Current Applications of Deep Learning and Radiomics on CT and CBCT for Maxillofacial Diseases
Abstract
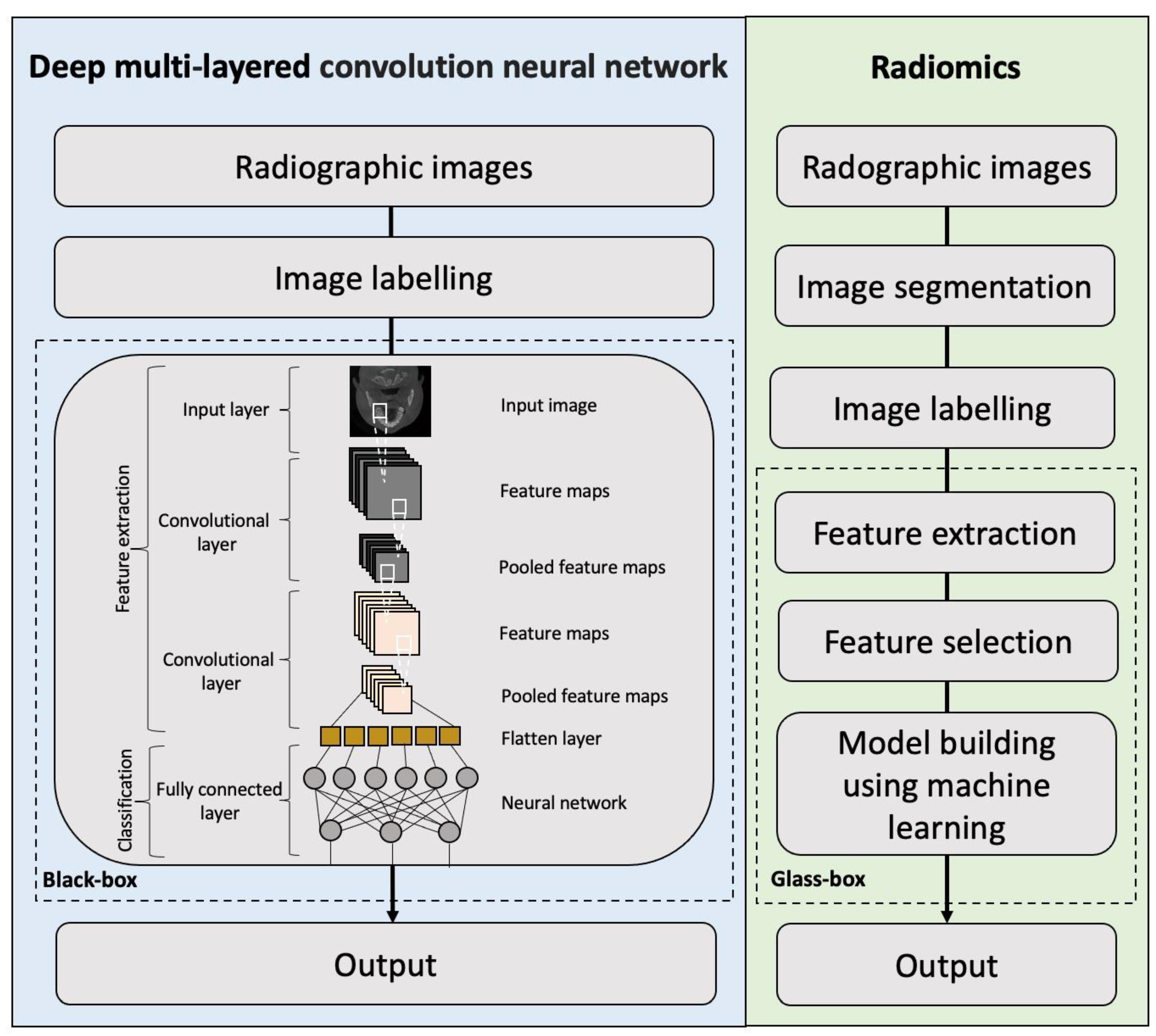
1. Introduction
2. Deep Learning and Radiomics on CT/CBCT for the Diagnosis and Management of Maxillofacial Diseases
2.1. Jaw Cysts and Tumors
2.2. Lymph Node Metastasis
2.3. Salivary Gland Diseases
2.4. Temporomandibular Joint Disorders
2.5. Maxillary Sinus Pathologies
2.6. Mandibular Fractures
2.7. Dentofacial Deformities and Malocclusion
| Author (Year) | Application | Imaging Modality | Model/Platform | Training and Validation Dataset | Test Dataset/Cross-validation | Execution Time | Performance | Major Findings | |
|---|---|---|---|---|---|---|---|---|---|
| Deep Learning | Manual/Semi-automatic Method | ||||||||
| Jaw cysts and tumors | |||||||||
| Lee et al. (2020) [16] | Detection, segmentation, and classification of OKCs, dentigerous and periapical cysts | Panoramic and CBCT images | CNN (Inception v3) | 912 panoramic and 789 CBCT images | 228 panoramic and 197 CBCT images | NA | Panoramic/CBCT AUC = 0.85/0.91 SEN = 88%/96% SPE = 77%/77% | NA | The model on CBCT images obtained higher diagnostic performance than the one on panoramic images. |
| Bispo et al. (2021) [17] | Differential diagnosis of ameloblastoma and OKCs | CT | CNN (Inception v3) | 2500 images augmented based on 350 slices from 40 scans of patients with ameloblastoma or OKCs | 2-fold CV with 5 iterations | NA | ACC = 90–92% | NA | The model obtained higher accuracy in identifying OKCs than ameloblastoma. |
| Chai et al. (2022) [18] | Classification of ameloblastoma and OKCs | CBCT | CNN (Inception v3) | 272 scans of patients with ameloblastoma or OKCs | 78 scans of patients with ameloblastoma or OKCs | Model/Senior/Junior OMF surgeons 36/1471/1113 s (78 scans) | ACC = 85% SEN = 87% SPE = 82% F1 = 85% | 7 senior/30 junior OMF surgeons ACC = 66%/59% SEN = 60%/64% SPE = 71%/53% F1 = 64%/61% | The model outperformed both senior and junior OMF surgeons. |
| Lymph node metastasis | |||||||||
| Ariji et al. (2019) [22] | Differentiation of metastatic cervical lymph nodes from negative lymph nodes in OSCC patients | Contrast-enhanced CT | CNN (AlexNet) | 441 cropped images including 127 metastatic and 314 non-metastatic lymph nodes from 45 OSCC patients | 5-fold CV | NA | AUC = 0.80 ACC = 78% SEN = 75% SPE = 81% PPV = 80% NPV = 77% | 2 radiologists AUC = 0.83 ACC = 83% SEN = 78% SPE = 89% PPV = 87% NPV = 80% | The model performed similarly to the radiologists. |
| Ariji et al. (2020) [23] | Differentiation between metastatic lymph nodes with and without extranodal extension in OSCC patients | Contrast-enhanced CT | CNN (AlexNet) | 80% of 703 cropped images including metastatic lymph nodes with or without extranodal extension from 51 OSCC patients | 20% of 703 cropped images | 11 s | AUC = 0.82 ACC = 84% SEN = 67% SPE = 90% PPV = 69% NPV = 89% | 4 Radiologists AUC = 0.52–63 ACC = 51–63% SEN = 42–55% SPE = 57–71% PPV = 52–66% NPV = 51–61% | The model outperformed 4 radiologists in identifying metastatic lymph nodes with extranodal extension. |
| Ariji et al. (2021) [24] | Detection of cervical lymph nodes in OSCC patients | Contrast-enhanced CT | CNN (DetectNet) | 320 image slices including 134 metastatic and 448 non-metastatic lymph nodes from 56 OSCC patients | 45 image slices including 25 metastatic and 69 non-metastatic lymph nodes from 56 OSCC patients | 8 s | SEN = 73% PPV = 96% F1 = 83% False positive rates per images = 4% | NA | The model has the potential to automatically detect cervical lymph nodes. |
| Ariji et al. (2022) [25] | Detection and segmentation of metastatic cervical lymph nodes in OSCC patients | Contrast-enhanced CT | CNN (U-Net) | 911 image slices including 134 metastatic and 446 non-metastatic lymph nodes from 59 OSCC patients | 72 image slices of 24 metastatic and 68 non-metastatic lymph nodes from 59 OSCC patients | 7 s | Detection AUC = 0.95 ACC = 96% SEN = 98% SPE = 95% Segmentation SEN = 74% PPV = 94% F1 = 83% | 2 radiologists Detection AUC = 0.90 ACC = 89% SEN = 94% SPE = 86% | The model outperformed 2 radiologists in detecting metastatic cervical lymph nodes while its segmentation accuracy should be improved. |
| Salivary gland diseases | |||||||||
| Kise et al. (2019) [30] | Diagnosis of Sjögren’s syndrome | CT | CNN (AlexNet) | 400 image slices from 20 scans of patients with Sjögren’s syndrome and 20 scans of individuals without parotid gland abnormalities | 100 image slices from 5 scans of patients with Sjögren’s syndrome and 5 scans of individuals without parotid gland abnormalities | NA | ACC = 0.96 SEN = 100% SPE = 92% | 3 experienced/3 inexperienced OMF radiologists ACC = 98%/84% SEN = 99%/78% SPE = 97%/89% | The model performed similarly to experienced radiologists and outperformed inexperienced radiologists. |
| Zhang et al. (2021) [32] | Classification between benign and malignant parotid gland tumors | CT | CNNs (Improved CNN, VGG16, InceptionV3, ResNet, and DenseNet) | 720 image slices (group 1) and 1050 image slices (group 2) | 180 image slices (group 1) and 270 image slices (group 2) | <1 min | Improved CNN on Group 1/2 ACC = 98%/78% SEN = 97%/77% SPE = 99%/79% PPV = 99%/79% F1 = 98%/78% | NA | The improved CNN model achieved the highest classification accuracy than other pre-trained CNN models. |
