Artificial Intelligence-Based Software with CE Mark for Chest X-ray Interpretation: Opportunities and Challenges
Abstract
1. Introduction
2. Materials and Methods
3. Results
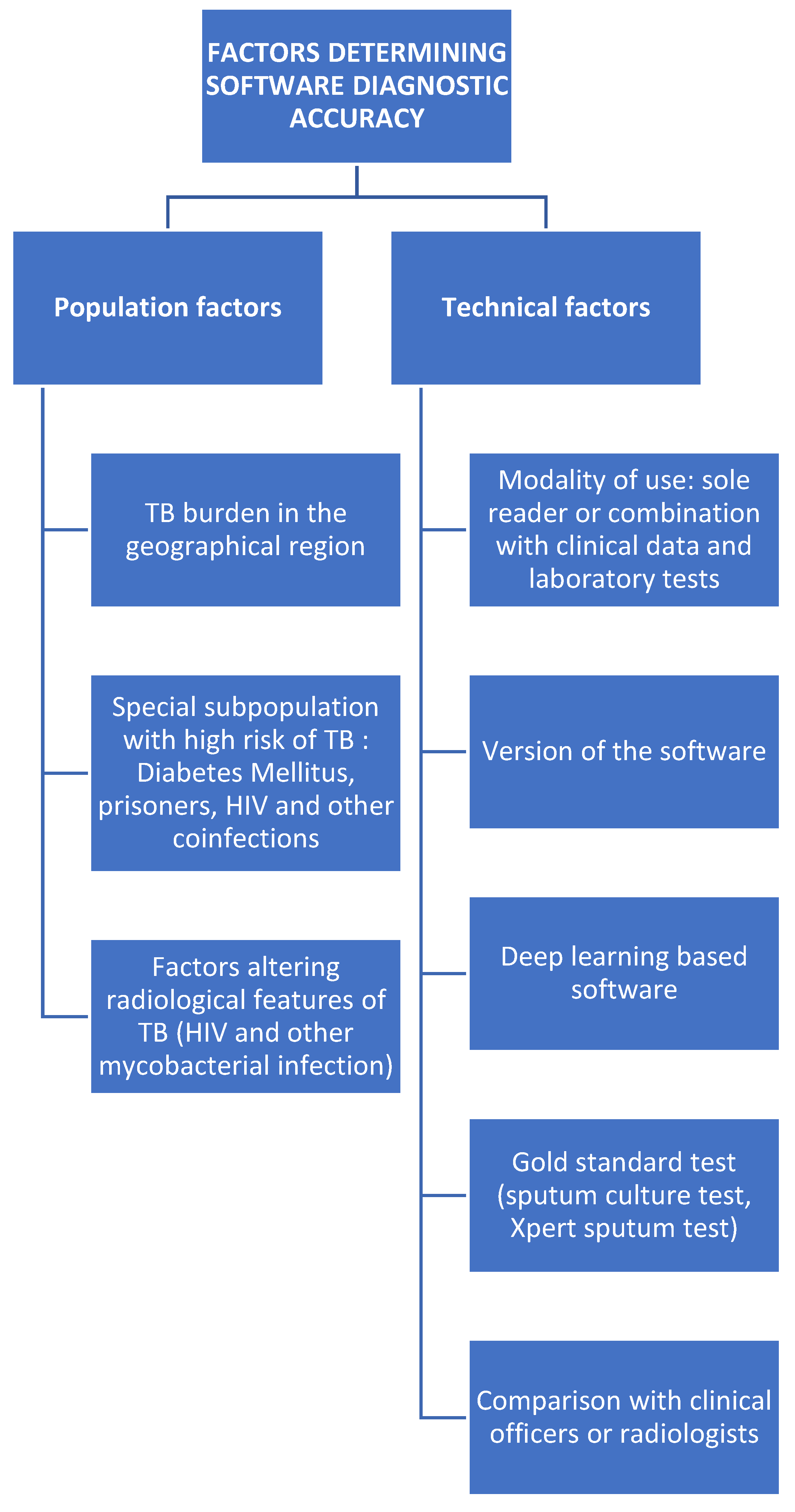
3.1. AI-Based Software for Tuberculosis-Related Abnormalities
3.2. AI-Based Software for Nodule and Lung Cancer Detection
3.3. AI-Based Software for Multiclass Findings and other Clinical Scenarios
3.4. Opportunities and Challenges
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Puddy, E.; Hill, C. Interpretation of the chest radiograph. Contin. Educ. Anaesth. Crit. Care Pain 2007, 7, 71–75. [Google Scholar] [CrossRef]
- Schalekamp, S.; Van Ginneken, B.; Karssemeijer, N.; Schaefer-Prokop, C.M. Chest Radiography: New Technological Developments and Their Applications. Semin. Respir. Crit. Care Med. 2014, 35, 3–16. [Google Scholar] [PubMed]
- Wright, F.W. Radiology of the Chest and Related Conditions: Together with an Extensive Illustrative Collection of Radiographs, Conventional and Computed Tomograms, Isotope Studies, MRs, etc.on CD-ROM. (Cross-Platform for Mac and Windows Version 3.1 or later), 1st ed.; CRC Press: London, UK, 2022. [Google Scholar]
