COVID-19 in Tunisia (North Africa): Seroprevalence of SARS-CoV-2 in the General Population of the Capital City Tunis
Abstract
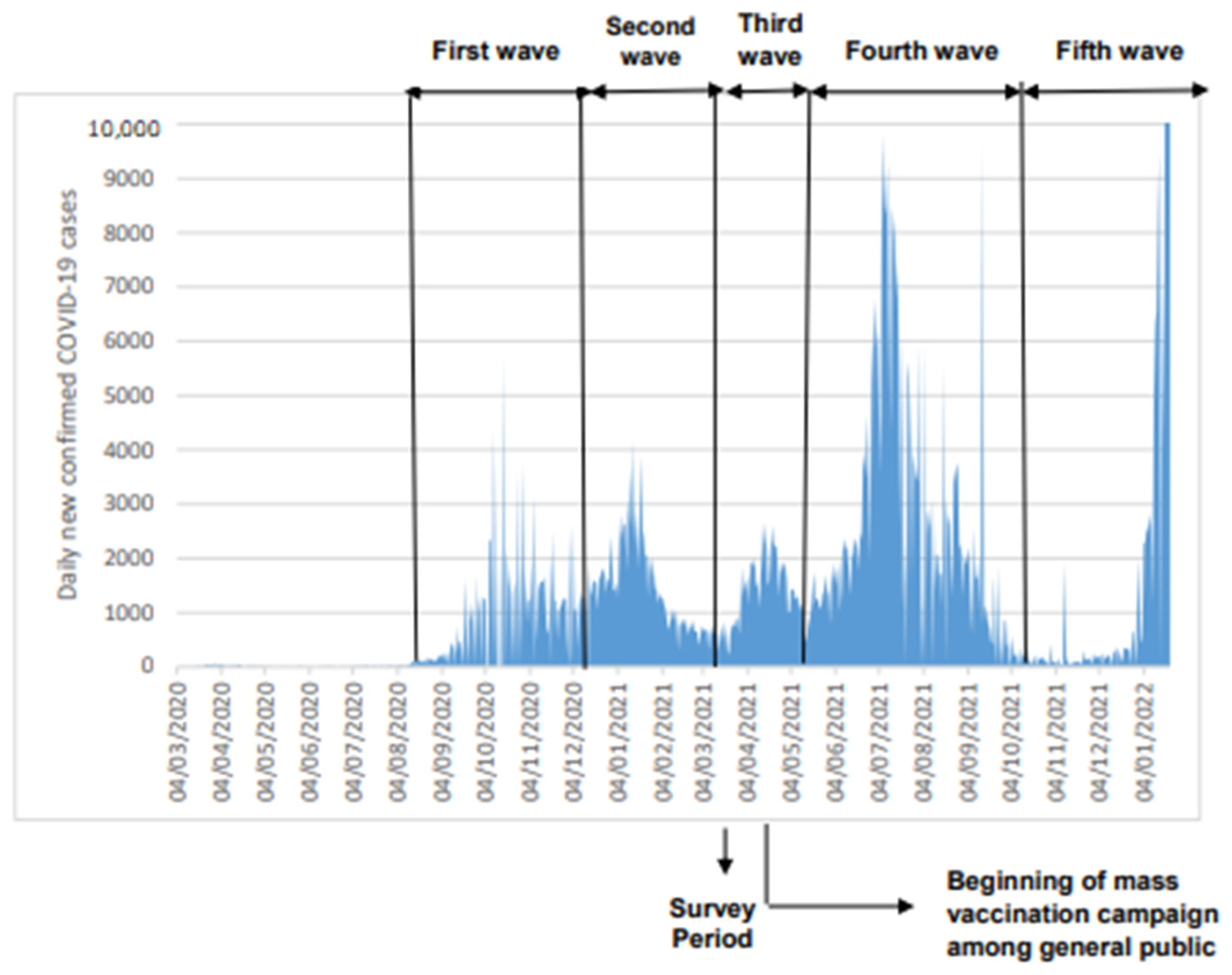
:1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Sampling Procedure
2.3. Sample Size
2.4. Data Collection
2.5. Data Analyses
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Questionnaire
| Household Report Form |
| First name–Last name | /______________________________________/ |
| Telephone number | /_____________________________________/ |
| Date of contact | ____/____/________ |
| Appointment date for the survey | ____/____/________ |
| Address | /__________________________________________ ___________________________________________/ | ||
| Dwelling type | □ Apartment □ Independent house | ||
| Number of rooms | _____ | ||
| Number of household members | _____ | ||
| First and last name of the household member | Sex | Age in years (months if <5 years) | Consent |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| □F □H | ____ years (____ months) | □Yes □ No | |
| Participant Report Form |
| First name–Last name | /______________________________________/ |
| Telephone number | /_____________________________________/ |
| Date of interview | ____/____/________ |
| First name–Last name | I__I__I__I__I__I__I__I__I__I__I__I__I__I__I__I__I__I__I__I__I__I |
| Sex | □ Male □ Female |
| Date of birth | ____/____/________ |
| Age in years (months if <5 years) | ____ years ____ months |
| Nationality | /______________________________________/ |
| Telephone number | /______________________________________/ |
| Profession/activity | □ Without activities/Retired □ Student/ Kindergarten Name and location of the school, university or kindergarten: □ Having an activity Nature of the activity/profession:…………………… Location of the work/activity:…………………………… |
| Morbid obesity (BMI> 40 kg/m2 if age ≥ 16 years) | □ Yes □ No □ Unknown |
| Cancer | □ Yes □ No □ Unknown |
| Diabetes | □ Yes □ No □ Unknown |
| HIV/other immune deficiency | □ Yes □ No □ Unknown |
| Heart disease | □ Yes □ No □ Unknown |
| Asthma | □ Yes □ No □ Unknown |
| Chronic respiratory disease (other than asthma) | □ Yes □ No □ Unknown |
| Chronic liver disease | □ Yes □ No □ Unknown |
| Blood disorder If yes, specify the disease: | □ Yes □ No □ Unknown |
| Chronic kidney disease | □ Yes □ No □ Unknown |
| Chronic neurological disorder | □ Yes □ No □ Unknown |
| Pregnancy | □ Yes □ No □ Unknown □ NA |
| If yes, specify:- Trimestre | □ First □ Second □ Third |
| - Estimated date of delivery | ____/___/________ |
| Other comorbidities | □ Yes □ No □ Unknown |
| If yes, specify: |
| Current tobacco smoking? | □ Yes □ No |
| Do you wear a face mask in public places? | □ Always □ Often □ Occasionally □ Never |
| Are you following recommended hand hygiene practices (using soap and/or hydroalcoholic solutions)? | □ Always □ Often □ Occasionally □ Never |
| Are you following the recommended physical distancing measures? | □ Always □ Often □ Occasionally □ Never |
| Since December 2019, have you traveled to a foreign country? | □ Yes □ No □ Unknown | |
| Since the begining of the COVID-19 pandemic (March 2020): | ||
| Seeking care in a health facility | □ Yes □ No □ Unknown | |
| What means of transportation do you usually use? | □Car □Public transport □Bicycle/motorcycle □Different means of transport □None | |
| A. Have you been tested for COVID-19? □ Yes □ No | |
| If No, have you presented any symptoms suggestive of Covid-19 since the diffusion of the coronavirus in Tunisia (mars 2020)? □ Yes□ No If yes, what symptoms? ………………………………..; If you answered Yes to question A, proceed to question B. If you answered No to question A, you do not have to answer the rest of the questions. | |
| B. Did you get a positive test result for COVID-19 □ Yes □ No If you answered No to question B, go to question C. If you answered Yes to question B, please answer questions: a, b, c, d. a. What was (were) the type(s) and result(s) of the diagnostic test(s) performed? □RT-PCR Result:…………………. □Rapid antigen test Result:………………… □Chest scan Result ………………… | |
| b. What was the date of COVID-19 diagnosis? c. What was the clinical form of COVID-19? | ____ / ____/ ________ □Symptomatic form □Asymptomatic form |