| Yuan et al. (2022) [31] | Classification between pleomorphic adenoma and malignant parotid gland tumors | CT | CNN (ResNet50) | 121 scans | 30 scans | NA | ACC = 90% | NA | The model achieved high accuracy in identifying malignant parotid gland tumors. |
| Temporomandibular disorders | |||||||||
| de Dumast et al. (2018) [40] | Classification of morphological variation in TMJ osteoarthritis | CBCT | Deep neural network | Scans of 259 condyles from 154/105 individuals with/without TMJ osteoarthritis | Scans of 34 condyles from 17/17 individuals with/without TMJ osteoarthritis. | NA | Agreement with two experts = 91% | NA | The model has the potential to assist clinicians in the diagnosis of TMJ osteoarthritis. |
| Kim et al. (2021) [39] | Segmentation and measurement of the cortical thickness of mandibular condyle head | CBCT | CNN (U-Net) | 11,776 image slices from 23 scans of individuals without pathological bony changes on the condyle head | 1024 image slices from 2 scans of individuals without pathological bony changes on the condyle head | 10–15 s | Marrow bone IoU = 0.87 HD = 0.93 mm Cortical bone IoU = 0.73 HD = 1.25 mm | NA | The model may contribute to automated quantitative analysis of the changes in bony structures of TMJ. |
| Le et al. (2021) [38] | Segmentation of mandibular ramus and condyle | CBCT | CNN (U-Net) | 90 scans of individuals with/without osteoarthritis, obtained from multiple centers | 19 scans of individuals with/without osteoarthritis, obtained from multiple centers | NA | AUC = 0.95 ACC = 100% SEN = 93% SPE = 100% F1 = 92% | NA | The model may facilitate treatment planning of TMJ degeneration. |
| Maxillary sinus | |||||||||
| Xu et al. (2020) [47] | Segmentation of the maxillary sinus | CT | CNN (V-Net) | 35 scans | 26 scans | <1 min | DSC = 0.94 IoU = 0.90 Precision = 94% | NA | The model achieved high segmentation accuracy. |
| Deng et al. (2020) [48] | Segmentation of the maxillary sinus | CT | CNN (BE-FNet) | 50 scans | 5-fold CV | 0.5 s | DSC = 0.95 VOE = 10.2% ASD = 2.9 mm | NA | The model achieved high segmentation accuracy. |
| Jung et al. (2021) [49] | Segmentation of maxillary sinus lesions | CBCT | CNN (3D nnU-Net) | 83 scans obtained from Korea University Anam Hospital | 20 scans obtained from Korea University Anam Hospital and 20 scans from Korea University Ansan Hospital | NA | Anam Hospital DSC = 0.76 Ansan Hospital DSC = 0.54 | NA | A lower segmentation accuracy of the model was found on external images. |
| Hung et al. (2022) [50] | Detection, segmentation, and measurement of the morphological changes of the sinus mucosa | CBCT | CNN (V-Net and SVR) | 347 low-dose scans of individuals with or without morphological changes of the maxillary sinus mucosa | 77 low-dose and 21 standard-dose scans of individuals with or without morphological changes of the maxillary sinus mucosa | NA | Low-dose scans AUC = 0.84–0.89 SEN = 79–81% SPE = 71–89% Standard-dose scans AUC = 0.89–0.93 SEN = 79–93% SPE = 89–93% | NA | The model performed similarly on both standard- and low-dose scans. |
| Fractures | |||||||||
| Wang et al. (2022) [53] | Detection and classification of mandibular fractures | CT | CNNs (U-Net and ResNet) | 278 scans | 408 scans | NA | AUC = 0.93–0.98 ACC = 94–98% SEN = 91–97% SPE = 91–99% | NA | The model may assist clinicians in timely and accurate detection of mandibular fractures. |
| Dentofacial deformities and malocclusion | |||||||||
| Kim et al. (2020) [66] | Classification of skeletal malocclusion | CBCT | Multi-channel CNNs | 173 scans of individuals with Class I, II, or III malocclusion | 45 scans of individuals with Class I, II, or III malocclusion | NA | ACC = 93–94% SEN = 95% PPV = 93–94% F1 = 94–95% | NA | The model may facilitate orthodontic and orthognathic evaluation to determine whether the patient needs surgical correction. |
| Ma et al. (2022) [68] | Prediction of skeletal changes after orthognathic surgery | CT | CNN | 50 pairs of preoperative and postoperative full skull scans | 6 pairs of preoperative and postoperative full skull scans | 43 s | Mean landmark localization deviation = 5.4 mm 74% of the predicted postoperative skull models was consistent with the ground truth | NA | The model may assist OMF surgeons in predicting postoperative skeletal changes for orthognathic surgical planning. |
| ter Horst et al. (2021) [67] | Prediction of virtual soft tissue profile after mandibular advancement surgery | 3D photographs and CBCT | Autoencoder-inspired neural network | 119 pairs of 3D photographs and CBCT scans of patients who underwent mandibular advancement surgery | 14 pairs of 3D photographs and CBCT scans of patients who underwent mandibular advancement surgery | NA | Mean absolute error 1 mm (lower face) 1.1 mm (lower lip) 1.4 mm (chin) | MTM-based soft-tissue simulations Mean absolute error 1.5 mm (lower face) 1.7 mm (lower lip) 2 mm (chin) | The model performed similarly to the MTM-based soft-tissue simulations, indicating that it may be useful for soft tissue profile prediction in orthognathic surgery. |
