Assessing the Effectiveness of Artificial Intelligence Models for Detecting Alveolar Bone Loss in Periodontal Disease: A Panoramic Radiograph Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Sources
2.3. Labeling and Training of Data
- Training group: 80% of the images (represents the data set used for training the model.)
- Validation group: 10% of the images (indicates the samples that are independent of the training of the model and should not be seen by the model during this period. The model is tested on this dataset to stop training or revise training variables.)
- Test group: 10% of the images (constitutes the data set in which the trained model is tested by using the training and validation data.)
2.4. Deep-Learning Algorithm
2.5. Statistical Analysis
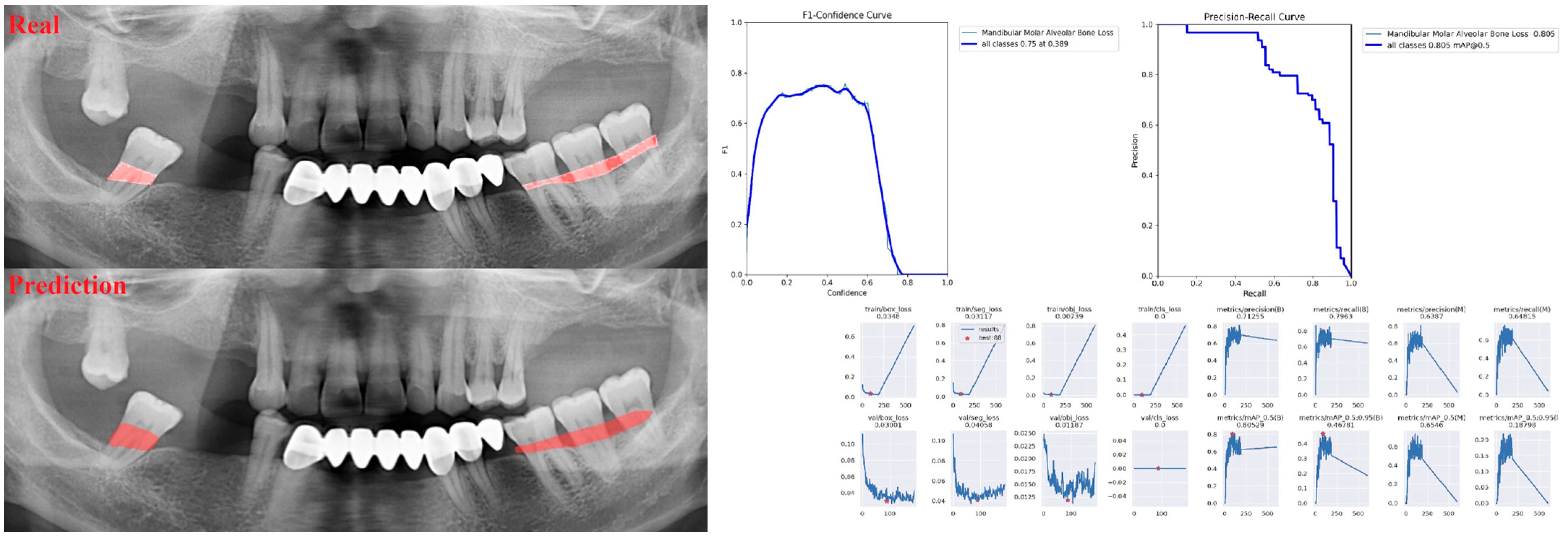
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Horner, K.; Drage, N.; Brettle, D. The Historical Perspective. In Twenty-First Century Imaging; Nairn, H.F., Ed.; Quintessence Publishing Co., Ltd.: Batavia, IL, USA, 2008; pp. 1–11. [Google Scholar]
- Ludlow, J.B.; Mol, A. Digital imaging. In Oral Radiology Principles and Interpretation; White, S.C., Ed.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 78–99. [Google Scholar]
- White, S.C.; Pharoah, M.J.; Frederiksen, N.L. Advanced imaging. In Oral Radiology Principles and Interpretation, 6th ed.; White, S.C., Ed.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 207–224. [Google Scholar]
- Kwon, T.; Lamster, I.B.; Levin, L. Current Concepts in the Management of Periodontitis. Int. Dent. J. 2020, 71, 462–476. [Google Scholar] [CrossRef] [PubMed]
- Lei, F.; Li, M.; Lin, T.; Zhou, H.; Wang, F.; Su, X. Treatment of inflammatory bone loss in periodontitis by stem cell-derived exosomes. Acta Biomater. 