Diagnostic Potential of Exosomal microRNAs in Colorectal Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Patients’ Characteristics
| Groups | N (Total n = 69) | Age (Years) (Mean/Range) |
|---|---|---|
| Healthy control | 26 | 58 (44–85) |
| Adenoma | 20 | 64 (27–79) |
| CRC | 23 | 64 (39–81) |
| Gender | Percentage | |
| Female | 24 | 34.8 |
| Male | 45 | 65.2 |
| UICC Stage | ||
| 1 | 3 | 13.1 |
| 2 | 4 | 17.4 |
| 3 | 7 | 30.4 |
| 4 | 9 | 39.1 |
| T-Stage | ||
| 1 | 1 | 4.3 |
| 2 | 3 | 13.1 |
| 3 | 15 | 65.2 |
| 4 | 4 | 17.4 |
| N-Stage | ||
| 0 | 8 | 34.8 |
| 1 | 5 | 21.7 |
| 2 | 10 | 43.5 |
| M-Stage | ||
| 0 | 14 | 60.9 |
| 1 | 9 | 39.1 |
2.3. MicroRNA Selection
2.4. Preparation of Blood Samples
2.5. Serum Circulating miRNA (c-miR) Isolation
2.6. Exosomal microRNA (exo-miR) Isolation
2.7. Measurement of exo-miR and c-miR Levels Using Real-Time Quantitative PCR
2.8. Statistical Analyses
3. Results
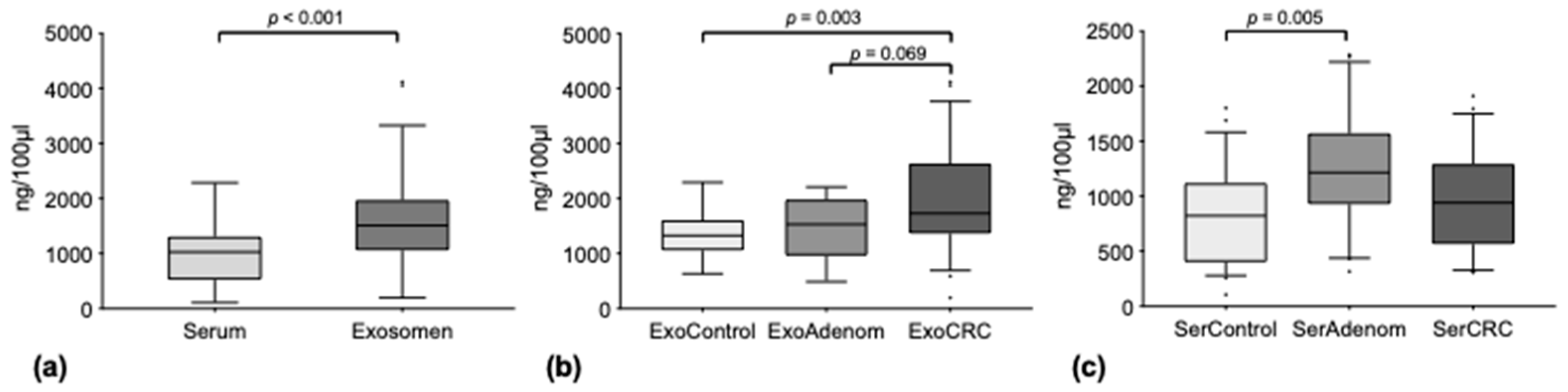
3.1. Circulating miRNAs Are Concentrated in Exosomes
3.2. MiRNA Levels in the Exosome Compartment Differentiate between Disease Groups
3.3. Free Circulating miRNA Levels in the Serum Have Lower Discriminating Potential
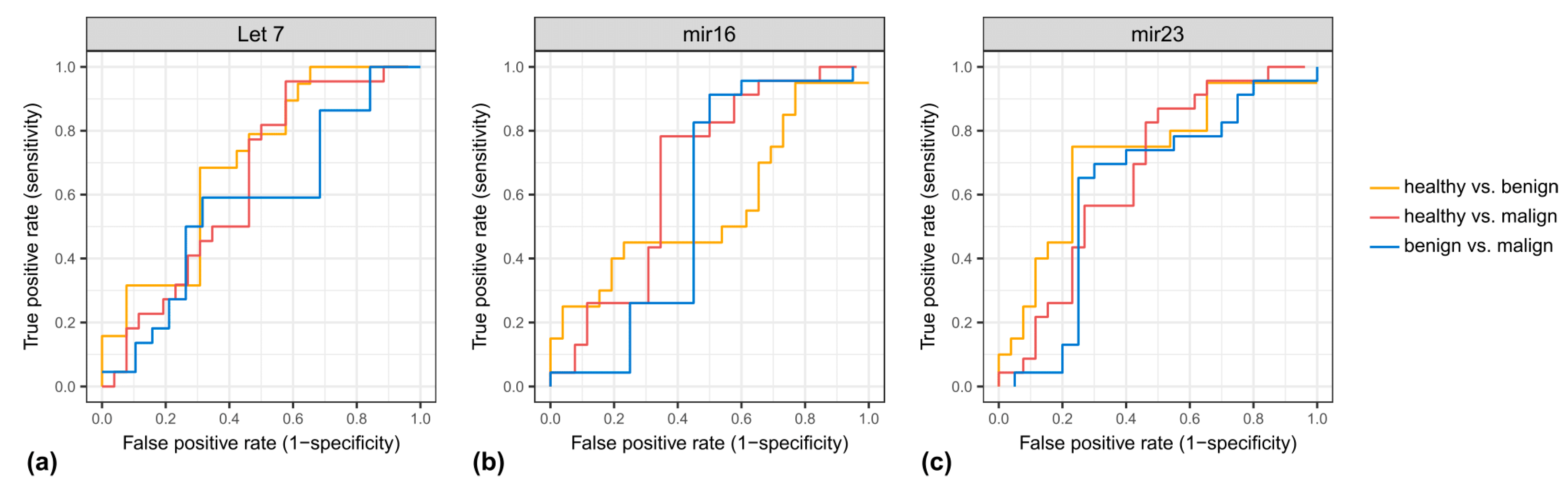
3.4. Tumor Marker
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Fearon, E.R. Molecular genetics of colorectal cancer. Annu. Rev. Pathol. 2011, 6, 479–507. [Google Scholar] [CrossRef] [PubMed]
- Brenner, H.; Kloor, M.; Pox, C.P. Colorectal cancer. Lancet 2014, 383, 1490–1502. [Google Scholar] [CrossRef]
- Burch, J.A.; Soares-Weiser, K.; St John, D.J.; Duffy, S.; Smith, S.; Kleijnen, J.; Westwood, M. Diagnostic accuracy of faecal occult blood tests used in screening for colorectal cancer: A systematic review. J. Med. Screen 2007, 14, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Robertson, D.J.; Lee, J.K.; Boland, C.R.; Dominitz, J.A.; Giardiello, F.M.; Johnson, D.A.; Kaltenbach, T.; Lieberman, D.; Levin, T.R.; Rex, D.K. Recommendations on Fecal Immunochemical Testing to Screen for Colorectal Neoplasia: A Consensus Statement by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology 2017, 152, 1217–1237. [Google Scholar] [CrossRef]
- Lieberman, D.A. Clinical practice. Screening for colorectal cancer. N. Engl. J. Med. 2009, 361, 1179–1187. [Google Scholar] [CrossRef]
- Burt, R.W.; Barthel, J.S.; Dunn, K.B.; David, D.S.; Drelichman, E.; Ford, J.M.; Giardiello, F.M.; Gruber, S.B.; Halverson, A.L.; Hamilton, S.R.; et al. NCCN clinical practice guidelines in oncology. Colorectal cancer screening. JNCCN J. Natl. Compr. Cancer Netw. 2010, 8, 8–61. [Google Scholar] [CrossRef]