| d. If you had a symptomatic form, did you experience the following symptoms? | |
| -Fever (≥38 °C) □ Yes □ No □ Unknown | |
| -Sore throa □ Yes □ No □ Unknown | |
| -Cough □ Yes □ No □ Unknown | |
| -Runny nose □ Yes □ No □ Unknown | |
| -Shortness of breath □ Yes □ No □ Unknown | |
| -Sweating □ Yes □ No □ Unknown | |
| -Chills □ Yes □ No □ Unknown | |
| -Vomiting □ Yes □ No □ Unknown | |
| -Nausea □ Yes □ No □ Unknown | |
| -Diarrhea □ Yes □ No □ Unknown | |
| -Abdominal pain □ Yes □ No □ Unknown | |
| -Chest pain □ Yes □ No □ Unknown | |
| -Headache □ Yes □ No □ Unknown | |
| -Skin rash □ Yes □ No □ Unknown | |
| -Conjunctivitis □ Yes □ No □ Unknown | |
| -Muscle ache □ Yes □ No □ Unknown | |
| -Joint pain □ Yes □ No □ Unknown | |
| -Loss of appetite □ Yes □ No □ Unknown | |
| -Loss of taste □ Yes □ No □ Unknown | |
| -Loss of smell □ Yes □ No □ Unknown | |
| -Nosebleed □ Yes □ No □ Unknown | |
| -Tiredness □ Yes □ No □ Unknown | |
| -Convulsions □ Yes □ No □ Unknown | |
| -Loss of consciousness □ Yes □ No □ Unknown | |
| -Other symptoms □ Yes □ No □ Unknown. If yes, specify | |
| C. If you have had one or more diagnostic test(s) for COVID-19 and they were all negative, specify: | |
| a. Why did you do the test: □ Travel abroad □ Contact with a confirmed case of COVID-19 □ Presence of signs suggestive of COVID-19, if so, specify the symptoms …………………………………………………………□ Other, Specify:………………… b. Date of the last test: ___/___/_________ c. Type of the last test: □RT-PCR □Rapid antigen test □Chest scan | |
| Thank you for your participation | |
Appendix B. Distribution of Study Subjects According to the Risk Factors for Exposure to SARS-CoV-2, Tunis, March–April 2021
| Variables | N | % |
| Respect of preventive measures (n = 1663) | ||
| Frequently | 1138 | 68.4 |
| Occasionally/Never | 525 | 31.6 |
| Travelling abroad since December, 2019 (n = 1676) | ||
| Yes | 52 | 3.1 |
| No | 1624 | 96.9 |
| Contact with a COVID-19 case within the household (n = 1676) | ||
| Yes | 1198 | 71.5 |
| No | 478 | 28.5 |
| Seeking care in a health facility since the beginning of the COVID-19 pandemic in Tunisia (Mars 2020) (n = 1667) | ||
| Yes | 909 | 54.5 |
| No | 758 | 45.5 |
| Means of transport used (n = 1632) | ||
| Car | 607 | 37.2 |
| Public transport | 441 | 27.0 |
| Bicycle/motorcycle | 14 | 0.9 |
| Different means of transport | 176 | 10.8 |
| None | 394 | 24.1 |
Appendix C. Some Previous SARS-CoV-2 Seroprevalence Surveys among General Public before the Beginning of Mass Vaccination Campaigns
| Country | References | Population Size | Study Period | Antibodies | Antibody Detection Tests | Seroprevalence (%) |
| France | Le Vu et al. [21] | 2879 | May 2020 | IgG anti-S b IgG anti-N c pseudo-neutralizing antibodies | In-house test (LuLISA) | 4.9 |
| UAE a | Alsuwaidi et al. [22] | 8831 | July August 2020 | IgG anti-S | Commercial test (Diasorin) | 10.4 |
| Peru, Iquitos | Álvarez-Antonio et al. [25] | 716 | July 2020 | IgG/IgM | Rapid test | 70.0 |
| Colombia, Monteria | Mattar et al. [26] | 1368 | August 2020 | IgG/IgM/IgA | Commercial test (ELISA) | 55.3 |
| Israel | Reicher et al. [20] | 54,357 | June-Sept 2020 | IgG anti-S | Commercial test (Diasorin) | 4.6 |
| South Sudan (Juba) | Wiens et al. [27] | 2214 | August-September 2020 | IgG anti-S-RBD d | Commercial test (Quantitative ELISA) | 22.3 |
| Oman | Al-Abri et al. [23] | 4064 | November 2020 | IgG anti-S | Commercial test (Diasorin) | 22.0 |
| Sri Lanka | Jeewandara et al. [24] | 2547 | January 2021 | IgA, IgM, IgG anti-RBD | Commercial test (Wantai SARS-CoV-2 Ab ELISA) | 24.5 |
| Sierra Leone | Barrie et al. [18] | 1893 | March 2021 | IgG/IgM | Commercial Rapid test | 2.6 |
| Zimbabwe | Fryatt et al. [19] | 2340 | Feb-April 2021 | IgG anti-N b | Commercial test Roche Elecsys | 53.0 |
| a: United Arab Emirates; b: Immunoglobuline G antibodies to the the spike protein; c: Immunoglobuline G antibodies to the nucleocapsid protein; d: Immunoglobuline G antibodies to the receptor-binding domain of the spike protein. | ||||||
References
- World Health Organization. Listings of WHO’s Response to COVID-19. Available online: https://www.who.int/news/item/29-06-2020-covidtimeline (accessed on 30 November 2021).