2022, 141, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Bui, F.Q.; Almeida-Da-Silva, C.L.C.; Huynh, B.; Trinh, A.; Liu, J.; Woodward, J.; Asadi, H.; Ojcius, D.M. Association between periodontal pathogens and systemic disease. Biomed. J. 2019, 42, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Alotaibi, G.; Awawdeh, M.; Farook, F.F.; Aljohani, M.; Aldhafiri, R.M.; Aldhoayan, M. Artificial intelligence (AI) diagnostic tools: Utilizing a convolutional neural network (CNN) to assess periodontal bone level radiographically—A retrospective study. BMC Oral Health 2022, 22, 399. [Google Scholar] [CrossRef]
- Leite, A.F.; Van Gerven, A.; Willems, H.; Beznik, T.; Lahoud, P.; Gaêta-Araujo, H.; Vranckx, M.; Jacobs, R. Artificial intelligence-driven novel tool for tooth detection and segmentation on panoramic radiographs. Clin. Oral Investig. 2020, 25, 2257–2267. [Google Scholar] [CrossRef]
- Jiang, L.; Chen, D.; Cao, Z.; Wu, F.; Zhu, H.; Zhu, F. A two-stage deep learning architecture for radiographic staging of periodontal bone loss. BMC Oral Health 2022, 22, 106. [Google Scholar] [CrossRef]
- Sen, D.; Chakrabarti, R.; Chatterjee, S.; Grewal, D.S.; Manrai, K. Artificial intelligence and the radiologist: The future in the Armed Forces Medical Services. BMJ Mil. Health 2019, 166, 254–256. [Google Scholar] [CrossRef]
- European Society of Radiology (ESR). What the radiologist should know about artificial intelligence—An ESR white paper. Insights Imaging 2019, 10, 44. [Google Scholar] [CrossRef]
- McCarthy, J.; Minsky, M.L.; Rochester, N.; Shannon, C.E. A Proposal for the Dartmouth Summer Research Project on Artificial Intelligence, August 31, 1955. AI Mag. 2006, 27, 12–14. [Google Scholar]
- Chartrand, G.; Cheng, P.M.; Vorontsov, E.; Drozdzal, M.; Turcotte, S.; Pal, C.J.; Kadoury, S.; Tang, A. Deep Learning: A Primer for Radiologists. RadioGraphics 2017, 37, 2113–2131. [Google Scholar] [CrossRef]
- King, B.F. Guest Editorial: Discovery and Artificial Intelligence. Am. J. Roentgenol. 2017, 209, 1189–1190. [Google Scholar] [CrossRef]