- Schmoll, H.J.; Van Cutsem, E.; Stein, A.; Valentini, V.; Glimelius, B.; Haustermans, K.; Nordlinger, B.; van de Velde, C.J.; Balmana, J.; Regula, J.; et al. ESMO Consensus Guidelines for management of patients with colon and rectal cancer. a personalized approach to clinical decision making. Ann. Oncol. 2012, 23, 2479–2516. [Google Scholar] [CrossRef]
- Duffy, M.J.; van Dalen, A.; Haglund, C.; Hansson, L.; Holinski-Feder, E.; Klapdor, R.; Lamerz, R.; Peltomaki, P.; Sturgeon, C.; Topolcan, O. Tumour markers in colorectal cancer: European Group on Tumour Markers (EGTM) guidelines for clinical use. Eur. J. Cancer 2007, 43, 1348–1360. [Google Scholar] [CrossRef]
- Mokhles, S.; Macbeth, F.; Farewell, V.; Fiorentino, F.; Williams, N.R.; Younes, R.N.; Takkenberg, J.J.; Treasure, T. Meta-analysis of colorectal cancer follow-up after potentially curative resection. Br. J. Surg. 2016, 103, 1259–1268. [Google Scholar] [CrossRef]
- Diaz, L.A., Jr.; Bardelli, A. Liquid biopsies: Genotyping circulating tumor DNA. J. Clin. Oncol. 2014, 32, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Iwakawa, H.O.; Tomari, Y. The Functions of MicroRNAs: mRNA Decay and Translational Repression. Trends Cell Biol. 2015, 25, 651–665. [Google Scholar] [CrossRef]
- Hollis, M.; Nair, K.; Vyas, A.; Chaturvedi, L.S.; Gambhir, S.; Vyas, D. MicroRNAs potential utility in colon cancer: Early detection, prognosis, and chemosensitivity. World J. Gastroenterol. 2015, 21, 8284–8292. [Google Scholar] [CrossRef] [PubMed]
- Cekaite, L.; Eide, P.W.; Lind, G.E.; Skotheim, R.I.; Lothe, R.A. MicroRNAs as growth regulators, their function and biomarker status in colorectal cancer. Oncotarget 2016, 7, 6476–6505. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Zhao, W.; Yu, H.; Wang, Y.; Liu, Y.; Xie, C. A Comprehensive Meta-Analysis of MicroRNAs for Predicting Colorectal Cancer. Medicine 2016, 95, e2738. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.; Tu, Y.; Zhu, Y.; Wang, Z.; Li, C.; Lao, L.; Wu, G. Predictive power of circulating miRNAs in detecting colorectal cancer. Tumour Biol. 2015, 36, 2559–2567. [Google Scholar] [CrossRef] [PubMed]
- Thery, C. Cancer: Diagnosis by extracellular vesicles. Nature 2015, 523, 161–162. [Google Scholar] [CrossRef]
- Hingorani, S.R. Intercepting Cancer Communiques: Exosomes as Heralds of Malignancy. Cancer Cell 2015, 28, 151–153. [Google Scholar] [CrossRef][Green Version]
