Autoimmune Connective Tissue Diseases-Related Pruritus: Proper Diagnosis and Possible Mechanisms
Abstract
:1. Introduction
2. Dermatomyositis (DM)
2.1. Prevalence and Characteristics of Pruritus
2.2. Mechanisms and Treatments
3. Cutaneous Lupus Erythematosus (CLE)
3.1. Prevelance and Characteristics of Pruritus
3.2. Mechanisms and Treatments
4. Systemic Sclerosis (SSc)
4.1. Prevalence and Charateristics of Pruritus
4.2. Mechanisms and Treatments
5. Morphea
5.1. Prevalance and Characteristics of Pruritus
5.2. Mechanisms and Treatments
6. Sjögren Syndrome (SS)
6.1. Prevalence and Characteristics of Pruritus
6.2. Mechanisms and Treatments
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Kahremany, S.; Hofmann, L.; Gruzman, A.; Cohen, G. Advances in understanding the initial steps of pruritoceptive itch: How the itch hits the switch. Int. J. Mol. Sci. 2020, 21, 4883. [Google Scholar] [CrossRef] [PubMed]
- Fowler, E.; Yosipovitch, G. A new generation of treatments for itch. Acta Derm. Venereol. 2020, 100, 37–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Argobi, Y.; Smith, G.P. Characterizing pruritus in autoimmune connective tissue diseases. J. Drugs. Dermatol. 2019, 18, 995–998. [Google Scholar]
- Das, S.; Bernstein, I.; Jacobe, H. Correlates of self-reported quality of life in adults and children with morphea. J. Am. Acad. Dermatol. 2014, 70, 904–910. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bali, G.; Kárpáti, S.; Sárdy, M.; Brodszky, V.; Hidvégi, B.; Rencz, F. Association between quality of life and clinical characteristics in patients with morphea. Qual. Life Res. 2018, 27, 2525–2532. [Google Scholar] [CrossRef] [PubMed]
- DeWane, M.E.; Waldman, R.; Lu, J. Dermatomyositis: Clinical features and pathogenesis. J. Am. Acad. Dermatol. 2020, 82, 267–281. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J. Pruritus in autoimmune connective tissue diseases. Ann. Transl. Med. 2021, 9, 441. [Google Scholar] [CrossRef]
- Shirani, Z.; Kucenic, M.; Carroll, C.; Fleischer, A., Jr.; Feldman, S.; Yosipovitch, G.; Jorizzo, J. Pruritus in adult dermatomyositis. Clin. Exp. Dermatol. 2004, 29, 273–276. [Google Scholar] [CrossRef]
- Goreshi, R.; Chock, M.; Foering, K.; Feng, R.; Okawa, J.; Rose, M.; Fiorentino, D.; Werth, V. Quality of life in dermatomyositis. J. Am. Acad. Dermatol. 2011, 65, 1107–1116. [Google Scholar] [CrossRef] [Green Version]
- Yosipovitch, G.; Tan, A.; LoSicco, K.; Manabat, C.G.; Kannagra, A.; Carroll, C.; Chan, Y.H.; Ng, P.; Jorizzo, J. A comparative study of clinical characteristics, work-up, treatment, and association to malignancy in dermatomyositis between two tertiary skin centers in the USA and singapore. Int. J. Dermatol. 2013, 52, 813–819. [Google Scholar] [CrossRef]
- Kim, H.J.; Zeidi, M.; Bonciani, D.; Pena, S.M.; Tiao, J.; Sahu, S.; Werth, V.P. Itch in dermatomyositis: The role of increased skin interleukin-31. Br. J. Dermatol. 2018, 179, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Kabashima, K.; Irie, H. Interleukin-31 as a clinical target for pruritus treatment. Front. Med. 2021, 8, 638325. [Google Scholar] [CrossRef] [PubMed]