| Lin et al. (2021) [69] | Assessment of facial symmetry before and after orthognathic surgery | CBCT | CNNs (VGG16, VGG19, ResNet50, and Xception) | 71 scans | 59 scans | NA | ACC 80% (VGG16) 86% (VGG19) 83% (ResNet50) 90% (Xception) | NA | The model trained with Xception achieved highest accuracy for facial symmetry assessment. |
| Image registration | |||||||||
| Chung et al. (2020) [65] | Registration between CBCT and optical dental model scans | CBCT and optical dental model scans | Deep pose regression neural networks and optimal cluster-based matching | 150 pairs of CBCT and optical maxillary model scans and 150 pairs of CBCT and mandibular model scans | 3-fold CV | 17.6 s | Mean distance errors 5.1 mm (surface) 1.8 mm (landmarks) | Conventional three-point registration Mean distance errors 9.6 mm (surface) 2.7 mm (landmarks) | The model is applicable to full-arch scanned models and can avoid metal artifacts during the matching procedures. |
| Jang et al. (2021) [64] | Registration between CBCT and intraoral scans | CBCT and intraoral scans | CNN | 71 maxillary or mandibular intraoral scans and the corresponding 49 CBCT scans | 22 pairs of CBCT and intraoral scans | NA | Mean distance errors 0.5 mm (surface) 0.2 mm (landmarks) | Manual registration Mean distance errors 1.7 mm (surface) 0.7 mm (landmarks) | The model outperformed the manual registration method. |
| Segmentation of maxillofacial structures | |||||||||
| Lo Giudice et al. (2021) [56] | Segmentation of the mandible | CBCT | CNN | 20 scans | 20 scans | 50 s | DSC = 0.97 Matching percentage = 89% | NA | The model may be useful in the planning of maxillofacial surgical procedures. |
| Xu et al. (2021) [63] | Segmentation of mandibles with/without tumor invasion | CT | CNN (3D V-Net) | 160 scans of 80 consisting of 80 MTI scans and 80 Non-MTI scans | 70 scans consisting of 35 MTI scans and 35 Non-MTI scans | 7.4 s | Non-MTI segmentation DSC = 0.98 IoU = 0.96 ASD = 0.06 mm HD = 0.48 mm MTI segmentation DSC = 0.97 IoU = 0.94 ASD = 0.16 mm HD = 1.16mm | NA | The model obtained high accuracy in segmenting mandibles with and without tumor invasion. |
| Sin et al. (2021) [59] | Segmentation of pharyngeal airway | CBCT | CNN (U-Net) | 260 scans | 46 scans | NA | DSC = 0.92 IoU = 0.99 | NA | The model can efficiently calculate the pharyngeal airway volume from CBCT images. |
| Orhan et al. (2022) [60] | Segmentation of the pharyngeal airway in OSA and non-OSA patients | CBCT | Diagnocat (a commercially available AI platform; https://diagnocat.com (accessed on 5 December 2022)) | NA | 200 scans of 100 OSA and 100 non-OSA patients, taken using 3 different CBCT scanners | NA | ICC between Diagnocat and radiologists = 0.97 | NA | Diagnocat performed similarly to radiologists and can efficiently calculate the pharyngeal airway volume in OSA and non-OSA patients. |
| Preda et al. (2022) [57] | Segmentation of the maxillofacial complex, including palatine, maxillary, zygomatic, nasal, and lacrimal bones | CBCT | CNN (U-Net) | 120 scans taken using two different scanners | 24 scans taken using two different scanners | Model 39 s Manual 133 min | DSC = 0.93 IoU = 0.86 95% HD = 0.62 mm RMS 0.5 mm | Semi-automated segmentation using Mimics DSC = 0.69 IoU = 0.53 95% HD = 2.78 mm RMS 1.76 mm | The model may improve the efficiency of the digital workflows for patient-specific treatment planning of maxillofacial surgical procedures. |
| Ezhov et al. (2021) [58] | Segmentation of teeth and jaws, numbering of teeth, detection of caries, periapical lesions, and periodontitis | CBCT | Diagnocat (a commercially available AI platform; https://diagnocat.com (accessed on 5 December 2022)) | 1346 scans taken using 17 scanners | 30 scans | With the aid of Diagnocat = 17.6 min Without the aid of Diagnocat = 18.7 min | Diagnocat SEN = 92% SPE = 99% 12 dentists with the aid of Diagnocat SEN = 85% SPE = 97% | 4 OMF radiologists SEN = 93–94% SPE = 99–100% 12 dentists without the aid of Diagnocat SEN = 77% SPE = 96% | Diagnocat performed similarly to four radiologists and improved twelve dentists’ performance |
| Jaskari et al. (2020) [61] | Segmentation of the mandibular canal | CBCT | CNN | 509 scans taken using two scanners | 15 scans | NA | MCD = 0.56 mm ASSD = 0.45 mm DSC = 0.57 (left) and 0.58 (right) HD = 1.40 (left) and 1.38 (right) | NA | The model may help to locate the inferior alveolar nerve for surgical planning |
| Lim et al. (2021) [62] | Segmentation of the mandibular canal | CBCT | CNN (3D nnU-Net) | 83 scans from Korea University Anam Hospital | 15, 20, and 20 scans from Korea University Anam Hospital (1), Korea University Ansan Hospital (2), and Korea University Guro Hospital (3) | Model 86 s Manual 125 s | Internal testing DSC = 0.58 (1) External testing DSC = 0.55 (2) DSC = 0.43 (3) | NA | The model may help to locate the inferior alveolar nerve for surgical planning |