- Matsumura, T.; Sugimachi, K.; Iinuma, H.; Takahashi, Y.; Kurashige, J.; Sawada, G.; Ueda, M.; Uchi, R.; Ueo, H.; Takano, Y.; et al. Exosomal microRNA in serum is a novel biomarker of recurrence in human colorectal cancer. Br. J. Cancer 2015, 113, 275–281. [Google Scholar] [CrossRef]
- Lin, J.; Li, J.; Huang, B.; Liu, J.; Chen, X.; Chen, X.M.; Xu, Y.M.; Huang, L.F.; Wang, X.Z. Exosomes: Novel biomarkers for clinical diagnosis. Sci. World J. 2015, 2015, 657086. [Google Scholar] [CrossRef]
- Brinton, L.T.; Sloane, H.S.; Kester, M.; Kelly, K.A. Formation and role of exosomes in cancer. Cell Mol. Life Sci. 2015, 72, 659–671. [Google Scholar] [CrossRef] [PubMed]
- Melo, S.A.; Luecke, L.B.; Kahlert, C.; Fernandez, A.F.; Gammon, S.T.; Kaye, J.; LeBleu, V.S.; Mittendorf, E.A.; Weitz, J.; Rahbari, N.; et al. Glypican-1 identifies cancer exosomes and detects early pancreatic cancer. Nature 2015, 523, 177–182. [Google Scholar] [CrossRef] [PubMed]
- El Andaloussi, S.; Mager, I.; Breakefield, X.O.; Wood, M.J. Extracellular vesicles: Biology and emerging therapeutic opportunities. Nat. Rev. Drug. Discov. 2013, 12, 347–357. [Google Scholar] [CrossRef]
- Taverna, S.; Giallombardo, M.; Pucci, M.; Flugy, A.; Manno, M.; Raccosta, S.; Rolfo, C.; De Leo, G.; Alessandro, R. Curcumin inhibits in vitro and in vivo chronic myelogenous leukemia cells growth: A possible role for exosomal disposal of miR-21. Oncotarget 2015, 6, 21918–21933. [Google Scholar] [CrossRef] [PubMed]
- Ogata-Kawata, H.; Izumiya, M.; Kurioka, D.; Honma, Y.; Yamada, Y.; Furuta, K.; Gunji, T.; Ohta, H.; Okamoto, H.; Sonoda, H.; et al. Circulating exosomal microRNAs as biomarkers of colon cancer. PLoS ONE 2014, 9, e92921. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Huang, Z.; Zhu, D.; Zhou, X.; Shan, X.; Qi, L.W.; Wu, L.; Cheng, W.; Zhu, J.; Zhang, L.; et al. A panel of microRNA signature in serum for colorectal cancer diagnosis. Oncotarget 2017, 8, 17081–17091. [Google Scholar] [CrossRef] [PubMed]
- Nedaeinia, R.; Manian, M.; Jazayeri, M.H.; Ranjbar, M.; Salehi, R.; Sharifi, M.; Mohaghegh, F.; Goli, M.; Jahednia, S.H.; Avan, A.; et al. Circulating exosomes and exosomal microRNAs as biomarkers in gastrointestinal cancer. Cancer Gene Ther. 2017, 24, 48–56. [Google Scholar] [CrossRef]
- Uratani, R.; Toiyama, Y.; Kitajima, T.; Kawamura, M.; Hiro, J.; Kobayashi, M.; Tanaka, K.; Inoue, Y.; Mohri, Y.; Mori, T.; et al. Diagnostic Potential of Cell-Free and Exosomal MicroRNAs in the Identification of Patients with High-Risk Colorectal Adenomas. PLoS ONE 2016, 11, e0160722. [Google Scholar] [CrossRef]
