Thoracic Aorta: Anatomy and Pathology
Abstract
1. Introduction
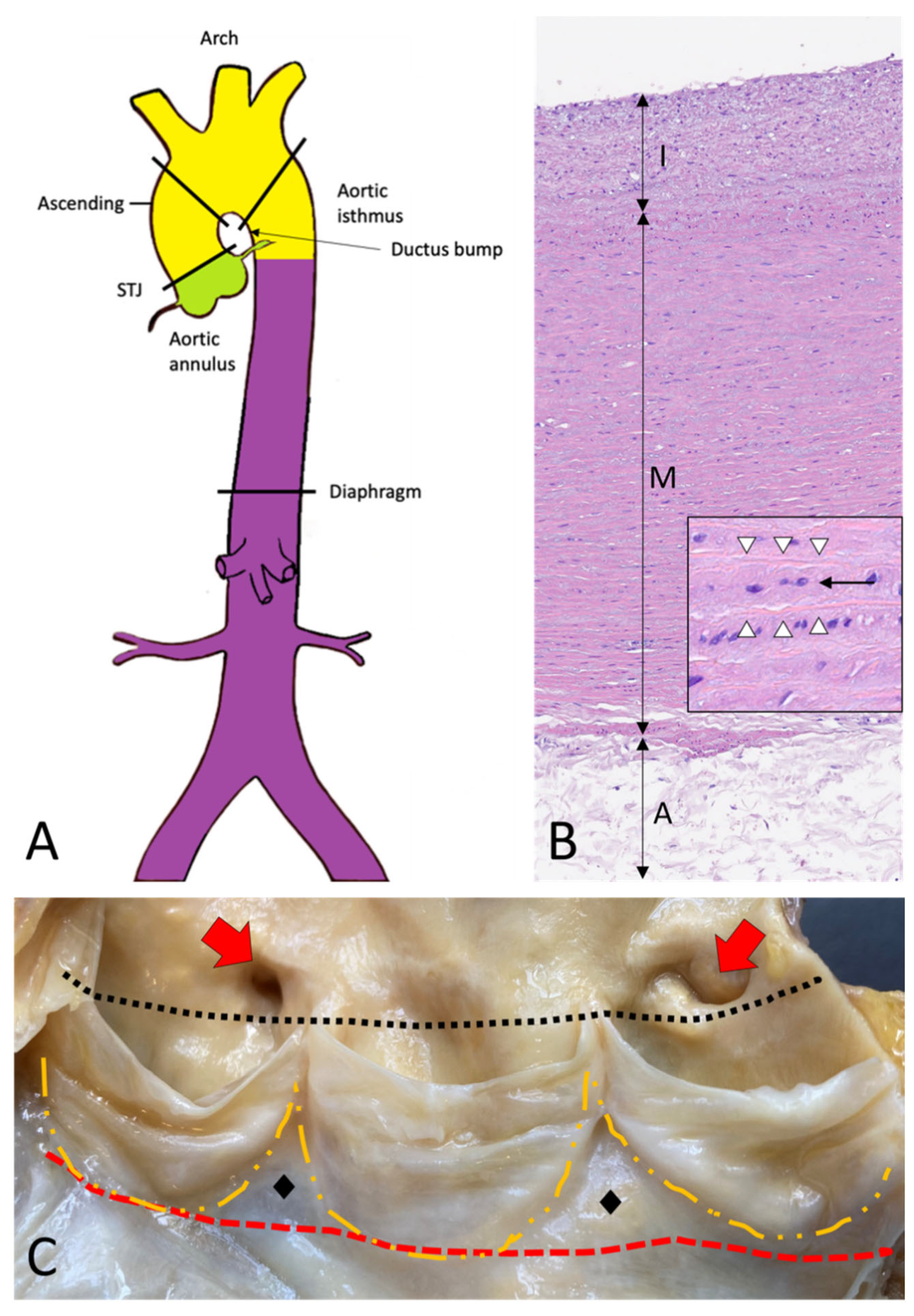
2. Topographic Anatomy of the Thoracic Aorta
3. Histology and Histogenesis of the Aorta
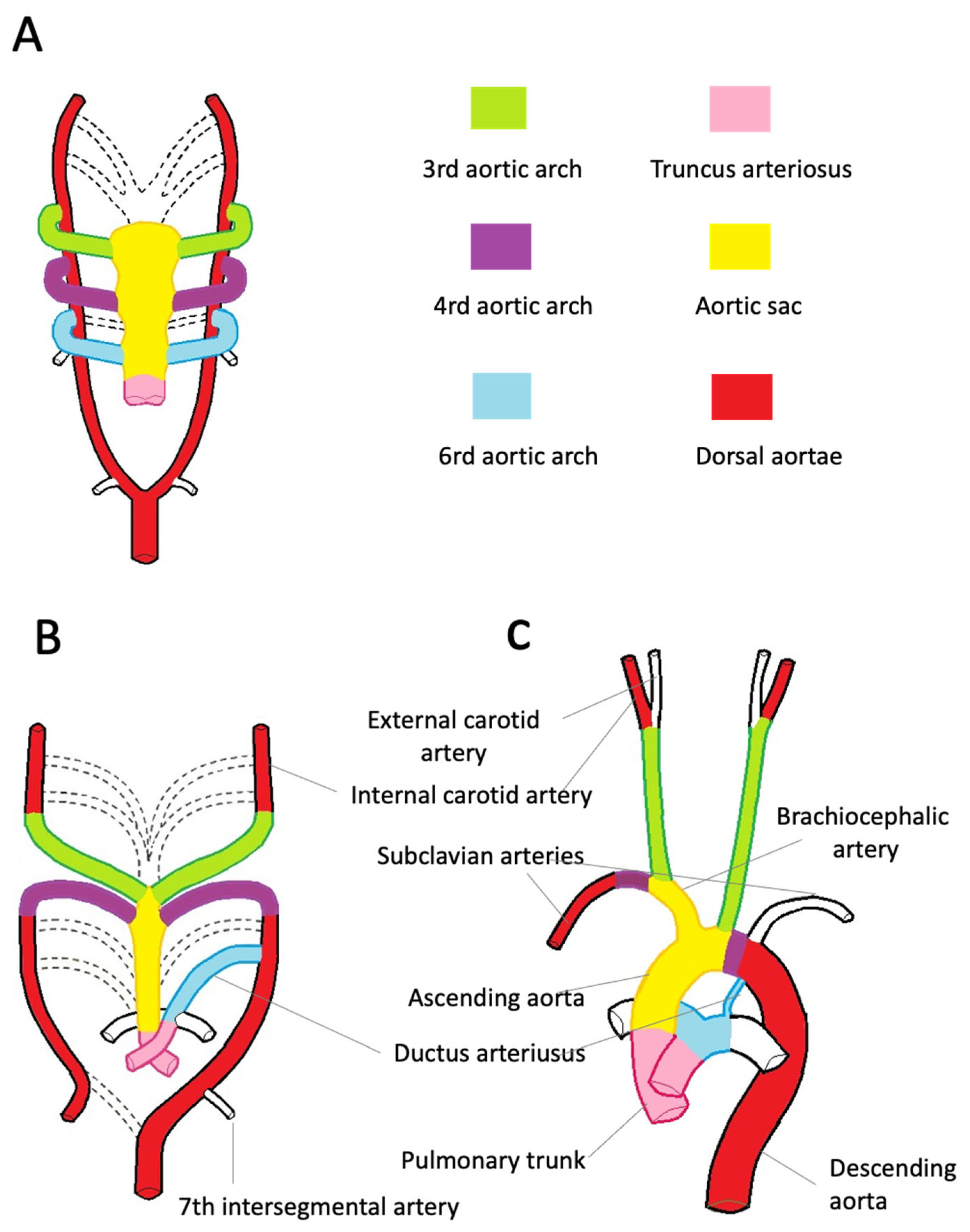
4. Embryology of the Aorta
4.1. Embryology of the Aortic Root
4.2. Embryology of the Ascending Aorta, Aortic Arch, and Descending Aorta
5. Congenital Disease of the Aorta
5.1. Aortic Root Malformations
5.2. Aortic Arch Malformations and Coarctation of the Aorta
6. Aortic Age- and Gender-Related Changes
7. Thoracic Aortic Aneurysm
7.1. Definition and Epidemiology
7.2. Etiology
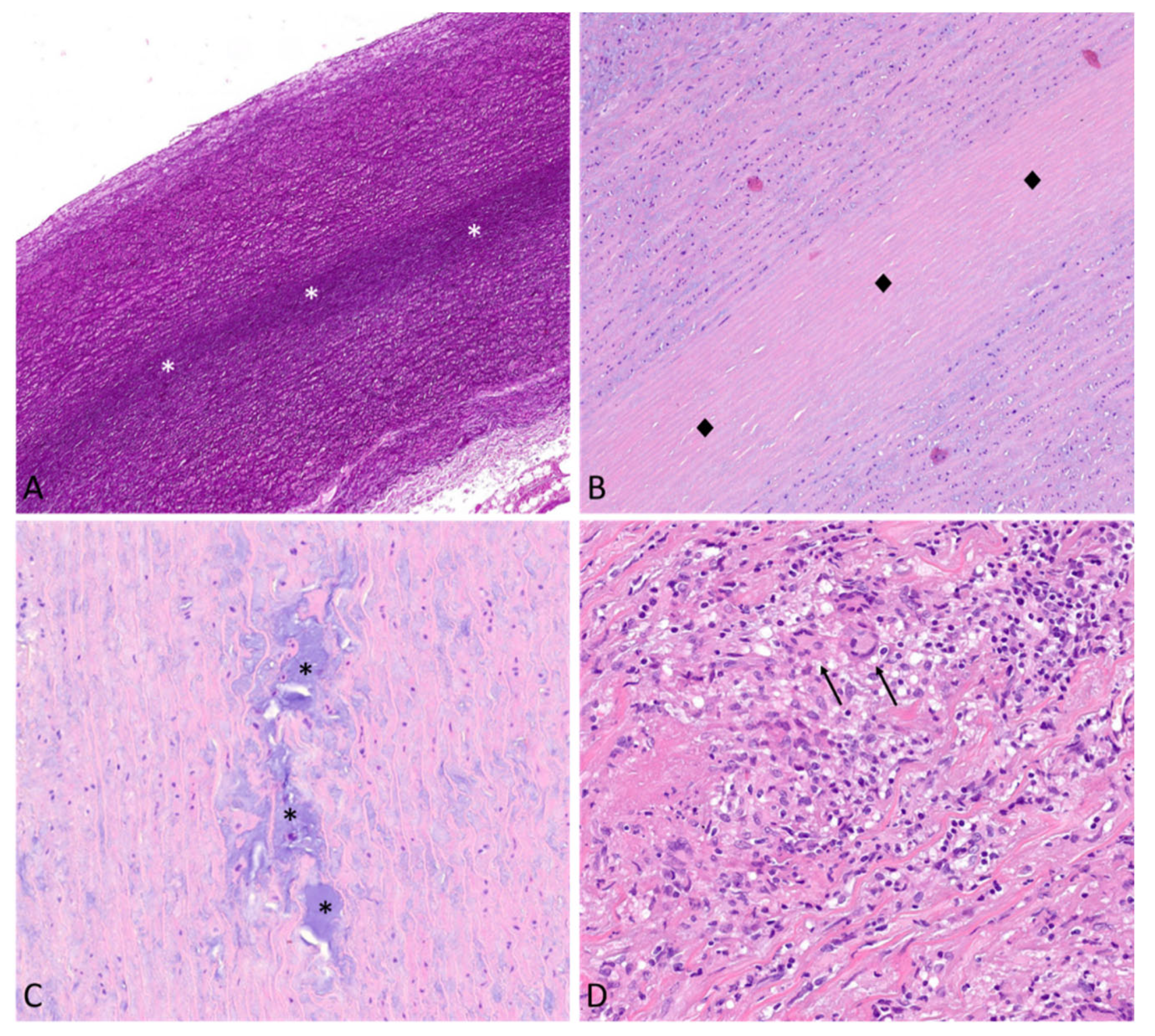
7.3. Morphologic Substrates of TAAs
7.3.1. Medial Non-Inflammatory Degenerative Disease
7.3.2. Inflammatory Aortic Disease
8. Acute Aortic Syndrome
8.1. Thoracic Aorta Dissection
8.1.1. Definition and Epidemiology
8.1.2. Risk Factors and Morphologic Substrates
8.2. Acute Aortic Syndromes Other Than AAD