- Lakhani, P.; Prater, A.B.; Hutson, R.K.; Andriole, K.P.; Dreyer, K.J.; Morey, J.; Prevedello, L.M.; Clark, T.J.; Geis, J.R.; Itri, J.N.; et al. Machine Learning in Radiology: Applications Beyond Image Interpretation. J. Am. Coll. Radiol. 2018, 15, 350–359. [Google Scholar] [CrossRef]
- Krittanawong, C. The rise of artificial intelligence and the uncertain future for physicians. Eur. J. Intern. Med. 2017, 48, e13–e14. [Google Scholar] [CrossRef]
- Hosny, A.; Parmar, C.; Quackenbush, J.; Schwartz, L.H.; Aerts, H.J.W.L. Artificial intelligence in radiology. Nat. Rev. Cancer 2018, 18, 500–510. [Google Scholar] [CrossRef]
- Duong, M.T.; Rauschecker, A.M.; Rudie, J.D.; Chen, P.-H.; Cook, T.S.; Bryan, R.N.; Mohan, S. Artificial intelligence for precision education in radiology. Br. J. Radiol. 2019, 92, 20190389. [Google Scholar] [CrossRef]
- Heo, M.-S.; Kim, J.-E.; Hwang, J.-J.; Han, S.-S.; Kim, J.-S.; Yi, W.-J.; Park, I.-W. Artificial intelligence in oral and maxillofacial radiology: What is currently possible? Dentomaxillofac. Radiol. 2021, 50, 20200375. [Google Scholar] [CrossRef]
- Giudice, A.L.; Ronsivalle, V.; Spampinato, C.; Leonardi, R. Fully automatic segmentation of the mandible based on convolutional neural networks (CNNs). Orthod. Craniofacial Res. 2021, 24, 100–107. [Google Scholar] [CrossRef]
- Leonardi, R.; Giudice, A.L.; Farronato, M.; Ronsivalle, V.; Allegrini, S.; Musumeci, G.; Spampinato, C. Fully automatic segmentation of sinonasal cavity and pharyngeal airway based on convolutional neural networks. Am. J. Orthod. Dentofac. Orthop. 2021, 159, 824–835.e1. [Google Scholar] [CrossRef]
- Harvey, S.; Patel, S. Guidelines and template for reporting on CBCT scans. Br. Dent. J. 2020, 228, 15–18. [Google Scholar] [CrossRef]
- Eshraghi, T.; McAllister, N.; McAllister, B. Clinical Applications of Digital 2-D and 3-D Radiography for the Periodontist. J. Evid. Based Dent. Pract. 2012, 12, 36–45. [Google Scholar] [CrossRef]