- Jones, C.M.; Buchlak, Q.D.; Oakden-Rayner, L.; Milne, M.; Seah, J.; Esmaili, N.; Hachey, B. Chest radiographs and machine learning-Past, present and future. J. Med. Imaging Radiat. Oncol. 2021, 65, 538–544. [Google Scholar] [CrossRef]
- Bruls, R.J.M.; Kwee, R.M. Workload for radiologists during on-call hours: Dramatic increase in the past 15 years. Insights Imaging 2020, 11, 121. [Google Scholar] [CrossRef] [PubMed]
- Winder, M.; Owczarek, A.J.; Chudek, J.; Pilch-Kowalczyk, J.; Baron, J. Are We Overdoing It? Changes in Diagnostic Imaging Workload during the Years 2010–2020 including the Impact of the SARS-CoV-2 Pandemic. Healthcare 2021, 9, 1557. [Google Scholar] [CrossRef]
- Xiong, Z.; Stiles, M.K.; Yao, Y.; Shi, R.; Nalar, A.; Hawson, J.; Lee, G.; Zhao, J. Automatic 3D Surface Reconstruction of the Left Atrium From Clinically Mapped Point Clouds Using Convolutional Neural Networks. Front. Physiol. 2022, 13, 880260. [Google Scholar] [CrossRef] [PubMed]
- Commission Notice—The ‘Blue Guide’ on the implementation of EU products rules 2016 (Text with EEA relevance) C/2016/1958 OJ C 272, 26.7.2016, p. 1–149 (BG, ES, CS, DA, DE, ET, EL, EN, FR, HR, IT, LV, LT, HU, MT, NL, PL, PT, RO, SK, SL, FI, SV). Available online: https://eur-lex.europa.eu/legal-content/HU/TXT/?uri=uriserv%3AOJ.C_.2016.272.01.0001.01.ENG&toc=OJ%3AC%3A2016%3A272%3ATOC (accessed on 5 March 2023).