| Author (Year) | Application | Imaging Modality | Image Dataset | Region of Interest for Feature Extraction | Data for Model Building | Machine Learning Approach | Validation Method | Performance of the Best Model(s) | Major Findings |
|---|---|---|---|---|---|---|---|---|---|
| Zhong et al. (2021) [27] | Prediction of cervical lymph node metastasis in patients with tongue cancer | Contrast-enhanced CT | 313 scans of patients with tongue cancer | Primary cancer | Radiomic features and clinical lymph node status | Artificial neural network | Hold-out validation (20%) | Model on radiomic features and clinical lymph node status AUC = 0.94 ACC = 84% SEN = 93% SPE = 77% Model on radiomic features AUC = 0.92 ACC = 86% SEN = 82% SPE = 89% | The model on radiomic features and clinical lymph node status achieved higher prediction accuracy than the one only on radiomic features. |
| Kubo et al. (2022) [26] | Prediction of occult cervical lymph node metastasis in patients with tongue cancer | Contrast-enhanced CT | 161 scans of tongue cancer patients with or without occult cervical lymph node metastasis | Cervical lymph nodes | Radiomic features | kNN, SVM, CART, RF, AdaBoost with/without SMOTE | 10-fold CV | Side level RF with SMOTE AUC = 0.92 ACC = 85% SEN = 82% PPV = 88% Region level SVM with SMOTE AUC = 0.98 ACC = 96% SEN = 95% PPV = 96% | The radiomics models may serve as useful tools to support clinical decision making in the management of patients with tongue cancer. |
| Morgan et al. (2021) [28] | Prediction of local failure in head and neck cancer | Contrast-enhanced CT and CBCT | Baseline CT scan, two CBCT scans at fractions 1 and 21 of radiotherapy from 90 head and neck SCC patients with or without local failure | All primary and nodal structures | Radiomic features and several clinical variables | Explainable boosting machine with 25 iterations | 5-fold CV | Fused ensemble model (primary/nodal structures) AUC = 0.87/0.91 SEN = 78%/100% SPE = 91%/68% | The model on radiomic features and clinical variables achieved the highest accuracy in predicting local failure in head and neck cancer. |
| Xu et al. (2021) [34] | Differentiation between benign and malignant parotid gland tumors | CT | 87 scans of patients with benign or malignant parotid gland tumor | Primary tumors | Radiomic features and radiological variables including the location and metastases of lymph nodes | SVM | Hold-out validation (38 scans) | The combined model AUC = 0.84 SEN = 82% SPE = 74% The model on radiomic features AUC = 0.77 SEN = 79% SPE = 89% | The combined model outperformed the models on individual radiomic features, lymph node location, or lymph node metastases. |
| Zhang et al. (2021) [33] | Differentiation between low- and high-grade mucoepidermoid carcinoma of the salivary glands | CT | 53 scans of patients with low or high grade mucoepidermoid carcinoma | Primary cancer | Radiomic features | Logistic regression | NA | AUC = 0.80 ACC = 78% SEN = 89% PPV = 67% | High-grade mucoepidermoid carcinomas may be associated with a low energy, high correlation texture, and high surface irregularity. |
| Liu et al. (2021) [35] | Differentiation between pleomorphic adenoma and Warthin tumors of the parotid glands | CT and MRI | 659 pairs of CT and MRI scans from patients with pleomorphic adenoma or Warthin tumors | Primary tumors | CT- and MRI-derived radiomic features | Logistic regression | NA | CT/MRI AUC = 0.88/0.91 ACC = 78%/84% SEN = 81%/85% SPE = 76%/83% PPV = 70%/77% NPV = 86%/89% | The model on MRI-derived radiomic features performed slightly higher than but not significantly differently from the model on CT-derived radiomic features. |
| Bianchi et al. (2020) [41] | Diagnosis of TMJ osteoarthritis | CBCT | 92 scans of subjects with or without TMJ osteoarthritis | Internal condylar lateral region | 20 radiomic and 25 biomolecular features, 5 clinical and 2 demographic variables | LR, RF, LightGBM, XGBoost with 10 iterations | 5-fold CV | XGBoost + LightGBM AUC = 0.87 ACC = 82% SEN = 84% F1 = 82% | The model may be helpful for screening individuals with early TMJ osteoarthritis. |
3. The Challenges and Prospects of Deep Learning and Radiomics on CT/CBCT for Maxillofacial Diseases
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Joda, T.; Yeung, A.W.K.; Hung, K.; Zitzmann, N.U.; Bornstein, M.M. Disruptive innovation in dentistry: What it is and what could be next. J. Dent. Res. 2021, 100, 448–453. [Google Scholar] [CrossRef]
- Schwendicke, F.; Samek, W.; Krois, J. Artificial intelligence in dentistry: Chances and challenges. J. Dent. Res. 2020, 99, 769–774. [Google Scholar] [CrossRef]
- Hung, K.F.; Ai, Q.Y.H.; Leung, Y.Y.; Yeung, A.W.K. Potential and impact of artificial intelligence algorithms in dento-maxillofacial radiology. Clin. Oral Investig. 2022, 26, 5535–5555. [Google Scholar] [CrossRef]
- Stone, P.; Brooks, R.; Brynjolfsson, E.; Calo, R.; Etzioni, O.; Hager, G.; Hirschberg, J.; Kalyanakrishnan, S.; Kamar, E.; Kraus, S.; et al. Artificial Intelligence and Life in 2030. One Hundred Year Study on Artificial Intelligence: Report of the 2015–2016 Study Panel; Stanford University: Stanford, CA, USA, 2016; Available online: http://ai100stanfordedu/2016-report (accessed on 5 December 2022).
