1. Introduction
Mural thrombus of the aorta is usually diagnosed by cross-sectional imaging in patients suffering from peripheral arterial embolism or as an incidental finding in asymptomatic patients [
1]. In a large autopsy series, mural thrombus of the aorta was found in 0.45% of cases [
2]. In abdominal aortic aneurysms, mural thrombus can be observed much more frequently (>90% in large aneurysms with indication for repair [
3]. After endovascular aortic repair (EVAR), wall-adherent thrombus is also a common finding (up to 20% of patients), and is mainly detected within the first year after implantation [
4]. However, the absolute risk of peripheral embolization related to mural thrombus within an abdominal aortic aneurysm or an EVAR-prosthesis seems to be rather low [
4].
Mobile or floating thrombus adherent to the arterial vessel wall (here referred to as wall adherent floating arterial structures, WAFAS) has been reported less frequently, so far predominantly described in the descending thoracic aorta and the carotid arteries [
5,
6]. The actual risk of peripheral embolization of aortic WAFAS is poorly defined, but may be substantially higher than that of non-mobile mural thrombus [
7]. In about 0.2–1.3% of patients suffering from ischemic stroke, an ipsilateral carotid free-floating thrombus can be found [
5,
8].
Diagnostic imaging of WAFAS is challenging. Colour duplex sonography is the primary diagnostic tool for evaluation of the abdominal aorta and the peripheral arteries and offers high spatial resolution as well as dynamic information on the blood flow. Contrast-enhanced ultrasound (CEUS) with second generation contrast agents provides additional information with regard to the detection of slow blood flow, vessel wall perfusion and intraluminal filling defects. B-Flow (BFl) is a patented technology developed by General Electric Medical Systems, Milwaukee, WI, USA (GE) [
9]. Unlike colour duplex sonography, BFl is not based on the Doppler principle. By using tissue-blood equalization technology, BFl detects moving objects by displaying the difference between two pictures of the object taken at different time points. Consequently, BFl can detect motion within anatomical structures with a spatial resolution similar to that of gray scale imaging. The magnitude of difference (as determined by the time interval between pictures and the speed of the object) is correlated to the brightness of the signal, making this method potentially useful for the detection of WAFAS [
9,
10].
Against this background, the present study aimed to determine the potential diagnostic yield of BFl in the detection of WAFAS in various anatomical locations and to compare BFl with CEUS and computed tomography angiography (CTA).
2. Patients and Methods
Consecutive patients with a sonographic diagnosis of WAFAS in any arterial segment diagnosed between January 2016 and December 2021 were retrospectively identified. Patient’s records were screened for clinical characteristics, sonographic findings (including BFl and CEUS) and CTA performed for the diagnostic workup of these vascular pathologies.
Two experienced vascular sonographers (C.L. and M.C.) consented the WAFAS-diagnosis based on independent review of recorded movies of ultrasound examinations of the abdominal aorta, the supraaortic arteries and the extremity arteries, including BFl. A characteristic BFl-finding was discovered and its potential diagnostic role for detection of WAFAS was analyzed in comparison to established imaging modalities.
All ultrasound examinations were conducted with a GE LOGIQ E9 ultrasound machine using a 4–10 MHz linear transducer (for examination of the supraaortic and extremity arteries) and/or a 2–6 MHz convex probe (for examination of the abdominal aorta and the iliac arteries). BFl was performed in all patients for assessment of the floating intraluminal structures in the longitudinal and transversal plane. For BFl, flash reduction was set on zero by default. The pulse repetition interval was 10 (linear probe) and 18 (convex probe), respectively. On a detection speckle reduction imaging strength scale ranging from 0 to 4, the values were set to 1 (linear probe) and 2 (convex probe), respectively. When CEUS was performed (on the same of the BFL-study), machine settings were adjusted appropriately (low MI imaging, as defined previously [
11] and movies of the respective vascular segment were recorded after injection of 1.2 mL sulfur hexafluoride intravenously.