- Bochkovskiy, A.; Wang, C.Y.; Liao, H.Y.M. YOLOv4: Optimal Speed and Accuracy of Object Detection. arXiv 2020, arXiv:200410934v1. [Google Scholar]
- Xu, R.; Lin, H.; Lu, K.; Cao, L.; Liu, Y. A Forest Fire Detection System Based on Ensemble Learning. Forests 2021, 12, 217. [Google Scholar] [CrossRef]
- Wang, C.Y.; Liao, H.Y.M.; Wu, Y.H.; Chen, P.Y.; Hsieh, J.W.; Yeh, I.H. CSPNet: A New Backbone that can Enhance Learning Capability of CNN. In Proceedings of the IEEE/CVF Conference on Computer Vision and Pattern Recognition Workshops (CVPRW), Seattle, WA, USA, 14–19 June 2020; pp. 390–391. [Google Scholar]
- Jones, O.T.; Matin, R.N.; van der Schaar, M.; Bhayankaram, K.P.; Ranmuthu, C.K.; Islam, M.S.; Behiyat, D.; Boscott, R.; Calanzani, N.; Emery, J.; et al. Artificial intelligence and machine learning algorithms for early detection of skin cancer in community and primary care settings: A systematic review. Lancet Digit. Health 2022, 4, e466–e476. [Google Scholar] [CrossRef] [PubMed]
- Revilla-León, M.; Gómez-Polo, M.; Vyas, S.; Barmak, B.A.; Galluci, G.O.; Att, W.; Krishnamurthy, V.R. Artificial intelligence applications in implant dentistry: A systematic review. J. Prosthet. Dent. 2021, 129, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Schwendicke, F.; Chaurasia, A.; Arsiwala, L.; Lee, J.-H.; Elhennawy, K.; Jost-Brinkmann, P.-G.; Demarco, F.; Krois, J. Deep learning for cephalometric landmark detection: Systematic review and meta-analysis. Clin. Oral Investig. 2021, 25, 4299–4309. [Google Scholar] [CrossRef]
- Abdalla-Aslan, R.; Yeshua, T.; Kabla, D.; Leichter, I.; Nadler, C. An artificial intelligence system using machine-learning for automatic detection and classification of dental restorations in panoramic radiography. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2020, 130, 593–602. [Google Scholar] [CrossRef]
- Chen, Y.W.; Stanley, K.; Att, W. Artificial intelligence in dentistry: Current applications and future perspectives. Quintessence Int. 2020, 51, 248–257. [Google Scholar] [CrossRef]