- World Health Organization. Weekly Epidemiological Update on COVID-19—30 March 2021. Available online: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19—31-march-2021 (accessed on 30 November 2021).
- WHO. WHO Coronavirus (COVID-19) Dashboard. 2021. Available online: https://covid19.who.int (accessed on 30 November 2021).
- National Observatory of New and Emerging Diseases. COVID-19 en Tunisie: Point de Situation a la Date du 07 Juin 2020. Available online: https://www.onmne.tn/?p=10229 (accessed on 30 November 2021).
- National Observatory of New and Emerging diseases. Point de Situation sur L’épidémie D’infections au Nouveau Coronavirus « COVID-19 » à la Date du 17 Mars 2020. Available online: https://www.onmne.tn/?p=10363 (accessed on 22 December 2021).
- National Observatory of New and Emerging Diseases. Bulletin de Veille COVID-19 a la Date du 12 Décembre 2021. Available online: https://www.onmne.tn/?p=15695 (accessed on 22 December 2021).
- Ministry of Health. Point de Situation en Tunisie—Coronavirus. Available online: http://coronavirus.rns.tn/point-de-situation-en-tunisie/ (accessed on 10 January 2022).
- McGowan, C.R.; Hellman, N.; Chowdhury, S.; Mannan, A.; Newell, K.; Cummings, R. COVID-19 testing acceptability and uptake amongst the Rohingya and host community in Camp 21, Teknaf, Bangladesh. Confl. Health 2020, 14, 74. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Song, Y.; Wang, Q.; Pu, J.; Sun, F.Y.; Zhang, Y.; Zhou, X.; Larson, H.J.; Hou, Z. Public Attitudes and Factors of COVID-19 Testing Hesitancy in the United Kingdom and China: Comparative Infodemiology Study. JMIR Infodemiol. 2021, 1, e26895. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Chen, Z.; Azman, A.S.; Deng, X.; Sun, R.; Zhao, Z.; Zheng, N.; Chen, X.; Lu, W.; Zhuang, T.; et al. Serological evidence of human infection with SARS-CoV-2: A systematic review and meta-analysis. Lancet Glob. Health 2021, 5, e598–e609. [Google Scholar] [CrossRef]
- World Health Organization. Population-Based Age-Stratified Seroepidemiological Investigation Protocol for COVID-19 Virus Infection; WHO: Geneva, Switzerland, 17 March 2020. [Google Scholar]
- National Institute of Statistics. Bulletin Mensuel de la Statistique, Septembre 2021. Available online: http://www.ins.tn/publication/bulletin-mensuel-de-la-statistique-septembre-2021 (accessed on 10 January 2022).
- Benabdessalem, C.; Marzouki, S.; Hamouda, W.B.; Trabelsi, K.; Boumaiza, M.; Hamouda, S.B.; Ouni, R.; Bchiri, S.; Chaaban, A.; Gdoura, M.; et al. COVID-19 in Tunisia (North Africa): IgG and IgG Subclass Antibody Responses to SARS-CoV-2 According to Disease Severity [Preprint]. 2022. Available online: https://www.medrxiv.org/content/10.1101/2022.03.01.22271696v1 (accessed on 5 March 2022).
- National Institute of Statistics. Recensement Général de la Population et de l’Habitat 2014. Available online: http://ins.tn/enquetes/recensement-general-de-la-population-et-de-lhabitat-2014 (accessed on 10 January 2022).
- Sempos, C.T.; Tian, L. Adjusting Coronavirus Prevalence Estimates for Laboratory Test Kit Error. Am. J. Epidemiol. 2020, 190, 109–115. [Google Scholar] [CrossRef]
- Ritchie, H.; Ortiz-Ospina, E.; Beltekian, D.; Mathieu, E.; Hasell, J.; Macdonald, B.; Giattino, C.; Appel, C.; Rodés-Guirao, L.; Roser, M. Coronavirus Pandemic (COVID-19). 2021. Available online: https://ourworldindata.org/coronavirus (accessed on 14 December 2021).
- Centers for Disease Control and Prevention. Delta Variant: What We Know about the Science. Available online: https://www.cdc.gov/coronavirus/2019-ncov/variants/delta-variant.html (accessed on 12 January 2022).