CTA images of the suspicious segments and, if indicated, of the upstream and/or downstream arterial tree, were independently reviewed by an experienced radiologist who was unaware of the clinical and sonographic data (D.P.-W.). Different CT scanners (Siemens SOMATOM Force, Siemens SOMATOM Definition AS+, GE Discovery CT750 HD, GE Optima CT660) were used for image acquisition with single (arterial) phase CTA or multiphasic CTA including an arterial phase. 1–1.5 mL/kg body weight iodinated contrast agent was administered at a flow rate of 3–5 mL/s followed by a 100 mL saline flush at 3–5 mL/s and the arterial phase images were acquired. The arterial phase was used for the assessment of wall adherent thrombus material.
Explorative data analysis was focussed on previous revascularization procedures and symptoms/signs of peripheral arterial embolism downstream to the vessel segments displaying the FLS. The most probable source of embolism in symptomatic patients suffering from arterial embolism was defined based on the results of cardiac diagnostics (Holter electrocardiogram, echocardiography) and sonographic and computed tomography imaging. Depending on the location of the WAFAS in the native abdominal aorta, in the limbs of EVAR prostheses, or in peripheral arteries (supraaortic arteries and extremity arteries) the cohort was divided into three subgroups. These were compared with regard to clinical and imaging findings.
3. Results
3.1. The Flashlight Sign and Cohort Characteristics
Twenty-eight patients were included in the analysis (85.7% men, mean age 72 ± 8.3 years), all of whom exhibited a BFl-based vascular ultrasound finding characterized by a fast moving, very bright intraluminal signal and indicating the presence of a WAFAS. Given the characteristic appearance, we termed this novel sonographic finding
flashlight sign (FLS) (
Figure 1,
Supplementary Video S1).
Seven patients in the cohort suffered from thromboembolic arterial events. In all of these patients the WAFAS, as visualized by the FLS in BFl, was deemed to be the most likely source of arterial embolism (
Table 1). In the remaining patients, the FLS was detected incidentally (
n = 7) or during routine surveillance following revascularization procedures (
n = 14). Characteristics of the subgroups with respect to the anatomic location of the FLS are outlined below and summarized in
Table 2.
Seventeen and thirteen out of 28 patients underwent CEUS and CTA, respectively. The mean time interval between the sonographic study (BFl with or without CEUS) and the CTA was 11.3 ± 15.2 days. When comparing BFl with CEUS and CTA, positive predictive values of the FLS for detection of WAFAS were 76.5% and 61.5%. However, there were remarkable differences between subgroups based on the affected arterial segments (native abdominal aorta, abdominal aorta after endovascular repair, peripheral arteries, see