- Kong, Z.; Ouyang, H.; Cao, Y.; Huang, T.; Ahn, E.; Zhang, M.; Liu, H. Automated periodontitis bone loss diagnosis in panoramic radiographs using a bespoke two-stage detector. Comput. Biol. Med. 2023, 152, 106374. [Google Scholar] [CrossRef]
- Berghuis, G.; Cosyn, J.; De Bruyn, H.; Hommez, G.; Dierens, M.; Christiaens, V. A controlled study on the diagnostic accuracy of panoramic and peri-apical radiography for detecting furcation involvement. BMC Oral. Health 2021, 21, 115. [Google Scholar] [CrossRef]
- Papapanou, P.N.; Tonetti, M.S. Diagnosis and epidemiology of periodontal osseous lesions. Periodontology 2000 2000, 22, 8–21. [Google Scholar] [CrossRef]
- Masthoff, M.; Gerwing, M.; Masthoff, M.; Timme, M.; Kleinheinz, J.; Berninger, M.; Heindel, W.; Wildgruber, M.; Schülke, C. Dental Imaging—A basic guide for the radiologist. RöFo Fortschr. Auf Geb. Röntgenstrahlen Bildgeb. Verfahr. 2019, 191, 192–198. [Google Scholar] [CrossRef]
- Li, H.; Zhou, J.; Zhou, Y.; Chen, Q.; She, Y.; Gao, F.; Xu, Y.; Chen, J.; Gao, X. An Interpretable Computer-Aided Diagnosis Method for Periodontitis from Panoramic Radiographs. Front. Physiol. 2021, 12, 556. [Google Scholar] [CrossRef]
- Stavropoulos, A.; Bertl, K.; Spineli, L.M.; Sculean, A.; Cortellini, P.; Tonetti, M. Medium- and long-term clinical benefits of periodontal regenerative/reconstructive procedures in intrabony defects: Systematic review and network meta-analysis of randomized controlled clinical studies. J. Clin. Periodontol. 2020, 48, 410–430. [Google Scholar] [CrossRef]
- Chang, H.-J.; Lee, S.-J.; Yong, T.-H.; Shin, N.-Y.; Jang, B.-G.; Kim, J.-E.; Huh, K.-H.; Lee, S.-S.; Heo, M.-S.; Choi, S.-C.; et al. Deep Learning Hybrid Method to Automatically Diagnose Periodontal Bone Loss and Stage Periodontitis. Sci. Rep. 2020, 10, 7531. [Google Scholar] [CrossRef]
- Preshaw, P.M. Detection and diagnosis of periodontal conditions amenable to prevention. BMC Oral Health 2015, 15, S5. [Google Scholar] [CrossRef]
