Is Panenteric PillcamTM Crohn’s Capsule Endoscopy Ready for Widespread Use? A Narrative Review
Abstract
1. Introduction
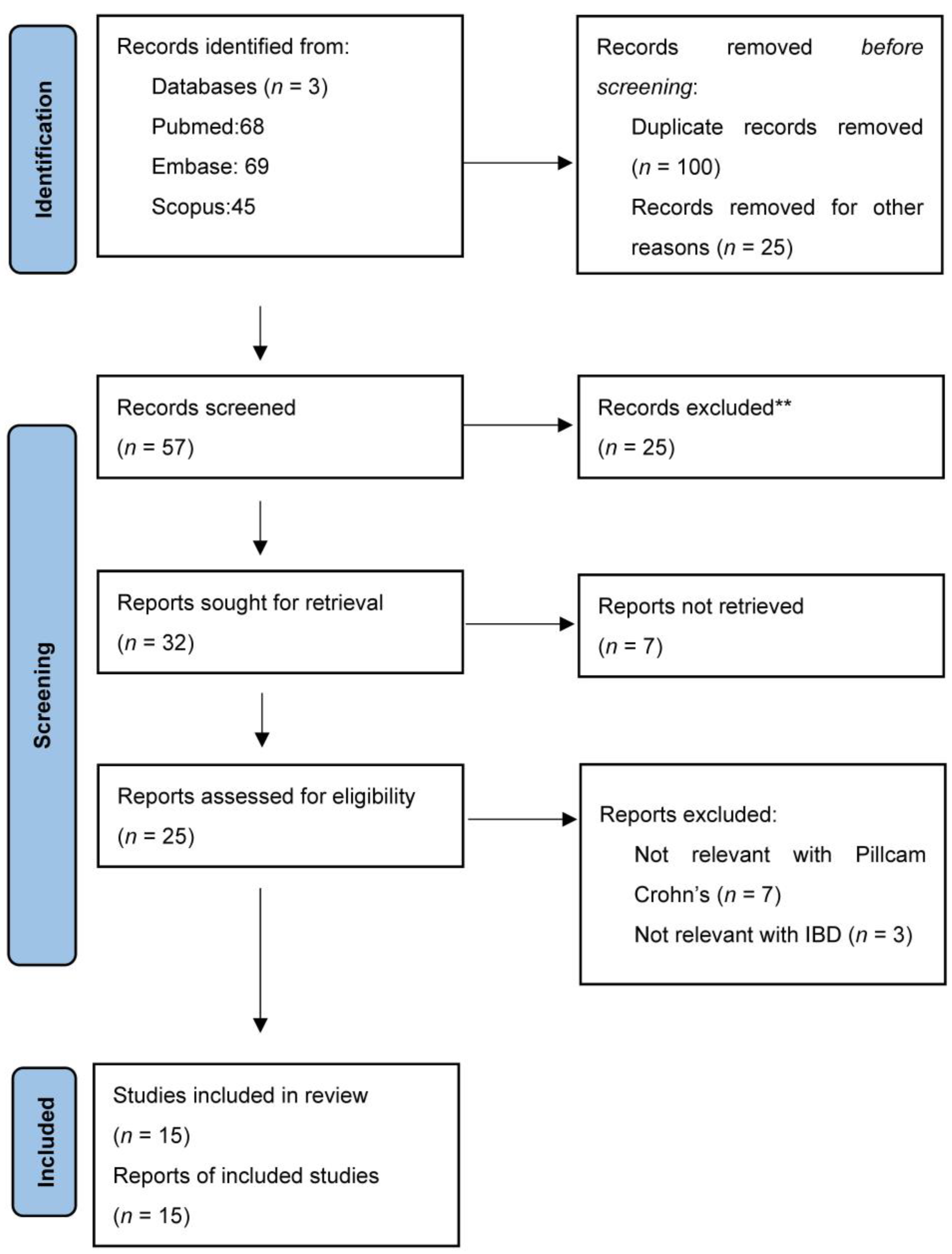
2. Materials and Methods
3. Results
3.1. Panenteric Capsule Endoscopy in Adults with Inflammatory Bowel Disease
3.2. Pantenteric Capsule Endoscopy in Paediatric Inflammatory Bowel Disease
3.3. Scoring Systems in PCC
3.3.1. Lewis Score (LS)
3.3.2. Pillcam Crohn’s Capsule Score
3.3.3. Capsule Endoscopy Crohn’s Disease Activity Index (CECDAI)
3.3.4. CECDALic Score
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nakamura, T.; Terano, A. Capsule endoscopy: Past, present, and future. J. Gastroenterol. 2008, 43, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Koffas, A.; Papaefthymiou, A.; Laskaratos, F.-M.; Kapsoritakis, A.; Epstein, O. Colon Capsule Endoscopy in the Diagnosis of Colon Polyps: Who Needs a Colonoscopy? Diagnostics 2022, 12, 2093. [Google Scholar] [CrossRef]
- Papaefthymiou, A.; Koffas, A.; Laskaratos, F.-M.; Epstein, O. Upper gastrointestinal video capsule endoscopy: The state of the art. Clin. Res. Hepatol. Gastroenterol. 2022, 46, 101798. [Google Scholar] [CrossRef]
- Koffas, A.; Laskaratos, F.-M.; Epstein, O. Training in video capsule endoscopy: Current status and unmet needs. World J. Gastrointest. Endosc. 2019, 11, 395–402. [Google Scholar] [CrossRef]
- Koulaouzidis, A.; Dabos, K.; Philipper, M.; Toth, E.; Keuchel, M. How should we do colon capsule endoscopy reading: A practical guide. Ther. Adv. Gastrointest. Endosc. 2021, 14, 26317745211001983. [Google Scholar] [CrossRef] [PubMed]
- Valdivia, P.C.; Elosua, A.; Houdeville, C.; Pennazio, M.; Fernández-Urién, I.; Dray, X.; Toth, E.; Eliakim, R.; Koulaouzidis, A. Clinical feasibility of panintestinal (or panenteric) capsule endoscopy: A systematic review. Eur. J. Gastroenterol. Hepatol. 2021, 33, 949–955. [Google Scholar] [CrossRef] [PubMed]
- Ribon, C.C.; Prieto-Frías, C.; Angos, R.; Betés, M.; Bayod, M.H.; Onandía, S.D.L.R.; Zozaya, F.; Fernandez-Calderon, M.; Lago, I.R.; Navas, M.M. Pan-enteric capsule for bleeding high-risk patients. Can we limit endoscopies? Rev. Esp. Enferm. Dig. 2021, 113, 580–584. [Google Scholar] [CrossRef]
- Tontini, G.E.; Rizzello, F.; Cavallaro, F.; Bonitta, G.; Gelli, D.; Pastorelli, L.; Salice, M.; Vecchi, M.; Gionchetti, P.; Calabrese, C. Usefulness of panoramic 344°-viewing in Crohn’s disease capsule endoscopy: A proof of concept pilot study with the novel PillCam™ Crohn’s system. BMC Gastroenterol. 2020, 20, 97. [Google Scholar] [CrossRef]
- Na, S.-Y.; Lim, Y.-J. Capsule Endoscopy in Inflammatory Bowel Disease: When? To Whom? Diagnostics 2021, 11, 2240. [Google Scholar] [CrossRef]
- Eliakim, R.; Yablecovitch, D.; Lahat, A.; Ungar, B.; Shachar, E.; Carter, D.; Selinger, L.; Neuman, S.; Ben-Horin, S.; Kopylov, U. A novel PillCam Crohn’s capsule score (Eliakim score) for quantification of mucosal inflammation in Crohn’s disease. United Eur. Gastroenterol. J. 2020, 8, 544–551. [Google Scholar] [CrossRef]