- Datsi, A.; Steinhoff, M.; Ahmad, F.; Alam, M.; Buddenkotte, J. Interleukin-31: The “itchy” cytokine in inflammation and therapy. Allergy 2021, 76, 2982–2997. [Google Scholar] [CrossRef] [PubMed]
- Hurliman, E.; Groth, D.; Wendelschafer-Crabb, G.; Kennedy, W.; Kavand, S.; Ericson, M.; Hordinsky, M. Small-fibre neuropathy in a patient with dermatomyositis and severe scalp pruritus. Br. J. Dermatol. 2016, 176, 209–211. [Google Scholar] [CrossRef] [Green Version]
- Raasing, L.R.M.; Vogels, O.J.M.; Veltkamp, M.; van Swol, C.F.P.; Grutters, J.C. Current view of diagnosing small fiber neuropathy. J. Neuromuscul. Dis. 2021, 8, 185–207. [Google Scholar] [CrossRef]
- Ständer, S.; Weisshaar, E.; Mettang, T.; Szepietowski, J.C.; Carstens, E.; Ikoma, A.; Bergasa, N.V.; Gieler, U.; Misery, L.; Wallengren, J.; et al. Clinical classification of itch: A position paper of the international forum for the study of itch. Acta Derm. Venereol. 2007, 87, 291–294. [Google Scholar] [CrossRef] [Green Version]
- Pereira, M.P.; Wiegmann, H.; Agelopoulos, K.; Ständer, S. Neuropathic itch: Routes to clinical diagnosis. Front. Med. 2021, 8, 641746. [Google Scholar] [CrossRef]
- Nguyen, T.P.; Bangert, C.; Biliciler, S.; Athar, P.; Sheikh, K. Dermatomyositis-associated sensory neuropathy: A unifying pathogenic hypothesis. J. Clin. Neuromuscul. Dis. 2014, 16, 7–11. [Google Scholar] [CrossRef]
- Kabashima, K.; Matsumura, T.; Komazaki, H.; Kawashima, M. Trial of nemolizumab and topical agents for atopic dermatitis with pruritus. N. Engl. J. Med. 2020, 383, 141–150. [Google Scholar] [CrossRef]
- Werth, V.P.; Hejazi, E.; Pena, S.M.; Haber, J.; Zeidi, M.; Reddy, N.; Okawa, J.; Feng, R.; Bashir, M.M.; Gebre, K.; et al. Safety and efficacy of lenabasum, a cannabinoid receptor type 2 agonist, in dermatomyositis patients with refractory skin disease a randomized clinical trial. J. Investig. Dermatol. 2022; online ahead of print. [Google Scholar] [CrossRef]
- Charlton, D.; Moghadam-Kia, S.; Smith, K.; Aggarwal, R.; English, J.R.; Oddis, C. Refractory cutaneous dermatomyositis with severe scalp pruritus responsive to apremilast. J. Clin. Rheumatol. 2021, 27, S561–S562. [Google Scholar] [CrossRef]
- Méndez-Flores, S.; Orozco-Topete, R.; Bermúdez-Bermejo, P.; Hernández-Molina, G. Pain and pruritus in cutaneous lupus: Their association with dermatologic quality of life and disease activity. Clin. Exp. Rheumatol. 2013, 31, 940–942. [Google Scholar] [PubMed]
- Samotij, D.; Szczęch, J.; Kushner, C.J.; Mowla, M.R.; Dańczak-Pazdrowska, A.; Antiga, E.; Chasset, F.; Furukawa, F.; Hasegawa, M.; Hashizume, H.; et al. Prevalence of pruritus in cutaneous lupus erythematosus: Brief report of a multicenter, multinational cross-sectional study. BioMed Res. Int. 2018, 2018, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Samotij, D.; Szczęch, J.; Antiga, E.; Bonciani, D.; Caproni, M.; Chasset, F.; Dańczak-Pazdrowska, A.; Furukawa, F.; Hasegawa, M.; Hashizume, H.; et al. Clinical characteristics of itch in cutaneous lupus erythematosus: A prospective, multicenter, multinational, cross-sectional study. Lupus 2021, 30, 1385–1393. [Google Scholar] [CrossRef]