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- d’amati, G.; Leone, O.; Nistri, S.; Roghi, A.; Angelini, A.; Basso, C.; Biagini, E.; Colombo, E.; Mele, D.; Pepe, G.; et al. Paradigm shifts in aortic pathology: Clinical and therapeutic implications. Biology and pathology of the aortic wall. G. Ital. Cardiol. 2013, 14, 97–109. [Google Scholar] [CrossRef]
- Virmani, R.; Sato, Y.; Sakamoto, A.; Romero, M.E.; Butany, J. Aneurysms of the aorta: Ascending, thoracic, and abdominal and their management. In Cardiovascular Pathology, 5th ed.; Academic Press: Cambridge, MA, USA, 2022; Volume 353–406, pp. 165–173. [Google Scholar]
- de Kerchove, L.; Jashari, R.; Boodhwani, M.; Duy, K.T.; Lengelé, B.; Gianello, P.; Nezhad, Z.M.; Astarci, P.; Noirhomme, P.; El Khoury, G. Surgical anatomy of the aortic root: Implication for valve-sparing reimplantation and aortic valve annuloplasty. J. Thorac. Cardiovasc. Surg. 2015, 149, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Khelil, N.; Sleilaty, G.; Palladino, M.; Fouda, M.; Escande, R.; Debauchez, M.; Di Centa, I.; Lansac, E. Surgical