan, R.; Klein, S.L. Impact of sex and gender on COVID-19 outcomes in Europe. Biol. Sex Differ. 2020, 11, 29. [Google Scholar] [CrossRef] [PubMed]





| Characteristics | Prior COVID-19 * | Seronegative n = 529 (%) | ||
|---|---|---|---|---|
| All n = 451 (%) | Symptomatic n = 204 (%) | Asymptomatic n = 247 (%) | ||
| Age (years) | 50 (36–54) | 46 (37–53) | 50 (35–55) | 47 (36–54) |
| 18–29 | 27 (5.9) | 10 (4.9) | 17 (6.8) | 37 (6.9) |
| 30–39 | 144 (31.9) | 70 (34.3) | 74 (29.9) | 158 (29.9) |
| 40–49 | 46 (10.2) | 29 (14.2) | 17 (6.8) | 81 (15.3) |
| 50–59 | 209 (46.3) | 88 (43.1) | 121 (48.9) | 220 (41.6) |
| 60–100 | 10 (2.2) | 3 (1.5) | 7 (2.8) | 16 (3.0) |
| Weight (Kg) | 74 (64–85) | 75 (65–85) | 74 (64–85) | 70 (62–80) d |
| Height (m) | 1.61 (1.55–1.68) | 1.62 (1.55–1.69) | 1.60 (1.55–1.67) | 1.60 (1.54–1.68) d |
| BMI (Kg/m2) ** | 28.3 (25.4–32.0) | 27.7 (25.1–32.5) | 28.6 (25.5–31.7) | 27.0 (24.4–30.1) d |
| Normal weight | 88 (19.5) | 46 (22.5) | 42 (17) | 148 (27.9) |
| Overweight | 169 (37.5) | 79 (38.7) | 90 (36.5) | 219 (41.4) |
| Class I obesity | 117 (25.9) a | 50 (24.5) | 67 (27.1) c | 101 (19.1) |
| Class II obesity | 30 (6.7) a | 15 (7.4) b | 15 (6.1) c | 16 (3.0) |
| Class III obesity | 13 (2.9) | 5 (2.5) | 8 (3.2) c | 10 (1.9) |
| Sex | ||||
| Women | 282 (62.5) | 123 (60.3) | 159 (64.4) | 343 (64.8) |
| IMen | 169 (37.5) | 81 (39.7) | 88 (35.6) | 186 (35.2) |
| Comorbidities | ||||
| Diabetes mellitus | 49 (10.9) | 20 (9.8) | 29 (11.7) | 58 (10.9) |
| Hypertension | 76 (16.9) | 32 (15.7) | 44 (17.8) | 88 (16.6) |
| Autoimmune disease | 13 (2.9) | 8 (3.9) | 5 (2.0) | 16 (3.0) |
| Other respiratory disease | 18 (3.9) | 10 (4.9) | 8 (3.2) | 23 (4.3) |
| Heart disease | 9 (1.9) | 6 (2.9) | 3 (1.2) | 7 (1.3) |
| HIV | 1 (0.2) | 1 (0.5) | 0 (0) | 4 (0.7) |
| Smoking history | ||||
| Never | 364 (80.7) | 164 (80.4) | 200 (80.9) | 437 (82.6) |
| Mild | 59 (13.1) a | 26 (12.7) b | 33 (13.4) c | 38 (7.1) |
| Moderate | 18 (3.9) | 8 (3.9) | 10 (4.1) | 32 (6.0) |
| Severe | 10 (2.2) | 6 (2.9) | 4 (1.7) | 22 (4.2) |
| Alcoholism | ||||
| Never | 321 (71.2) | 143 (70.1) | 178 (72.1) | 335 (63.3) |
| Mild | 121 (26.8) a | 57 (27.9) | 64 (25.9) c | 169 (31.9) |
| Moderate | 8 (1.8) a | 3 (1.5) | 5 (2.0) | 21 (3.9) |
| Severe | 1 (0.2) | 1 (0.5) | 0 (0) | 4 (0.7) |
| Vaccination center location | ||||
| Chalco | 94 (20.8) | 47 (23.0) | 47 (19.0) | 118 (22.3) |
| Toluca | 69 (15.3) | 35 (17.2) | 34 (13.7) | 135 (25.5) |
| Zinacantepec | 94 (20.8) | 29 (14.2) | 65 (26.3) | 77 (14.6) |
| Ecatepec | 75 (16.6) | 37 (18.1) | 38 (15.4) | 104 (19.7) |
| Acolman | 119 (26.4) | 56 (27.5) | 63 (25.5) | 95 (17.9) |
| Characteristic | Prior COVID-19 vs. Seronegative | Asymptomatic vs. Seronegative | Symptomatic vs. Seronegative | Symptomatic vs. Asymptomatic |
|---|---|---|---|---|
| OR (95% CI); p-Value | OR (95% CI); p-Value | OR (95% CI); p-Value | OR (95% CI); p-Value | |
| Unadjusted analysis | ||||
| Normal weight | Ref | |||