- Tekatas, A.; Tekatas, D.D.; Solmaz, V.; Karaca, T.; Pamuk, O.N. Small fiber neuropathy and related factors in patients with systemic lupus erythematosus; the results of cutaneous silent period and skin biopsy. Adv. Rheumatol. 2020, 60, 31. [Google Scholar] [CrossRef]
- González Sánchez, M.; Llorente Ayuso, L.; Ruiz Morales, J.; Martínez Salio, A. Neuropathic pruritus as the presenting symptom of lupus transverse myelitis. J. Neurol. Sci. 2016, 368, 138–139. [Google Scholar] [CrossRef] [PubMed]
- Konda, D.; Chandrashekar, L.; Rajappa, M.; Kattimani, S.; Thappa, D.M.; Ananthanarayanan, P.H. Serotonin and interleukin-6: Association with pruritus severity, sleep quality and depression severity in prurigo nodularis. Asian J. Psychiatry 2015, 17, 24–28. [Google Scholar] [CrossRef] [PubMed]
- Keshari, S.; Sipayung, A.D.; Hsieh, C.C.; Su, L.J.; Chiang, Y.R.; Chang, H.C.; Yang, W.C.; Chuang, T.H.; Chen, C.L.; Huang, C.M. IL-6/p-BTK/p-ERK signaling mediates calcium phosphate-induced pruritus. FASEB J. 2019, 33, 12036–12046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rincón, M.; Anguita, J.; Nakamura, T.; Fikrig, E.; Flavell, R.A. Interleukin (IL)-6 directs the differentiation of IL-4-producing CD4+ T cells. J. Exp. Med. 1997, 185, 461–469. [Google Scholar] [CrossRef] [Green Version]
- Huang, H.-T.; Chen, J.-M.; Guo, J.; Lan, Y.; Wei, Y.-S. The association of interleukin-31 polymorphisms with interleukin-31 serum levels and risk of systemic lupus erythematosus. Rheumatol. Int. 2016, 36, 799–805. [Google Scholar] [CrossRef]
- Guo, J.; Xiang, Y.; Peng, Y.-F.; Huang, H.-T.; Lan, Y.; Wei, Y.-S. The association of novel IL-33 polymorphisms with sIL-33 and risk of systemic lupus erythematosus. Mol. Immunol. 2016, 77, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Xu, W.D.; Zhu, Y.; Wen, P.F.; Leng, R.X.; Pan, H.F.; Ye, D.Q. Serum levels of cytokines in systemic lupus erythematosus. Z. Fur Rheumatol. 2013, 73, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Wong, L.-S.; Yen, Y.-T.; Lee, C.-H. The implications of pruritogens in the pathogenesis of atopic dermatitis. Int. J. Mol. Sci. 2021, 22, 7227. [Google Scholar] [CrossRef] [PubMed]
- Holme, S.A.; Holmes, S.C. Hydroxychloroquine-induced pruritus. Acta Derm. Venereol. 1999, 79, 333. [Google Scholar] [PubMed] [Green Version]
- Ajayi, A.A.L. Itching, chloroquine, and malaria: A review of recent molecular and neuroscience advances and their contribution to mechanistic understanding and therapeutics of chronic non-histaminergic pruritus. Int. J. Dermatol. 2018, 58, 880–891. [Google Scholar] [CrossRef] [PubMed]
