Features of Extrinsic Plantar Muscles in Patients with Plantar Fasciitis by Ultrasound Imaging: A Retrospective Case Control Research
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design
2.2. Participants
2.3. Ultrasonography Examination
2.4. Image Analysis
2.5. Data Analysis
3. Results
4. Discussion
4.1. Limitations and Future Lines
4.2. Clinical Applications
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Reeves, N.D.; Maganaris, C.N.; Narici, M.V. Ultrasonographic assessment of human skeletal muscle size. Eur. J. Appl. Physiol. 2004, 91, 116–118. [Google Scholar] [CrossRef] [PubMed]
- Weide, G.; van der Zwaard, S.; Huijing, P.A.; Jaspers, R.T.; Harlaar, J. 3D Ultrasound Imaging: Fast and Cost-effective Morphometry of Musculoskeletal Tissue. J. Vis. Exp. 2017, 129, 55493. [Google Scholar] [CrossRef] [Green Version]
- Ríos-Díaz, J.; del Baño-Aledo, M.E.; Tembl-Ferrairó, J.I.; Chumillas, M.J.; Vázquez-Costa, J.F.; Martínez-Payá, J.J. Quantitative neuromuscular ultrasound analysis as biomarkers in amyotrophic lateral sclerosis. Eur. Radiol. 2019, 29, 4266–4275. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, J.L.; Ellis, R.; Hodges, P.W.; Osullivan, C.; Hides, J.; Carnero, S.F.; Arias-Buria, J.L.; Teyhen, D.S.; Stokes, M.J. Imaging with ultrasound in physical therapy: What is the PT’s scope of practice? A competency-based educational model and training recommendations. Br. J. Sports Med. 2019, 53, 1447–1453. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, M.; Fukumoto, Y.; Kobayashi, M.; Kawasaki, T.; Maegawa, S.; Ibuki, S.; Ichihashi, N. Quantity and Quality of the Lower Extremity Muscles in Women with Knee Osteoarthritis. Ultrasound Med. Biol. 2015, 41, 2567–2574. [Google Scholar] [CrossRef]
- Lobo, C.C.; Morales, C.R.; Sanz, D.R.; Corbalan, I.S.; Marin, A.G.; Lopez, D.L. Ultrasonography Comparison of Peroneus Muscle Cross-sectional Area in Subjects with or without Lateral Ankle Sprains. J. Manip. Physiol. Ther. 2016, 39, 635–644. [Google Scholar] [CrossRef] [Green Version]
- Romero-Morales, C.; Martín-Llantino, P.J.; Calvo-Lobo, C.; Almazán-Polo, J.; López-López, D.; De-La-Cruz-Torres, B.; Palomo-López, P.; Rodríguez-Sanz, D. Intrinsic foot muscles morphological modifications in patients with Achilles tendinopathy: A novel case-control research study. Phys. Ther. Sport 2019, 40, 208–212. [Google Scholar] [CrossRef]