- Majtner, T.; Brodersen, J.B.; Herp, J.; Kjeldsen, J.; Halling, M.L.; Jensen, M.D. A deep learning framework for autonomous detection and classification of Crohn’s disease lesions in the small bowel and colon with capsule endoscopy. Endosc. Int. Open 2021, 9, E1361–E1370. [Google Scholar] [CrossRef]
- Leighton, J.A.; Helper, D.J.; Gralnek, I.M.; Dotan, I.; Fernandez-Urien, I.; Lahat, A.; Malik, P.; Mullin, G.E.; Rosa, B. Comparing diagnostic yield of a novel pan-enteric video capsule endoscope with ileocolonoscopy in patients with active Crohn’s disease: A feasibility study. Gastrointest. Endosc. 2017, 85, 196–205.e1. [Google Scholar] [CrossRef]
- Eliakim, R.; Spada, C.; Lapidus, A.; Eyal, I.; Pecere, S.; Fernández-Urién, I.; Lahat, A.; Costamagna, G.; Schwartz, A.; Ron, Y.; et al. Evaluation of a new pan-enteric video capsule endoscopy system in patients with suspected or established inflammatory bowel disease—feasibility study. Endosc. Int. Open 2018, 6, E1235–E1246. [Google Scholar] [CrossRef]
- Adler, S.N.; Lama, Y.G.; Royo, V.M.; Ferrer, C.S.; Schwartz, A.; Shitrit, A.B.-G. Comparison of small-bowel colon capsule endoscopy system to conventional colonoscopy for the evaluation of ulcerative colitis activity. Endosc. Int. Open 2019, 7, E1253–E1261. [Google Scholar] [CrossRef]
- Bruining, D.H.; Oliva, S.; Fleisher, M.R.; Fischer, M.; Fletcher, J.G. Panenteric capsule endoscopy versus ileocolonoscopy plus magnetic resonance enterography in Crohn’s disease: A multicentre, prospective study. BMJ Open Gastroenterol. 2020, 7, e000365. [Google Scholar] [CrossRef]
- Tai, F.W.D.; Ellul, P.; Elosua, A.; Fernandez-Urien, I.; Tontini, G.E.; Elli, L.; Eliakim, R.; Kopylov, U.; Koo, S.; Parker, C.; et al. Panenteric capsule endoscopy identifies proximal small bowel disease guiding upstaging and treatment intensification in Crohn’s disease: A European multicentre observational cohort study. United Eur. Gastroenterol. J. 2021, 9, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Volkers, A.; Bossuyt, P.; de Jong, J.; Pouillon, L.; Gecse, K.; Duijvestein, M.; Ponsioen, C.; D’haens, G.; Löwenberg, M. Assessment of endoscopic response using pan-enteric capsule endoscopy in Crohn’s disease; the Sensitivity to Change (STOC) study. Scand. J. Gastroenterol. 2022, 57, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Oliva, S.; Aloi, M.; Viola, F.; Mallardo, S.; Civitelli, F.; Maccioni, F.; Hassan, C.; Papoff, P.; Cucchiara, S.; Cohen, S.A. A Treat to Target Strategy Using Panenteric Capsule Endoscopy in Pediatric Patients with Crohn’s Disease. Clin. Gastroenterol. Hepatol. 2019, 17, 2060–2067.e1. [Google Scholar] [CrossRef] [PubMed]