- Van Leeuwen, K.G.; Schalekamp, S.; Rutten, M.J.; van Ginneken, B.; de Rooij, M. Artificial intelligence in radiology: 100 commercially available products and their scientific evidence. Eur. Radiol. 2021, 31, 3797–3804. [Google Scholar] [CrossRef]
- Schwartz, W.S.; Moyer, R.E. The management of massive tuberculous pneumonia; a therapeutic review of seventy-five cases. Am. Rev. Tuberc. 1951, 64, 41–49. [Google Scholar]
- WHO. Chest Radiography Intuberculosis Detection: Summary of Current WHO Recommendations and Guidance on Programmatic Approaches; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Murphy, K.; Habib, S.S.; Zaidi, S.M.A.; Khowaja, S.; Khan, A.; Melendez, J.; Scholten, E.T.; Amad, F.; Schalekamp, S.; Verhagen, M.; et al. Computer aided detection of tuberculosis on chest radiographs: An evaluation of the CAD4TB v6 system. Sci. Rep. 2020, 10, 5492. [Google Scholar] [CrossRef]
- Qin, Z.Z.; Barrett, R.; Ahmed, S.; Sarker, M.S.; Paul, K.; Adel, A.S.S.; Banu, S.; Creswell, J. Comparing different versions of computer-aided detection products when reading chest X-rays for tuberculosis. PLoS Digit. Health 2022, 1, e0000067. [Google Scholar] [CrossRef]
- Khan, F.A.; Majidulla, A.; Tavaziva, G.; Nazish, A.; Abidi, S.K.; Benedetti, A.; Menzies, D.; Johnston, J.C.; Khan, A.J.; Saeed, S. Chest x-ray analysis with deep learning-based software as a triage test for pulmonary tuberculosis: A prospective study of diagnostic accuracy for culture-confirmed disease. Lancet Digit. Health 2020, 2, e573–e581. [Google Scholar] [CrossRef]
- Fehr, J.; Konigorski, S.; Olivier, S.; Gunda, R.; Surujdeen, A.; Gareta, D.; Smit, T.; Baisley, K.; Moodley, S.; Moosa, Y.; et al. Computer-aided interpretation of chest radiography reveals the spectrum of tuberculosis in rural South Africa. NPJ Digit. Med. 2021, 4, 106. [Google Scholar] [CrossRef]
- Koesoemadinata, R.C.; Kranzer, K.; Livia, R.; Susilawati, N.; Annisa, J.; Soetedjo, N.N.M.; Ruslami, R.; Philipsen, R.; van Ginneken, B.; Soetikno, R.D.; et al. Computer-assisted chest radiography reading for tuberculosis screening in people living with diabetes mellitus. Int. J. Tuberc. Lung Dis. 2018, 22, 1088–1094. [Google Scholar] [CrossRef]
- Wali, A.; Safdar, N.; Manair, R.; Khan, M.D.; Khan, A.; Kurd, S.A.; Khalil, L. Early TB case detection by community-based mobile X-ray screening and Xpert testing in Balochistan. Public Health Action 2021, 11, 174–179. [Google Scholar] [CrossRef]
- Odume, B.; Chukwu, E.; Fawole, T.; Nwokoye, N.; Ogbudebe, C.; Chukwuogo, O.; Useni, S.; Dim, C.; Ubochioma, E.; Nongo, D.; et al. Portable digital X-ray for TB pre-diagnosis screening in rural communities in Nigeria. Public Health Action 2022, 12, 85–89. [Google Scholar]
- Philipsen, R.H.H.M.; Sánchez, C.I.; Maduskar, P.; Melendez, J.; Peters-Bax, L.; Peter, J.G.; Dawson, R.; Theron, G.; Dheda, K.; van Ginneken, B. Automated chest-radiography as a triage for Xpert testing in resource-constrained settings: A prospective study of diagnostic accuracy and costs. Sci. Rep. 2015, 5, 12215. [Google Scholar] [CrossRef] [PubMed]