anatomy of the aortic annulus: Landmarks for external annuloplasty in aortic valve repair. Ann. Thorac. Surg. 2015, 99, 1220–1226. [Google Scholar] [CrossRef] [PubMed]
- Wolinsky, H.; Glagov, S. Comparison of abdominal and thoracic aortic medial structure in mammals. Deviation of man from the usual pattern. Circ. Res. 1969, 25, 677–686. [Google Scholar] [CrossRef]
- Cheung, C.; Bernardo, A.S.; Trotter, M.W.; Pedersen, R.A.; Sinha, S. Generation of human vascular smooth muscle subtypes provides insight into embryological origin-dependent disease susceptibility. Nat. Biotechnol. 2012, 30, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Majesky, M.W. Developmental basis of vascular smooth muscle diversity. Arter. Thromb. Vasc. Biol. 2007, 27, 1248–1258. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Rowitch, D.H.; Soriano, P.; McMahon, A.P.; Sucov, H.M. Fate of the mammalian cardiac neural crest. Development 2000, 127, 1607–1616. [Google Scholar] [CrossRef]
- Wolinsky, H. Comparison of medial growth of human thoracic and abdominal aortas. Circ. Res. 1970, 27, 531–538. [Google Scholar] [CrossRef]
- Rathke, H. Ueber die Entwickelung der Arterien welche bie den Saugethieren von den Bogen der Aorta ausgehen. Arch. Anat. 1843, 9, 270–302. [Google Scholar]
- Kau, T.; Sinzig, M.; Gasser, J.; Lesnik, G.; Rabitsch, E.; Celedin, S.; Eicher, W.; Illiasch, H.; Hausegger, K.A. Aortic development and anomalies. Semin. Interv. Radiol. 2007, 24, 141–152. [Google Scholar] [CrossRef]
- Kaukis, R.; Kaoukis, R.; Dieter, R.S.; IOkundaye, I.; Dauzvardis, M.; Frysztak, R.J.; Ibrahim, W.; Pyle, M.J. Embryology and Anatomy of the Aorta in: Diseases of Aorta, 3rd ed.; Springer Nature: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Schoenwolf, G.; Bleyl, S.; Brauer, P.; Francis-West, P. Larsen’s Human Embryology, 6th ed.; Churchill Livingstone Elsevier: London, UK, 2020. [Google Scholar]
- Khalid, N.; Bordoni, B. Embryology, Great Vessel. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK545254/ (accessed on 1 April 2023).