- Barrie, M.B.; Lakoh, S.; Kelly, J.D.; Kanu, J.S.; Squire, J.S.; Koroma, Z.; Bah, S.; Sankoh, O.; Brima, A.; Ansumana, R.; et al. SARS-CoV-2 antibody prevalence in Sierra Leone, March 2021: A cross-sectional, nationally representative, age-stratified serosurvey. BMJ Glob. Health 2021, 6, e007271. [Google Scholar] [CrossRef]
- Fryatt, A.; Simms, V.; Bandason, T.; Redzo, N.; Olaru, I.D.; Ndhlovu, C.E.; Mujuru, H.; Rusakaniko, S.; Hoelscher, M.; Rubio-Acero, R.; et al. Community SARS-CoV-2 seroprevalence before and after the second wave of SARS-CoV-2 infection in Harare, Zimbabwe. eClinicalMedicine 2021, 41, 101172. [Google Scholar] [CrossRef]
- Reicher, S.; Ratzon, R.; Ben-Sahar, S.; Hermoni-Alon, S.; Mossinson, D.; Shenhar, Y.; Friger, M.; Lustig, Y.; Alroy-Preis, S.; Anis, E.; et al. Nationwide seroprevalence of antibodies against SARS-CoV-2 in Israel. Eur. J. Epidemiol. 2021, 36, 727–734. [Google Scholar] [CrossRef]
- Le Vu, S.; Jones, G.; Anna, F.; Rose, T.; Richard, J.-B.; Bernard-Stoecklin, S.; Goyard, S.; Demeret, C.; Helynck, O.; Escriou, N.; et al. Prevalence of SARS-CoV-2 antibodies in France: Results from nationwide serological surveillance. Nat. Commun. 2021, 12, 3025. [Google Scholar] [CrossRef]
- Alsuwaidi, A.R.; I Al Hosani, F.; Al Memari, S.; Narchi, H.; Wareth, L.A.; Kamal, H.; Al Ketbi, M.; Al Baloushi, D.; Elfateh, A.; Khudair, A.; et al. Seroprevalence of COVID-19 infection in the Emirate of Abu Dhabi, United Arab Emirates: A population-based cross-sectional study. Int. J. Epidemiol. 2021, 50, 1077–1090. [Google Scholar] [CrossRef]
- Al-Abri, S.S.; Al-Wahaibi, A.; Al-Kindi, H.; Kurup, P.J.; Al-Maqbali, A.; Al-Mayahi, Z.; Al-Tobi, M.H.; Al-Katheri, S.H.; Albusaidi, S.; Al-Sukaiti, M.H.; et al. Seroprevalence of SARS-CoV-2 antibodies in the general population of Oman: Results from four successive nationwide sero-epidemiological surveys. Int. J. Infect. Dis. 2021, 112, 269–277. [Google Scholar] [CrossRef]
- Jeewandara, C.; Guruge, D.; Abyrathna, I.S.; Danasekara, S.; Gunasekera, B.; Pushpakumara, P.D.; Madhusanka, D.; Jayathilaka, D.; Ranasinghe, T.; Somathilake, G.; et al. Seroprevalence of SARS-CoV-2 Infection in the Colombo Municipality Region, Sri Lanka. Front. Public Health 2021, 9, 724398. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Antonio, C.; Meza-Sánchez, G.; Calampa, C.; Casanova, W.; Carey, C.; Alava, F.; Rodríguez-Ferrucci, H.; Quispe, A.M. Seroprevalence of anti-SARS-CoV-2 antibodies in Iquitos, Peru in July and August, 2020: A population-based study. Lancet Glob. Health 2021, 9, e925–e931. [Google Scholar] [CrossRef]
- Mattar, S.; Alvis-Guzman, N.; Garay, E.; Rivero, R.; García, A.; Botero, Y.; Miranda, J.; Galeano, K.; De La Hoz, F.; Martínez, C.; et al. Severe Acute Respiratory Syndrome Coronavirus 2 Seroprevalence Among Adults in a Tropical City of the Caribbean Area, Colombia: Are We Much Closer to Herd Immunity Than Developed Countries? Open Forum Infect. Dis. 2020, 7, ofaa550. [Google Scholar] [CrossRef] [PubMed]
- Wiens, K.E.; Mawien, P.N.; Rumunu, J.; Slater, D.; Jones, F.K.; Moheed, S.; Caflish, A.; Bior, B.K.; Jacob, I.A.; Lako, R.L.L.; et al. Seroprevalence of Anti-SARS-CoV-2 IgG Antibodies in Juba, South Sudan: A Population-Based Study. Emerg. Infect. Dis. 2021, 27, 1598–1606. [Google Scholar] [CrossRef] [PubMed]
- Poustchi, H.; Darvishian, M.; Mohammadi, Z.; Shayanrad, A.; Delavari, A.; Bahadorimonfared, A.; Eslami, S.; Javanmard, S.H.; Shakiba, E.; Somi, M.H.; et al. SARS-CoV-2 antibody seroprevalence in the general population and high-risk occupational groups across 18 cities in Iran: A population-based cross-sectional study. Lancet Infect. Dis. 2020, 21, 473–481. [Google Scholar] [CrossRef]
- Mulenga, L.B.; Hines, J.Z.; Fwoloshi, S.; Chirwa, L.; Siwingwa, M.; Yingst, S.; Wolkon, A.; Barradas, D.T.; Favaloro, J.; Zulu, J.E.; et al. Prevalence of SARS-CoV-2 in six districts in Zambia in July, 2020: A cross-sectional cluster sample survey. Lancet Glob. Health 2021, 9, e773–e781. [Google Scholar] [CrossRef]
- Nkuba, A.N.; Makiala, S.M.; Guichet, E.; Tshiminyi, P.M.; Bazitama, Y.M.; Yambayamba, M.K.; Kazenza, B.M.; Kabeya, T.M.; Matungulu, E.B.; Baketana, L.K.; et al. High Prevalence of Anti–Severe Acute Respiratory Syndrome Coronavirus 2 (Anti–SARS-CoV-2) Antibodies After the First Wave of Coronavirus Disease 2019 (COVID-19) in Kinshasa, Democratic Republic of the Congo: Results of a Cross-sectional Household-Based Survey. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2021, 74, 882–890. [Google Scholar] [CrossRef]
- Mwananyanda, L.; Gill, C.J.; MacLeod, W.; Kwenda, G.; Pieciak, R.; Mupila, Z.; Lapidot, R.; Mupeta, F.; Forman, L.; Ziko, L.; et al. Covid-19 deaths in Africa: Prospective systematic postmortem surveillance study. BMJ 2021, 372, n334. [Google Scholar] [CrossRef]