- Melendez, J.; Hogeweg, L.; Sánchez, C.I.; Philipsen, R.H.H.M.; Aldridge, R.; Hayward, A.C.; Abubakar, I.; van Ginneken, B.; Story, A. Accuracy of an automated system for tuberculosis detection on chest radiographs in high-risk screening. Int. J. Tuberc. Lung Dis. 2018, 22, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.Z.; Sander, M.S.; Rai, B.; Titahong, C.N.; Sudrungrot, S.; Laah, S.N.; Adhikari, L.M.; Carter, E.J.; Puri, L.; Codlin, A.J.; et al. Using artificial intelligence to read chest radiographs for tuberculosis detection: A multi-site evaluation of the diagnostic accuracy of three deep learning systems. Sci. Rep. 2019, 9, 15000. [Google Scholar] [CrossRef]
- Philipsen, R.H.H.M.; Sánchez, C.I.; Melendez, J.; Lew, W.J.; Van Ginneken, B. Automated chest X-ray reading for tuberculosis in the Philippines to improve case detection: A cohort study. Int. J. Tuberc. Lung Dis. 2019, 23, 805–810. [Google Scholar] [CrossRef]
- Steiner, A.; Mangu, C.; Hombergh, J.V.D.; van Deutekom, H.; van Ginneken, B.; Clowes, P.; Mhimbira, F.; Mfinanga, S.; Rachow, A.; Reither, K.; et al. Screening for pulmonary tuberculosis in a Tanzanian prison and computer-aided interpretation of chest X-rays. Public Health Action 2015, 5, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.D.S.; de Oliveira, R.D.; Lemos, E.F.; Lima, F.; Cohen, T.; Cords, O.; Martinez, L.; Gonçalves, C.; Ko, A.; Andrews, J.R.; et al. Yield, Efficiency, and Costs of Mass Screening Algorithms for Tuberculosis in Brazilian Prisons. Clin. Infect. Dis. 2021, 72, 771–777. [Google Scholar] [CrossRef] [PubMed]
- Wali, A.; Khan, D.; Safdar, N.; Shawani, Z.; Fatima, R.; Yaqoob, A.; Qadir, A.; Ahmed, S.; Rashid, H.; Ahmed, B.; et al. Prevalence of tuberculosis, HIV/AIDS, and hepatitis; in a prison of Balochistan: A cross-sectional survey. BMC Public Health 2019, 19, 1631. [Google Scholar] [CrossRef] [PubMed]
- Breuninger, M.; Van Ginneken, B.; Philipsen, R.H.H.M.; Mhimbira, F.; Hella, J.J.; Lwilla, F.; Hombergh, J.V.D.; Ross, A.; Jugheli, L.; Wagner, D.; et al. Diagnostic Accuracy of Computer-Aided Detection of Pulmonary Tuberculosis in Chest Radiographs: A Validation Study from Sub-Saharan Africa. PLoS ONE 2014, 9, e106381. [Google Scholar] [CrossRef]
- Qin, Z.Z.; Ahmed, S.; Sarker, M.S.; Paul, K.; Adel, A.S.S.; Naheyan, T.; Barrett, R.; Banu, S.; Creswell, J. Tuberculosis detection from chest x-rays for triaging in a high tuberculosis-burden setting: An evaluation of five artificial intelligence algorithms. Lancet Digit. Health 2021, 3, e543–e554. [Google Scholar] [CrossRef]
- Nishtar, T.; Burki, S.; Ahmad, F.S.; Ahmad, T. Diagnostic accuracy of computer aided reading of chest x-ray in screening for pulmonary tuberculosis in comparison with Gene-Xpert. Pak. J. Med Sci. 2021, 38, 62–68. [Google Scholar] [CrossRef]
- Maduskar, P.; Muyoyeta, M.; Ayles, H.; Hogeweg, L.; Peters-Bax, L.; Van Ginneken, B. Detection of tuberculosis using digital chest radiography: Automated reading vs. interpretation by clinical officers. Int. J. Tuberc. Lung Dis. 2013, 17, 1613–1620. [Google Scholar]
- Melendez, J.; Philipsen, R.H.H.M.; Chanda-Kapata, P.; Sunkutu, V.; Kapata, N.; Van Ginneken, B. Automatic versus human reading of chest X-rays in the Zambia National Tuberculosis Prevalence Survey. Int. J. Tuberc. Lung Dis. 2017, 21, 880–886. [Google Scholar] [CrossRef] [PubMed]
- Del Ciello, A.; Franchi, P.; Contegiacomo, A.; Cicchetti, G.; Bonomo, L.; Larici, A.R. Missed lung cancer: When, where, and why? Diagn. Interv. Radiol. 2017, 23, 118–126. [Google Scholar] [CrossRef]