- Bravo-Jaimes, K.; Prakash, S.K. Genetics in bicuspid aortic valve disease: Where are we? Prog. Cardiovasc. Dis. 2020, 63, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Kong, W.K.F.; Bax, J.J.; Michelena, H.I.; Delgado, V. Sex differences in bicuspid aortic valve disease. Prog. Cardiovasc. Dis. 2020, 63, 452–456. [Google Scholar] [CrossRef]
- Curfman, G. Bicuspid Aortic Valve-A Common Form of Structural Heart Disease. JAMA 2021, 325, 540–541. [Google Scholar] [CrossRef]
- Michelena, H.I.; Della Corte, A.; Evangelista, A.; Maleszewski, J.J.; Edwards, W.D.; Roman, M.J.; Devereux, R.B.; Fernández, B.; Asch, F.M.; Barker, A.J.; et al. International Consensus Statement on Nomenclature and Classification of the Congenital Bicuspid Aortic Valve and Its Aortopathy, for Clinical, Surgical, Interventional and Research Purposes. Radiol. Cardiothorac. Imaging 2021, 3, e200496. [Google Scholar] [CrossRef] [PubMed]
- Backer, C.L.; Mongé, M.C.; Popescu, A.R.; Eltayeb, O.M.; Rastatter, J.C.; Rigsby, C.K. Vascular rings. Semin. Pediatr. Surg. 2016, 25, 165–175. [Google Scholar] [CrossRef]
- Yoshimura, N.; Fukahara, K.; Yamashita, A.; Doi, T.; Yamashita, S.; Homma, T.; Yokoyama, S.; Aoki, M.; Higashida, A.; Shimada, Y.; et al. Congenital vascular ring. Surg. Today 2020, 50, 1151–1158. [Google Scholar] [CrossRef] [PubMed]
- Backer, C.L.; Mavroudis, C. Congenital Heart Surgery Nomenclature and Database Project: Vascular rings, tracheal stenosis, pectus excavatum. Ann. Thorac. Surg. 2000, 69 (Suppl. S4), S308–S318. [Google Scholar] [CrossRef]
- Bin Bae, S.; Kang, E.-J.; Choo, K.S.; Lee, J.; Kim, S.H.; Lim, K.J.; Kwon, H. Aortic Arch Variants and Anomalies: Embryology, Imaging Findings, and Clinical Considerations. J. Cardiovasc. Imaging 2022, 30, 231–262. [Google Scholar] [CrossRef]
- Torok, R.D.; Torok, R.D.; Campbell, M.J.; Fleming, G.A.; Hill, K.D. Coarctation of the Aorta in: Diseases of Aorta, 3rd ed.; Springer Nature: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Law, M.A.; Tivakaran, V.S. Coarctation of the Aorta. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK430913/ (accessed on 31 March 2023).
- Pirruccello, J.P.; Rämö, J.T.; Choi, S.H.; Chaffin, M.D.; Kany, S.; Nekoui, M.; Chou, E.L.; Jurgens, S.J.; Friedman, S.F.; Juric, D.; et al. The Genetic Determinants of Aortic Distention. J. Am. Coll. Cardiol. 2023, 81, 1320–1335. [Google Scholar] [CrossRef]
- Cheung, K.; Boodhwani, M.; Chan, K.L.; Beauchesne, L.; Dick, A.; Coutinho, T. Thoracic Aortic Aneurysm Growth: Role of Sex and Aneurysm Etiology. J. Am. Heart Assoc. 2017, 6, e003792. [Google Scholar] [CrossRef]
- Chung, J.; Stevens, L.-M.; Ouzounian, M.; El-Hamamsy, I.; Bouhout, I.; Dagenais, F.; Cartier, A.; Peterson, M.D.; Boodhwani, M.; Guo, M.H.; et al. Sex-Related Differences in Patients Undergoing Thoracic Aortic Surgery. Circulation 2019, 139, 1177–1184. [Google Scholar] [CrossRef]
- Chung, J.; Coutinho, T.; Chu, M.W.A.; Ouzounian, M. Sex differences in thoracic aortic disease: A review of the literature and a call to action. J. Thorac. Cardiovasc. Surg. 2020, 160, 656–660. [Google Scholar] [CrossRef]
- Sokolis, D.P.; Iliopoulos, D.C. Impaired mechanics and matrix metalloproteinases/inhibitors expression in female ascending thoracic aortic aneurysms. J. Mech. Behav. Biomed. Mater. 2014, 34, 154–164. [Google Scholar] [CrossRef]
- Bossone, E.; Eagle, K.A. Epidemiology and management of aortic disease: Aortic aneurysms and acute aortic syndromes. Nat. Rev. Cardiol. 2021, 18, 331–348. [Google Scholar] [CrossRef]