- Ing, A.J.; Cocks, C.; Green, J.P. COVID-19: In the footsteps of Ernest Shackleton. Thorax 2020, 75, 693–694. [Google Scholar] [CrossRef]
- George, C.E.; Inbaraj, L.R.; Chandrasingh, S.; de Witte, L.P. High seroprevalence of COVID-19 infection in a large slum in South India; what does it tell us about managing a pandemic and beyond? Epidemiol. Infect. 2021, 149, 1–19. [Google Scholar] [CrossRef]
- McArthur, L.; Sakthivel, D.; Ataide, R.; Chan, F.; Richards, J.S.; Narh, C.A. Review of Burden, Clinical Definitions, and Management of COVID-19 Cases. Am. J. Trop. Med. Hyg. 2020, 103, 625–638. [Google Scholar] [CrossRef] [PubMed]
- Byambasuren, O.; Dobler, C.C.; Bell, K.; Rojas, D.P.; Clark, J.; McLaws, M.-L.; Glasziou, P. Comparison of seroprevalence of SARS-CoV-2 infections with cumulative and imputed COVID-19 cases: Systematic review. PLoS ONE 2021, 16, e0248946. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.L. COVID-19 will continue but the end of the pandemic is near. Lancet 2022, 399, 417–419. [Google Scholar] [CrossRef]
- Mossong, J.; Hens, N.; Jit, M.; Beutels, P.; Auranen, K.; Mikolajczyk, R.; Massari, M.; Salmaso, S.; Tomba, G.S.; Wallinga, J.; et al. Social Contacts and Mixing Patterns Relevant to the Spread of Infectious Diseases. PLoS Med. 2008, 5, e74. [Google Scholar] [CrossRef] [PubMed]
- Rumain, B.; Schneiderman, M.; Geliebter, A. Prevalence of COVID-19 in adolescents and youth compared with older adults in states experiencing surges. PLoS ONE 2021, 16, e0242587. [Google Scholar] [CrossRef] [PubMed]
- Dowell, A.C.; Butler, M.S.; Jinks, E.; Tut, G.; Lancaster, T.; Sylla, P.; Begum, J.; Bruton, R.; Pearce, H.; Verma, K.; et al. Children develop robust and sustained cross-reactive spike-specific immune responses to SARS-CoV-2 infection. Nat. Immunol. 2021, 23, 40–49. [Google Scholar] [CrossRef]
- Shakiba, M.; Nazemipour, M.; Salari, A.; Mehrabian, F.; Nazari, S.S.H.; Rezvani, S.M.; Ghasempour, Z.; Heidarzadeh, A.; Mansournia, M.A. Seroprevalence of SARS-CoV-2 in Guilan Province, Iran, April 2020. Emerg. Infect. Dis. 2021, 27, 636–638. [Google Scholar] [CrossRef]
- Stringhini, S.; Wisniak, A.; Piumatti, G.; Azman, A.S.; Lauer, S.A.; Baysson, H.; De Ridder, D.; Petrovic, D.; Schrempft, S.; Marcus, K.; et al. Seroprevalence of anti-SARS-CoV-2 IgG antibodies in Geneva, Switzerland (SEROCoV-POP): A population-based study. Lancet 2020, 396, 313–319. [Google Scholar] [CrossRef]
- Pollán, M.; Pérez-Gómez, B.; Pastor-Barriuso, R.; Oteo, J.; Hernán, M.A.; Perez-Olmeda, M.; Sanmartín, J.L.; Fernández-García, A.; Cruz, I.; de Larrea, N.F.; et al. Prevalence of SARS-CoV-2 in Spain (ENE-COVID): A nationwide, population-based seroepidemiological study. Lancet 2020, 396, 535–544. [Google Scholar] [CrossRef]
- Murhekar, M.V.; Bhatnagar, T.; Thangaraj, J.W.V.; Saravanakumar, V.; Kumar, M.S.; Selvaraju, S.; Rade, K.; Kumar, C.G.; Sabarinathan, R.; Turuk, A.; et al. SARS-CoV-2 seroprevalence among the general population and healthcare workers in India, December 2020–January 2021. Int. J. Infect. Dis. 2021, 108, 145–155. [Google Scholar] [CrossRef]
- Indenbaum, V.; Lustig, Y.; Mendelson, E.; Hershkovitz, Y.; Glatman-Freedman, A.; Keinan-Boker, L.; Bassal, R. Under-diagnosis of SARS-CoV-2 infections among children aged 0–15 years, a nationwide seroprevalence study, Israel, January 2020 to March 2021. Eurosurveillance 2021, 26, 2101040. [Google Scholar] [CrossRef]
- Ulyte, A.; Radtke, T.; Abela, I.A.; Haile, S.R.; Berger, C.; Huber, M.; Schanz, M.; Schwarzmueller, M.; Trkola, A.; Fehr, J.; et al. Clustering and longitudinal change in SARS-CoV-2 seroprevalence in school children in the canton of Zurich, Switzerland: Prospective cohort study of 55 schools. BMJ 2021, 372, n616. [Google Scholar] [CrossRef] [PubMed]
- Bundle, N.; Dave, N.; Pharris, A.; Spiteri, G.; Deogan, C.; Suk, J.E. Study group members COVID-19 trends and severity among symptomatic children aged 0–17 years in 10 European Union countries, 3 August 2020 to 3 October 2021. Eurosurveillance 2021, 26, 2101098. [Google Scholar] [CrossRef] [PubMed]
- Cloete, J.; Kruger, A.; Masha, M.; Plessis, N.M.D.; Mawela, D.; Tshukudu, M.; Manyane, T.; Komane, L.; Venter, M.; Jassat, W.; et al. Rapid Rise in Paediatric COVID-19 Hospitalisations during the Early Stages of the Omicron Wave, Tshwane District, South Africa [Preprint]. 2021. Available online: https://www.medrxiv.org/content/10.1101/2021.12.21.21268108v1#:~:text=Background%20South%20Africa%20reported%20a,COVID%2D19%2Dassociated%20hospitalisations (accessed on 15 January 2022).