- Leite, A.F.; Vasconcelos, K.F.; Willems, H.; Jacobs, R. Radiomics and machine learning in oral healthcare. Proteom. Clin. Appl. 2020, 14, e1900040. [Google Scholar] [CrossRef]
- van Timmeren, J.E.; Cester, D.; Tanadini-Lang, S.; Alkadhi, H.; Baessler, B. Radiomics in medical imaging-“how-to” guide and critical reflection. Insights Imaging 2020, 11, 91. [Google Scholar] [CrossRef]
- Roy, S.; Whitehead, T.D.; Quirk, J.D.; Salter, A.; Ademuyiwa, F.O.; Li, S.; An, H.; Shoghi, K.I. Optimal co-clinical radiomics: Sensitivity of radiomic features to tumour volume, image noise and resolution in co-clinical T1-weighted and T2-weighted magnetic resonance imaging. EBioMedicine 2020, 59, 102963. [Google Scholar] [CrossRef]
- Roy, S.; Whitehead, T.D.; Li, S.; Ademuyiwa, F.O.; Wahl, R.L.; Dehdashti, F.; Shoghi, K.I. Co-clinical FDG-PET radiomic signature in predicting response to neoadjuvant chemotherapy in triple-negative breast cancer. Eur. J. Nucl. Med. Mol. Imaging 2021, 49, 550–562. [Google Scholar] [CrossRef]
- Hung, K.; Yeung, A.W.K.; Tanaka, R.; Bornstein, M.M. Current applications, opportunities, and limitations of AI for 3D imaging in dental research and practice. Int. J. Environ. Res. Public Health 2020, 17, 4424. [Google Scholar] [CrossRef] [PubMed]
- Hung, K.; Montalvao, C.; Tanaka, R.; Kawai, T.; Bornstein, M.M. The use and performance of artificial intelligence applications in dental and maxillofacial radiology: A systematic review. Dentomaxillofac. Radiol. 2020, 49, 20190107. [Google Scholar] [CrossRef]
- Poedjiastoeti, W.; Suebnukarn, S. Application of convolutional neural network in the diagnosis of jaw tumors. Healthc. Inform. Res. 2018, 24, 236–241. [Google Scholar] [CrossRef]
- Kwon, O.; Yong, T.H.; Kang, S.R.; Kim, J.E.; Huh, K.H.; Heo, M.S. Automatic diagnosis for cysts and tumors of both jaws on panoramic radiographs using a deep convolution neural network. Dentomaxillofac. Radiol. 2020, 49, 20200185. [Google Scholar] [CrossRef]
- Endres, M.G.; Hillen, F.; Salloumis, M.; Sedaghat, A.R.; Niehues, S.M.; Quatela, O.; Hanken, H.; Smeets, R.; Beck-Broichsitter, B.; Rendenbach, C.; et al. Development of a deep learning algorithm for periapical disease detection in dental radiographs. Diagnostics 2020, 10, 430. [Google Scholar] [CrossRef]
- Ariji, Y.; Yanashita, Y.; Kutsuna, S.; Muramatsu, C.; Fukuda, M.; Kise, Y.; Nozawa, M.; Kuwada, C.; Fujita, H.; Katsumata, A.; et al. Automatic detection and classification of radiolucent lesions in the mandible on panoramic radiographs using a deep learning object detection technique. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2019, 128, 424–430. [Google Scholar] [CrossRef]
- Watanabe, H.; Ariji, Y.; Fukuda, M.; Kuwada, C.; Kise, Y.; Nozawa, M.; Sugita, Y.; Ariji, E. Deep learning object detection of maxillary cyst-like lesions on panoramic radiographs: Preliminary study. Oral Radiol. 2021, 37, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, D.H.; Jeong, S.N. Diagnosis of cystic lesions using panoramic and cone beam computed tomographic images based on deep learning neural network. Oral Dis. 2020, 26, 152–158. [Google Scholar] [CrossRef]
- Bispo, M.S.; Pierre Júnior, M.L.G.Q.; Apolinário Jr, A.L.; Dos Santos, J.N.; Junior, B.C.; Neves, F.S.; Crusoé-Rebello, I. Computer tomographic differential diagnosis of ameloblastoma and odontogenic keratocyst: Classification using a convolutional neural network. Dentomaxillofac. Radiol. 2021, 50, 20210002. [Google Scholar] [CrossRef] [PubMed]
- Chai, Z.K.; Mao, L.; Chen, H.; Sun, T.G.; Shen, X.M.; Liu, J.; Sun, Z.J. Improved diagnostic accuracy of ameloblastoma and odontogenic keratocyst on cone-beam CT by artificial intelligence. Front Oncol. 2022, 11, 793417. [Google Scholar] [CrossRef] [PubMed]
- Negi, A.; Puri, A.; Gupta, R.; Nangia, R.; Sachdeva, A.; Mittal, M. Comparison of immunohistochemical expression of antiapoptotic protein survivin in normal oral mucosa, oral leukoplakia, and oral squamous cell carcinoma. Patholog. Res. Int. 2015, 2015, 840739. [Google Scholar] [CrossRef] [PubMed]
- Hanai, N.; Asakage, T.; Kiyota, N.; Homma, A.; Hayashi, R. Controversies in relation to neck management in N0 early oral tongue cancer. Jpn. J. Clin. Oncol. 2019, 49, 297–305. [Google Scholar] [CrossRef]
- van den Brekel, M.W.; Stel, H.V.; Castelijns, J.A.; Nauta, J.J.; van der Waal, I.; Valk, J.; Meyer, C.J.; Snow, G.B. Cervical lymph node metastasis: Assessment of radiologic criteria. Radiology 1990, 177, 379–384. [Google Scholar] [CrossRef]
- Ariji, Y.; Fukuda, M.; Kise, Y.; Nozawa, M.; Yanashita, Y.; Fujita, H.; Katsumata, A.; Ariji, E. Contrast-enhanced computed tomography image assessment of cervical lymph node metastasis in patients with oral cancer by using a deep learning system of artificial intelligence. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2019, 127, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Ariji, Y.; Sugita, Y.; Nagao, T.; Nakayama, A.; Fukuda, M.; Kise, Y.; Nozawa, M.; Nishiyama, M.; Katumata, A.; Ariji, E. CT evaluation of extranodal extension of cervical lymph node metastases in patients with oral squamous cell carcinoma using deep learning classification. Oral Radiol. 2020, 36, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Ariji, Y.; Fukuda, M.; Nozawa, M.; Kuwada, C.; Goto, M.; Ishibashi, K.; Nakayama, A.; Sugita, Y.; Nagao, T.; Ariji, E. Automatic detection of cervical lymph nodes in patients with oral squamous cell carcinoma using a deep learning technique: A preliminary study. Oral Radiol. 2021, 37, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Ariji, Y.; Kise, Y.; Fukuda, M.; Kuwada, C.; Ariji, E. Segmentation of metastatic cervical lymph nodes from CT images of oral cancers using deep-learning technology. Dentomaxillofac. Radiol. 2022, 51, 20210515. [Google Scholar] [CrossRef]