- Brodersen, J.B.; Andersen, K.W.; Jensen, M.D. Adherence to the bowel cleansing regimen for pan-enteric capsule endoscopy in patients with suspected Crohn’s disease and factors affecting the image quality. Scand. J. Gastroenterol. 2022, 57, 501–506. [Google Scholar] [CrossRef]
- Rosa, B.; Pinho, R.; De Ferro, S.M.; Almeida, N.; Cotter, J.; Saraiva, M.M. Endoscopic Scores for Evaluation of Crohn’s Disease Activity at Small Bowel Capsule Endoscopy: General Principles and Current Applications. GE—Port. J. Gastroenterol. 2015, 23, 36–41. [Google Scholar] [CrossRef]
- Yablecovitch, D.; Lahat, A.; Neuman, S.; Levhar, N.; Avidan, B.; Ben-Horin, S.; Eliakim, R.; Kopylov, U. The Lewis score or the capsule endoscopy Crohn’s disease activity index: Which one is better for the assessment of small bowel inflammation in established Crohn’s disease? Ther. Adv. Gastroenterol. 2018, 11, 1756283X17747780. [Google Scholar] [CrossRef] [PubMed]
- Arieira, C.; Magalhães, R.; De Castro, F.D.; Carvalho, P.B.; Rosa, B.; Moreira, M.J.; Cotter, J. CECDAIic—A new useful tool in pan-intestinal evaluation of Crohn’s disease patients in the era of mucosal healing. Scand. J. Gastroenterol. 2019, 54, 1326–1330. [Google Scholar] [CrossRef] [PubMed]
- Tamilarasan, A.G.; Tran, Y.; Paramsothy, S.; Leong, R. The diagnostic yield of pan-enteric capsule endoscopy in inflammatory bowel disease: A systematic review and meta-analysis. J. Gastroenterol. Hepatol. 2022, 37, 2207–2216. [Google Scholar] [CrossRef] [PubMed]
- Tziortziotis, I.; Laskaratos, F.-M.; Coda, S. Role of Artificial Intelligence in Video Capsule Endoscopy. Diagnostics 2021, 11, 1192. [Google Scholar] [CrossRef] [PubMed]
| Author (Year) | Study Population | Study Design | Topic | Findings |
|---|---|---|---|---|
| Leighton et al., 2017 [12] | 114 | Prospective, multicentre | Comparison of diagnostic yield of PCE vs. IC for active CD lesions. | The diagnostic yield rate for active CD lesions was 83.3% for PCE and 69.7% for IC. PCE detection rates for active CD in the TI, caecum, ascending colon, transverse colon, descending/sigmoid colon, and rectum were 70, 38, 36, 30, 39, and 29%. IC was less sensitive with detection rates of 54, 26, 34, 24, 31, and 25%, respectively. |
| Eliakim et al., 2018 [13] | 41 | Prospective, multicentre | Feasibility study with a primary endpoint of successful video creation and report creation. The secondary endpoints were entire bowel inspection, duration of reading time, video quality, and adverse events. | All 41 videos met the primary endpoint. There was no capsule retention. Bowel coverage was graded 6.7 ± 0.6 and 6.1 ± 1.3 (1–7, unconfident–confident), image quality was graded 6.1 ± 0.8 (1–7, poor–excellent), and reading time was graded 3.7 ± 1.4 (1–7, very short to very long). |
| Adler et al., 2019 [14] | 30 | Prospective, multicentre | Comparison of PCE and colonoscopy in UC. | Moderate agreement for disease extent in UC in 56.5% of cases (kappa coefficient 0.42) with very good agreement for assessment of UC disease activity based on Mayo endoscopic subscore in 95.7% of cases (kappa coefficient 0.86). |