- Tsoromokos, N.; Parinussa, S.; Claessen, F.; Moin, D.A.; Loos, B.G. Estimation of Alveolar Bone Loss in Periodontitis Using Machine Learning. Int. Dent. J. 2022, 72, 621–627. [Google Scholar] [CrossRef]
- Lee, C.; Kabir, T.; Nelson, J.; Sheng, S.; Meng, H.; Van Dyke, T.E.; Walji, M.F.; Jiang, X.; Shams, S. Use of the deep learning approach to measure alveolar bone level. J. Clin. Periodontol. 2021, 49, 260–269. [Google Scholar] [CrossRef]
- Moran, M.B.H.; Faria, M.; Giraldi, G.; Bastos, L.; Inacio, B.D.S.; Conci, A. On using convolutional neural networks to classify periodontal bone destruction in periapical radiographs. In Proceedings of the IEEE International Conference on Bioinformatics and Biomedicine (BIBM), Seoul, Republic of Korea, 16–19 December 2020; pp. 2036–2039. [Google Scholar] [CrossRef]
- Lee, J.-H.; Kim, D.-H.; Jeong, S.-N.; Choi, S.-H. Diagnosis and prediction of periodontally compromised teeth using a deep learning-based convolutional neural network algorithm. J. Periodontal Implant Sci. 2018, 48, 114–123. [Google Scholar] [CrossRef]
- Krois, J.; Ekert, T.; Meinhold, L.; Golla, T.; Kharbot, B.; Wittemeier, A.; Dörfer, C.; Schwendicke, F. Deep Learning for the Radiographic Detection of Periodontal Bone Loss. Sci. Rep. 2019, 9, 8495. [Google Scholar] [CrossRef]
- Thanathornwong, B.; Suebnukarn, S. Automatic detection of periodontal compromised teeth in digital panoramic radiographs using faster regional convolutional neural networks. Imaging Sci. Dent. 2020, 50, 169–174. [Google Scholar] [CrossRef]
- Chang, J.; Chang, M.-F.; Angelov, N.; Hsu, C.-Y.; Meng, H.-W.; Sheng, S.; Glick, A.; Chang, K.; He, Y.-R.; Lin, Y.-B.; et al. Application of deep machine learning for the radiographic diagnosis of periodontitis. Clin. Oral Investig. 2022, 26, 6629–6637. [Google Scholar] [CrossRef] [PubMed]
