Introducing the Sanguis-Filum for Detection of Gastric Mucosal Lesions Prior to Endoscopy: A Study Protocol
Abstract
:1. Introduction
2. Experimental Design and Materials
2.1. Description of the S-Filum
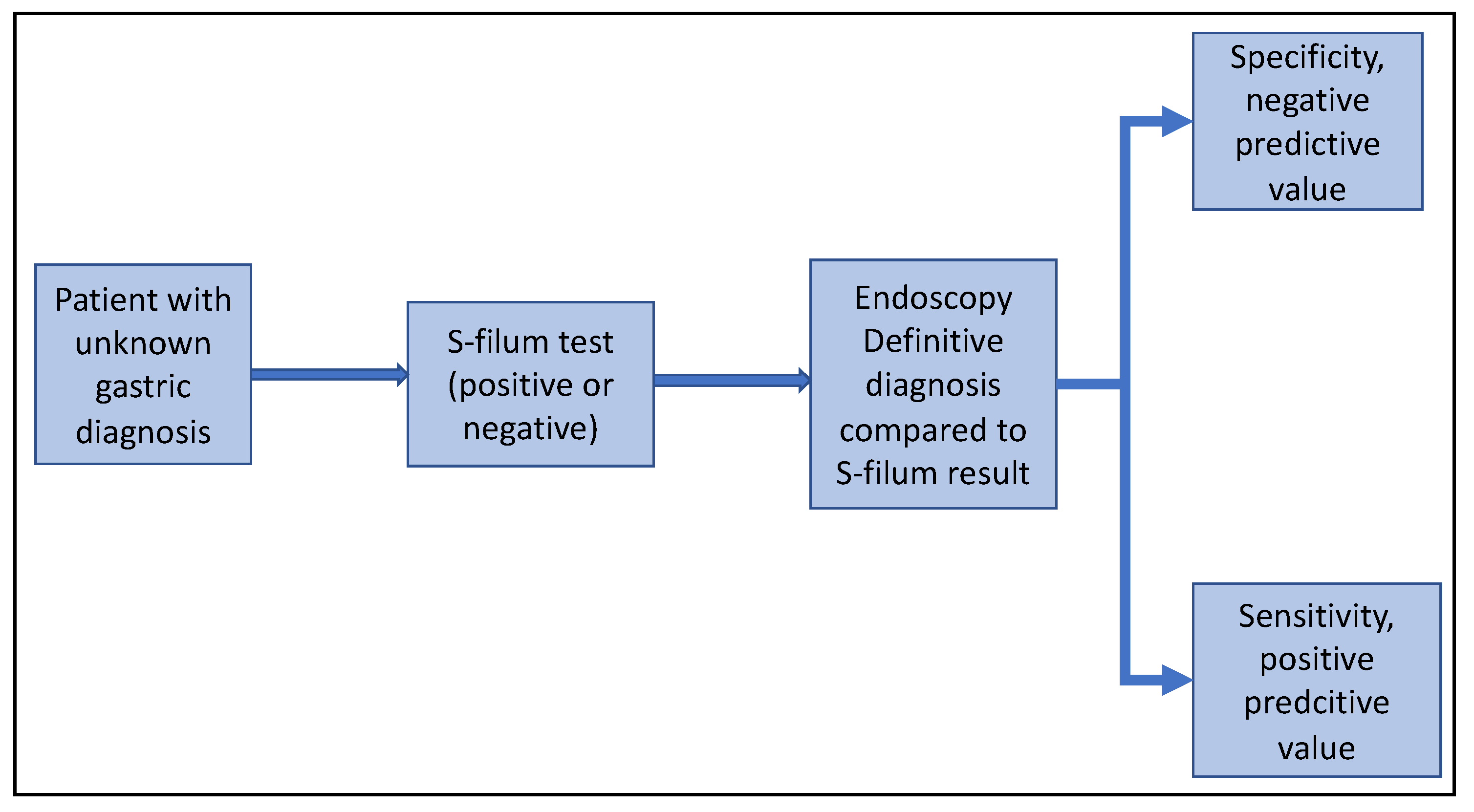
2.2. Study Design
2.3. Sample Size Calculation
2.4. Data Collection, Management and Analysis
3. Preliminary Work Carried Out to Design the S-Filum
4. Results from the S-Filum Testing on Volunteers
5. Expected Results from This Study
6. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Ferlay, J.; Ervik, M.; Lam, F.; Colombet, M.; Mery, L.; Piñeros, M.; Znaor, A.; Soerjomataram, I.; Bray, F. Global Cancer Observatory: Cancer Today. Lyon, France: International Agency for Research on Cancer. 2020. Available online: https://gco.iarc.fr/today (accessed on 16 December 2021).