- Bradley, S.H.; Bhartia, B.S.; Callister, M.E.; Hamilton, W.T.; Hatton, N.L.F.; Kennedy, M.P.; Mounce, L.T.; Shinkins, B.; Wheatstone, P.; Neal, R.D. Chest X-ray sensitivity and lung cancer outcomes: A retrospective observational study. Br. J. Gen. Pract. 2021, 71, e862–e868. [Google Scholar] [CrossRef]
- Gampala, S.; Vankeshwaram, V.; Gadula, S.S.P. Is Artificial Intelligence the New Friend for Radiologists? A Review Article. Cureus 2020, 12, e11137. [Google Scholar] [CrossRef]
- Nam, J.G.; Park, S.; Hwang, E.J.; Lee, J.H.; Jin, K.-N.; Lim, K.Y.; Vu, T.H.; Sohn, J.H.; Hwang, S.; Goo, J.M.; et al. Development and Validation of Deep Learning–based Automatic Detection Algorithm for Malignant Pulmonary Nodules on Chest Radiographs. Radiology 2019, 290, 218–228. [Google Scholar] [CrossRef] [PubMed]
- Sim, Y.; Chung, M.J.; Kotter, E.; Yune, S.; Kim, M.; Do, S.; Han, K.; Kim, H.; Yang, S.; Lee, D.-J.; et al. Deep Convolutional Neural Network–based Software Improves Radiologist Detection of Malignant Lung Nodules on Chest Radiographs. Radiology 2020, 294, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Homayounieh, F.; Digumarthy, S.; Ebrahimian, S.; Rueckel, J.; Hoppe, B.F.; Sabel, B.O.; Conjeti, S.; Ridder, K.; Sistermanns, M.; Wang, L.; et al. An Artificial Intelligence–Based Chest X-ray Model on Human Nodule Detection Accuracy From a Multicenter Study. JAMA Netw. Open 2021, 4, e2141096. [Google Scholar] [CrossRef]
- Kim, H.; Park, C.M.; Goo, J.M. Test-retest reproducibility of a deep learning–based automatic detection algorithm for the chest radiograph. Eur. Radiol. 2020, 30, 2346–2355. [Google Scholar] [CrossRef]
- Kim, E.Y.; Kim, Y.J.; Choi, W.J.; Lee, G.P.; Choi, Y.R.; Jin, K.N.; Cho, Y.J. Performance of a deep-learning algorithm for referable thoracic abnormalities on chest radiographs: A multicenter study of a health screening cohort. PLoS ONE 2021, 16, e0246472. [Google Scholar]
- Liang, C.H.; Liu, Y.C.; Wu, M.T.; Garcia-Castro, F.; Alberich-Bayarri, A.; Wu, F.Z. Identifying pulmonary nodules or masses on chest radiography using deep learning: External validation and strategies to improve clinical practice. Clin. Radiol. 2020, 75, 38–45. [Google Scholar] [CrossRef]
- Teng, P.H.; Liang, C.H.; Lin, Y.; Alberich-Bayarri, A.; González, R.L.; Li, P.W.; Weng, Y.H.; Chen, Y.T.; Lin, C.H.; Chou, K.J.; et al. Performance and educational training of radiographers in lung nodule or mass detection: Retrospective comparison with different deep learning algorithms. Medicine 2021, 100, e26270. [Google Scholar] [CrossRef] [PubMed]
- Tam, M.; Dyer, T.; Dissez, G.; Morgan, T.N.; Hughes, M.; Illes, J.; Rasalingham, R. Augmenting lung cancer diagnosis on chest radiographs: Positioning artificial intelligence to improve radiologist performance. Clin. Radiol. 2021, 76, 607–614. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.K.; Austin, J.H.M.; White, C.S.; Patel, P.; Haramati, L.B.; Pearson, G.D.N.; Shiau, M.C.; Berkmen, Y.M. Missed Non–Small Cell Lung Cancer: Radiographic Findings of Potentially Resectable Lesions Evident Only in Retrospect. Radiology 2003, 226, 235–241. [Google Scholar] [CrossRef]