- Kurt, S.; Çelίk, Ö.; Bayrakdar, I.; Orhan, K.; Bίlgίr, E.; Odabas, A.; Aslan, A.F. Determination Alveolar Bone Loss Using Artificial Intelligence System on Den-tal Panoramic Radiography. Cumhur. Dent. J. 2020, 23, 318–324. [Google Scholar] [CrossRef]
- Kim, J.; Lee, H.-S.; Song, I.-S.; Jung, K.-H. DeNTNet: Deep Neural Transfer Network for the detection of periodontal bone loss using panoramic dental radiographs. Sci. Rep. 2019, 9, 17615. [Google Scholar] [CrossRef] [PubMed]
- Helmi, M.F.; Huang, H.; Goodson, J.M.; Hasturk, H.; Tavares, M.; Natto, Z.S. Prevalence of periodontitis and alveolar bone loss in a patient population at Harvard School of Dental Medicine. BMC Oral Health 2019, 19, 254. [Google Scholar] [CrossRef] [PubMed]
- Wylleman, A.; Van Der Veken, D.; Teughels, W.; Quirynen, M.; Laleman, I. Alveolar bone level at deciduous molars in Flemish children: A retrospective, radiographic study. J. Clin. Periodontol. 2020, 47, 660–667. [Google Scholar] [CrossRef]
- Castro, L.O.; Castro, I.O.; de Alencar, A.H.G.; Valladares-Neto, J.; Estrela, C. Cone beam computed tomography evaluation of distance from cementoenamel junction to alveolar crest before and after nonextraction orthodontic treatment. Angle Orthod. 2015, 86, 543–549. [Google Scholar] [CrossRef]
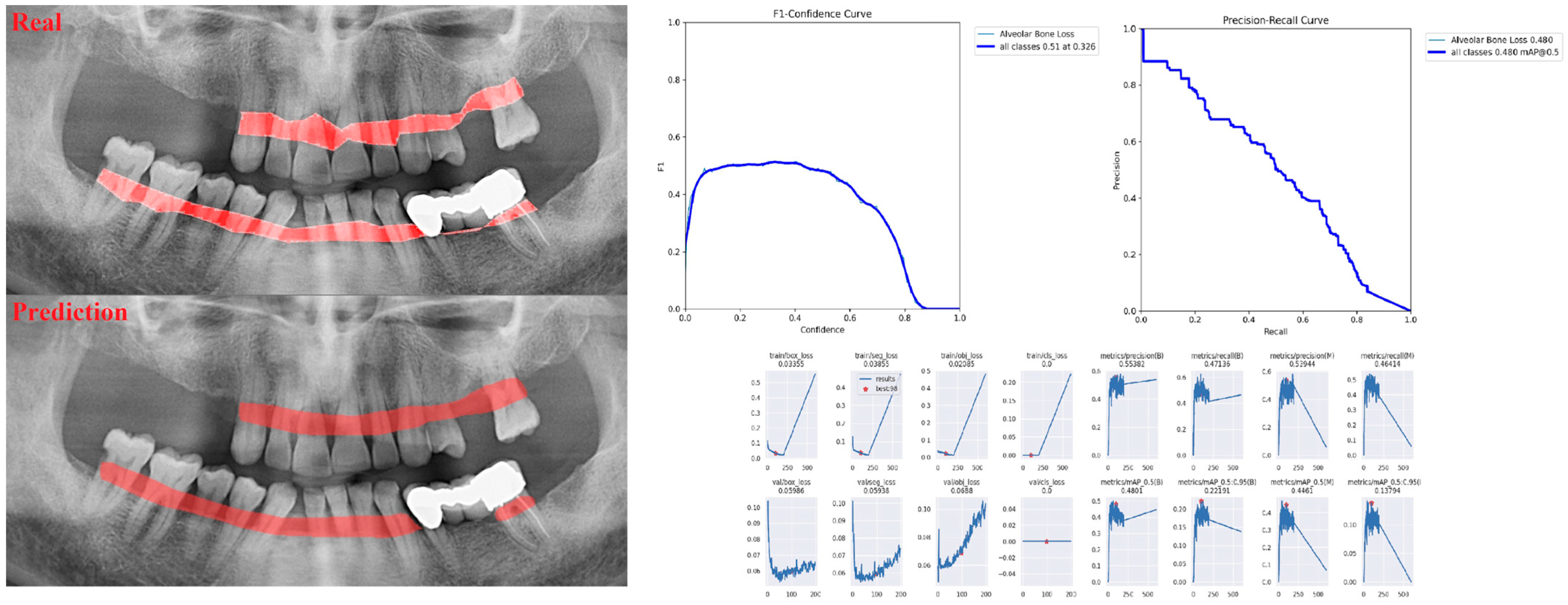
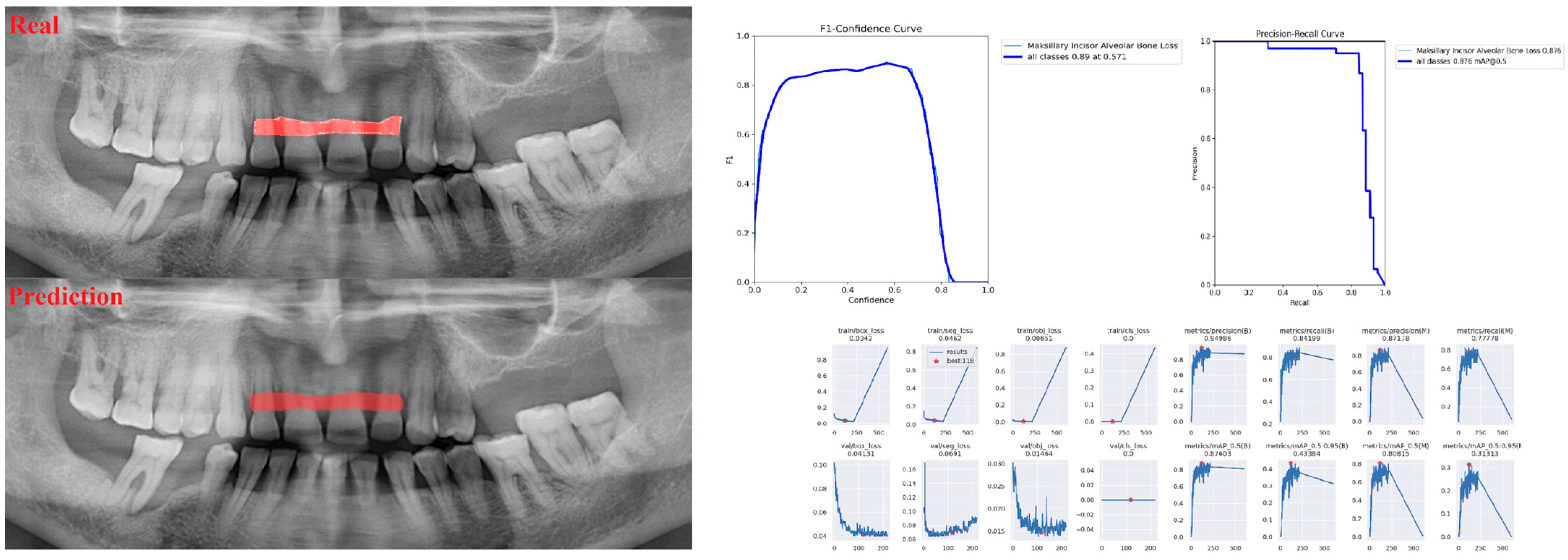
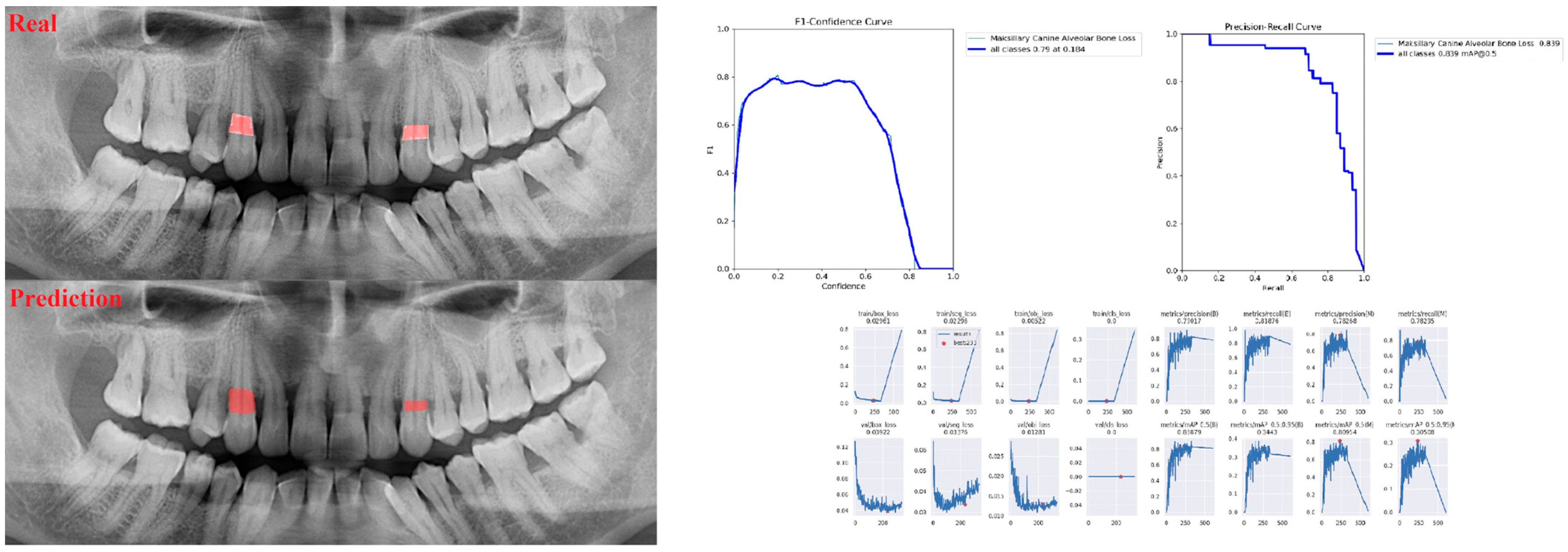
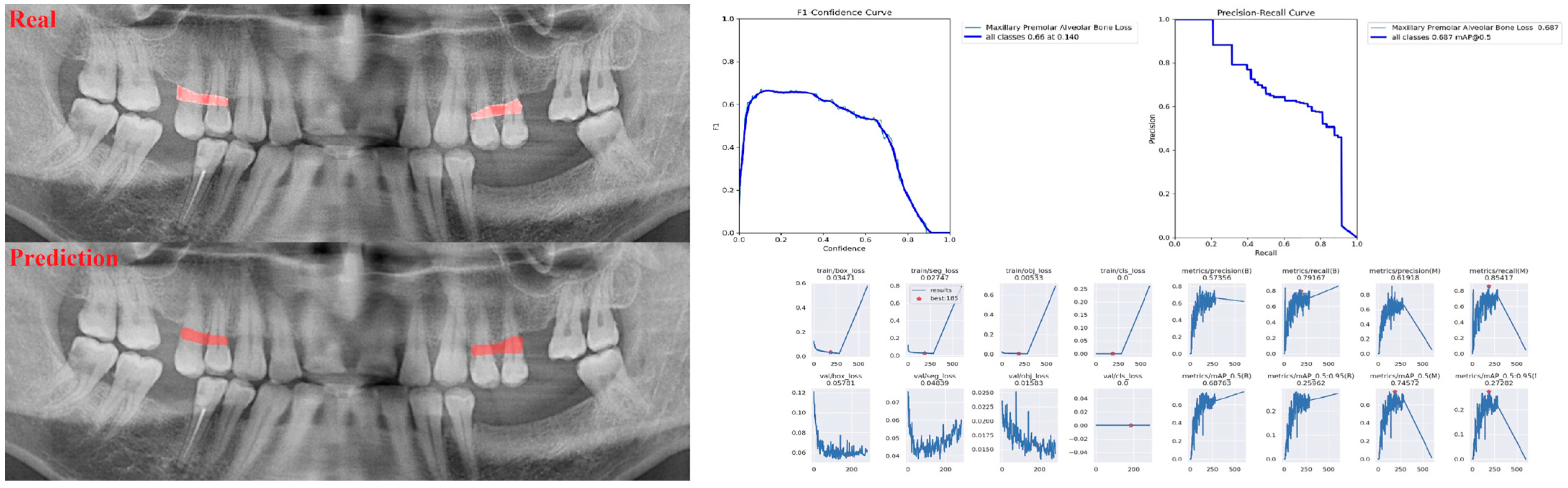
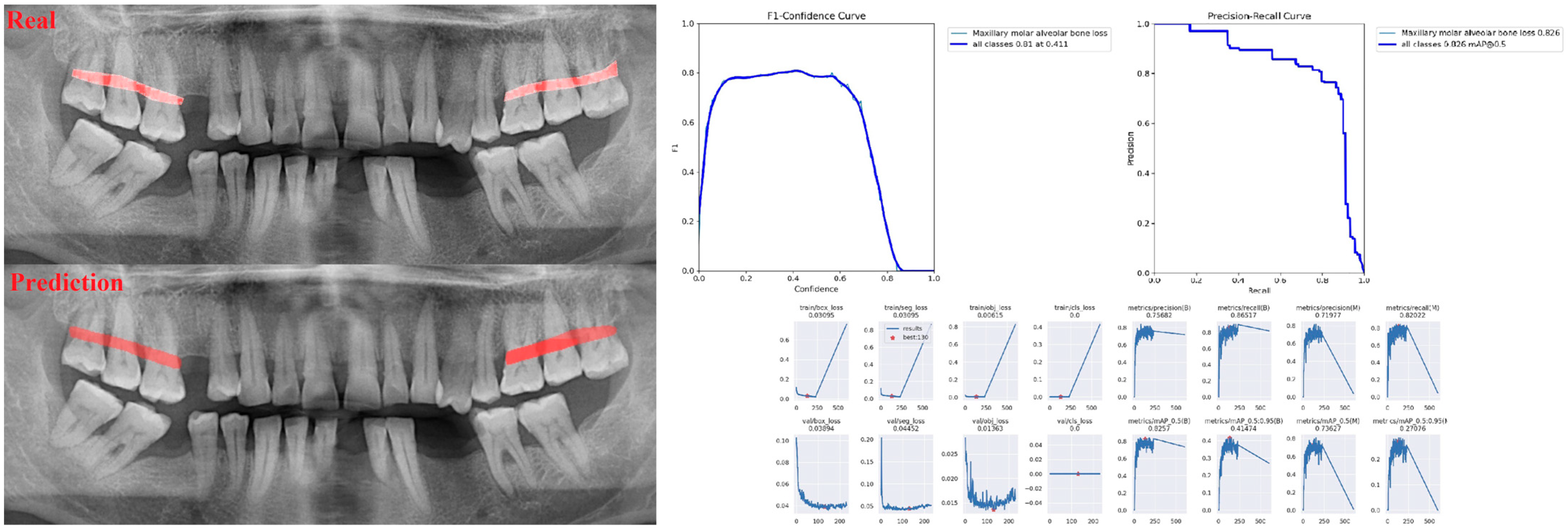
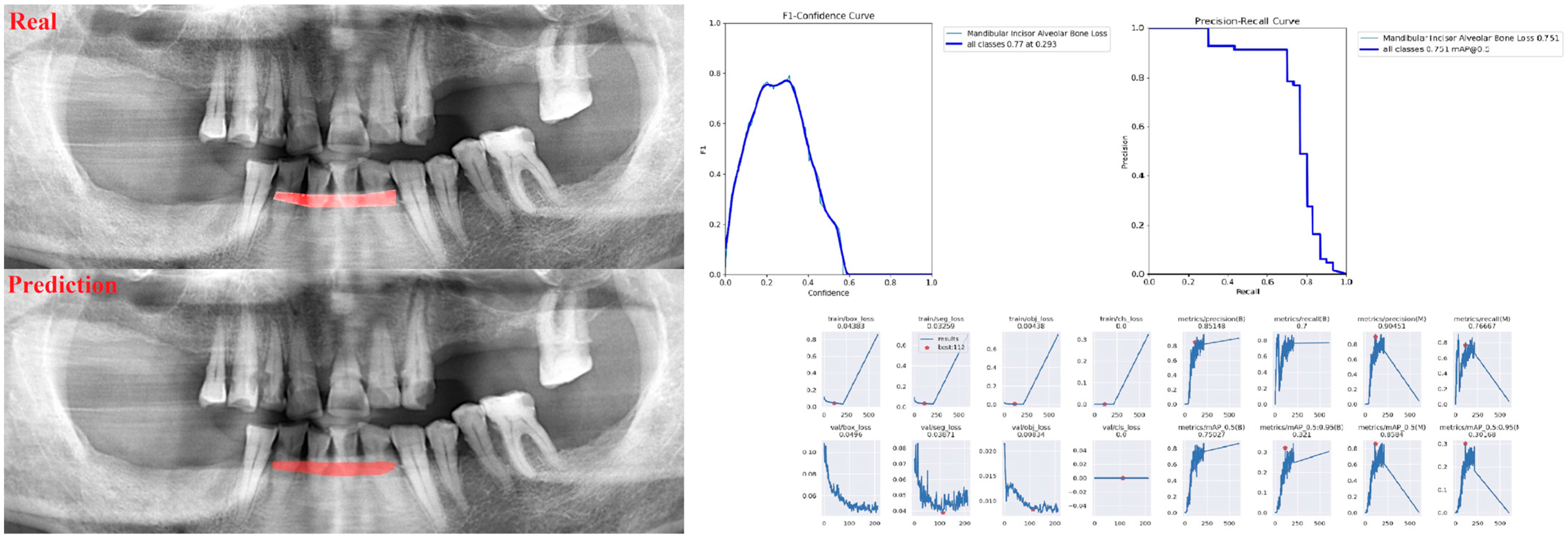
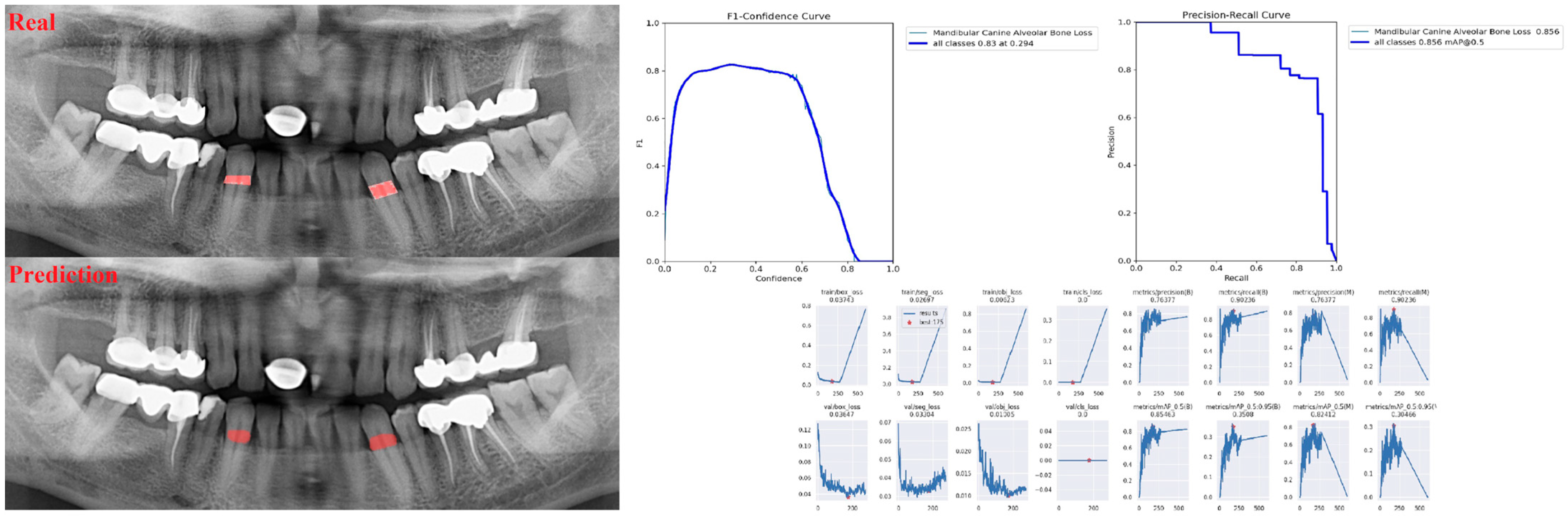
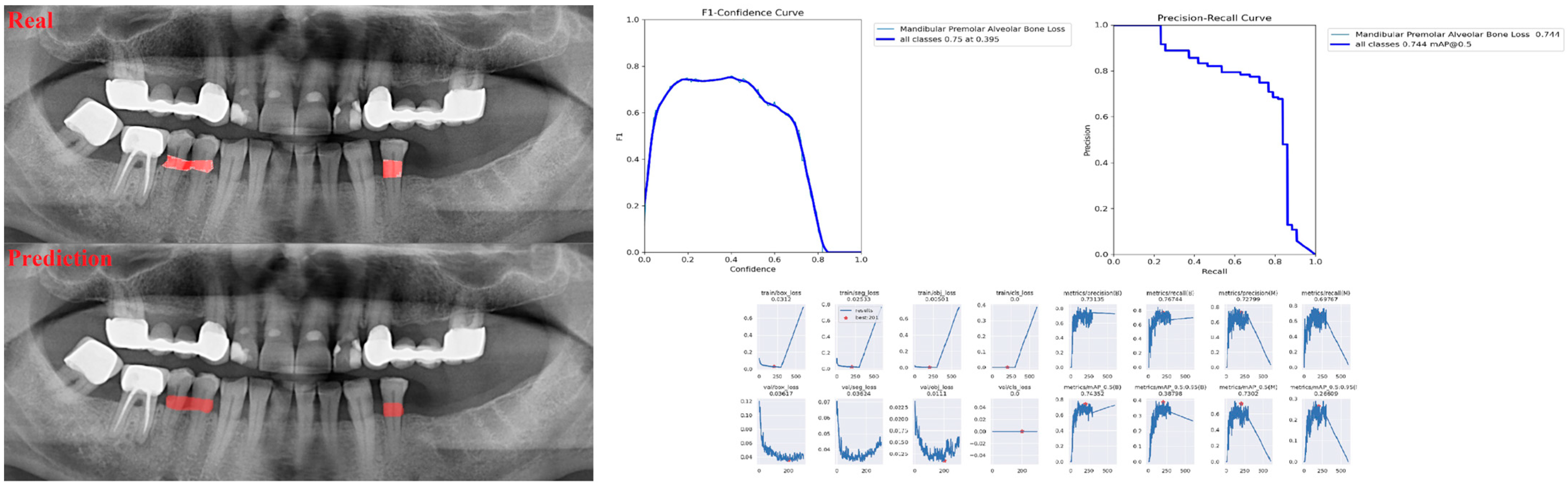
| Panoramic Images | |||
|---|---|---|---|
| Diagnoses | Training Group | Testing Group | Validation Group |
| Alveolar Bone Loss (ABL) | 549 | 68 | 68 |
| Maxillary | |||
| Incisor ABL | 352 | 43 | 44 |
| Canine ABL | 253 | 31 | 32 |
| Premolar ABL | 284 | 35 | 35 |
| Molar ABL | 445 | 55 | 56 |
| Mandibular | |||
| Incisor ABL | 238 | 29 | 30 |
| Canine ABL | 214 | 26 | 27 |
| Premolar ABL | 233 | 29 | 29 |