Table 2).
3.2. Patients with Pathologies of the Native Abdominal Aorta
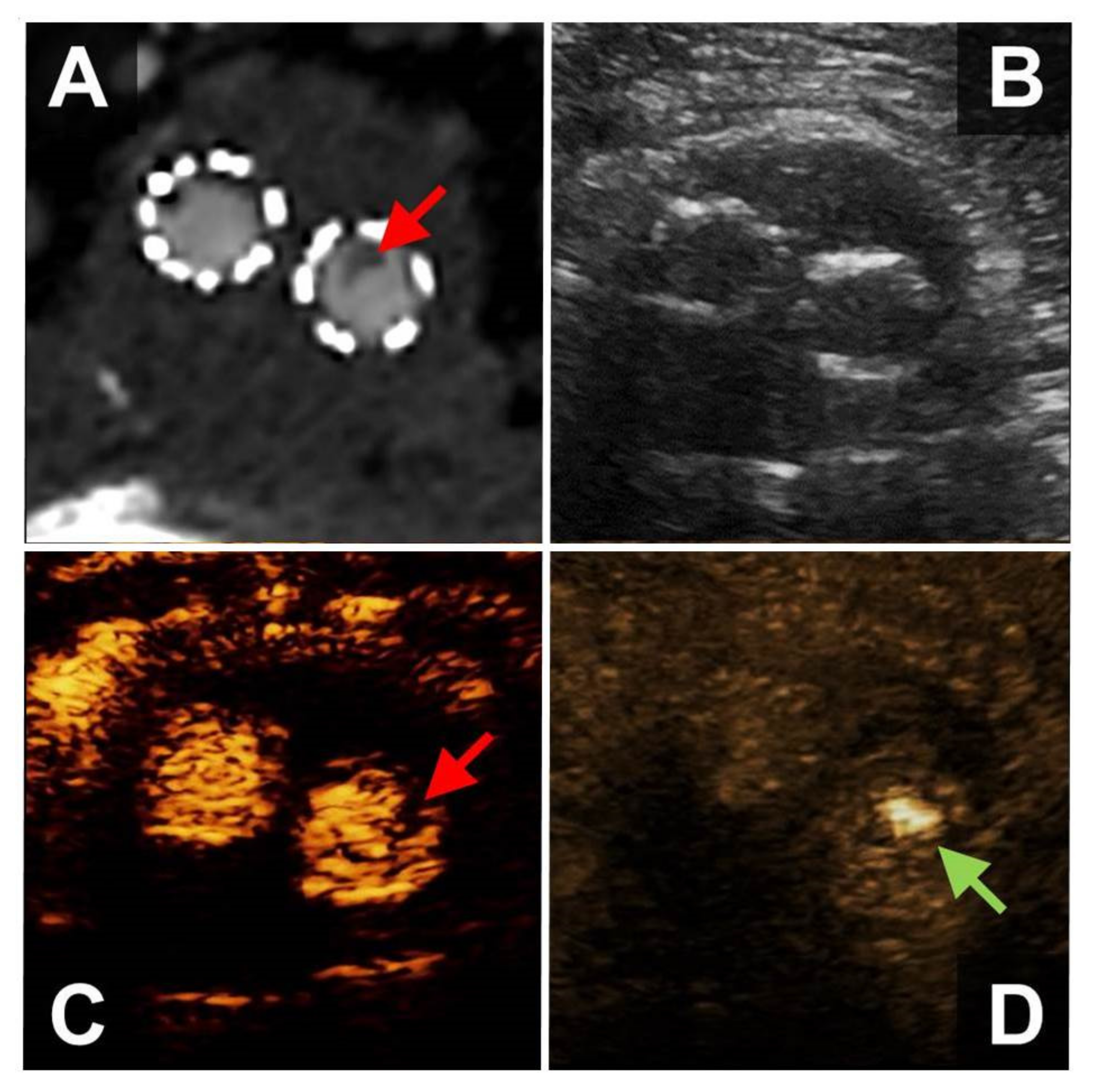
The FLS was detected in eight male patients in the native infrarenal aorta, three of whom had abdominal aortic aneurysms (
Figure 1,
Video S1). One of these patients suffered from unilateral embolic popliteal artery occlusion and in two patients the FLS was detected after preceding endovascular procedures involving catheter passage of the abdominal aorta. In four patients, the FLS was detected incidentally. Another female patient who had undergone the open repair of the infrarenal aorta decades ago presented with thrombotic material in the suprarenal aorta and bilateral renal artery stenoses; in this patient an FLS was detected incidentally in the suprarenal aorta.
CEUS-imaging was performed in only one patient but was negative regarding the presence of a WAFAS. CTA was performed in six patients, proving the presence of wall adherent intraluminal structures surrounded by contrast agent, indicative of WAFAS.
3.3. Patients after Endovascular Aortic Repair (EVAR)
The FLS was found in 10 male patients who had undergone EVAR of an infrarenal abdominal aortic aneurysm, four of whom presented with embolic occlusions of the popliteal and/or below the knee arteries. In six patients, the FLS was detected during routine surveillance after EVAR (
Figure 2,
Video S2).
The FLS was detected in the right and left EVAR limb in four patients, respectively, and two patients exhibited the FLS in both EVAR limbs. All embolic occlusions were located ipsilaterally to the FLS (three patients with unilateral embolism, one patient with bilateral FLS who developed metachronous bilateral popliteal artery embolism). In another patient who underwent endovascular thrombectomy for right-sided embolic popliteal artery occlusion, the FLS of both EVAR limbs disappeared after four weeks of anticoagulation with low molecular weight heparin. After discontinuation of low molecular weight heparin, the patient suffered from embolic re-occlusion of the right popliteal artery, and a prominent FLS corresponding to a large floating intraluminal thrombus was detected in the right EVAR limb.
Nine patients also underwent CEUS-imaging, which detected intraluminal floating structures in six patients (66%). CTA was performed in eight patients and allowed for the detection of wall-adherent structures within the EVAR-prosthesis in seven cases.