- Pattanaik, D.; Brown, M.; Postlethwaite, B.C.; Postlethwaite, A.E. Pathogenesis of systemic sclerosis. Front. Immunol. 2015, 6, 272. [Google Scholar] [CrossRef] [Green Version]
- Pearson, D.R.; Werth, V.P.; Pappas-Taffer, L. Systemic sclerosis: Current concepts of skin and systemic manifestations. Clin. Dermatol. 2018, 36, 459–474. [Google Scholar] [CrossRef]
- Bassel, M.; Hudson, M.; Taillefer, S.S.; Schieir, O.; Baron, M.; Thombs, B.D. Frequency and impact of symptoms experienced by patients with systemic sclerosis: Results from a canadian national survey. Rheumatology 2010, 50, 762–767. [Google Scholar] [CrossRef] [Green Version]
- Razykov, I.; Thombs, B.D.; Hudson, M.; Bassel, M.; Baron, M. Prevalence and clinical correlates of pruritus in patients with systemic sclerosis. Arthritis Rheumatol. 2009, 61, 1765–1770. [Google Scholar] [CrossRef]
- El-Baalbaki, G.; Razykov, I.; Hudson, M.; Bassel, M.; Baron, M.; Thombs, B.D. Association of pruritus with quality of life and disability in systemic sclerosis. Arthritis Care Res. 2010, 62, 1489–1495. [Google Scholar] [CrossRef]
- Razykov, I.; Levis, B.; Hudson, M.; Baron, M.; Thombs, B.D. Prevalence and clinical correlates of pruritus in patients with systemic sclerosis: An updated analysis of 959 patients. Rheumatology 2013, 52, 2056–2061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stull, C.; Weaver, L.; Valdes-Rodriguez, R.; Naramala, S.; Lavery, M.; Chan, Y.; Mendoza, F.; Yosipovitch, G. Characteristics of chronic itch in systemic sclerosis: A cross-sectional survey. Acta Derm. Venereol. 2018, 98, 793–794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Théréné, C.; Brenaut, E.; Sonbol, H.; Pasquier, E.; Saraux, A.; Devauchelle, V.; Le Moigne, E.; Misery, L.; Abasq-Thomas, C. Itch and systemic sclerosis: Frequency, clinical characteristics and consequences. Br. J. Dermatol. 2017, 176, 1392–1393. [Google Scholar] [CrossRef] [PubMed]
- Gourier, G.; Théréné, C.; Mazeas, M.; Abasq-Thomas, C.; Brenaut, E.; Huet, F.; Sonbol, H.; Campillo, E.; Lemerle, J.; Pasquier, E.; et al. Clinical characteristics of pruritus in systemic sclerosis vary according to the autoimmune subtype. Acta Derm. Venereol. 2018, 98, 735–741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Racine, M.; Hudson, M.; Baron, M.; Nielson, W.R.; Pope, J.; Markland, J.; Robinson, D.; Jones, N.; Khalidi, N.; Docherty, P.; et al. The impact of pain and itch on functioning and health-related quality of life in systemic sclerosis: An exploratory study. J. Pain Symptom Manag. 2016, 52, 43–53. [Google Scholar] [CrossRef] [Green Version]
- Milette, K.; Hudson, M.; Korner, A.; Baron, M.; Thombs, B.D. Sleep disturbances in systemic sclerosis: Evidence for the role of gastrointestinal symptoms, pain and pruritus. Rheumatology 2013, 52, 1715–1720. [Google Scholar] [CrossRef] [Green Version]
- Anzelc, M.; Burkhart, C.G. Pain and pruritus: A study of their similarities and differences. Int. J. Dermatol. 2019, 59, 159–164. [Google Scholar] [CrossRef]