- Boutefeu, J.M.; Moret, P.R.; Hahn, C.; Hauf, E. Aneurysms of the sinus of Valsalva. Report of seven cases and review of the literature. Am. J. Med. 1978, 65, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Sakakibara, S.; Konno, S. Congenital aneurysm of the sinus of Valsalva. Anatomy and classification. Am. Heart J. 1962, 63, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Newton, E.R.; Akerman, A.W.; Strassle, P.D.; Kibbe, M.R. Association of Fluoroquinolone Use with Short-term Risk of Develop- ment of Aortic Aneurysm. JAMA Surg. 2021, 156, 264–272. [Google Scholar] [CrossRef]
- Chen, S.W.; Chan, Y.H.; Chien-Chia Wu, V.; Cheng, Y.T.; Chen, D.Y.; Lin, C.P.; Hung, K.C.; Chang, S.H.; Chu, P.H.; Chou, A.H. Effects of fluoroquinolones on outcomes of patients with aortic dissection or aneurysm. J. Am. Coll. Cardiol. 2021, 77, 1875–1887. [Google Scholar] [CrossRef]
- Bennett, C.L.; Witherspoon, B.J.; Knopf, K.B. An evaluation of reports of ciprofloxacin, levofloxacin, and moxifloxacin-association neuropsychiatric toxicities, long-term disability, and aortic aneurysms/dissections disseminated by the Food and Drug Administration and the European Medicines Agency. Expert Opin. Drug Saf. 2019, 18, 1055–1063. [Google Scholar] [CrossRef]
- Jondeau, G.; Boileau, C. Familial thoracic aortic aneurysms. Curr. Opin. Cardiol. 2014, 29, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Pyeritz, R.E. Heritable thoracic aortic disorders. Curr. Opin. Cardiol. 2014, 29, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Monda, E.; Lioncino, M.; Verrillo, F.; Rubino, M.; Caiazza, M.; Mauriello, A.; Guarnaccia, N.; Fusco, A.; Cirillo, A.; Covino, S.; et al. The Role of Genetic Testing in Patients with Heritable Thoracic Aortic Diseases. Diagnostics 2023, 13, 772. [Google Scholar] [CrossRef] [PubMed]
- De Cario, R.; Giannini, M.; Cassioli, G.; Kura, A.; Gori, A.M.; Marcucci, R.; Nistri, S.; Pepe, G.; Giusti, B.; Sticchi, E. Tracking an Elusive Killer: State of the Art of Molecular-Genetic Knowledge and Laboratory Role in Diagnosis and Risk Stratification of Thoracic Aortic Aneurysm and Dissection. Diagnostics 2022, 12, 1785. [Google Scholar] [CrossRef] [PubMed]
- Gäbel, G.; Northoff, B.H.; Balboa, A.; Agic, M.B.; Petri, M.; Busch, A.; Maegdefessel, L.; Mahlmann, A.; Ludwig, S.; Teupser, D.; et al. Parallel Murine and Human Aortic Wall Genomics Reveals Metabolic Reprogramming as Key Driver of Abdominal Aortic Aneurysm Progression. J. Am. Hear. Assoc. 2021, 10, e020231. [Google Scholar] [CrossRef]
- Oller, J.; Gabandé-Rodríguez, E.; Ruiz-Rodríguez, M.J.; Desdín-Micó, G.; Aranda, J.F.; Rodrigues-Diez, R.; Ballesteros-Martínez, C.; Blanco, E.M.; Roldan-Montero, R.; Acuña, P.; et al. Extracellular Tuning of Mitochondrial Respiration Leads to Aortic Aneurysm. Circulation 2021, 143, 2091–2109. [Google Scholar] [CrossRef]
- Van Der Pluijm, I.; Burger, J.; Van Heijningen, P.M.; Ijpma, A.; Van Vliet, N.; Milanese, C.; Schoonderwoerd, K.; Sluiter, W.; Ringuette, L.-J.; Dekkers, D.H.W.; et al. Decreased mitochondrial respiration in aneurysmal aortas of Fibulin-4 mutant mice is linked to PGC1A regulation. Cardiovasc. Res. 2018, 114, 1776–1793. [Google Scholar] [CrossRef]
- Oller, J.; Gabandé-Rodríguez, E.; Roldan-Montero, R.; Ruiz-Rodríguez, M.J.; Redondo, J.M.; Martín-Ventura, J.L.; Mittelbrunn, M. Rewiring Vascular Metabolism Prevents Sudden Death due to Aortic Ruptures—Brief Report. Arter. Thromb. Vasc. Biol. 2022, 42, 462–469. [Google Scholar] [CrossRef]