3.4. Patients with Pathologies of the Peripheral Arteries
The FLS was detected in peripheral arteries of 10 patients (seven men and three women), including the femoral arteries in six patients, the carotid arteries in two patients, and the brachiocephalic artery and axillary artery in one patient, respectively. In two patients, the FLS-positive lesion was considered the most probable source of symptomatic arterial embolism. One female patient with right-sided retinal artery occlusion had a large arteriosclerotic plaque with WAFAS of the brachiocephalic trunk. Another male patient suffered from left hand ischemia due to an arterial embolism originating from a stenotic arteriosclerotic lesion of the ipsilateral axillary artery containing a WAFAS. It is worthy of note that in six patients the FLS was detected after revascularization procedures (two FLS of the common femoral artery which had been used as an access vessel for endovascular procedures, three FLS of the superficial femoral artery after endovascular recanalization, and one FLS after carotid endarterectomy). Two FLS were found incidentally in patients with severe calcifying arteriosclerosis (origin of the internal carotid artery and of the superficial femoral artery, respectively).
CEUS-imaging and CTA was performed in three patients, respectively. In two CEUS-examinations but in none of the CTA-examinations the WAFAS could be confirmed.
3.5. Clinical Impact of the FLS
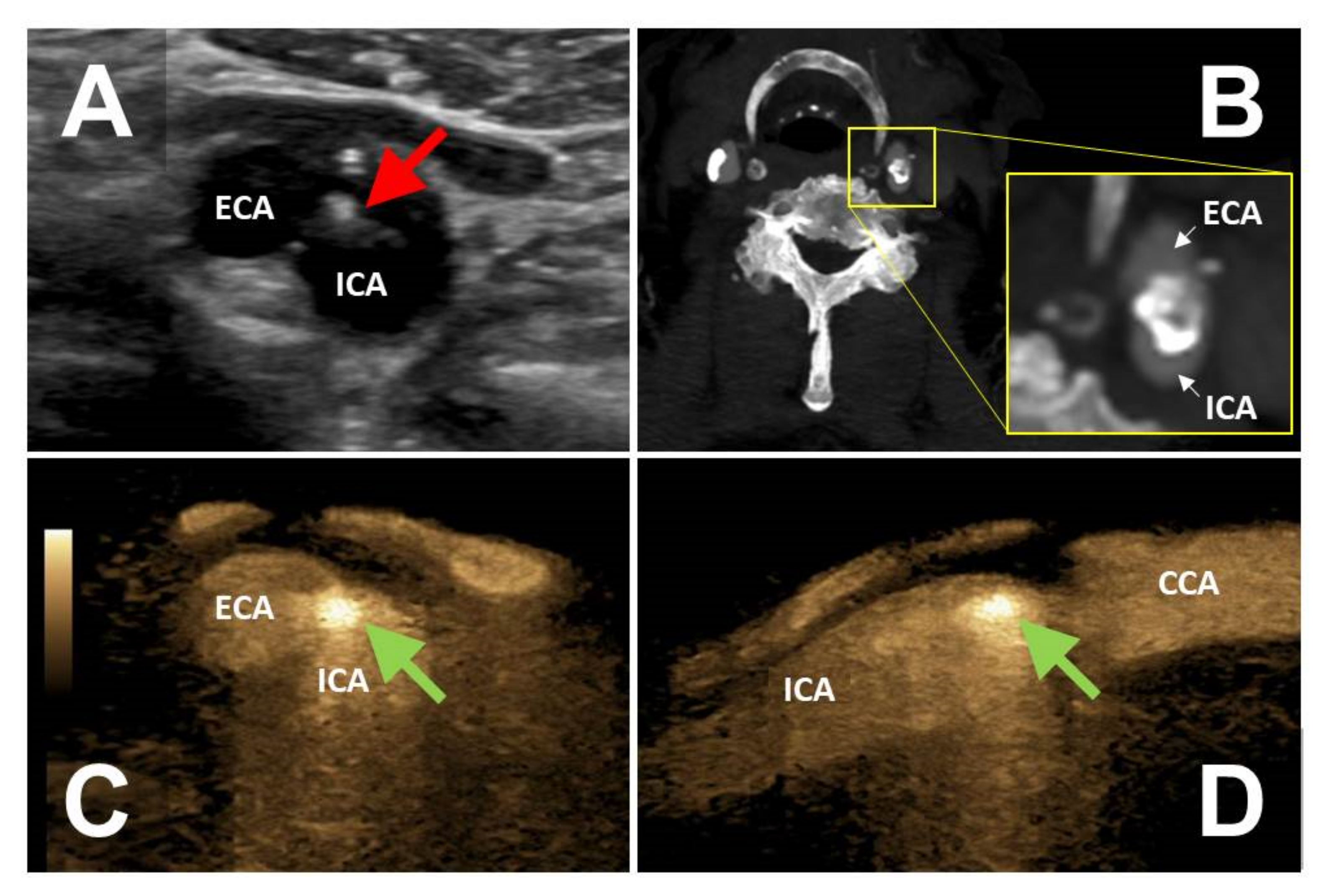
In six patients the FLS had a direct, additional impact on further patient management, including the initiation of anticoagulation in four patients and the initiation of antiplatelet therapy in two patients. One asymptomatic patient with a FLS corresponding to WAFAS adherent to a mid-grade internal carotid artery stenosis (
Figure 3) underwent successful carotid endarterectomy. The above-mentioned patient with recurrent popliteal artery embolism originating from a floating intraluminal thrombus located in the EVAR limbs underwent endovascular re-lining of the prosthesis with covered stent grafts.