- Rawla, P.; Barsouk, A. Epidemiology of gastric cancer: Global trends, risk factors and prevention. Prz. Gastroenterol. 2019, 14, 26–38. [Google Scholar] [CrossRef]
- Hong, S.; Won, Y.J.; Park, Y.R.; Jung, K.W.; Kong, H.J.; Lee, E.S.; Community of Population-Based Regional Cancer Registries. Cancer Statistics in Korea: Incidence, Mortality, Survival, and Prevalence in 2017. Cancer Res Treat. 2020, 52, 335–350. [Google Scholar] [CrossRef]
- Katai, H.; Ishikawa, T.; Akazawa, K.; Isobe, Y.; Miyashiro, I.; Oda, I.; Tsujitani, S.; Ono, H.; Tanabe, S.; Fukagawa, T.; et al. Five-year survival analysis of surgically resected gastric cancer cases in Japan: A retrospective analysis of more than 100,000 patients from the nationwide registry of the Japanese Gastric Cancer Association (2001–2007). Gastric Cancer 2018, 21, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Tuo, J.Y.; Bi, J.H.; Yuan, H.Y.; Jiang, Y.F.; Ji, X.W.; Li, H.L.; Xiang, Y.B. Trends of stomach cancer survival: A systematic review of survival rates from population-based cancer registration. J. Dig. Dis. 2022, 23, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.W.; Wang, C.C.; Hung, W.C.; Liu, Y.H.; Sung, W.W.; Tsai, M.C. Improvement in the Mortality-to-Incidence Ratios for Gastric Cancer in Developed Countries with High Health Expenditures. Front. Public Health 2021, 9, 713895. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Hendifar, A.; Lenz, C.; Togawa, K.; Lenz, F.; Lurje, G.; Pohl, A.; Winder, T.; Ning, Y.; Groshen, S.; et al. Survival of metastatic gastric cancer: Significance of age, sex and race/ethnicity. J. Gastrointest. Oncol. 2011, 2, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Waddingham, W.; Nieuwenburg, S.A.V.; Carlson, S.; Rodriguez-Justo, M.; Spaander, M.; Kuipers, E.J.; Jansen, M.; Graham, D.G.; Banks, M. Recent advances in the detection and management of early gastric cancer and its precursors. Front. Gastroenterol. 2020, 12, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Mwachiro, M.; Topazian, H.M.; Kayamba, V.; Mulima, G.; Ogutu, E.; Erkie, M.; Lenga, G.; Mutie, T.; Mukhwana, E.; Desalegn, H.; et al. Gastrointestinal endoscopy capacity in Eastern Africa. Endosc. Int. Open 2021, 9, E1827–E1836. [Google Scholar] [CrossRef] [PubMed]
- Kayamba, V.; Kelly, P. Delayed referral for diagnostic endoscopy is a contributing factor to late gastric cancer diagnosis in Zambia. Health Press 2019, 3, 14–19. [Google Scholar]
- Rasmussen, S.; Haastrup, P.F.; Balasubramaniam, K.; Christensen, R.D.; Søndergaard, J.; Jarbøl, D.E. Predictive values of upper gastrointestinal cancer alarm symptoms in the general population: A nationwide cohort study. BMC Cancer 2018, 18, 440. [Google Scholar] [CrossRef] [Green Version]
- Maconi, G.; Manes, G.; Porro, G.B. Role of symptoms in diagnosis and outcome of gastric cancer. World J. Gastroenterol. 2008, 14, 1149–1155. [Google Scholar] [CrossRef] [PubMed]
- Bowles, M.J.; Benjamin, I.S. ABC of the upper gastrointestinal tract: Cancer of the stomach and pancreas. BMJ 2001, 323, 1413–1416. [Google Scholar] [CrossRef] [PubMed]
- Yao, K.; Uedo, N.; Kamada, T.; Hirasawa, T.; Nagahama, T.; Yoshinaga, S.; Oka, M.; Inoue, K.; Mabe, K.; Yao, T.; et al. Guidelines for endoscopic diagnosis of early gastric cancer. Dig. Endosc. 2020, 32, 663–698. [Google Scholar] [CrossRef] [PubMed]