- Nagtegaal, I.; Arends, M.; Salto-Tellez, M. Colorectal Adenocarcinoma: Tumours of the Colon and Rectum. In World Health Organization Classification of Tumours of the Digestive System; IARC Press: Basel, Switzerland, 2019; pp. 177–187. [Google Scholar]
- Brierley, J.D.; Gospodarowicz, M.K.; Wittekind, C. TNM Classification of Malignant Tumours; John Wiley & Sons: Hoboken, NY, USA, 2016. [Google Scholar]
- Conteduca, V.; Sansonno, D.; Russi, S.; Dammacco, F. Precancerous colorectal lesions (Review). Int. J. Oncol. 2013, 43, 973–984. [Google Scholar] [CrossRef]
- King, C.E.; Wang, L.; Winograd, R.; Madison, B.B.; Mongroo, P.S.; Johnstone, C.N.; Rustgi, A.K. LIN28B fosters colon cancer migration, invasion and transformation through let-7-dependent and -independent mechanisms. Oncogene 2011, 30, 4185–4193. [Google Scholar] [CrossRef]
- Akao, Y.; Nakagawa, Y.; Naoe, T. let-7 microRNA functions as a potential growth suppressor in human colon cancer cells. Biol. Pharm. Bull. 2006, 29, 903–906. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Wang, Z.; Liao, B.Y.; Yu, L.; Gao, X.; Lu, S.; Wang, S.; Dai, Z.; Zhang, X.; Chen, Q.; et al. Human miR-1228 as a stable endogenous control for the quantification of circulating microRNAs in cancer patients. Int. J. Cancer 2014, 135, 1187–1194. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Chen, F.; Zhang, J.; Zhang, Q.; Lin, J. Exosome analysis: A promising biomarker system with special attention to saliva. J. Membr. Biol. 2014, 247, 1129–1136. [Google Scholar] [CrossRef]
- Ng, E.K.; Chong, W.W.; Jin, H.; Lam, E.K.; Shin, V.Y.; Yu, J.; Poon, T.C.; Ng, S.S.; Sung, J.J. Differential expression of microRNAs in plasma of patients with colorectal cancer: A potential marker for colorectal cancer screening. Gut 2009, 58, 1375–1381. [Google Scholar] [CrossRef] [PubMed]
- Inoue, A.; Yamamoto, H.; Uemura, M.; Nishimura, J.; Hata, T.; Takemasa, I.; Ikenaga, M.; Ikeda, M.; Murata, K.; Mizushima, T.; et al. MicroRNA-29b is a Novel Prognostic Marker in Colorectal Cancer. Ann. Surg. Oncol. 2015, 22 (Suppl. 3), S1410–S1418. [Google Scholar] [CrossRef] [PubMed]
- Aherne, S.T.; Madden, S.F.; Hughes, D.J.; Pardini, B.; Naccarati, A.; Levy, M.; Vodicka, P.; Neary, P.; Dowling, P.; Clynes, M. Circulating miRNAs miR-34a and miR-150 associated with colorectal cancer progression. BMC Cancer 2015, 15, 329. [Google Scholar] [CrossRef] [PubMed]
- Gungormez, C.; Teker, E.; Atmanoglu, S.; Borazan, E. miRNA Profile and Bioinformatic Analysis for Diagnosis in Patients with Stage IIIA Colon Cancer. Biochem. Genet. 2022, 60, 191–203. [Google Scholar] [CrossRef]