- Kubo, K.; Kawahara, D.; Murakami, Y.; Takeuchi, Y.; Katsuta, T.; Imano, N.; Nishibuchi, I.; Saito, A.; Konishi, M.; Kakimoto, N.; et al. Development of a radiomics and machine learning model for predicting occult cervical lymph node metastasis in patients with tongue cancer. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2022, 134, 93–101. [Google Scholar] [CrossRef]
- Zhong, Y.W.; Jiang, Y.; Dong, S.; Wu, W.J.; Wang, L.X.; Zhang, J.; Huang, M.W. Tumor radiomics signature for artificial neural network-assisted detection of neck metastasis in patient with tongue cancer. J. Neuroradiol. 2022, 49, 213–218. [Google Scholar] [CrossRef]
- Morgan, H.E.; Wang, K.; Dohopolski, M.; Liang, X.; Folkert, M.R.; Sher, D.J.; Wang, J. Exploratory ensemble interpretable model for predicting local failure in head and neck cancer: The additive benefit of CT and intra-treatment cone-beam computed tomography features. Quant. Imaging Med. Surg. 2021, 11, 4781–4796. [Google Scholar] [CrossRef]
- Gündüz, E.; Alçin, Ö.F.; Kizilay, A.; Piazza, C. Radiomics and deep learning approach to the differential diagnosis of parotid gland tumors. Curr. Opin. Otolaryngol. Head Neck Surg. 2022, 30, 107–113. [Google Scholar] [CrossRef]
- Kise, Y.; Ikeda, H.; Fujii, T.; Fukuda, M.; Ariji, Y.; Fujita, H.; Katsumata, A.; Ariji, E. Preliminary study on the application of deep learning system to diagnosis of Sjögren’s syndrome on CT images. Dentomaxillofac. Radiol. 2019, 48, 20190019. [Google Scholar] [CrossRef]
- Yuan, J.; Fan, Y.; Lv, X.; Chen, C.; Li, D.; Hong, Y.; Wang, Y. Research on the practical classification and privacy protection of CT images of parotid tumors based on ResNet50 model. J. Phys. Conf. Ser. 2020 2022, 1576, 012040. [Google Scholar] [CrossRef]
- Zhang, H.; Lai, H.; Wang, Y.; Lv, X.; Hong, Y.; Peng, J.; Zhang, Z.; Chen, C.; Chen, C. Research on the classification of benign and malignant parotid tumors based on transfer learning and a convolutional neural network. IEEE Access 2021, 9, 40360–40371. [Google Scholar] [CrossRef]
- Zhang, M.H.; Hasse, A.; Carroll, T.; Pearson, A.T.; Cipriani, N.A.; Ginat, D.T. Differentiating low and high grade mucoepidermoid carcinoma of the salivary glands using CT radiomics. Gland Surg. 2021, 10, 1646–1654. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Shu, Z.; Song, G.; Liu, Y.; Pang, P.; Wen, X.; Gong, X. The role of preoperative computed tomography radiomics in distinguishing benign and malignant tumors of the parotid gland. Front. Oncol. 2021, 11, 634452. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zheng, J.; Lu, X.; Wang, Y.; Meng, F.; Zhao, J.; Guo, C.; Yu, L.; Zhu, Z.; Zhang, T. Radiomics-based comparison of MRI and CT for differentiating pleomorphic adenomas and Warthin tumors of the parotid gland: A retrospective study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2021, 131, 591–599. [Google Scholar] [CrossRef]
- Li, D.T.S.; Leung, Y.Y. Temporomandibular disorders: Current concepts and controversies in diagnosis and management. Diagnostics 2021, 11, 459. [Google Scholar] [CrossRef]
- Ottersen, M.K.; Abrahamsson, A.K.; Larheim, T.A.; Arvidsson, L.Z. CBCT characteristics and interpretation challenges of temporomandibular joint osteoarthritis in a hand osteoarthritis cohort. Dentomaxillofac. Radiol. 2019, 48, 20180245. [Google Scholar] [CrossRef]
- Le, C.; Deleat-Besson, R.; Prieto, J.; Brosset, S.; Dumont, M.; Zhang, W.; Cevidanes, L.; Bianchi, J.; Ruellas, A.; Gomes, L.; et al. Automatic segmentation of mandibular ramus and condyles. In Proceedings of the 2021 43rd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Guadalajara, Mexico, 1–5 November 2021; pp. 2952–2955. [Google Scholar]
- Kim, Y.H.; Shin, J.Y.; Lee, A.; Park, S.; Han, S.S.; Hwang, H.J. Automated cortical thickness measurement of the mandibular condyle head on CBCT images using a deep learning method. Sci. Rep. 2021, 11, 14852. [Google Scholar] [CrossRef]
- de Dumast, P.; Mirabel, C.; Cevidanes, L.; Ruellas, A.; Yatabe, M.; Ioshida, M.; Ribera, N.T.; Michoud, L.; Gomes, L.; Huang, C.; et al. A web-based system for neural network based classification in temporomandibular joint osteoarthritis. Comput. Med. Imaging Graph. 2018, 67, 45–54. [Google Scholar] [CrossRef]
- Bianchi, J.; de Oliveira Ruellas, A.C.; Gonçalves, J.R.; Paniagua, B.; Prieto, J.C.; Styner, M.; Li, T.; Zhu, H.; Sugai, J.; Giannobile, W.; et al. Osteoarthritis of the Temporomandibular Joint can be diagnosed earlier using biomarkers and machine learning. Sci. Rep. 2020, 10, 8012. [Google Scholar] [CrossRef]
- Hung, K.F.; Hui, L.L.; Leung, Y.Y. Patient-specific estimation of the bone graft volume needed for maxillary sinus floor elevation: A radiographic study using cone-beam computed tomography. Clin. Oral Investig. 2022, 26, 3875–3884. [Google Scholar] [CrossRef]
- Yeung, A.W.K.; Hung, K.F.; Li, D.T.S.; Leung, Y.Y. The use of CBCT in evaluating the health and pathology of the maxillary sinus. Diagnostics 2022, 12, 2819. [Google Scholar] [CrossRef]
- Hung, K.; Hui, L.; Yeung, A.W.K.; Wu, Y.; Hsung, R.T.; Bornstein, M.M. Volumetric analysis of mucous retention cysts in the maxillary sinus: A retrospective study using cone-beam computed tomography. Imaging Sci. Dent. 2021, 51, 117–127. [Google Scholar] [CrossRef]
- Hung, K.; Montalvao, C.; Yeung, A.W.K.; Li, G.; Bornstein, M.M. Frequency, location, and morphology of accessory maxillary sinus ostia: A retrospective study using cone beam computed tomography (CBCT). Surg. Radiol. Anat. 2020, 42, 219–228. [Google Scholar] [CrossRef]
- Yeung, A.W.K.; Colsoul, N.; Montalvao, C.; Hung, K.; Jacobs, R.; Bornstein, M.M. Visibility, location, and morphology of the primary maxillary sinus ostium and presence of accessory ostia: A retrospective analysis using cone beam computed tomography (CBCT). Clin. Oral Investig. 2019, 23, 3977–3986. [Google Scholar] [CrossRef]