| Overweight | 1.29(0.93–1.80); 0.1230 | 1.44(0.95–2.20); 0.0850 | 1.16(0.76–1.76); 0.4860 | 0.80(0.47–1.34); 0.4010 |
| Obesity | 2.11(1.49–3.01); <0.0001 | 2.49(1.61–3.86); <0.0001 | 1.77(1.14–2.75); 0.0110 | 0.71(0.42–1.19); 0.1990 |
| Class Obesity | ||||
| Class I Obesity | 1.94(1.33–2.83); <0.0001 | 2.33(1.47–3.70); <0.0001 | 1.59(0.99–2.55); 0.0540 | 0.68(0.39–1.18); 0.1760 |
| Class II Obesity | 3.15(1.62–6.11); 0.0010 | 3.30(1.50–7.23); 0.0030 | 3.01(1.38–6.56); 0.0050 | 0.91(0.39–2.09); 0.8300 |
| Class III Obesity | 2.18(0.92–5.19); 0.0770 | 2.81(1.04–7.59); 0.0400 | 1.60(0.52–4.94); 0.4070 | 0.57(0.17–1.88); 0.3570 |
| Adjusted analysis | ||||
| Normal weight | Ref | |||
| Overweight | 1.30(0.93–1.82); 0.1210 | 1.45(0.94–2.24); 0.0850 | 1.16(0.76–1.78); 0.4710 | 0.79(0.46–1.34); 0.3830 |
| Obesity | 2.18(1.51–3.16); <0.0001 | 2.58(1.63–4.09); <0.0001 | 1.88(1.18–2.98); 0.0070 | 0.71(0.41–1.23); 0.2320 |
| Class Obesity | ||||
| Class I Obesity | 1.99(1.35–2.95); 0.0010 | 2.44(1.50–3.98); <0.0001 | 1.64(1.002–2.69); 0.0490 | 0.65(0.36–1.17); 0.1580 |
| Class II Obesity | 3.02(1.54–5.91); 0.0010 | 3.12(1.38–7.02); 0.0060 | 3.02(1.37–6.62); 0.0060 | 0.95(0.40–2.25); 0.9100 |
| Class III Obesity | 2.46(0.99–6.10); 0.0520 | 3.12(1.08–8.94); 0.0340 | 1.82(0.56–5.88); 0.3150 | 0.56(0.16–1.91); 0.3570 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Montes-Herrera, D.; Muñoz-Medina, J.E.; Fernandes-Matano, L.; Salas-Lais, A.G.; Hernández-Cueto, M.D.L.Á.; Santacruz-Tinoco, C.E.; Monroy-Muñoz, I.E.; Angeles-Martínez, J. Association of Obesity with SARS-CoV-2 and Its Relationship with the Humoral Response Prior to Vaccination in the State of Mexico: A Cross-Sectional Study. Diagnostics 2023, 13, 2630. https://doi.org/10.3390/diagnostics13162630
Montes-Herrera D, Muñoz-Medina JE, Fernandes-Matano L, Salas-Lais AG, Hernández-Cueto MDLÁ, Santacruz-Tinoco CE, Monroy-Muñoz IE, Angeles-Martínez J. Association of Obesity with SARS-CoV-2 and Its Relationship with the Humoral Response Prior to Vaccination in the State of Mexico: A Cross-Sectional Study. Diagnostics. 2023; 13(16):2630. https://doi.org/10.3390/diagnostics13162630
Chicago/Turabian StyleMontes-Herrera, Daniel, José Esteban Muñoz-Medina, Larissa Fernandes-Matano, Angel Gustavo Salas-Lais, Ma. De Los Ángeles Hernández-Cueto, Clara Esperanza Santacruz-Tinoco, Irma Eloisa Monroy-Muñoz, and Javier Angeles-Martínez. 2023. "Association of Obesity with SARS-CoV-2 and Its Relationship with the Humoral Response Prior to Vaccination in the State of Mexico: A Cross-Sectional Study" Diagnostics 13, no. 16: 2630. https://doi.org/10.3390/diagnostics13162630
APA StyleMontes-Herrera, D., Muñoz-Medina, J. E., Fernandes-Matano, L., Salas-Lais, A. G., Hernández-Cueto, M. D. L. Á., Santacruz-Tinoco, C. E., Monroy-Muñoz, I. E., & Angeles-Martínez, J. (2023). Association of Obesity with SARS-CoV-2 and Its Relationship with the Humoral Response Prior to Vaccination in the State of Mexico: A Cross-Sectional Study. Diagnostics, 13(16), 2630. https://doi.org/10.3390/diagnostics13162630