- Romero-Morales, C.; Martin-Llantino, P.J.; Calvo-Lobo, C.; Sanchez-Gomez, R.; Lopez-Lopez, D.; Pareja-Galeano, H.; Rodríguez-Sanz, D. Ultrasound evaluation of extrinsic foot muscles in patients with chronic non-insertional Achilles tendinopathy: A case-control study. Phys. Ther. Sport 2019, 37, 44–48. [Google Scholar] [CrossRef]
- Molinari, F.; Caresio, C.; Acharya, U.R.; Mookiah, M.R.K.; Minetto, M.A. Advances in quantitative muscle ultrasonography using texture analysis of ultrasound images. Ultrasound Med. Biol. 2015, 41, 2520–2532. [Google Scholar] [CrossRef]
- Caresio, C.; Molinari, F.; Emanuel, G.; Minetto, M.A. Muscle echo intensity: Reliability and conditioning factors. Clin. Physiol. Funct. Imaging 2015, 35, 393–403. [Google Scholar] [CrossRef]
- Wong, V.; Spitz, R.W.; Bell, Z.W.; Viana, R.B.; Chatakondi, R.N.; Abe, T.; Loenneke, J.P. Exercise induced changes in echo intensity within the muscle: A brief review. J. Ultrasound 2020, 23, 457–472. [Google Scholar] [CrossRef]
- Wilkinson, T.J.; Ashman, J.; Baker, L.A.; Watson, E.L.; Smith, A.C. Quantitative Muscle Ultrasonography Using 2D Textural Analysis: A Novel Approach to Assess Skeletal Muscle Structure and Quality in Chronic Kidney Disease. Ultrason. Imaging 2021, 43, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, L.; Gu, S.; Sun, J.; Qin, Z.; Yue, J.; Zhong, Y.; Ding, N.; Gao, R. Comparative effectiveness of extracorporeal shock wave, ultrasound, low-level laser therapy, noninvasive interactive neurostimulation, and pulsed radiofrequency treatment for treating plantar fasciitis: A systematic review and network meta-analysis. Medicine 2018, 97, e12819. [Google Scholar] [CrossRef] [PubMed]
- Moyne-Bressand, S.; Dhieux, C.; Dousset, E.; Decherchi, P. Effectiveness of Foot Biomechanical Orthoses to Relieve Patients Suffering from Plantar Fasciitis: Is the Reduction of Pain Related to Change in Neural Strategy? Biomed. Res. Int. 2018, 2018, 3594150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choudhary, R.; Kunal, K. Modifiable Risk Factors of Plantar Fasciitis in Non-Athletic Patients and Proposal of a New Objective Assessment System—RKISP. Rev. Bras. Ortop. 2021, 56, 368–371. [Google Scholar]