- Xu, J.; Wang, S.; Zhou, Z.; Liu, J.; Jiang, X.; Chen, X. Automatic CT image segmentation of maxillary sinus based on VGG network and improved V-Net. Int. J. Comput. Assist. Radiol. Surg. 2020, 15, 1457–1465. [Google Scholar] [CrossRef]
- Deng, Z.; Wang, B.; Zhu, Z. BE-FNet: 3D bounding box estimation feature pyramid network for accurate and efficient maxillary sinus segmentation. Math. Probl. Eng. 2020, 2020, 5689301. [Google Scholar] [CrossRef]
- Jung, S.K.; Lim, H.K.; Lee, S.; Cho, Y.; Song, I.S. Deep active learning for automatic segmentation of maxillary sinus lesions using a convolutional neural network. Diagnostics 2021, 11, 688. [Google Scholar] [CrossRef]
- Hung, K.F.; Ai, Q.Y.H.; King, A.D.; Bornstein, M.M.; Wong, L.M.; Leung, Y.Y. Automatic detection and segmentation of morphological changes of the maxillary sinus mucosa on cone-beam computed tomography images using a three-dimensional convolutional neural network. Clin. Oral Investig. 2022, 26, 3987–3998. [Google Scholar] [CrossRef]
- Hung, K.F.; Hui, L.; Yeung, A.W.K.; Jacobs, R.; Leung, Y.Y.; Bornstein, M.M. An analysis of patient dose received during cone-beam computed tomography in relation to scan settings and imaging indications as seen in a dental institution in order to establish institutional diagnostic reference levels. Dentomaxillofac. Radiol. 2022, 51, 20200529. [Google Scholar] [CrossRef]
- Bornstein, M.M.; Yeung, A.W.K.; Tanaka, R.; von Arx, T.; Jacobs, R.; Khong, P.L. Evaluation of health or pathology of bilateral maxillary sinuses in patients referred for cone beam computed tomography using a low-dose protocol. Int. J. Periodontics Restor. Dent. 2018, 38, 699–710. [Google Scholar] [CrossRef]
- Wang, X.; Xu, Z.; Tong, Y.; Xia, L.; Jie, B.; Ding, P.; Bai, H.; Zhang, Y.; He, Y. Detection and classification of mandibular fracture on CT scan using deep convolutional neural network. Clin. Oral Investig. 2022, 26, 4593–4601. [Google Scholar] [CrossRef]
- Apostolakis, D.; Michelinakis, G.; Kamposiora, P.; Papavasiliou, G. The current state of computer assisted orthognathic surgery: A narrative review. J. Dent. 2022, 119, 104052. [Google Scholar] [CrossRef]
- Shujaat, S.; Bornstein, M.M.; Price, J.B.; Jacobs, R. Integration of imaging modalities in digital dental workflows—Possibilities, limitations, and potential future developments. Dentomaxillofac. Radiol. 2021, 50, 20210268. [Google Scholar] [CrossRef]
- Lo Giudice, A.; Ronsivalle, V.; Spampinato, C.; Leonardi, R. Fully automatic segmentation of the mandible based on convolutional neural networks (CNNs). Orthod. Craniofac. Res. 2021, 24 (Suppl. S2), 100–107. [Google Scholar] [CrossRef]
- Preda, F.; Morgan, N.; Van Gerven, A.; Nogueira-Reis, F.; Smolders, A.; Wang, X.; Nomidis, S.; Shaheen, E.; Willems, H.; Jacobs, R. Deep convolutional neural network-based automated segmentation of the maxillofacial complex from cone-beam computed tomography: A validation study. J. Dent. 2022, 124, 104238. [Google Scholar] [CrossRef]
- Ezhov, M.; Gusarev, M.; Golitsyna, M.; Yates, J.M.; Kushnerev, E.; Tamimi, D.; Aksoy, S.; Shumilov, E.; Sanders, A.; Orhan, K. Clinically applicable artificial intelligence system for dental diagnosis with CBCT. Sci. Rep. 2021, 11, 15006. [Google Scholar] [CrossRef]
- Sin, Ç.; Akkaya, N.; Aksoy, S.; Orhan, K.; Öz, U. A deep learning algorithm proposal to automatic pharyngeal airway detection and segmentation on CBCT images. Orthod. Craniofac. Res. 2021, 24 (Suppl. S2), 117–123. [Google Scholar] [CrossRef]
- Orhan, K.; Shamshiev, M.; Ezhov, M.; Plaksin, A.; Kurbanova, A.; Ünsal, G.; Gusarev, M.; Golitsyna, M.; Aksoy, S.; Mısırlı, M.; et al. AI-based automatic segmentation of craniomaxillofacial anatomy from CBCT scans for automatic detection of pharyngeal airway evaluations in OSA patients. Sci. Rep. 2022, 12, 11863. [Google Scholar] [CrossRef]
- Jaskari, J.; Sahlsten, J.; Järnstedt, J.; Mehtonen, H.; Karhu, K.; Sundqvist, O.; Hietanen, A.; Varjonen, V.; Mattila, V.; Kaski, K. Deep learning method for mandibular canal segmentation in dental cone beam computed tomography volumes. Sci. Rep. 2020, 10, 5842. [Google Scholar] [CrossRef]
- Lim, H.K.; Jung, S.K.; Kim, S.H.; Cho, Y.; Song, I.S. Deep semi-supervised learning for automatic segmentation of inferior alveolar nerve using a convolutional neural network. BMC Oral Health 2021, 21, 630. [Google Scholar] [CrossRef]
- Xu, J.; Liu, J.; Zhang, D.; Zhou, Z.; Zhang, C.; Chen, X. A 3D segmentation network of mandible from CT scan with combination of multiple convolutional modules and edge supervision in mandibular reconstruction. Comput. Biol. Med. 2021, 138, 104925. [Google Scholar] [CrossRef]
- Jang, T.J.; Yun, H.S.; Kim, J.E.; Lee, S.H.; Seo, J.K. Fully automatic integration of dental CBCT images and full-arch intraoral impressions with stitching error correction via individual tooth segmentation and identification. arXiv 2021, arXiv:2112.01784. [Google Scholar]
- Chung, M.; Lee, J.; Song, W.; Song, Y.; Yang, L.-H.; Lee, J.; Shin, Y.-G. Automatic registration between dental cone-beam CT and scanned surface via deep pose regression neural networks and clustered similarities. IEEE Trans. Med. Imaging 2020, 39, 3900–3909. [Google Scholar] [CrossRef]
- Kim, I.; Misra, D.; Rodriguez, L.; Gill, M.; Liberton, D.K.; Almpani, K.; Lee, J.S.; Antani, S. Malocclusion classification on 3D cone-beam CT craniofacial images using multi-channel deep learning models. In Proceedings of the 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Montreal, QC, Canada, 20–24 July 2020; pp. 1294–1298. [Google Scholar]