| Bruining et al., 2020 [15] | 158 | Prospective, multicentre | This study assessed the accuracy and safety of PCE in CD compared with IC and/or MRE. | PCE was equally sensitive to MRE and/or IC for active enteric inflammation (94 vs. 100%, p = 0.125) and more specific (74 vs. 22%, p = 0.001). The sensitivity of PCE was superior to that of MRE for enteric inflammation in the proximal small bowel (97 vs. 71%, p = 0.021) and similar to that of MRE and/or IC in the TI and colon. |
| Tontini et al., 2020 [8] | 41 | Prospective, multicentre | Comparison of diagnostic performance of PCC (with two cameras offering a 344° panoramic view) vs. the standard 172° view (one camera) in suspected or known CD. | PCC study completion rate of 90%. Compared with the standard 172°-view capsule, the panoramic 344°-view capsule showed that more patients had a relevant lesion (56.1% vs. 39.0%; p = 0.023), resulting in higher Lewis scores (222.8 vs. 185.7; p = 0.031), improving their clinical management (48.8% vs. 31.7%, p = 0.023) |
| Eliakim et al., 2020 [10] | 41 | Prospective, single-centre | The study evaluated the development of a novel Pillcam Crohn’s capsule score for the quantification of inflammation in the small bowel and colon in patients with CD | There was a high interrater reliability coefficient between the two readers for Lewis inflammatory and PillCamTM Crohn’s score (0.9, p < 0.0001 for both). The correlation between PillCamTM Crohn’s score and faecal calprotectin was stronger than for Lewis score (r = 0.32 and 0.54, respectively, p = 0.001 for both). |
| Majter et al., 2021 [11] | 38 | Prospective, multicentre | Detection and classification of CD using PCE using a deep learning framework | Deep learning approaches in PCE led to identification of ulcers with sensitivity of 95.4% and specificity of 98.4%. The diagnostic accuracy was 98.5% for the small bowel and 98.1% for the colon |
| Tai et al., 2021 [16] | 93 | Multicentre, observational study | PCC was used to evaluate the extent and severity of CD. The feasibility, safety, and impact on patient outcomes were also examined. | In 85% of cases, the examination was complete, and the PCC resulted in change of clinical management in 38.7% of patients. The Montreal classification was upstaged in 33.8% of patients with established CD, and mucosal healing was demonstrated in 15.5%. In 12.7% of patients, PCC upstaged the small bowel disease and predicted escalation of treatment. |
| Volkers et al., 2022 [17] | 22 | Prospective, multicentre | PCE was used to measure changes in mucosal disease activity before and after (8–12 weeks) starting biologic treatment in CD patients. | Endoscopic remission (absence of ulcers) was observed in 6 of 22 (27%) patients. 3 of 22 patients (59%) responded endoscopically, (50% decrease in SES-CD and CDEIS scores compared to baseline). No adverse effects were observed. |