- Zheng, G.; Du, L.; Yang, X.; Zhang, X.; Wang, L.; Yang, Y.; Li, J.; Wang, C. Serum microRNA panel as biomarkers for early diagnosis of colorectal adenocarcinoma. Br. J. Cancer 2014, 111, 1985–1992. [Google Scholar] [CrossRef]
- Kara, M.; Yumrutas, O.; Ozcan, O.; Celik, O.I.; Bozgeyik, E.; Bozgeyik, I.; Tasdemir, S. Differential expressions of cancer-associated genes and their regulatory miRNAs in colorectal carcinoma. Gene 2015, 567, 81–86. [Google Scholar] [CrossRef]
- Matsuzaki, J.; Suzuki, H. Role of MicroRNAs-221/222 in Digestive Systems. J. Clin. Med. 2015, 4, 1566–1577. [Google Scholar] [CrossRef]
- Bitarte, N.; Bandres, E.; Boni, V.; Zarate, R.; Rodriguez, J.; Gonzalez-Huarriz, M.; Lopez, I.; Javier Sola, J.; Alonso, M.M.; Fortes, P.; et al. MicroRNA-451 is involved in the self-renewal, tumorigenicity, and chemoresistance of colorectal cancer stem cells. Stem Cells 2011, 29, 1661–1671. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Xue, J.Q.; Han, S.J.; Qian, S.Y.; Zhang, W.H. Circulating microRNA-451 as a predictor of resistance to neoadjuvant chemotherapy in breast cancer. Cancer Biomark. 2016, 16, 395–403. [Google Scholar] [CrossRef]
- Huang, J.Y.; Zhang, K.; Chen, D.Q.; Chen, J.; Feng, B.; Song, H.; Chen, Y.; Zhu, Z.; Lu, L.; De, W.; et al. MicroRNA-451: Epithelial-mesenchymal transition inhibitor and prognostic biomarker of hepatocelluar carcinoma. Oncotarget 2015, 6, 18613–18630. [Google Scholar] [CrossRef] [PubMed]
- Shivapurkar, N.; Weiner, L.M.; Marshall, J.L.; Madhavan, S.; Deslattes Mays, A.; Juhl, H.; Wellstein, A. Recurrence of early stage colon cancer predicted by expression pattern of circulating microRNAs. PLoS ONE 2014, 9, e84686. [Google Scholar] [CrossRef]
- Sanders, I.; Holdenrieder, S.; Walgenbach-Brunagel, G.; von Ruecker, A.; Kristiansen, G.; Muller, S.C.; Ellinger, J. Evaluation of reference genes for the analysis of serum miRNA in patients with prostate cancer, bladder cancer and renal cell carcinoma. Int. J. Urol. 2012, 19, 1017–1025. [Google Scholar] [CrossRef]
- Sauer, E.; Madea, B.; Courts, C. An evidence based strategy for normalization of quantitative PCR data from miRNA expression analysis in forensically relevant body fluids. Forensic Sci. Int. Genet. 2014, 11, 174–181. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Ishikawa, H.; Yamada, H.; Taromaru, N.; Kondo, K.; Nagura, A.; Yamazaki, M.; Ando, Y.; Munetsuna, E.; Suzuki, K.; Ohashi, K.; et al. Stability of serum high-density lipoprotein-microRNAs for preanalytical conditions. Ann. Clin. Biochem. 2017, 54, 134–142. [Google Scholar] [CrossRef]