- Kayamba, V.; Zyambo, K.; Kelly, P. Presence of blood in gastric juice: A sensitive marker for gastric cancer screening in a poor resource setting. PLoS ONE 2018, 13, e0205185. [Google Scholar] [CrossRef]
- Mathes, T.; Pieper, D. An algorithm for the classification of study designs to assess diagnostic, prognostic and predictive test accuracy in systematic reviews. Syst. Rev. 2019, 8, 226. [Google Scholar] [CrossRef] [Green Version]
- Kayamba, V.; Shibemba, A.; Zyambo, K.; Heimburger, D.C.; Morgan, D.; Kelly, P. HIV related hypochlorhydria does not appear to respond to anti-retroviral therapy in Zambian adults: A case control study. Pan Afr. Med. J. 2018, 31, 128. [Google Scholar] [CrossRef]
- Matsuoka, T.; Yashiro, M. Biomarkers of gastric cancer: Current topics and future perspective. World J. Gastroenterol. 2018, 24, 2818–2832. [Google Scholar] [CrossRef]
- González, C.A.; Agudo, A. Carcinogenesis, prevention and early detection of gastric cancer: Where we are and where we should go. Int. J. Cancer 2012, 130, 745–753. [Google Scholar] [CrossRef]
- Gong, E.J.; Ahn, J.Y.; Jung, H.Y.; Lim, H.; Choi, K.S.; Lee, J.H.; Kim, D.H.; Choi, K.D.; Song, H.J.; Lee, G.H.; et al. Risk factors and clinical outcomes of gastric cancer identified by screening endoscopy: A case-control study. J. Gastroenterol. Hepatol. 2014, 29, 301–309. [Google Scholar] [CrossRef]
- Chung, S.J.; Park, M.J.; Kang, S.J.; Kang, H.Y.; Chung, G.E.; Kim, S.G.; Jung, H.C. Effect of annual endoscopic screening on clinicopathologic characteristics and treatment modality of gastric cancer in a high-incidence region of Korea. Int. J. Cancer 2012, 131, 2376–2384. [Google Scholar] [CrossRef]
- Kim, G.H.; Bang, S.J.; Ende, A.R.; Hwang, J.H. Is screening and surveillance for early detection of gastric cancer needed in Korean Americans? Korean J. Intern. Med. 2015, 30, 747–758. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.G.; Kong, S.H.; Oh, S.Y.; Lee, K.G.; Suh, Y.S.; Yang, J.Y.; Choi, J.; Kim, S.G.; Kim, J.S.; Kim, W.H.; et al. Effects of screening on gastric cancer management: Comparative analysis of the results in 2006 and in 2011. J. Gastric Cancer 2014, 14, 129–134. [Google Scholar] [CrossRef] [Green Version]
- Latorre, S.G.; Álvarez, O.J.; Ivanovic-Zuvic, S.D.; Valdivia, C.G.; Margozzini, M.P.; Chianale, B.J.; Miquel, P.J.F. Coverage of diagnostic upper gastrointestinal endoscopy for the prevention of gastric cancer in Chile: Results from the 2009–2010 National Health Survey. Rev. Med. Chil. 2015, 143, 1198–1205. [Google Scholar]
- Jung, H.M.; Lee, J.S.; Lairson, D.R.; Kim, Y. The effect of national cancer screening on disparity reduction in cancer stage at diagnosis by income level. PLoS ONE 2015, 10, e0136036. [Google Scholar] [CrossRef]
- Venerito, M.; Goni, E.; Malfertheiner, P. Helicobacter pylori screening: Options and challenges. Expert Rev. Gastroenterol. Hepatol. 2016, 10, 497–503. [Google Scholar] [CrossRef]
- Kayamba, V.; Butt, J.; Varga, M.; Shibemba, A.; Piazuelo, M.B.; Wilson, K.T.; Zyambo, K.; Mwakamui, S.; Mulenga, C.; Waterboer, T.; et al. Serum antibodies to Helicobacter pylori antigens are associated with active gastric inflammation but not gastric cancer in patients seen at the University Teaching Hospital in Lusaka, Zambia. Malawi Med. J. 2022, 34, 17–24. [Google Scholar] [CrossRef]