| Molar ABL | 277 | 34 | 35 |
| Measurement Value | |||
|---|---|---|---|
| Sensitivity | Precision | F1 Score | |
| Alveolar Bone Loss (ABL) | 0.75 | 0.76 | 0.76 |
| Maxillary | |||
| Incisor ABL | 1 | 0.90 | 0.95 |
| Canine ABL | 0.88 | 0.81 | 0.84 |
| Premolar ABL | 0.94 | 0.69 | 0.80 |
| Molar ABL | 0.87 | 0.96 | 0.91 |
| Mandibular | |||
| Incisor ABL | 0.83 | 0.89 | 0.86 |
| Canine ABL | 0.92 | 0.78 | 0.84 |
| Premolar ABL | 0.83 | 0.75 | 0.79 |
| Molar ABL | 0.85 | 0.73 | 0.79 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uzun Saylan, B.C.; Baydar, O.; Yeşilova, E.; Kurt Bayrakdar, S.; Bilgir, E.; Bayrakdar, İ.Ş.; Çelik, Ö.; Orhan, K. Assessing the Effectiveness of Artificial Intelligence Models for Detecting Alveolar Bone Loss in Periodontal Disease: A Panoramic Radiograph Study. Diagnostics 2023, 13, 1800. https://doi.org/10.3390/diagnostics13101800
Uzun Saylan BC, Baydar O, Yeşilova E, Kurt Bayrakdar S, Bilgir E, Bayrakdar İŞ, Çelik Ö, Orhan K. Assessing the Effectiveness of Artificial Intelligence Models for Detecting Alveolar Bone Loss in Periodontal Disease: A Panoramic Radiograph Study. Diagnostics. 2023; 13(10):1800. https://doi.org/10.3390/diagnostics13101800
Chicago/Turabian StyleUzun Saylan, Bilge Cansu, Oğuzhan Baydar, Esra Yeşilova, Sevda Kurt Bayrakdar, Elif Bilgir, İbrahim Şevki Bayrakdar, Özer Çelik, and Kaan Orhan. 2023. "Assessing the Effectiveness of Artificial Intelligence Models for Detecting Alveolar Bone Loss in Periodontal Disease: A Panoramic Radiograph Study" Diagnostics 13, no. 10: 1800. https://doi.org/10.3390/diagnostics13101800
APA StyleUzun Saylan, B. C., Baydar, O., Yeşilova, E., Kurt Bayrakdar, S., Bilgir, E., Bayrakdar, İ. Ş., Çelik, Ö., & Orhan, K. (2023). Assessing the Effectiveness of Artificial Intelligence Models for Detecting Alveolar Bone Loss in Periodontal Disease: A Panoramic Radiograph Study. Diagnostics, 13(10), 1800. https://doi.org/10.3390/diagnostics13101800