- Tsui, N.B.; Ng, E.K.; Lo, Y.M. Stability of endogenous and added RNA in blood specimens, serum, and plasma. Clin. Chem. 2002, 48, 1647–1653. [Google Scholar] [CrossRef]
- Gallo, A.; Tandon, M.; Alevizos, I.; Illei, G.G. The majority of microRNAs detectable in serum and saliva is concentrated in exosomes. PLoS ONE 2012, 7, e30679. [Google Scholar] [CrossRef]
- Tanaka, Y.; Kamohara, H.; Kinoshita, K.; Kurashige, J.; Ishimoto, T.; Iwatsuki, M.; Watanabe, M.; Baba, H. Clinical impact of serum exosomal microRNA-21 as a clinical biomarker in human esophageal squamous cell carcinoma. Cancer 2013, 119, 1159–1167. [Google Scholar] [CrossRef] [PubMed]
- Thakur, B.K.; Zhang, H.; Becker, A.; Matei, I.; Huang, Y.; Costa-Silva, B.; Zheng, Y.; Hoshino, A.; Brazier, H.; Xiang, J.; et al. Double-stranded DNA in exosomes: A novel biomarker in cancer detection. Cell Res. 2014, 24, 766–769. [Google Scholar] [CrossRef] [PubMed]
- Bettegowda, C.; Sausen, M.; Leary, R.J.; Kinde, I.; Wang, Y.; Agrawal, N.; Bartlett, B.R.; Wang, H.; Luber, B.; Alani, R.M.; et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci. Transl. Med. 2014, 6, 224ra224. [Google Scholar] [CrossRef] [PubMed]
- Sohn, W.; Kim, J.; Kang, S.H.; Yang, S.R.; Cho, J.Y.; Cho, H.C.; Shim, S.G.; Paik, Y.H. Serum exosomal microRNAs as novel biomarkers for hepatocellular carcinoma. Exp. Mol. Med. 2015, 47, e184. [Google Scholar] [CrossRef] [PubMed]
- Clancy, C.; Khan, S.; Glynn, C.L.; Holian, E.; Dockery, P.; Lalor, P.; Brown, J.A.; Joyce, M.R.; Kerin, M.J.; Dwyer, R.M. Screening of exosomal microRNAs from colorectal cancer cells. Cancer Biomark. 2016, 17, 427–435. [Google Scholar] [CrossRef]
- Diamantopoulos, M.A.; Kontos, C.K.; Kerimis, D.; Papadopoulos, I.N.; Scorilas, A. Upregulated miR-16 expression is an independent indicator of relapse and poor overall survival of colorectal adenocarcinoma patients. Clin. Chem. Lab. Med. 2017, 55, 737–747. [Google Scholar] [CrossRef]
- Qian, J.; Jiang, B.; Li, M.; Chen, J.; Fang, M. Prognostic significance of microRNA-16 expression in human colorectal cancer. World J. Surg. 2013, 37, 2944–2949. [Google Scholar] [CrossRef]
- Xiao, G.; Tang, H.; Wei, W.; Li, J.; Ji, L.; Ge, J. Aberrant Expression of MicroRNA-15a and MicroRNA-16 Synergistically Associates with Tumor Progression and Prognosis in Patients with Colorectal Cancer. Gastroenterol. Res. Pract. 2014, 2014, 364549. [Google Scholar] [CrossRef]