- Schalekamp, S.; van Ginneken, B.; Meiss, L.; Peters-Bax, L.; Quekel, L.G.; Snoeren, M.M.; Tiehuis, A.M.; Wittenberg, R.; Karssemeijer, N.; Schaefer-Prokop, C.M. Bone suppressed images improve radiologists’ detection performance for pulmonary nodules in chest radiographs. Eur. J. Radiol. 2013, 82, 2399–2405. [Google Scholar] [CrossRef]
- Schalekamp, S.; Karssemeijer, N.; Cats, A.M.; De Hoop, B.; Geurts, B.H.; Berger-Hartog, O.; van Ginneken, B.; Schaefer-Prokop, C.M. The Effect of Supplementary Bone-Suppressed Chest Radiographs on the Assessment of a Variety of Common Pulmonary Abnormalities: Results of an Observer Study. J. Thorac. Imaging 2016, 31, 119–125. [Google Scholar] [CrossRef]
- Freedman, M.T.; Lo, S.-C.B.; Seibel, J.C.; Bromley, C.M. Lung Nodules: Improved Detection with Software That Suppresses the Rib and Clavicle on Chest Radiographs. Radiology 2011, 260, 265–273. [Google Scholar] [CrossRef]
- Schalekamp, S.; Van Ginneken, B.; Koedam, E.; Snoeren, M.M.; Tiehuis, A.M.; Wittenberg, R.; Karssemeijer, N.; Schaefer-Prokop, C.M. Computer-aided Detection Improves Detection of Pulmonary Nodules in Chest Radiographs beyond the Support by Bone-suppressed Images. Radiology 2014, 272, 252–261. [Google Scholar] [CrossRef] [PubMed]
- Schalekamp, S.; Van Ginneken, B.; Heggelman, B.; Imhof-Tas, M.; Somers, I.; Brink, M.; Spee, M.; Schaefer-Prokop, C.; Karssemeijer, N. New methods for using computer-aided detection information for the detection of lung nodules on chest radiographs. Br. J. Radiol. 2014, 87, 20140015. [Google Scholar] [CrossRef]
- Park, S.; Lee, S.M.; Lee, K.H.; Jung, K.-H.; Bae, W.; Choe, J.; Seo, J.B. Deep learning-based detection system for multiclass lesions on chest radiographs: Comparison with observer readings. Eur. Radiol. 2019, 30, 1359–1368. [Google Scholar] [CrossRef]
- Wisbach, G.G.; Sise, M.J.; Sack, D.I.; Swanson, S.M.; Sundquist, S.M.; Paci, G.M.; Kingdon, K.M.; Kaminski, S.S. What is the role of chest X-ray in the initial assessment of stable trauma patients? J. Trauma 2007, 62, 74–78; discussion 78–79. [Google Scholar] [CrossRef] [PubMed]
- Gipson, J.; Tang, V.; Seah, J.; Kavnoudias, H.; Zia, A.; Lee, R.; Mitra, B.; Clements, W. Diagnostic accuracy of a commercially available deep-learning algorithm in supine chest radiographs following trauma. Br. J. Radiol. 2022, 95, 20210979. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.J.; Nam, J.G.; Lim, W.H.; Park, S.J.; Jeong, Y.S.; Kang, J.H.; Hong, E.K.; Kim, T.M.; Goo, J.M.; Park, S.; et al. Deep Learning for Chest Radiograph Diagnosis in the Emergency Department. Radiology 2019, 293, 573–580. [Google Scholar] [CrossRef]
- Jones, C.M.; Danaher, L.; Milne, M.R.; Tang, C.; Seah, J.; Oakden-Rayner, L.; Johnson, A.; Buchlak, Q.D.; Esmaili, N. Assessment of the effect of a comprehensive chest radiograph deep learning model on radiologist reports and patient outcomes: A real-world observational study. BMJ Open 2021, 11, e052902. [Google Scholar] [CrossRef]
- Shin, H.J.; Son, N.-H.; Kim, M.J.; Kim, E.-K. Diagnostic performance of artificial intelligence approved for adults for the interpretation of pediatric chest radiographs. Sci. Rep. 2022, 12, 10215. [Google Scholar] [CrossRef]