3.6. Follow Up
Repeated BFl-imaging was performed in 15 patients, with a mean follow-up time of 10 months (range 1 to 35 months). Five patients underwent revascularization of the arterial segment displaying the FLS, resulting in the disappearance of the FLS in all of them. The FLS persisted in five patients (EVAR, n = 3; native abdominal aorta, n = 2) and disappeared in five patients (EVAR, n = 1; peripheral arteries, n = 4) treated conservatively.
4. Discussion
Based on a novel sonographic sign in BFl, which we termed FLS, we characterized the so far largest cohort of patients with WAFAS in various locations. The FLS may indicate an increased recurrence risk in patients who suffered from symptomatic embolism of the arterial tree downstream to the lesion and thus may have a direct impact on the patient’s treatment. Whether an incidentally found FLS also predicts an increased risk of arterio-arterial embolism in asymptomatic patients remains to be determined.
We compared BFl-findings with CEUS and CTA in a considerable subset of our patients. Some studies used CEUS for assessment of mural thrombus volume [
12] and mural thrombus vascularization [
13] in abdominal aortic aneurysms, but to our knowledge sonographic studies on WAFAS of the abdominal aorta have not been published before. We found that in the abdominal aorta, CEUS was less sensitive than CTA for the detection of WAFAS. Since such lesions in this anatomic location may have a significant clinical impact (high rate of embolic events, particularly in EVAR-patients), BFl appears as an important tool in addition to CEUS to increase the diagnostic accuracy of multimodal sonography.
By contrast, CTA was less sensitive than sonography (CEUS, BFl) for detection of WAFAS of the peripheral arteries in a limited number of our patients. Time-resolved imaging data are not available with routine CTA acquisitions. In view of this, the ability of dynamic blood flow characterization with high temporal and spatial resolution is a major strength of multimodal vascular sonography including BFl. However, the evidence regarding the frequency and the clinical impact of sonographically detected WAFAS of the peripheral arteries is sparse. Vassileva et al. reported a few cases of free-floating thrombus of the nonstenotic carotid artery detected by colour duplex sonography in ischemic stroke patients [
8]. Tatheishi et al. described the “snake fang sign”, characterized by a protruding carotid plaque which was associated with free floating thrombus and ischemic stroke [
14]. Our findings emphasize the advantages of a detailed sonographic examination of the peripheral arteries in cases of arterial embolism. The follow-up of WAFAS in any peripheral arterial segment can easily be performed with multimodal sonography including BFl, bearing the potential to reduce radiation exposure associated with CTA-examinations.
BFl, introduced more than 20 years ago, so far has been applied in a variety of indications, including the assessment of transplant vascularization (e.g., kidney transplants) and the dignity assessment of thyroid nodules. Studies with a focus on vascular pathologies aimed on grading of carotid stenosis [
15,
16] and on detection of cervical artery dissection and fibromuscular dysplasia [
17,
18]. A single study evaluated the diagnostic value of BFl for assessment of the lower extremity arteries and bypass grafts in patients with peripheral arterial disease [
19]. In all the above mentioned vascular indications, an additional diagnostic yield of BFl has not yet been established. The addition of BFl, for example, only slightly improved the diagnostic accuracy of sonography for the diagnosis of cervical artery dissections [
16]. Based on our results, we believe that the BFl-based FLS for detection of WAFAS is an important adjunct finding to established vascular imaging modalities. Whether other Doppler-independent sonographic techniques of flow detection such as superb microvascular imaging [
20] offer a similar diagnostic yield for diagnosis of WAFAS remains to be determined.