- Lemont, H.; Ammirati, K.M.; Usen, N. Plantar fasciitis: A degenerative process (fasciosis) without inflammation. J. Am. Podiatr. Med. Assoc. 2003, 93, 234–237. [Google Scholar] [CrossRef] [PubMed]
- Cotchett, M.; Rathleff, M.S.; Dilnot, M.; Landorf, K.B.; Morrissey, D.; Barton, C. Lived experience and attitudes of people with plantar heel pain: A qualitative exploration. J. Foot Ankle Res. 2020, 13, 12. [Google Scholar] [CrossRef] [Green Version]
- Schwartz, E.N.; Su, J. Plantar fasciitis: A concise review. Perm. J. 2014, 18, e105–e107. [Google Scholar] [CrossRef] [Green Version]
- Crofts, G.; Angin, S.; Mickle, K.J.; Hill, S.; Nester, C.J. Reliability of ultrasound for measurement of selected foot structures. Gait Posture 2014, 39, 35–39. [Google Scholar] [CrossRef]
- Otis, J.C.; Deland, J.T.; Lee, S.; Gordon, J. Peroneus brevis is a more effective evertor than peroneus longus. Foot Ankle Int. 2004, 25, 242–246. [Google Scholar] [CrossRef]
- Balen, P.F.; Helms, C.A. Association of posterior tibial tendon injury with spring ligament injury, sinus tarsi abnormality, and plantar fasciitis on MR imaging. AJR Am. J. Roentgenol. 2001, 176, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Huerta, J.P. The effect of the gastrocnemius on the plantar fascia. Foot Ankle Clin. 2014, 19, 701–718. [Google Scholar] [CrossRef] [PubMed]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thong-On, S.; Bovonsunthonchai, S.; Vachalathiti, R.; Intiravoranont, W.; Suwannarat, S.; Smith, R. Effects of Strengthening and Stretching Exercises on the Temporospatial Gait Parameters in Patients with Plantar Fasciitis: A Randomized Controlled Trial. Ann. Rehabil. Med. 2019, 43, 662–676. [Google Scholar] [CrossRef]
- Renan-Ordine, R.; Alburquerque-Sendín, F.; De Souza, D.P.R.; Cleland, J.A.; Fernández-De-Las-Penas, C. Effectiveness of myofascial trigger point manual therapy combined with a self-stretching protocol for the management of plantar heel pain: A randomized controlled trial. J. Orthop. Sports Phys. Ther. 2011, 41, 43–50. [Google Scholar] [CrossRef] [Green Version]