- World Health Organization. COVID-19 Disease in Children and Adolescents: Scientific Brief, 29 September 2021. Available online: https://www.who.int/publications-detail-redirect/WHO-2019-nCoV-Sci_Brief-Children_and_adolescents-2021.1 (accessed on 23 January 2022).
- Bobrovitz, N.; Arora, R.K.; Cao, C.; Boucher, E.; Liu, M.; Donnici, C.; Yanes-Lane, M.; Whelan, M.; Perlman-Arrow, S.; Chen, J.; et al. Global seroprevalence of SARS-CoV-2 antibodies: A systematic review and meta-analysis. PLoS ONE 2021, 16, e0252617. [Google Scholar] [CrossRef] [PubMed]
- Hallal, P.C.; Hartwig, F.P.; Horta, B.L.; Silveira, M.F.; Struchiner, C.J.; Vidaletti, L.P.; Neumann, N.A.; Pellanda, L.C.; Dellagostin, O.A.; Burattini, M.N.; et al. SARS-CoV-2 antibody prevalence in Brazil: Results from two successive nationwide serological household surveys. Lancet Glob. Health 2020, 8, e1390–e1398. [Google Scholar] [CrossRef]
- Tuells, J.; Egoavil, C.M.; Pena Pardo, M.A.; Montagud, A.C.; Montagud, E.; Caballero, P.; Zapater, P.; Puig-Barberá, J.; Hurtado-Sanchez, J.A. Seroprevalence Study and Cross-Sectional Survey on COVID-19 for a Plan to Reopen the University of Alicante (Spain). Int. J. Environ. Res. Public Health 2021, 18, 1908. [Google Scholar] [CrossRef]
- Paleiron, N.; Mayet, A.; Marbac, V.; Perisse, A.; Barazzutti, H.; Brocq, F.-X.; Janvier, F.; Dautzenberg, B.; Bylicki, O. Impact of Tobacco Smoking on the Risk of COVID-19: A Large Scale Retrospective Cohort Study. Nicotine Tob. Res. Off. J. Soc. Res. Nicotine Tob. 2021, 23, 1398–1404. [Google Scholar] [CrossRef]
- Changeux, J.-P.; Amoura, Z.; Rey, F.A.; Miyara, M. A nicotinic hypothesis for Covid-19 with preventive and therapeutic implications. Comptes Rendus. Biol. 2020, 343, 33–39. [Google Scholar] [CrossRef]
- Clift, A.K.; von Ende, A.; Tan, P.S.; Sallis, H.M.; Lindson, N.; Coupland, C.A.C.; Munafò, M.R.; Aveyard, P.; Hippisley-Cox, J.; Hopewell, J.C. Smoking and COVID-19 outcomes: An observational and Mendelian randomisation study using the UK Biobank cohort. Thorax 2021, 77, 65–73. [Google Scholar] [CrossRef]
- Patanavanich, R.; Glantz, S.A. Smoking is associated with worse outcomes of COVID-19 particularly among younger adults: A systematic review and meta-analysis. BMC Public Health 2021, 21, 1–9. [Google Scholar] [CrossRef]
- Tude ComCor sur Les Lieux de Contamination au SARS-CoV-2: Où Les Français S’infectent-Ils? Available online: https://www.pasteur.fr/fr/espace-presse/documents-presse/etude-comcor-lieux-contamination-au-sars-cov-2-ou-francais-s-infectent-ils (accessed on 19 January 2022).
- Burbelo, P.D.; Riedo, F.X.; Morishima, C.; Rawlings, S.; Smith, D.; Das, S.; Strich, J.R.; Chertow, D.S.; Davey, R.T.; Cohen, J.I. Sensitivity in Detection of Antibodies to Nucleocapsid and Spike Proteins of Severe Acute Respiratory Syndrome Coronavirus 2 in Patients With Coronavirus Disease 2019. J. Infect. Dis. 2020, 222, jiaa273. [Google Scholar] [CrossRef] [PubMed]
- Gallais, F.; Gantner, P.; Bruel, T.; Velay, A.; Planas, D.; Wendling, M.-J.; Bayer, S.; Solis, M.; Laugel, E.; Reix, N.; et al. Evolution of antibody responses up to 13 months after SARS-CoV-2 infection and risk of reinfection. EBioMedicine 2021, 71, 103561. [Google Scholar] [CrossRef] [PubMed]
- Schoenhals, M.; Rabenindrina, N.; Rakotondramanga, J.M.; Dussart, P.; Randremanana, R.; Heraud, J.-M.; Andriamandimby, S.F.; Sahondranirina, P.H.; Vololoniaina, M.C.A.; Randriatsarafara, F.M.; et al. SARS-CoV-2 antibody seroprevalence follow-up in Malagasy blood donors during the 2020 COVID-19 Epidemic. EBioMedicine 2021, 68, 103419. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Science Brief: SARS-CoV-2 Infection-Induced and Vaccine-Induced Immunity. Available online: https://www.cdc.gov/coronavirus/2019-ncov/science/science-briefs/vaccine-induced-immunity.html (accessed on 22 January 2022).