| Oliva et al., 2020 [18] | Prospective, multicentre | PCC was used to evaluate the extent and severity of paediatric IBD population. | At baseline, active inflammation was seen in 34 patients (71%), in 22 patients (46%) at week 24, and in 18 patients (39%) at week 52. PCC led to treatment change in 34 patients (71%) at baseline and 11 patients (23%) at 24 weeks. |
| Author (Year) | Bowel Prep | Dietary Instructions | Prokinetic Used | Boosters Used |
|---|---|---|---|---|
| Leigthton et al., 2017 [12] | 4 L PEG | Clear liquid diet for 24 h | Metoclopramide 10 mg (optional) | Suprep+ 2 L of water+ bisacodyl |
| Eliakim et al., 2018 [13] | 4 L PEG | Clear liquid diet for 24 h | Metoclopramide 10 mg (optional) | Suprep/Picosalax + bisacodyl |
| Adler et al., 2019 [14] | 3 L PEG | Low residue diet for 12 h, fasting for 12 h | Metoclopramide 10 mg (optional) | Half bottle (88 mL) of sodium sulphate, potassium sulphate, and magnesium sulphate solution |
| Bruining et al., 2020 [15] | 4 L PEG | Clear liquid diet for 24 h | Metoclopramide 10 mg or erythromycin 250 mg (optional) | Suprep |
| Tontini et al., 2020 [8] | 2 L PEG | Clear liquid diet for 24 h, fasting for 12 h | None | None |
| Eliakim et al., 2020 [10] | 4 L PEG | Clear liquid diet for 24 h | Metoclopramide 10 mg (optional) | Suprep/Picosalax + bisacodyl |
| Majter et al., 2021 [11] | 4 L PEG | Overnight fasting | None | None |
| Tai et al., 2021 [16] | NA | NA | NA | NA |
| Volkers et al., 2022 [17] | 3 L PEG (split dose) + 10 mg bisacodyl 2 days before the procedure | Low-fibre diet 2 days prior to test | None | Phosphoral (sodium phosphate) or picoprep (sodium picosulphate and magnesium citrate) with 1–2 L of clear fluid afterwards |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toskas, A.; Laskaratos, F.-M.; Coda, S.; Banerjee, S.; Epstein, O. Is Panenteric PillcamTM Crohn’s Capsule Endoscopy Ready for Widespread Use? A Narrative Review. Diagnostics 2023, 13, 2032. https://doi.org/10.3390/diagnostics13122032
Toskas A, Laskaratos F-M, Coda S, Banerjee S, Epstein O. Is Panenteric PillcamTM Crohn’s Capsule Endoscopy Ready for Widespread Use? A Narrative Review. Diagnostics. 2023; 13(12):2032. https://doi.org/10.3390/diagnostics13122032
Chicago/Turabian StyleToskas, Alexandros, Faidon-Marios Laskaratos, Sergio Coda, Saswata Banerjee, and Owen Epstein. 2023. "Is Panenteric PillcamTM Crohn’s Capsule Endoscopy Ready for Widespread Use? A Narrative Review" Diagnostics 13, no. 12: 2032. https://doi.org/10.3390/diagnostics13122032
APA StyleToskas, A., Laskaratos, F.-M., Coda, S., Banerjee, S., & Epstein, O. (2023). Is Panenteric PillcamTM Crohn’s Capsule Endoscopy Ready for Widespread Use? A Narrative Review. Diagnostics, 13(12), 2032. https://doi.org/10.3390/diagnostics13122032