Some limitations of our study warrant consideration, including retrospective data collection, the limited number of patients and the lack of complementary imaging in some cases. We are not able to provide accurate values of diagnostic accuracy of the imaging modalities analysed since we only analysed patients with an established diagnosis of WAFAS. Besides the above mentioned methodological limitations, BFl can be hampered similarly to all other ultrasound technologies by patient-related factors such as obesity, vessel calcification and intestinal gas overlay. Finally, BFl is only available with GE ultrasound machines.
In summary, we propose a clinically useful, BFl-based sonographic sign, termed FLS, for detection of WAFAS of the abdominal aorta and the peripheral arteries.
Supplementary Materials
The following supporting information can be downloaded at:
https://www.mdpi.com/article/10.3390/diagnostics12071708/s1. Video S1: Movie (B-Flow, longitudinal section) corresponding to the images presented in
Figure 1. Wall-adherent floating arterial structure (WAFAS), depicted by a fast moving, bright intraluminal signal (flashlight sign, FLS) within an aortic aneurysm with mural thrombus. Video S2: Movie (B-Flow, longitudinal section) corresponding to the images presented in
Figure 2. A wall-adherent, floating arterial structure (WAFAS) in an EVAR limb is uncovered by a flashlight sign (FLS). The transducer is then repositioned into the transversal plane, locating the FLS in the left limb of the prosthesis (displayed on the right side of the screen).
Author Contributions
Conceptualization, C.L. and M.C.; methodology, C.L., U.H. and M.C.; validation, U.H., J.S. and M.C.; formal analysis, C.L., D.P.-W. and M.C.; investigation, C.L., D.P.-W. and J.S.; writing—original draft preparation, C.L. and M.C.; visualization, C.L. and D.P.-W.; supervision, J.S., U.H. and M.C.; project administration, C.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Ethical review and approval were waived due to the retrospective nature of this study.
Informed Consent Statement
Informed consent was waived except for those patients whose medical images are depicted in this paper.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare that they have no conflict of interest.
References
- Meyermann, K.; Trani, J.; Caputo, F.J.; Lombardi, J.V. Descending thoracic aortic mural thrombus presentation and treatment strategies. J. Vasc. Surg. 2017, 66, 931–936. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Machleder, H.I.; Takiff, H.; Lois, J.F.; Holburt, E. Aortic mural thrombus: An occult source of arterial thromboembolism. J. Vasc. Surg. 1986, 4, 473–478. [Google Scholar] [CrossRef]
- Hans, S.S.; Jareunpoon, O.; Balasubramaniam, M.; Zelenock, G.B. Size and location of thrombus in intact and ruptured abdominal aortic aneurysms. J. Vasc. Surg. 2005, 41, 584–588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perini, P.; Massoni, C.B.; Azzarone, M.; Ucci, A.; Rossi, G.; Gallitto, E.; Freyrie, A. Significance and Risk Factors for Intraprosthetic Mural Thrombus in Abdominal Aortic Endografts: A Systematic Review and Meta-Analysis. Ann. Vasc. Surg. 2018, 53, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Fridman, S.; Lownie, S.P.; Mandzia, J. Diagnosis and management of carotid free-floating thrombus: A systematic literature review. Int. J. Stroke 2019, 14, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Tsilimparis, N.; Hanack, U.; Pisimisis, G.; Yousefi, S.; Wintzer, C.; Rückert, R.I. Thrombus in the non-aneurysmal, non-atherosclerotic descending thoracic aorta—An unusual source of arterial embolism. Eur. J. Vasc. Endovasc. Surg. 2011, 41, 450–457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, S.; Yu, J.; Zeng, W.; Yang, L.; Teng, L.; Cui, Y.; Shi, H. Aortic floating thrombus detected by computed tomography angiography incidentally: Five cases and a literature review. J. Thorac. Cardiovasc. Surg. 2017, 153, 791–803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vassileva, E.; Daskalov, M.; Stamenova, P. Free-floating thrombus in stroke patients with nonstenotic internal carotid artery—An ultrasonographic study. J. Clin. Ultrasound 2014, 43, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.; Adams, Q. White Paper B-Flow Technology. 2012. Available online: www.logiqclub.net/emea/white-papers (accessed on 10 May 2022).