| Characteristics | El Omrane (%) | La Goulette (%) | Both (%) |
|---|---|---|---|
| Total | 851 (50.8) | 825 (49.2) | 1676 (100.0) |
| Sex | n = 851 | n = 825 | n = 1676 |
| Male | 327 (38.4) | 301 (36.5) | 628 (37.5) |
| Female | 524 (61.6) | 524 (63.5) | 1048 (62.5) |
| Age | n = 851 | n = 825 | n = 1676 |
| <10 | 49 (5.8) | 33 (4.0) | 82 (4.9) |
| (10–20) | 118 (13.9) | 93 (11.3) | 211 (12.6) |
| (20–30) | 88 (10.3) | 94 (11.4) | 182 (10.9) |
| (30–40) | 98 (11.5) | 130 (15.8) | 228 (13.6) |
| (40–50) | 122 (14.3) | 151 (18.3) | 273 (16.3) |
| (50–60) | 143 (16.8) | 133 (16.1) | 276 (16.5) |
| ≥60 | 233 (27.4) | 191 (23.2) | 424 (25.3) |
| Occupation | n = 848 | n = 823 | n = 1671 |
| Without professional and educational activities | 441 (52.0) | 330 (40.1) | 771 (46.1) |
| Employee/student | 407 (48.0) | 493 (59.9) | 900 (53.9) |
| Comorbidities | n = 850 | n = 823 | n = 1673 |
| Yes | 339 (39.9) | 353 (42.9) | 692 (41.4) |
| No | 511 (60.1) | 470 (57.1) | 981 (58.6) |
| Current tobacco use (n = 1670) | n = 847 | n = 823 | n = 1670 |
| Yes | 185 (21.8) | 189 (23.0) | 374 (22.4) |
| No | 662 (78.2) | 634 (77.0) | 1296 (77.6) |
| Dwelling type (n = 1642) | n = 849 | n = 793 | n = 1642 |
| Apartment | 11 (1.3) | 139 (17.5) | 150 (9.1) |
| Independent house | 838 (98.7) | 654 (82.5) | 1492 (90.9) |
| El Omrane % (95% CI) * | La Goulette % (95% CI) | Both % (95% CI) | |
|---|---|---|---|
| Unweighted seroprevalence | |||
| Anti-S-RBD IgG † antibodies | 32.0 (28.9–35.2) | 29.8 (26.8–33.0) | 30.9 (28.7–33.1) |
| Anti-N IgG § antibodies | 33.6 (30.5–36.8) | 26.3 (23.4–29.4) | 30.0 (27.9–32.2) |
| Anti-S-RBD IgG antibodies and Anti-N IgG antibodies | 24.2 (21.4–27.2) | 22.2 (19.5–25.1) | 23.2 (21.2–25.3) |
| Anti-S-RBD IgG antibodies and/or Anti-N IgG antibodies | 41.5 (38.2–44.8) | 33.9 (30.8–37.2) | 37.8 (35.5–40.1) |
| Weighted seroprevalence | |||
| Anti-S-RBD IgG antibodies | 30.8 (27.3–34.5) | 27.4 (23.5–31.7) | 29.1 (26.5–32.0) |
| Anti-N IgG antibodies | 34.1 (30.3–38.1) | 26.2 (20.7–32.6) | 30.2 (26.9–33.8) |
| Anti-S-RBD IgG antibodies and Anti-N IgG antibodies | 23.1 (19.9–26.6) | 19.7 (16.4–23.5) | 21.4 (19.1–24.0) |
| Anti-S-RBD IgG antibodies and/or Anti-N IgG antibodies | 41.9 (38.0–45.9) | 34.0 (28.5–39.9) | 38.0 (34.6–41.5) |
| The weighted and test-performance adjusted seroprevalence | |||
| Anti-S-RBD IgG antibodies | 27.0 (23.1–31.2) | 23.1 (18.7–28.1) | 25.1 (22.2–28.4) |
| Anti-N IgG antibodies | 31.1 (26.8–35.7) | 22.1 (15.7–29.4) | 26.6 (22.9–30.8) |
| Variables | N | Anti SARS-CoV-2 Positive | Seropositivity Prevalence % (95% CI) * | Weighted Seropositivity Prevalence % (95% CI) | Crude ORc † (95% CI) | p-Value |
|---|---|---|---|---|---|---|
| Sex | NS | |||||
| Male | 628 | 236 | 37.6 (33.9–49.4) | 39.8 (33.4–46.5) | 1.1 (0.9–1.4) | |
| Female | 1048 | 397 | 37.9 (34.9–40.9) | 36.9 (33.3–40.7) | 1 | |
| Age | <10−3 | |||||
| <10 | 82 | 42 | 51.2 (40.6–61.7) | 51.1 (35.2–66.8) | 1.9 (1.2–2.9) | |
| (10–20) | 211 | 101 | 47.9 (41.2–54.6) | 46.0 (39.0–53.2] | 1.4 (0.9–2.1] | |
| (20–30) | 182 | 50 | 27.5 (21.5–34.4) | 27.4 (21.4–34.4) | 0.6 (0.4–0.9) | |
| (30–40) | 228 | 73 | 32.0 (26.3–38.3) | 32.2 (26.4–38.6) | 0.9 (0.6–1.3) | |
| (40–50) | 273 | 102 | 37.4 (31.8–43.2) | 38.1 (32.4–44.1) | 1.1 (0.7–1.6) | |
| (50–60) | 276 | 108 | 39.1 (33.6–45.0) | 37.7 (32.1–43.6) | 1.1 (0.7–1.6) | |
| ≥60 | 424 | 157 | 37.0 (32.6–41.7) | 37.1 (32.6–41.8) | 1 | |
| Occupation | NS | |||||
| Without professional and educational activities | 771 | 293 | 38.0 (34.6–41.5) | 39.7 (35.2–44.5) | 1 | |
| Employee/student | 900 | 338 | 37.6 (34.4–40.8) | 37.0 (32.5–41.8) | 0.9 (0.7–1.1) | |
| Comorbidities | NS | |||||
| Yes | 692 | 263 | 38.0 (34.4–49.7) | 36.5 (32.1–41.1) | 0.9 (0.7–1.1) | |
| No | 981 | 369 | 37.6 (34.6–40.7) | 38.7 (34.2–43.3) | 1 | |
| Current tobacco use | <10−3 | |||||
| Yes | 374 | 98 | 26.2 (22.0–30.9) | 25.7 (21.2–30.8) | 0.5 (0.4–0.6) | |
| No | 1296 | 530 | 40.9 (38.2–43.6) | 41.2 (37.2–45.3) | 1 | |
| Respect of preventive measures | NS | |||||