- Xiang, B.; Abudupataer, M.; Liu, G.; Zhou, X.; Liu, D.; Zhu, S.; Ming, Y.; Yin, X.; Yan, S.; Sun, Y.; et al. Ciprofloxacin exacerbates dysfunction of smooth muscle cells in a microphysiological model of thoracic aortic aneurysm. JCI Insight 2023, 8, e161729. [Google Scholar] [CrossRef]
- Moullan, N.; Mouchiroud, L.; Wang, X.; Ryu, D.; Williams, E.G.; Mottis, A.; Jovaisaite, V.; Frochaux, M.V.; Quiros, P.M.; Deplancke, B.; et al. Tetracyclines Disturb Mitochondrial Function across Eukaryotic Models: A Call for Caution in Biomedical Research. Cell Rep. 2015, 10, 1681–1691. [Google Scholar] [CrossRef]
- Stone, J.R.; Bruneval, P.; Angelini, A.; Bartoloni, G.; Basso, C.; Batoroeva, L.; Buja, L.M.; Butany, J.; D’Amati, G.; Fallon, J.T.; et al. Consensus statement on surgical pathology of the aorta from the Society for Cardiovascular Pathology and the Association for European Cardiovascular Pathology: I. Inflammatory diseases. Cardiovasc. Pathol. 2015, 24, 267–278. [Google Scholar] [CrossRef]
- Halushka, M.K.; Angelini, A.; Bartoloni, G.; Basso, C.; Batoroeva, L.; Bruneval, P.; Buja, L.M.; Butany, J.; D’Amati, G.; Fallon, J.T.; et al. Consensus statement on surgical pathology of the aorta from the Society for Cardiovascular Pathology and the Association for European Cardiovascular Pathology: II. Noninflammatory degenerative diseases—Nomenclature and diagnostic criteria. Cardiovasc. Pathol. 2016, 25, 247–257. [Google Scholar] [CrossRef]
- Leone, O.; Corsini, A.; Pacini, D.; Corti, B.; Lorenzini, M.; Laus, V.; Foà, A.; Reggiani, M.L.B.; Di Marco, L.; Rapezzi, C. The complex interplay among atherosclerosis, inflammation, and degeneration in ascending thoracic aortic aneurysms. J. Thorac. Cardiovasc. Surg. 2020, 160, 1434–1443.e6. [Google Scholar] [CrossRef] [PubMed]
- Waters, K.M.; Rooper, L.; Guajardo, A.; Halushka, M.K. Histopathologic differences partially distinguish syndromic aortic diseases. Cardiovasc. Pathol. 2017, 30, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Amemiya, K.; Mousseaux, E.; Ishibashi-Ueda, H.; Achouh, P.; Ochiai, M.; Bruneval, P. Impact of histopathological changes in ascending aortic diseases. Int. J. Cardiol. 2020, 311, 91–96. [Google Scholar] [CrossRef]
- Amemiya, K.; Ishibashi-Ueda, H.; Mousseaux, E.; Achouh, P.; Ochiai, M.; Bruneval, P. Comparison of the damage to aorta wall in aortitis versus noninflammatory degenerative aortic diseases. Cardiovasc. Pathol. 2021, 52, 107329. [Google Scholar] [CrossRef]
- Lindeman, J.H.; Rabelink, T.J.; van Bockel, J.H. Immunosuppression and the abdominal aortic aneurysm: Doctor Jekyll or Mister Hyde? Circulation 2011, 124, e463–e465. [Google Scholar] [CrossRef]
- Stone, J.R. Diseases of small and medium-sized blood vessels. In Cardiovascular Pathology, 5th ed.; Academic Press: Cambridge, MA, USA, 2022; pp. 307–351. [Google Scholar]
- Ozawa, M.; Fujinaga, Y.; Asano, J.; Nakamura, A.; Watanabe, T.; Ito, T.; Muraki, T.; Hamano, H.; Kawa, S. Clinical features of IgG4-related periaortitis/periarteritis based on the analysis of 179 patients with IgG4-related disease: A case-control study. Arthritis Res. Ther. 2017, 19, 223. [Google Scholar] [CrossRef]
- Koo, B.S.; Koh, Y.W.; Hong, S.; Kim, Y.J.; Kim, Y.-G.; Lee, C.-K.; Yoo, B. Frequency of immunoglobulin G4-related aortitis in cases with aortic resection and their clinical characteristics compared to other aortitises. Int. J. Rheum. Dis. 2014, 17, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Nikiphorou, E.; Galloway, J.; Fragoulis, G.E. Overview of IgG4-related aortitis and periaortitis. A decade since their first description. Autoimmun. Rev. 2020, 19, 102694. [Google Scholar] [CrossRef]