- Weskott, H.P. B-flow—A new method for detecting blood flow. Ultraschall Med. 2000, 21, 59–65. [Google Scholar] [PubMed]
- Sidhu, P.S.; Cantisani, V.; Dietrich, C.F.; Gilja, O.H.; Saftoiu, A.; Bartels, E.; Bertolotto, M.; Calliada, F.; Clevert, D.-A.; Cosgrove, D.; et al. The EFSUMB Guidelines and Recommendations for the Clinical Practice of Contrast-Enhanced Ultrasound (CEUS) in Non-Hepatic Applications: Update 2017 (Long Version). Ultraschall Med. Eur. J. Ultrasound 2018, 39, e2–e44. [Google Scholar] [CrossRef] [Green Version]
- Ghulam, Q.M.; Bredahl, K.; Sandholt, B.; Taudorf, M.; Lönn, L.; Rouet, L.; Sillesen, H.; Eiberg, J.P. Contrast Enhanced Three Dimensional Ultrasound for Intraluminal Thrombus Assessment in Abdominal Aortic Aneurysms. Eur. J. Vasc. Endovasc. Surg. 2018, 56, 673–680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Łukasiewicz, A.; Garkowski, A.; Rutka, K.; Janica, J.; Łebkowska, U. Evaluation of the thrombus of abdominal aortic aneurysms using contrast enhanced ultrasound—Preliminary results. Sci. Rep. 2016, 6, 34152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tateishi, Y.; Tsujino, A.; Hamabe, J.; Tasaki, O.; Morikawa, M.; Hayashi, T.; Horie, N.; Hayashi, K.; Suyama, K.; Nagata, I. “Snake Fang” Sign without Carotid Stenosis on Duplex Ultrasonography Indicates High Risk of Artery-to-Artery Embolic Stroke. J. Neuroimaging 2014, 24, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Clevert, D.-A.; Johnson, T.; Jung, E.M.; Flach, P.M.; Strautz, T.I.; Ritter, G.; Gallegos, M.T.; Kubale, R.; Becker, C.; Reiser, M. Color Doppler, power Doppler and B-flow ultrasound in the assessment of ICA stenosis: Comparison with 64-MD-CT angiography. Eur. Radiol. 2006, 17, 2149–2159. [Google Scholar] [CrossRef] [PubMed]
- Finkenzeller, T.; Tacke, J.; Clevert, D.A.; Jung, W.; Kubale, R.; Schreyer, A.; Feuerbach, S.; Jung, E.M. Quantification of extracranial ICA stenoses with vessel ultrasound by CCDS and B-flow in comparison to 64-slice multidector CTA, contrast-enhanced MRA and DSA. Ultraschall Med. 2008, 29, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Clevert, D.-A.; Jung, E.M.; Johnson, T.; Kubale, R.; Rupp, N.; Schoenberg, S.O.; Reiser, M. Cervical artery dissection: Improved diagnosis by B-Flow ultrasound. Clin. Hemorheol. Microcirc. 2007, 36, 141–153. [Google Scholar] [PubMed]
- Cutler, J.J.; Campo, N.; Koch, S. B-Flow and B-Mode Ultrasound Imaging in Carotid Fibromuscular Dysplasia. J. Neuroimaging 2018, 28, 269–272. [Google Scholar] [CrossRef] [PubMed]
- D’Abate, F.; Ramachandran, V.; Young, M.A.; Farrah, J.; Ahmed, M.H.; Jones, K.; Hinchliffe, R.J. B-Flow Imaging in Lower Limb Peripheral Arterial Disease and Bypass Graft Ultrasonography. Ultrasound Med. Biol. 2016, 42, 2345–2351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sato, W.; Suto, Y.; Yamanaka, T.; Watanabe, H. An advanced ultrasound application used to assess peripheral vascular diseases: Superb microvascular imaging. J. Echocardiogr. 2021, 19, 150–157. [Google Scholar] [CrossRef] [PubMed]
| Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).