- You, C.; Liang, H.; Sun, W.; Li, J.; Liu, Y.; Fan, Q.; Zhang, H.; Yue, X.; Li, J.; Chen, X.; et al. Deregulation of the miR-16-KRAS axis promotes colorectal cancer. Sci. Rep. 2016, 6, 37459. [Google Scholar] [CrossRef]
- Yong, F.L.; Law, C.W.; Wang, C.W. Potentiality of a triple microRNA classifier: miR-193a-3p, miR-23a and miR-338-5p for early detection of colorectal cancer. BMC Cancer 2013, 13, 280. [Google Scholar] [CrossRef]
- Jahid, S.; Sun, J.; Edwards, R.A.; Dizon, D.; Panarelli, N.C.; Milsom, J.W.; Sikandar, S.S.; Gumus, Z.H.; Lipkin, S.M. miR-23a promotes the transition from indolent to invasive colorectal cancer. Cancer Discov. 2012, 2, 540–553. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Yuan, Y.; Cho, J.H.; McClarty, S.; Baxter, D.; Galas, D.J. Comparing the MicroRNA spectrum between serum and plasma. PLoS ONE 2012, 7, e41561. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.J.; Lin, C.Z.; Zhang, H.H.; Qian, J.; Zhong, L.; Xu, N. Detection of let-7a microRNA by real-time PCR in colorectal cancer: A single-centre experience from China. J. Int. Med. Res. 2007, 35, 716–723. [Google Scholar] [CrossRef] [PubMed]
- Ghanbari, R.; Mosakhani, N.; Sarhadi, V.K.; Armengol, G.; Nouraee, N.; Mohammadkhani, A.; Khorrami, S.; Arefian, E.; Paryan, M.; Malekzadeh, R.; et al. Simultaneous Underexpression of let-7a-5p and let-7f-5p microRNAs in Plasma and Stool Samples from Early Stage Colorectal Carcinoma. Biomark Cancer 2015, 7, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Dutta, A. The tumor suppressor microRNA let-7 represses the HMGA2 oncogene. Genes Dev. 2007, 21, 1025–1030. [Google Scholar] [CrossRef]
- Dou, R.; Nishihara, R.; Cao, Y.; Hamada, T.; Mima, K.; Masuda, A.; Masugi, Y.; Shi, Y.; Gu, M.; Li, W.; et al. MicroRNA let-7, T Cells, and Patient Survival in Colorectal Cancer. Cancer Immunol. Res. 2016, 4, 927–935. [Google Scholar] [CrossRef]
- Ohshima, K.; Inoue, K.; Fujiwara, A.; Hatakeyama, K.; Kanto, K.; Watanabe, Y.; Muramatsu, K.; Fukuda, Y.; Ogura, S.; Yamaguchi, K.; et al. Let-7 microRNA family is selectively secreted into the extracellular environment via exosomes in a metastatic gastric cancer cell line. PLoS ONE 2010, 5, e13247. [Google Scholar] [CrossRef]
- Tsukamoto, M.; Iinuma, H.; Yagi, T.; Matsuda, K.; Hashiguchi, Y. Circulating Exosomal MicroRNA-21 as a Biomarker in Each Tumor Stage of Colorectal Cancer. Oncology 2017, 92, 360–370. [Google Scholar] [CrossRef]
- Cho, K.R.; Vogelstein, B. Genetic alterations in the adenoma--carcinoma sequence. Cancer 1992, 70, 1727–1731. [Google Scholar] [CrossRef]