- Dunning, J.; Butts, R.; Mourad, F.; Young, I.; Flannagan, S.; Perreault, T. Dry needling: A literature review with implications for clinical practice guidelines. Phys. Ther. Rev. 2014, 19, 252–265. [Google Scholar] [CrossRef]
- Johnson, A.W.; Bruening, D.A.; Violette, V.A.; Perkins, K.V.; Thompson, C.L.; Ridge, S.T. Ultrasound Imaging Is Reliable for Tibialis Posterior Size Measurements. J. Ultrasound Med. 2020, 39, 2305–2312. [Google Scholar] [CrossRef]
- Narici, M.V.; Binzoni, T.; Hiltbrand, E.; Fasel, J.; Terrier, F.; Cerretelli, P. In vivo human gastrocnemius architecture with changing joint angle at rest and during graded isometric contraction. J. Physiol. 1996, 496 Pt 1, 287–297. [Google Scholar] [CrossRef]
- Sarafraz, H.; Hadian, M.R.; Yazdi, N.A.; Olyaei, G.; Bagheri, H.; Jalaie, S.; Rasouli, O. Test-retest reliability of nerve and muscle morphometric characteristics utilizing ultrasound imaging in individuals with unilateral sciatica and controls. Chiropr. Man. Ther. 2018, 26, 47. [Google Scholar] [CrossRef]
- Calhoun, J.H.; Li, F.; Ledbetter, B.R.; Viegas, S.F. A comprehensive study of pressure distribution in the ankle joint with inversion and eversion. Foot Ankle Int. 1994, 15, 125–133. [Google Scholar] [CrossRef]
- Brockett, C.L.; Chapman, G.J. Biomechanics of the ankle. Orthop. Trauma 2016, 30, 232–238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kimizuka, M.; Kurosawa, H.; Fukubayashi, T. Load-bearing pattern of the ankle joint. Arch. Orthop. Trauma Surg. 1980, 96, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.S.B.; Zambardino, J.A.; Banning, V.A. Transverse-plane mechanics at the knee and tibia in runners with and without a history of achilles tendonopathy. J. Orthop. Sports Phys. Ther. 2008, 38, 761–767. [Google Scholar] [CrossRef] [Green Version]
- Maffulli, N.; Longo, U.G.; Kadakia, A.; Spiezia, F. Achilles tendinopathy. Foot Ankle Surg. 2020, 26, 240–249. [Google Scholar] [CrossRef]
- Stecco, C.; Corradin, M.; Macchi, V.; Morra, A.; Porzionato, A.; Biz, C.; De Caro, R. Plantar fascia anatomy and its relationship with Achilles tendon and paratenon. J. Anat. 2013, 223, 665–676. [Google Scholar] [CrossRef]