- Kim, E.Y.; Kim, Y.J.; Choi, W.-J.; Jeon, J.S.; Kim, M.Y.; Oh, D.H.; Jin, K.N.; Cho, Y.J. Concordance rate of radiologists and a commercialized deep-learning solution for chest X-ray: Real-world experience with a multicenter health screening cohort. PLoS ONE 2022, 17, e0264383. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Wang, D.; Tong, X.; Liu, T.; Zhang, S.; Huang, J.; Zhang, L.; Chen, L.; Fan, H.; et al. Artificial Intelligence for COVID-19: A Systematic Review. Front. Med. 2021, 8, 704256. [Google Scholar] [CrossRef]
- Khan, M.; Mehran, M.T.; Haq, Z.U.; Ullah, Z.; Naqvi, S.R.; Ihsan, M.; Abbass, H. Applications of artificial intelligence in COVID-19 pandemic: A comprehensive review. Expert Syst. Appl. 2021, 185, 115695. [Google Scholar] [CrossRef] [PubMed]
- Mushtaq, J.; Pennella, R.; Lavalle, S.; Colarieti, A.; Steidler, S.; Martinenghi, C.M.A.; Palumbo, D.; Esposito, A.; Rovere-Querini, P.; Tresoldi, M.; et al. Initial chest radiographs and artificial intelligence (AI) predict clinical outcomes in COVID-19 patients: Analysis of 697 Italian patients. Eur. Radiol. 2020, 31, 1770–1779. [Google Scholar] [CrossRef]
- Tovar, M.A.; Puma, D.; Palomino, S.; Peinado, J.; Llanos, F.; Martinelli, C.; Jimenez, J.; Calderon, R.; Yuen, C.M.; Lecca, L. Integrated screening and testing for TB and COVID-19 in Peru. Public Health Action 2022, 12, 7–9. [Google Scholar] [CrossRef] [PubMed]
- Schalekamp, S.; Van Ginneken, B.; Berk, I.A.H.V.D.; Hartmann, I.J.C.; Snoeren, M.M.; Odink, A.E.; Van Lankeren, W.; Pegge, S.A.H.; Schijf, L.J.; Karssemeijer, N.; et al. Bone Suppression Increases the Visibility of Invasive Pulmonary Aspergillosis in Chest Radiographs. PLoS ONE 2014, 9, e108551. [Google Scholar] [CrossRef] [PubMed]
- Fanni, S.C.; Shalaby, S.M.; Neri, E. The Role of Artificial Intelligence (AI) in the Management of Geriatric Patients. In Imaging in Geriatrics; Guglielmi, G., Maas, M., Eds.; Springer International Publishing: Cham, Switzerland, 2023; pp. 445–456. [Google Scholar]
- Coppola, F.; Faggioni, L.; Gabelloni, M.; De Vietro, F.; Mendola, V.; Cattabriga, A.; Cocozza, M.A.; Vara, G.; Piccinino, A.; Lo Monaco, S.; et al. Human, All Too Human? An All-Around Appraisal of the "Artificial Intelligence Revolution" in Medical Imaging. Front. Psychol. 2021, 12, 710982. [Google Scholar]
- Daneshjou, R.; Smith, M.P.; Sun, M.D.; Rotemberg, V.; Zou, J. Lack of Transparency and Potential Bias in Artificial Intelligence Data Sets and Algorithms: A Scoping Review. JAMA Derm. 2021, 157, 1362–1369. [Google Scholar] [CrossRef]
- Erondu, O.F. Challenges and Peculiarities of Paediatric Imaging, in Medical Imaging in Clinical Practice; IntechOpen: London, UK, 2013. [Google Scholar]
| Software Name | Company | Disease Targeted | Population | Processing Time |
|---|---|---|---|---|
| ClearRead XRay | Riverain Technologies | Lung cancer | Adult, not pediatric | 1–10 min |
| ChestLink | Oxipit | 75 different pathologies | Older than 18 years old | 10–60 s |
| CAD4TB | Thirona | Tuberculosis | All chest X-rays | 3–10 s |
| AI-Rad Companion Chest X-ray | Siemens Healthineers | Pulmonary lesions, pleural effusion, pneumothorax, consolidation, atelectasis | All upright chest X-rays | 1–10 min |
| SenseCare-Chest DR Pro | SenseTime | Pneumonia, tuberculosis, pneumothorax, pleural effusion, cardiomegaly, rib fractures | All chest X-rays | 3–10 s |
| Annalise Enterprise CXR | annalise.ai | 124 findings present in the emergent, urgent, and non-urgent care settings | Patients over 16 years of age | 3–10 s |