- Bateman, A.C. Pathology of serrated colorectal lesions. J. Clin. Pathol. 2014, 67, 865–874. [Google Scholar] [CrossRef]
- Leggett, B.; Whitehall, V. Role of the Serrated Pathway in Colorectal Cancer Pathogenesis. Gastroenterology 2010, 138, 2088–2100. [Google Scholar] [CrossRef] [PubMed]
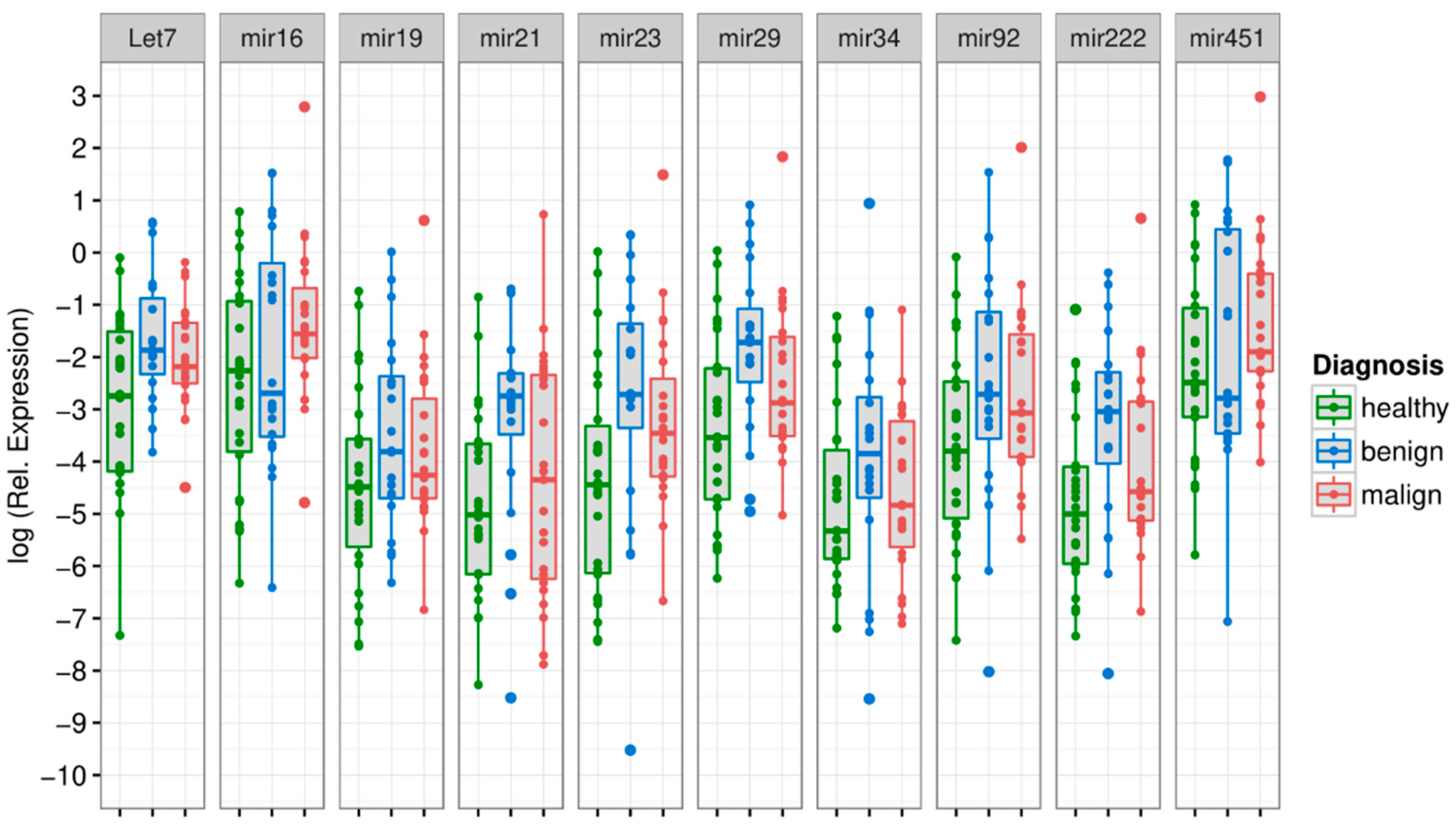
| miRNA | Healthy vs. Adenoma | Healthy vs. CRC | Adenoma vs. CRC |
|---|---|---|---|
| Let7 | 0.008 * | 0.035 * | 0.333 |
| mir16 | 0.323 | 0.022 * | 0.368 |
| mir19 | 0.098 | 0.174 | 0.626 |
| mir21 | 0.006 * | 0.444 | 0.076 |
| mir23 | 0.025 * | 0.039 * | 0.509 |
| mir29 | 0.005 * | 0.079 | 0.167 |
| mir34 | 0.207 | 0.783 | 0.298 |
| mir92 | 0.093 | 0.063 | 0.844 |
| mir222 | 0.010 * | 0.110 | 0.213 |
| mir451 | 0.516 | 0.072 | 0.466 |
| miRNA | Healthy (neg.) vs. CRC (pos.) | |
|---|---|---|
| AUC | SN@95%SP | |
| Let7 | 0.64 | 4.5% |
| mir16 | 0.67 | 4.3% |
| mir19 | 0.60 | 4.3% |
| mir21 | 0.55 | 8.7% |
| mir23 | 0.67 | 4.3% |
| mir29 | 0.64 | 4.3% |
| mir34 | 0.55 | 4.5% |
| mir92 | 0.64 | 8.7% |
| mir222 | 0.63 | 13.0% |
| mir451 | 0.65 | 4.3% |
| Combinations | AUC | SN@95%SP |
|---|---|---|
| 10 miRNAs | 0.90 | 65.2 |
| 5 miRNAs: mir16, mir19, mir21, mir34, mir222 | 0.87 | 56.5 |
| 3 miRNAs: mir16, mir19, mir34 | 0.80 | 30.4 |
| 2 miRNAs: mir16, mir34 | 0.74 | 21.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dohmen, J.; Semaan, A.; Kobilay, M.; Zaleski, M.; Branchi, V.; Schlierf, A.; Hettwer, K.; Uhlig, S.; Hartmann, G.; Kalff, J.C.; et al. Diagnostic Potential of Exosomal microRNAs in Colorectal Cancer. Diagnostics 2022, 12, 1413. https://doi.org/10.3390/diagnostics12061413
Dohmen J, Semaan A, Kobilay M, Zaleski M, Branchi V, Schlierf A, Hettwer K, Uhlig S, Hartmann G, Kalff JC, et al. Diagnostic Potential of Exosomal microRNAs in Colorectal Cancer. Diagnostics. 2022; 12(6):1413. https://doi.org/10.3390/diagnostics12061413
Chicago/Turabian StyleDohmen, Jonas, Alexander Semaan, Makbule Kobilay, Martin Zaleski, Vittorio Branchi, Anja Schlierf, Karina Hettwer, Steffen Uhlig, Gunther Hartmann, Jörg C. Kalff, and et al. 2022. "Diagnostic Potential of Exosomal microRNAs in Colorectal Cancer" Diagnostics 12, no. 6: 1413. https://doi.org/10.3390/diagnostics12061413
APA StyleDohmen, J., Semaan, A., Kobilay, M., Zaleski, M., Branchi, V., Schlierf, A., Hettwer, K., Uhlig, S., Hartmann, G., Kalff, J. C., Matthaei, H., Lingohr, P., & Holdenrieder, S. (2022). Diagnostic Potential of Exosomal microRNAs in Colorectal Cancer. Diagnostics, 12(6), 1413. https://doi.org/10.3390/diagnostics12061413