- Heckmatt, J.Z.; Dubowitz, V.; Leeman, S. Detection of pathological change in dystrophic muscle with B-scan ultrasound imaging. Lancet 1980, 8183, 1389–1390. [Google Scholar] [CrossRef]
- Pillen, S.; Tak, R.O.; Zwarts, M.J.; Lammens, M.M.Y.; Verrijp, K.N.; Arts, I.M.P.; van der Laak, J.A.; Hoogerbrugge, P.M.; van Engelen, B.G.M.; Verrips, A. Skeletal muscle ultrasound: Correlation between fibrous tissue and echo intensity. Ultrasound Med. Biol. 2009, 35, 443–446. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Payá, J.J.; Ríos-Díaz, J.; Medina-Mirapeix, F.; Vázquez-Costa, J.F.; del Baño-Aledo, M.E. Monitoring Progression of Amyotrophic Lateral Sclerosis Using Ultrasound Morpho-Textural Muscle Biomarkers: A Pilot Study. Ultrasound Med. Biol. 2018, 44, 102–109. [Google Scholar] [CrossRef]
- Giacchino, M.; Caresio, C.; Gorji, N.E.; Molinari, F.; Massazza, G.; Minetto, M.A. Quantitative analysis of patellar tendon size and structure in asymptomatic professional players: Sonographic study. Muscles Ligaments Tendons J. 2017, 7, 449–458. [Google Scholar] [CrossRef]
- Duenwald, S.; Kobayashi, H.; Frisch, K.; Lakes, R.; Vanderby, R. Ultrasound echo is related to stress and strain in tendon. J. Biomech. 2011, 44, 424–429. [Google Scholar] [CrossRef] [Green Version]
- Frisch, K.E.; Marcu, D.; Baer, G.S.; Thelen, D.G.; Vanderby, R. Influence of tendon tears on ultrasound echo intensity in response to loading. J. Biomech. 2014, 47, 3813–3819. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martinez-Paya, J.J.; Del Bano-Aledo, M.E.; Rios-Diaz, J.; Tembl-Ferrairo, J.I.; Vazquez-Costa, J.F.; Medina-Mirapeix, F. Muscular Echovariation: A New Biomarker in Amyotrophic Lateral Sclerosis. Ultrasound Med. Biol. 2017, 43, 1153–1162. [Google Scholar] [CrossRef] [PubMed]
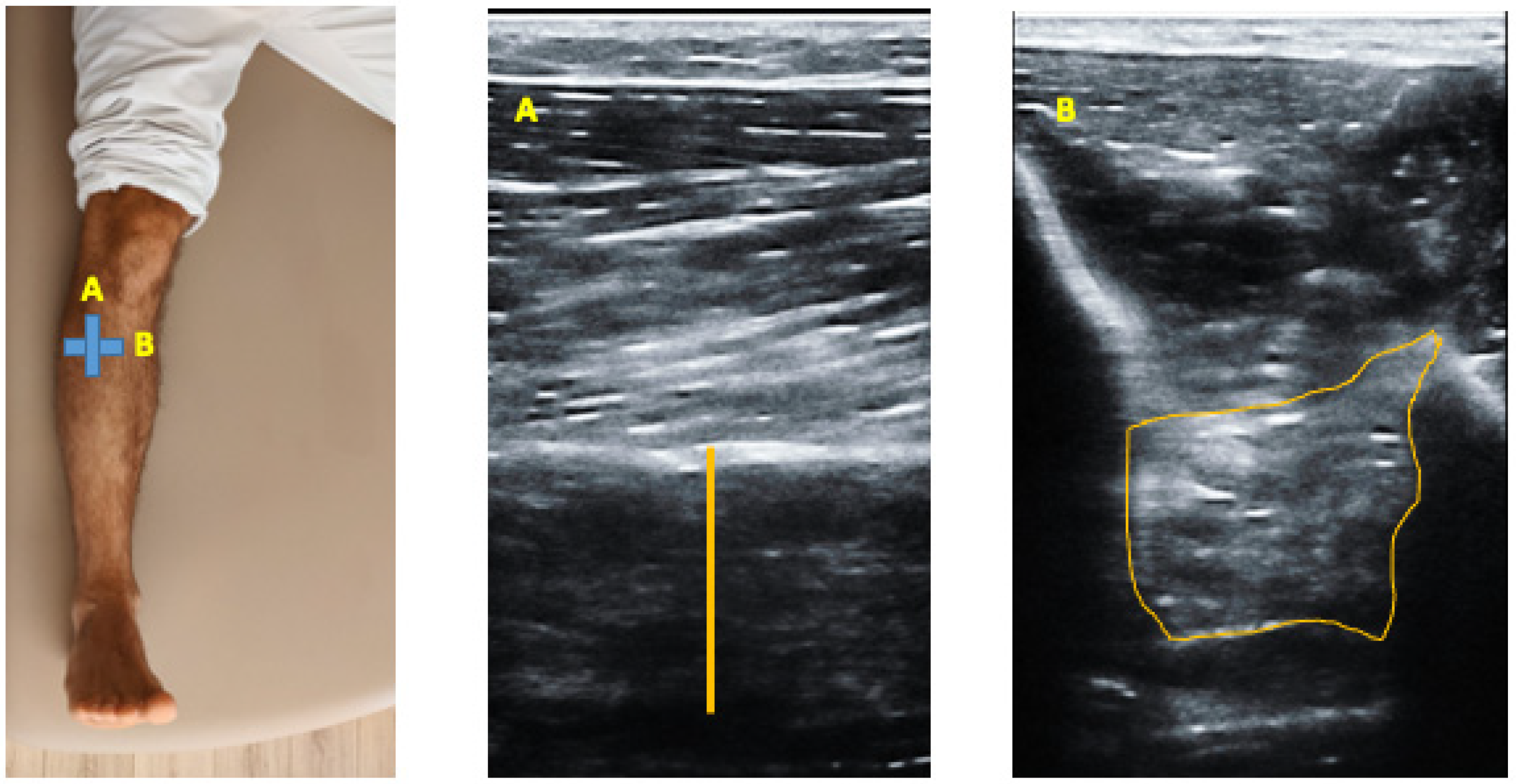
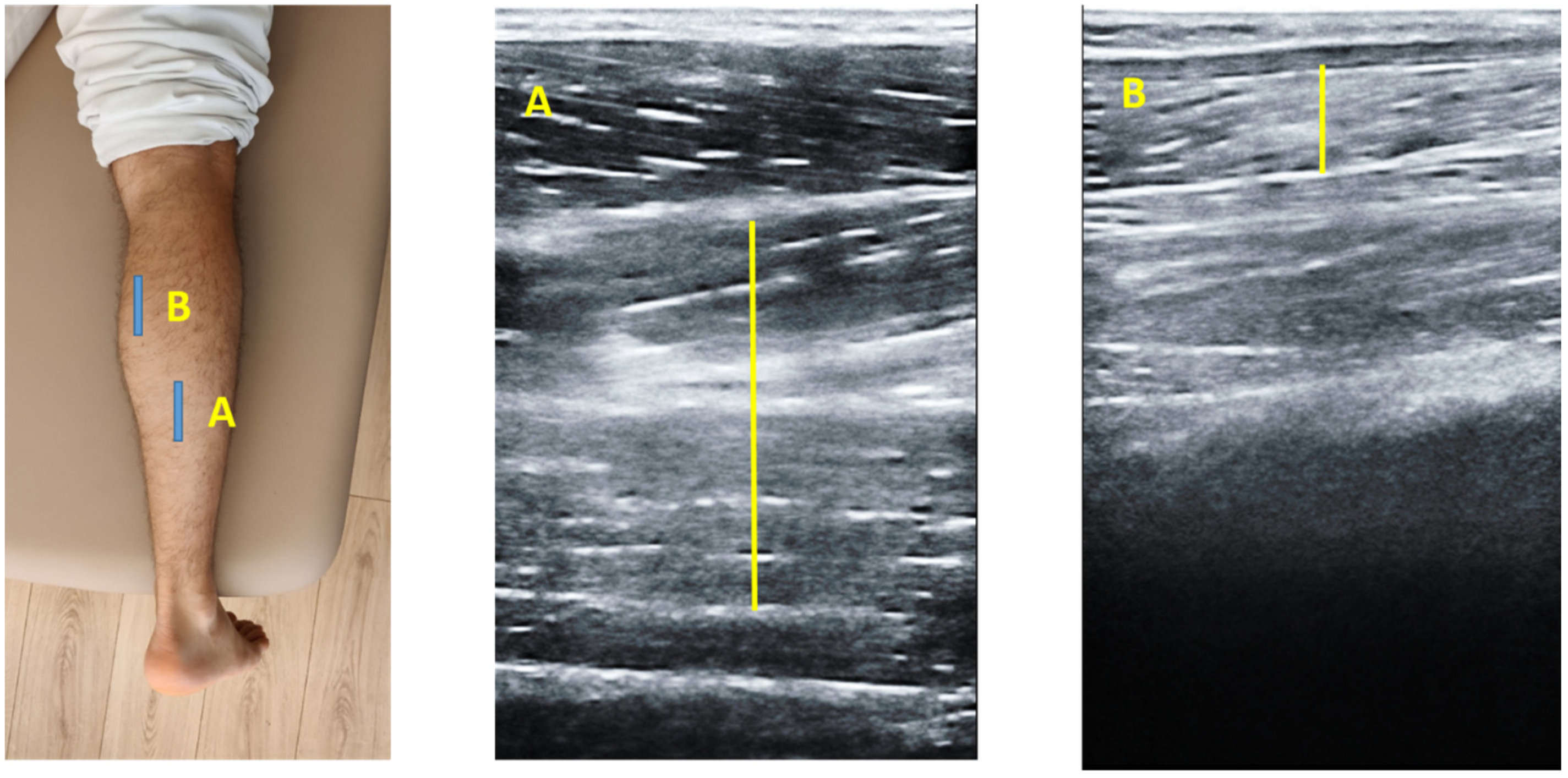
| Data | Total Sample (n = 42) | Plantar Fasciitis (n = 21) | Controls (n = 21) | p-Value Cases vs. Controls |
|---|---|---|---|---|
| Age, y | 39.23 ± 11.10 | 42.54 ± 11.57 | 36.95 ± 11.19 | 0.115 |
| Weight, kg | 77.80 ± 14.87 | 79.90 ± 27.00 | 75.13 ± 9.88 | 0.293 |
| Height, m | 1.72 ± 6.05 | 1.71 ± 6.3 | 1.76 ± 5.30 | 0.001 |