- ter Horst, R.; van Weert, H.; Loonen, T.; Bergé, S.; Vinayahalingam, S.; Baan, F.; Maal, T.; de Jong, G.; Xi, T. Three-dimensional virtual planning in mandibular advancement surgery: Soft tissue prediction based on deep learning. J. Craniomaxillofac. Surg. 2021, 49, 775–782. [Google Scholar] [CrossRef]
- Ma, Q.; Kobayashi, E.; Fan, B.; Hara, K.; Nakagawa, K.; Masamune, K.; Sakuma, I.; Suenaga, H. Machine-learning-based approach for predicting postoperative skeletal changes for orthognathic surgical planning. Int. J. Med. Robot. 2022, 18, e2379. [Google Scholar] [CrossRef]
- Lin, H.H.; Chiang, W.C.; Yang, C.T.; Cheng, C.T.; Zhang, T.; Lo, L.J. On construction of transfer learning for facial symmetry assessment before and after orthognathic surgery. Comput. Methods Programs Biomed. 2021, 200, 105928. [Google Scholar] [CrossRef]
- Nishiyama, M.; Ishibashi, K.; Ariji, Y.; Fukuda, M.; Nishiyama, W.; Umemura, M.; Katsumata, A.; Fujita, H.; Ariji, E. Performance of deep learning models constructed using panoramic radiographs from two hospitals to diagnose fractures of the mandibular condyle. Dentomaxillofac. Radiol. 2021, 50, 20200611. [Google Scholar] [CrossRef]
- Krois, J.; Cantu, A.G.; Chaurasia, A.; Patil, R.; Chaudhari, P.K.; Gaudin, R.; Gehrung, S.; Schwendicke, F. Generalizability of deep learning models for dental image analysis. Sci. Rep. 2021, 11, 6102. [Google Scholar] [CrossRef]
- Rischke, R.; Schneider, L.; Müller, K.; Samek, W.; Schwendicke, F.; Krois, J. Federated learning in dentistry: Chances and challenges. J. Dent. Res. 2022, 101, 1269–1273. [Google Scholar] [CrossRef]
- Roy, S.; Meena, T.; Lim, S.J. Demystifying supervised learning in healthcare 4.0: A new reality of transforming diagnostic medicine. Diagnostics 2022, 12, 2549. [Google Scholar] [CrossRef]
- Pfaehler, E.; Zhovannik, I.; Wei, L.; Boellaard, R.; Dekker, A.; Monshouwer, R.; El Naqa, I.; Bussink, J.; Gillies, R.; Wee, L.; et al. A systematic review and quality of reporting checklist for repeatability and reproducibility of radiomic features. Phys. Imaging Radiat. Oncol. 2021, 20, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Xue, C.; Yuan, J.; Lo, G.G.; Chang, A.T.Y.; Poon, D.M.C.; Wong, O.L.; Zhou, Y.; Chu, W.C.W. Radiomics feature reliability assessed by intraclass correlation coefficient: A systematic review. Quant. Imaging Med. Surg. 2021, 11, 4431–4460. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Ai, Q.Y.H.; Wong, L.M.; Green, C.; Qamar, S.; So, T.Y.; Vlantis, A.C.; King, A.D. Radiomics for discriminating benign and malignant salivary gland tumors; which radiomic feature categories and MRI sequences should be used? Cancers 2022, 14, 5804. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhou, Y.; Wang, X.; Zhang, Y.; Ma, C.; Liu, B.; Kong, Q.; Yue, N.; Xu, Z.; Nie, K. Reproducibility and repeatability of CBCT-derived radiomics features. Front. Oncol. 2021, 11, 773512. [Google Scholar] [CrossRef]
- Wong, L.M.; Ai, Q.Y.H.; Zhang, R.; Mo, F.; King, A.D. Radiomics for discrimination between early-stage nasopharyngeal carcinoma and benign hyperplasia with stable feature selection on MRI. Cancers 2022, 14, 3433. [Google Scholar] [CrossRef]
- Bagher-Ebadian, H.; Siddiqui, F.; Liu, C.; Movsas, B.; Chetty, I.J. On the impact of smoothing and noise on robustness of CT and CBCT radiomics features for patients with head and neck cancers. Med. Phys. 2017, 44, 1755–1770. [Google Scholar] [CrossRef]
- Sellami, S.; Bourbonne, V.; Hatt, M.; Tixier, F.; Bouzid, D.; Lucia, F.; Pradier, O.; Goasduff, G.; Visvikis, D.; Schick, U. Predicting response to radiotherapy of head and neck squamous cell carcinoma using radiomics from cone-beam CT images. Acta Oncol. 2022, 61, 73–80. [Google Scholar] [CrossRef]
- Foy, J.J.; Robinson, K.R.; Li, H.; Giger, M.L.; Al-Hallaq, H.; Armato, S.G. Variation in algorithm implementation across radiomics software. J. Med. Imaging (Bellingham) 2018, 5, 044505. [Google Scholar] [CrossRef]
- Bianchi, J.; Gonçalves, J.R.; Ruellas, A.C.O.; Vimort, J.B.; Yatabe, M.; Paniagua, B.; Hernandez, P.; Benavides, E.; Soki, F.N.; Cevidanes, L.H.S. Software comparison to analyze bone radiomics from high resolution CBCT scans of mandibular condyles. Dentomaxillofac. Radiol. 2019, 48, 20190049. [Google Scholar] [CrossRef]
- Zwanenburg, A.; Vallières, M.; Abdalah, M.A.; Aerts, H.J.W.L.; Andrearczyk, V.; Apte, A.; Ashrafinia, S.; Bakas, S.; Beukinga, R.J.; Boellaard, R.; et al. The image biomarker standardization initiative: Standardized quantitative radiomics for high-throughput image-based phenotyping. Radiology 2020, 295, 328–338. [Google Scholar] [CrossRef]
- Hung, K.F.; Yeung, A.W.K.; Bornstein, M.M.; Schwendicke, F. Personalized dental medicine, artificial intelligence, and their relevance for dentomaxillofacial imaging. Dentomaxillofac. Radiol. 2023, 52, 20220335. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hung, K.F.; Ai, Q.Y.H.; Wong, L.M.; Yeung, A.W.K.; Li, D.T.S.; Leung, Y.Y. Current Applications of Deep Learning and Radiomics on CT and CBCT for Maxillofacial Diseases. Diagnostics 2023, 13, 110. https://doi.org/10.3390/diagnostics13010110
Hung KF, Ai QYH, Wong LM, Yeung AWK, Li DTS, Leung YY. Current Applications of Deep Learning and Radiomics on CT and CBCT for Maxillofacial Diseases. Diagnostics. 2023; 13(1):110. https://doi.org/10.3390/diagnostics13010110
Chicago/Turabian StyleHung, Kuo Feng, Qi Yong H. Ai, Lun M. Wong, Andy Wai Kan Yeung, Dion Tik Shun Li, and Yiu Yan Leung. 2023. "Current Applications of Deep Learning and Radiomics on CT and CBCT for Maxillofacial Diseases" Diagnostics 13, no. 1: 110. https://doi.org/10.3390/diagnostics13010110
APA StyleHung, K. F., Ai, Q. Y. H., Wong, L. M., Yeung, A. W. K., Li, D. T. S., & Leung, Y. Y. (2023). Current Applications of Deep Learning and Radiomics on CT and CBCT for Maxillofacial Diseases. Diagnostics, 13(1), 110. https://doi.org/10.3390/diagnostics13010110