| Frequently | 1138 | 457 | 40.2 (37.3–43.0) | 39.2 (36.1–42.4) | 1 | |
| Occasionally/ Never | 525 | 169 | 32.2 (28.1–36.3) | 35.7 (28.5–43.6) | 0.9 (0.7–1.1) | |
| Travelling abroad since December, 2019 | NS | |||||
| Yes | 12 | 3 | 25.0 (8.9–53.2) | 31.1 (19.1–46.2) | 0.7 (0.4–1.3) | |
| No | 1664 | 630 | 37.9 (35.6–40.2) | 38.2 (34.8–41.8) | 1 | |
| Contact with a COVID-19 case within the household | <10−3 | |||||
| Yes | 1198 | 510 | 42.6 (39.8–45.4) | 42.7 (38.6–47.0) | 2.3 (1.8–2.9) | |
| No | 478 | 123 | 25.7 (22.0–29.8) | 24.7 (20.5–29.4) | 1 | |
| Seeking care in a health facility since the beginning of the COVID-19 pandemic in Tunisia (Mars 2020) | NS | |||||
| Yes | 909 | 347 | 38.2 (35.1–49.4) | 36.4 (32.8–40.2) | 0.9 (0.7–1.1) | |
| No | 758 | 281 | 37.1 (33.7–40.6) | 39.5 (34.1–45.3) | 1 | |
| Means of transport used | NS | |||||
| Car | 607 | 229 | 37.7 (33.9–41.6) | 38.6 (31.9–45.7) | 1.0 (0.8–1.3) | |
| Public transport | 441 | 164 | 37.2 (32.8–49.8) | 35.8 (30.4–41.5) | 0.9 (0.7–1.2) | |
| Bicycle/motorcycle | 14 | 3 | 21.4 (7.8–47.6) | 22.9 (7.0–53.9) | 0.5 (0.1–1.7) | |
| Different means of transport | 176 | 71 | 40.3 (33.4–47.7) | 43.1 (34.6–52.1) | 1.2 (0.8–1.8) | |
| None | 394 | 150 | 38.1 (33.4–42.9) | 38.3 (32.8–44.0) | 1 | |
| Previous diagnosis of COVID-19 infection | <10−3 | |||||
| Yes | 73 | 56 | 76.7 (65.8–84.9) | 71.6 (58.4–81.9) | 4.3 (2.4–7.9) | |
| No | 1603 | 577 | 36.0 (33.7–38.4) | 36.7 (33.2–40.3) | 1 | |
| History of COVID-19 related symptoms | <10−3 | |||||
| Yes | 254 | 148 | 58.3 (52.1–64.2) | 52.6 (45.5–59.6) | 2.0 (1.5–2.6) | |
| No | 1408 | 481 | 34.2 (31.7–36.7) | 35.5 (31.7–39.5) | 1 | |
| Dwelling type | NS | |||||
| Apartment | 150 | 49 | 32.7 (25.7–40.5) | 33.0 (25.2–42.0) | 0.8 (0.5–1.2) | |
| Independent house | 1492 | 576 | 38.6 (36.2–41.1) | 38.8 (35.1–42.5) | 1 |
| Variables | ORa * | (95% CI) † | p-Value |
|---|---|---|---|
| Current tobacco use | |||
| Yes | 0.6 | (0.5–0.8) | 0.001 |
| No | 1 | ||
| Previous diagnosis of COVID-19 infection | |||
| Yes | 3.1 | (1.6–5.8) | <10−3 |
| No | 1 | ||
| History of COVID-19 related symptoms | <10−3 | ||
| Yes | 1.8 | (1.3–2.5) | |
| No | 1 | ||
| Age | 0.03 | ||
| <10 | 1.9 | (1.2–2.9) | |
| (10–20) | 1.4 | (0.9–2.1) | |
| (20–30) | 0.6 | (0.4–0.9) | |
| (30–40) | 0.9 | (0.6–1.3) | |
| (40–50) | 1.0 | (0.7–1.6) | |
| (50–60) | 1.1 | (0.7–1.6) | |
| ≥ 60 | 1 | ||
| Contact with a COVID-19 case within the household | <10−3 | ||
| Yes | 2.1 | (1.3–2.5) | |
| No | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cherif, I.; Kharroubi, G.; Chaabane, S.; Yazidi, R.; Dellagi, M.; Snoussi, M.A.; Salem, S.; Marzouki, S.; Kammoun Rebai, W.; Rourou, S.; et al. COVID-19 in Tunisia (North Africa): Seroprevalence of SARS-CoV-2 in the General Population of the Capital City Tunis. Diagnostics 2022, 12, 971. https://doi.org/10.3390/diagnostics12040971
Cherif I, Kharroubi G, Chaabane S, Yazidi R, Dellagi M, Snoussi MA, Salem S, Marzouki S, Kammoun Rebai W, Rourou S, et al. COVID-19 in Tunisia (North Africa): Seroprevalence of SARS-CoV-2 in the General Population of the Capital City Tunis. Diagnostics. 2022; 12(4):971. https://doi.org/10.3390/diagnostics12040971
Chicago/Turabian StyleCherif, Ines, Ghassen Kharroubi, Sana Chaabane, Rihab Yazidi, Mongi Dellagi, Mohamed Ali Snoussi, Sadok Salem, Soumaya Marzouki, Wafa Kammoun Rebai, Samia Rourou, and et al. 2022. "COVID-19 in Tunisia (North Africa): Seroprevalence of SARS-CoV-2 in the General Population of the Capital City Tunis" Diagnostics 12, no. 4: 971. https://doi.org/10.3390/diagnostics12040971
APA StyleCherif, I., Kharroubi, G., Chaabane, S., Yazidi, R., Dellagi, M., Snoussi, M. A., Salem, S., Marzouki, S., Kammoun Rebai, W., Rourou, S., Dellagi, K., Barbouche, M. R., Benabdessalem, C., Ben Ahmed, M., & Bettaieb, J. (2022). COVID-19 in Tunisia (North Africa): Seroprevalence of SARS-CoV-2 in the General Population of the Capital City Tunis. Diagnostics, 12(4), 971. https://doi.org/10.3390/diagnostics12040971