- Giuggioli, D.; Lumetti, F.; Colaci, M.; Fallahi, P.; Antonelli, A.; Ferri, C. Rituximab in the treatment of patients with systemic sclerosis. Our experience and review of the literature. Autoimmun. Rev. 2015, 14, 1072–1078. [Google Scholar] [CrossRef]
- Denton, C.P.; Hughes, M.; Gak, N.; Vila, J.; Buch, M.H.; Chakravarty, K.; Fligelstone, K.; Gompels, L.L.; Griffiths, B.; Herrick, A.L.; et al. BSR and BHPR guideline for the treatment of systemic sclerosis. Rheumatology 2016, 55, 1906–1910. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, E.; Lim, G.; Ross, S.E. Evaluation of therapies for peripheral and neuraxial opioid-induced pruritus based on molecular and cellular discoveries. Anesthesiology 2021, 135, 350–365. [Google Scholar] [CrossRef]
- Frech, T.; Novak, K.; Revelo, M.P.; Murtaugh, M.; Markewitz, B.; Hatton, N.; Scholand, M.B.; Frech, E.; Markewitz, D.; Sawitzke, A.D. Low-dose naltrexone for pruritus in systemic sclerosis. Int. J. Rheumatol. 2011, 2011, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Bhattacharyya, S.; Marangoni, R.G.; Carns, M.; Dennis-Aren, K.; Yeldandi, A.; Wei, J.; Varga, J. The JAK/STAT pathway is activated in systemic sclerosis and is effectively targeted by tofacitinib. J. Scleroderma Relat. Disord 2019, 5, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Junttila, I.S. Tuning the cytokine responses: An update on interleukin (IL)-4 and IL-13 receptor complexes. Front. Immunol. 2018, 9, 888. [Google Scholar] [CrossRef] [PubMed]
- Oetjen, L.K.; Mack, M.R.; Feng, J.; Whelan, T.M.; Niu, H.; Guo, C.J.; Chen, S.; Trier, A.M.; Xu, A.Z.; Tripathi, S.V.; et al. Sensory neurons co-opt classical immune signaling pathways to mediate chronic itch. Cell 2017, 171, 217–228.e213. [Google Scholar] [CrossRef] [Green Version]
- Ju, T.; Labib, A.; Vander Does, A.; Yosipovitch, G. Topical janus kinase-signal transducers and activators of transcription inhibitor tofacitinib is effective in reducing nonatopic dermatitis chronic itch: A case series. J. Am. Acad. Dermatol. 2022, 87, 400–403, online ahead of print. [Google Scholar] [CrossRef]
- Abbas, L.; Joseph, A.; Kunzler, E.; Jacobe, H.T. Morphea: Progress to date and the road ahead. Ann. Transl. Med. 2021, 9, 437. [Google Scholar] [CrossRef]
- Lis-Święty, A.; Skrzypek-Salamon, A.; Ranosz-Janicka, I.; Brzezińska-Wcisło, L. Associations between disease activity/severity and damage and health-related quality of life in adult patients with localized scleroderma—A comparison of loscat and visual analogue scales. J. Clin. Med. 2020, 9, 756. [Google Scholar] [CrossRef] [Green Version]
- Lis-Święty, A.; Skrzypek-Salamon, A.; Ranosz-Janicka, I.; Brzezińska-Wcisło, L. Health-related quality of life and its influencing factors in adult patients with localized scleroderma—A cross-sectional study. Health Qual. Life Outcomes 2020, 18, 1–10. [Google Scholar] [CrossRef]