| Chest | MSK AI | Arterys | Fracture, dislocation, elbow joint effusion, pleural effusion, pulmonary nodule, pulmonary opacity, pneumothorax | Emergency department population | Not available |
| Chest X-ray Classifier | Quibim | 14 different findings | All chest X-rays | 10–60 s |
| qXR | Qure.ai | Tuberculosis, COVID-19, signs seen in Lung Parenchyma, Pleura, Mediastinum, Cardiac and bones | All chest X-rays | 10–60 s |
| TIRESYA | Digitec | Unspecified | Unspecified | Not available |
| Milvue Suite | Milvue | Bone fracture, pleural effusion, lung opacity, elbow joint effusion, lung nodule, pneumothorax, dislocation | All chest X-rays | 10–60 s |
| CheXVision | XVision | 17 pathologies | All chest X-rays of patients above 16 years old | 3–10 s |
| CAD4COVID-XRay | Thirona | COVID-19 | All chest X-rays | 3–10 s |
| Chest Solution | Nanox.AI | Pneumothorax, pleural effusion | Unspecified | Not available |
| VUNO Med®-Chest X-ray™ | VUNO | Nodule/Mass, Consolidation, Interstitial Opacity, Pneumothorax, Pleural Effusion | All chest X-rays | <3 s |
| InferRead DR Tuberculosis | Infervision | Tuberculosis | Unspecified | Not available |
| InferRead DR Chest | Infervision | Lung cancer, pneumothorax, fracture, tuberculosis, lung infection, aortic calcification, cord imaging, heart shadow enlargement, pleural effusion. | Any | Not available |
| X1 | Visionairy Health | 12 different findings | Unspecified | Not available |
| Auto Lung Nodule Detection | Samsung Electronics | Lung cancer | Unspecified | <3 s |
| Pneumothorax (Ptx) | Aidoc | Pneumothorax | Radiograph | 1–10 min |
| Red Dot | Behold.ai | Pneumothorax | Unspecified | Not available |
| JLD-02K | JLK Inc. | Lung cancer | All Chest X-rays | 3–10 s |
| Lunit INSIGHT CXR | Lunit | 11 different findings | Patients aged 14 years or older | 3–10 s |
| Critical Care Suite | GE Healthcare | Pneumothorax | Adult-size patients. Suspected of pneumothorax or intubated. | <3 s |
| ChestView | GLEAMER | Pneumothorax, pleural effusion, alveolar syndrome, nodule, mediastinal mass | All Chest X-rays | 10–60 s |
| ChestEye Quality | Oxipit | 75 different pathologies | Patients over 18 years old | 3–10 s |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fanni, S.C.; Marcucci, A.; Volpi, F.; Valentino, S.; Neri, E.; Romei, C. Artificial Intelligence-Based Software with CE Mark for Chest X-ray Interpretation: Opportunities and Challenges. Diagnostics 2023, 13, 2020. https://doi.org/10.3390/diagnostics13122020
Fanni SC, Marcucci A, Volpi F, Valentino S, Neri E, Romei C. Artificial Intelligence-Based Software with CE Mark for Chest X-ray Interpretation: Opportunities and Challenges. Diagnostics. 2023; 13(12):2020. https://doi.org/10.3390/diagnostics13122020
Chicago/Turabian StyleFanni, Salvatore Claudio, Alessandro Marcucci, Federica Volpi, Salvatore Valentino, Emanuele Neri, and Chiara Romei. 2023. "Artificial Intelligence-Based Software with CE Mark for Chest X-ray Interpretation: Opportunities and Challenges" Diagnostics 13, no. 12: 2020. https://doi.org/10.3390/diagnostics13122020
APA StyleFanni, S. C., Marcucci, A., Volpi, F., Valentino, S., Neri, E., & Romei, C. (2023). Artificial Intelligence-Based Software with CE Mark for Chest X-ray Interpretation: Opportunities and Challenges. Diagnostics, 13(12), 2020. https://doi.org/10.3390/diagnostics13122020