- Kayamba, V.; Asombang, A.W.; Mudenda, V.; Lisulo, M.M.; Sinkala, E.; Mwanamakondo, S.; Mweemba, I.; Kelly, P. Gastric adenocarcinoma in Zambia: A case-control study of HIV, lifestyle risk factors, and biomarkers of pathogenesis. S. Afr. Med. J. 2013, 103, 255–259. [Google Scholar] [CrossRef] [Green Version]
- Kishikawa, H.; Kimura, K.; Takarabe, S.; Kaida, S.; Nishida, J. Helicobacter pylori Antibody Titer and Gastric Cancer Screening. Dis. Markers 2015, 2015, 156719. [Google Scholar] [CrossRef] [Green Version]
- Syrjänen, K. A Panel of Serum Biomarkers (GastroPanel®) in Non-invasive Diagnosis of Atrophic Gastritis. Systematic Review and Meta-analysis. Anticancer Res. 2016, 36, 5133–5144. [Google Scholar] [CrossRef] [Green Version]
- Lin, J.S.; Perdue, L.A.; Henrikson, N.B.; Bean, S.I.; Blasi, P.R. Screening for Colorectal Cancer: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA 2021, 325, 1978–1998. [Google Scholar] [CrossRef]
- Allard, J.; Cosby, R.; Del Giudice, M.E.; Irvine, E.J.; Morgan, D.; Tinmouth JAllard, J.; Cosby, R.; Del Giudice, M.E.; Irvine, E.J.; Morgan, D.; et al. Gastroscopy following a positive fecal occult blood test and negative colonoscopy: Systematic review and guideline. Can. J. Gastroenterol. 2010, 24, 113–120. [Google Scholar] [CrossRef]
- Nakama, H.; Zhang, B. Immunochemical fecal occult blood test is inadequate for screening test of stomach cancer. Dig. Dis. Sci. 2000, 45, 2195–2198. [Google Scholar] [CrossRef]
- Konrad, G.; Katz, A. Are medication restrictions before FOBT necessary? Practical advice based on a systematic review of the literature. Can. Fam. Physician 2012, 58, 939–948. [Google Scholar]

| H20 | H202 | Guaiacum | Blood | Result | |
|---|---|---|---|---|---|
| Tube 1 | + | + | – | – | No colour change |
| Tube 2 | – | + | – | + | No colour change |
| Tube 3 | – | + | + | – | No colour change |
| Tube 4 | – | – | + | + | No colour change |
| Tube 5 | – | + | + | + | Colour change to blue |
| Tube 6 | + | + | + | + | Colour change to blue |
| pH of the Solution | Volume of Blood Added in μL | |||||
|---|---|---|---|---|---|---|
| 1 | 5 | 10 | 20 | 30 | 40 | |
| pH 0 | – | – | – | – | – | – |
| pH 1 | – | – | – | – | – | + |
| pH 2 | – | – | + | + | + | + |
| pH 3 | – | + | + | + | + | + |
| pH 4 | – | + | + | + | + | + |
| pH 5 | – | + | + | + | + | + |
| pH 6 | – | + | + | + | + | + |
| pH 7 | – | + | + | + | + | + |
| Tube (pH) | Dissolving Time (min) |
|---|---|
| 0 | 23 |
| 1 | 25 |
| 2 | 29 |
| 3 | 29 |
| 4 | 30 |
| 5 | 31 |
| 6 | 29 |
| 7 | 28 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kayamba, V.; Kelly, P. Introducing the Sanguis-Filum for Detection of Gastric Mucosal Lesions Prior to Endoscopy: A Study Protocol. Diagnostics 2022, 12, 1160. https://doi.org/10.3390/diagnostics12051160
Kayamba V, Kelly P. Introducing the Sanguis-Filum for Detection of Gastric Mucosal Lesions Prior to Endoscopy: A Study Protocol. Diagnostics. 2022; 12(5):1160. https://doi.org/10.3390/diagnostics12051160
Chicago/Turabian StyleKayamba, Violet, and Paul Kelly. 2022. "Introducing the Sanguis-Filum for Detection of Gastric Mucosal Lesions Prior to Endoscopy: A Study Protocol" Diagnostics 12, no. 5: 1160. https://doi.org/10.3390/diagnostics12051160
APA StyleKayamba, V., & Kelly, P. (2022). Introducing the Sanguis-Filum for Detection of Gastric Mucosal Lesions Prior to Endoscopy: A Study Protocol. Diagnostics, 12(5), 1160. https://doi.org/10.3390/diagnostics12051160