- Kroft, E.B.M.; Jong, E.M.G.J.d.; Evers, A.W.M. Psychological distress in patients with morphea and eosinophilic fasciitis. Arch. Dermatol. 2009, 145, 1017–1022. [Google Scholar] [CrossRef] [Green Version]
- Narbutt, J.; Hołdrowicz, A.; Lesiak, A. Morphea—Selected local treatment methods and their effectiveness. Reumatologia 2017, 55, 305–313. [Google Scholar] [CrossRef] [Green Version]
- Frez, M.L.F.; Eusebio-Alpapara, K.M.V.; Pastorfide, G.C.; Castillo, R.L.; Tioleco, G.S. Treatment of morphea with narrowband ultraviolet b: A case series. J. Phil. Dermatol. Soc. 2019, 11, 36–50. [Google Scholar]
- Kreuter, A.; Hyun, J.; Stücker, M.; Sommer, A.; Altmeyer, P.; Gambichler, T. A randomized controlled study of low-dose UVA1, medium-dose UVA1, and narrowband UVB phototherapy in the treatment of localized scleroderma. J. Am. Acad. Dermatol. 2006, 54, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Legat, F.J. The antipruritic effect of phototherapy. Front. Med. 2018, 5, 333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stausbøl-Grøn, B.; Olesen, A.; Deleuran, B.; Deleuran, M. Abatacept is a promising treatment for patients with disseminated morphea profunda: Presentation of two cases. Acta Derm. Venereol. 2011, 91, 686–688. [Google Scholar] [CrossRef] [Green Version]
- Negrini, S.; Emmi, G.; Greco, M.; Borro, M.; Sardanelli, F.; Murdaca, G.; Indiveri, F.; Puppo, F. Sjögren’s syndrome: A systemic autoimmune disease. Clin. Exp. Med. 2021, 22, 9–25. [Google Scholar] [CrossRef]
- Bernacchi, E.; Amato, L.; Parodi, A.; Cottoni, F.; Rubegni, P.; Pitá, O.D.; Papini, M.; Rebora, A.; Bombardieri, S.; Fabbri, P. Sjögren’s syndrome: A retrospective review of the cutaneous features of 93 patients by the italian group of immunodermatology. Clin. Exp. Rheumatol. 2004, 22, 55–62. [Google Scholar]
- Valdes-Rodriguez, R.; Rowe, B.; Lee, H.; Moldovan, T.; Chan, Y.; Blum, M.; Yosipovitch, G. Chronic pruritus in primary sjögren’s syndrome: Characteristics and effect on quality of life. Acta Derm. Venereol. 2017, 97, 385–386. [Google Scholar] [CrossRef] [Green Version]
- Katayama, I.; Yokozeki, H.; Nishioka, K. Impaired sweating as an exocrine manifestation in sjögren’s syndrome. Br. J. Dermatol. 1995, 133, 716–720. [Google Scholar] [CrossRef]
- Bernacchi, E.; Bianchi, B.; Amato, L.; Tavoni, A.; Giorgini, S.; Bombardieri, S.; Fabbri, P. Xerosis in primary sjögren syndrome: Immunohistochemical and functional investigations. J. Dermatol. Sci. 2005, 39, 53–55. [Google Scholar] [CrossRef]
- Parisis, D.; Chivasso, C.; Perret, J.; Soyfoo, M.S.; Delporte, C. Current state of knowledge on primary sjögren’s syndrome, an autoimmune exocrinopathy. J. Clin. Med. 2020, 9, 2299. [Google Scholar] [CrossRef]