- Botelho-Nevers, E.; Fournier, P.-E.; Richet, H.; Fenollar, F.; Lepidi, H.; Foucault, C.; Branchereau, A.; Piquet, P.; Maurin, M.; Raoult, D. Coxiella urnetiid infection of aortic aneurysms or vascular grafts: Report of 30 new cases and evaluation of outcome. Eur. J. Clin. Microbiol. Infect. Dis. 2007, 26, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Howard, D.P.; Banerjee, A.; Fairhead, J.F.; Perkins, J.; Silver, L.E.; Rothwell, P.M. Population-based study of incidence and outcome of acute aortic dissection and premorbid risk factor control: 10-year results from the Oxford Vascular Study. Circulation 2013, 127, 2031–2037. [Google Scholar] [CrossRef] [PubMed]
- Acosta, S.; Gottsäter, A. Stable population-based incidence of acute type A and B aortic dissection. Scand. Cardiovasc. J. 2019, 53, 274–279. [Google Scholar] [CrossRef]
- Isselbacher, E.M.; Preventza, O.; Black, I.J.H., 3rd; Augoustides, J.G.; Beck, A.W.; Bolen, M.A.; Braverman, A.C.; Bray, B.E.; Brown-Zimmerman, M.M.; Chen, E.P.; et al. 2022 ACC/AHA Guideline for the Diagnosis and Management of Aortic Disease: A Report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. Circulation 2022, 146, e334–e482. [Google Scholar] [CrossRef] [PubMed]
- Leone, O.; Pacini, D.; Foà, A.; Corsini, A.; Agostini, V.; Corti, B.; Di Marco, L.; Leone, A.; Lorenzini, M.; Reggiani, L.B.; et al. Redefining the histopathologic profile of acute aortic syndromes: Clinical and prognostic implications. J. Thorac. Cardiovasc. Surg. 2018, 156, 1776–1785.e6. [Google Scholar] [CrossRef] [PubMed]
- AlOmari, I.B.; Hamirani, Y.S.; Madera, G.; Tabe, C.; Akhtar, N.; Raizada, V. Aortic Intramural Hematoma and Its Complications. Circulation 2014, 129, 711–716. [Google Scholar] [CrossRef]
- Dev, R.; Gitanjali, K.; Anshuman, D. Demystifying penetrating atherosclerotic ulcer of aorta: Unrealized tyrant of senile aortic changes. J. Cardiovasc. Thorac. Res. 2021, 13, 1–14. [Google Scholar] [CrossRef]
| Granulomatous/Giant Cell | |
|---|---|
| Non-infectious Giant cell arteritis Takayasu’s arteritis Granulomatosis with polyangiitis Sarcoidosis Rheumatoid arthritis | Infectious Syphilitic aortitis Mycobacterial Fungal infection |
| Lymphoplasmacytic | |
| Non-infectious IgG4-related disease Ankylosing spondylitis Systemic lupus erythematosus | Infectious Syphilitic aortitis |
| Suppurative | |
| Non-infectious | Infectious Staphylococcus, Streptococcus, Salmonella, Pseudomonas, fungal infections |
| Mixed | |
| Non-infectious Behçet’s disease | Non-infectious |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
di Gioia, C.R.T.; Ascione, A.; Carletti, R.; Giordano, C. Thoracic Aorta: Anatomy and Pathology. Diagnostics 2023, 13, 2166. https://doi.org/10.3390/diagnostics13132166
di Gioia CRT, Ascione A, Carletti R, Giordano C. Thoracic Aorta: Anatomy and Pathology. Diagnostics. 2023; 13(13):2166. https://doi.org/10.3390/diagnostics13132166
Chicago/Turabian Styledi Gioia, Cira Rosaria Tiziana, Andrea Ascione, Raffaella Carletti, and Carla Giordano. 2023. "Thoracic Aorta: Anatomy and Pathology" Diagnostics 13, no. 13: 2166. https://doi.org/10.3390/diagnostics13132166
APA Styledi Gioia, C. R. T., Ascione, A., Carletti, R., & Giordano, C. (2023). Thoracic Aorta: Anatomy and Pathology. Diagnostics, 13(13), 2166. https://doi.org/10.3390/diagnostics13132166