| BMI, kg/m2 | 26.07 ± 4.80 | 27.66 ± 5.27 | 24.27 ± 3.44 | 0.017 |
| Measurement | Plantar Fasciitis (n = 32) | Controls (n = 32) | p-Value |
|---|---|---|---|
| Distance (cm) | |||
| TP CSA | 1.43 ± 0.65 | 1.76 ± 0.56 | 0.375 |
| TP Th | 1.13 ± 0.30 | 1.17 ± 0.15 | 0.076 |
| GM Th | 1.57 ± 0.56 | 1.66 ± 0.34 | 0.679 |
| Soleus Th | 0.68 ± 0.21 | 0.71 ± 0.14 | 0.512 |
| MG EI | 104.14 ± 17.19 | 96.98 ± 14.48 | 0.015 |
| MG EV | 15.59 ± 6.42 | 19.42 ± 6.42 | 0.002 |
| Soleus EI | 85.52 ± 15.09 | 92.66 ± 15.49 | 0.012 |
| Soleus EV | 12.17 ± 4.66 | 12.49 ± 4.25 | 0.690 |
| TP EI | 88.04 ± 14.01 | 84.56 ± 13.74 | 0.173 |
| TP EV | 10.83 ± 3.23 | 12.81 ± 12.15 | 0.225 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Canosa-Carro, L.; López-López, D.; García-Sanz, F.; Díaz-Meco-Conde, R.; García-Bermejo, P.; de-la-Cruz-Torres, B.; Marszalek, J.; Romero-Morales, C. Features of Extrinsic Plantar Muscles in Patients with Plantar Fasciitis by Ultrasound Imaging: A Retrospective Case Control Research. Diagnostics 2022, 12, 897. https://doi.org/10.3390/diagnostics12040897
Canosa-Carro L, López-López D, García-Sanz F, Díaz-Meco-Conde R, García-Bermejo P, de-la-Cruz-Torres B, Marszalek J, Romero-Morales C. Features of Extrinsic Plantar Muscles in Patients with Plantar Fasciitis by Ultrasound Imaging: A Retrospective Case Control Research. Diagnostics. 2022; 12(4):897. https://doi.org/10.3390/diagnostics12040897
Chicago/Turabian StyleCanosa-Carro, Lorena, Daniel López-López, Fernando García-Sanz, Raquel Díaz-Meco-Conde, Paula García-Bermejo, Blanca de-la-Cruz-Torres, Jolanta Marszalek, and Carlos Romero-Morales. 2022. "Features of Extrinsic Plantar Muscles in Patients with Plantar Fasciitis by Ultrasound Imaging: A Retrospective Case Control Research" Diagnostics 12, no. 4: 897. https://doi.org/10.3390/diagnostics12040897
APA StyleCanosa-Carro, L., López-López, D., García-Sanz, F., Díaz-Meco-Conde, R., García-Bermejo, P., de-la-Cruz-Torres, B., Marszalek, J., & Romero-Morales, C. (2022). Features of Extrinsic Plantar Muscles in Patients with Plantar Fasciitis by Ultrasound Imaging: A Retrospective Case Control Research. Diagnostics, 12(4), 897. https://doi.org/10.3390/diagnostics12040897