- Descamps, E.; Henry, J.; Labeyrie, C.; Adams, D.; Ghaidaa, A.N.; Vandendries, C.; Adam, C.; Aiello, D.; Mariette, X.; Seror, R. Small fiber neuropathy in sjögren syndrome: Comparison with other small fiber neuropathies. Muscle Nerve 2019, 61, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Rosen, J.D.; Fostini, A.C.; Yosipovitch, G. Diagnosis and management of neuropathic itch. Dermatol. Clin. 2018, 36, 213–224. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Yang, Z.; Qin, B.; Zhong, R. Primary sjögren’s syndrome and malignancy risk: A systematic review and meta-analysis. Ann. Rheum. Dis. 2014, 73, 1151–1156. [Google Scholar] [CrossRef] [PubMed]
- Brito-Zerón, P.; Kostov, B.; Fraile, G.; Caravia-Durán, D.; Maure, B.; Rascón, F.-J.; Zamora, M.; Casanovas, A.; Lopez-Dupla, M.; Ripoll, M.; et al. Characterization and risk estimate of cancer in patients with primary sjögren syndrome. J. Hematol. Oncol. 2017, 10, 90. [Google Scholar] [CrossRef] [Green Version]
- Yosipovitch, G. Chronic pruritus: A paraneoplastic sign. Dermatol. Ther. 2010, 23, 590–596. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.; Kuruvilla, M.; Pai, G.; Dinesh, M. Cutaneous manifestations of non-Hodgkin’s lymphoma. Indian J. Dermatol. Venereol. Leprol. 2003, 69, 12–15. [Google Scholar]
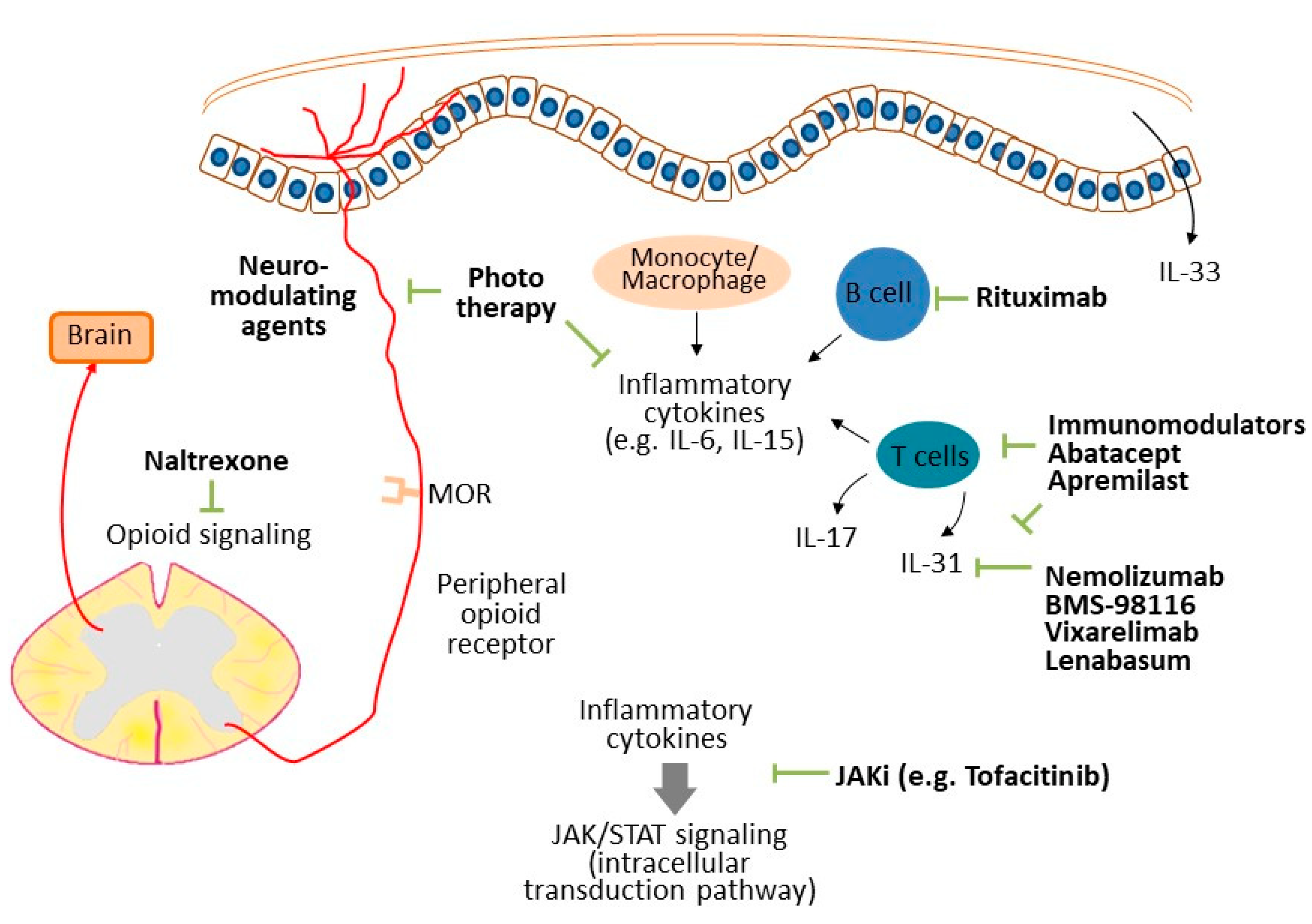
| Proposed Mechanisms | Potential Targeted Treatments |
|---|---|
| Dermatomyositis | |
| IL-31 [11] | Nemolizumab, BMS-98116, vixarelimab, lenabasum |
| Small-fiber neuropathy [14] | Neuro-modulating agents (e.g., gabapentin, pregabalin) |
| Inflammatory components | Tacrolimus [10], apremilast [21] |
| Cutaneous lupus erythematosus | |
| Small-fiber neuropathy [25] | Neuro-modulating agents (e.g., gabapentin, pregabalin) |
| Neuropathic itch [26] | Neuro-modulating agents (e.g., gabapentin, pregabalin) |
| IL-6 [25], IL-33 [30], IL-31 [31] | Topical corticosteroid, calcineurin inhibitor, methotrexate, azathioprine |
| Systemic sclerosis | |
| Xerosis [42] | Emollient [49] |
| IL-6, IL-15, IL-23, IL-17 [48] | Rituximab [48] |
| Opioid signaling | Naltrexone [51] |
| JAK/STAT signaling [52] | JAK inhibitor (e.g., tofacitinib) [52] |
| Morphea | |
| Inflammatory components | Phototherapy [61,62], abatacept [64] |
| Sjögren syndrome | |
| Xerosis [67] | Emollient |
| Small-fiber neuropathy [71] | Neuro-modulating agents (e.g., gabapentin, pregabalin) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wong, L.-S.; Yen, Y.-T. Autoimmune Connective Tissue Diseases-Related Pruritus: Proper Diagnosis and Possible Mechanisms. Diagnostics 2022, 12, 1772. https://doi.org/10.3390/diagnostics12071772
Wong L-S, Yen Y-T. Autoimmune Connective Tissue Diseases-Related Pruritus: Proper Diagnosis and Possible Mechanisms. Diagnostics. 2022; 12(7):1772. https://doi.org/10.3390/diagnostics12071772
Chicago/Turabian StyleWong, Lai-San, and Yu-Ta Yen. 2022. "Autoimmune Connective Tissue Diseases-Related Pruritus: Proper Diagnosis and Possible Mechanisms" Diagnostics 12, no. 7: 1772. https://doi.org/10.3390/diagnostics12071772
APA StyleWong, L.-S., & Yen, Y.-T. (2022). Autoimmune Connective Tissue Diseases-Related Pruritus: Proper Diagnosis and Possible Mechanisms. Diagnostics, 12(7), 1772. https://doi.org/10.3390/diagnostics12071772






