Impaired Expression of Membrane Type-2 and Type-3 Matrix Metalloproteinases in Endometriosis but Not in Adenomyosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Ethical Approval
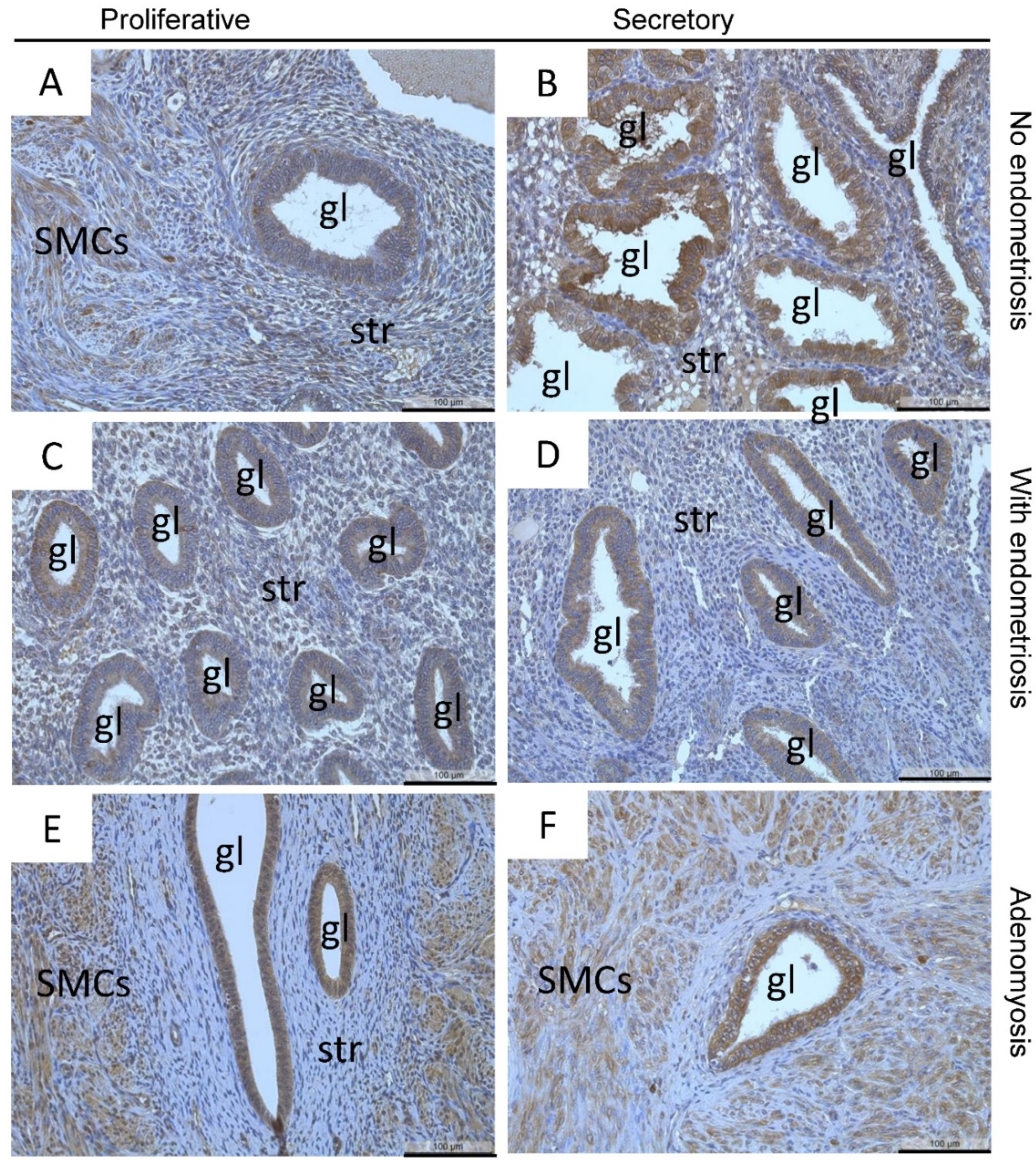
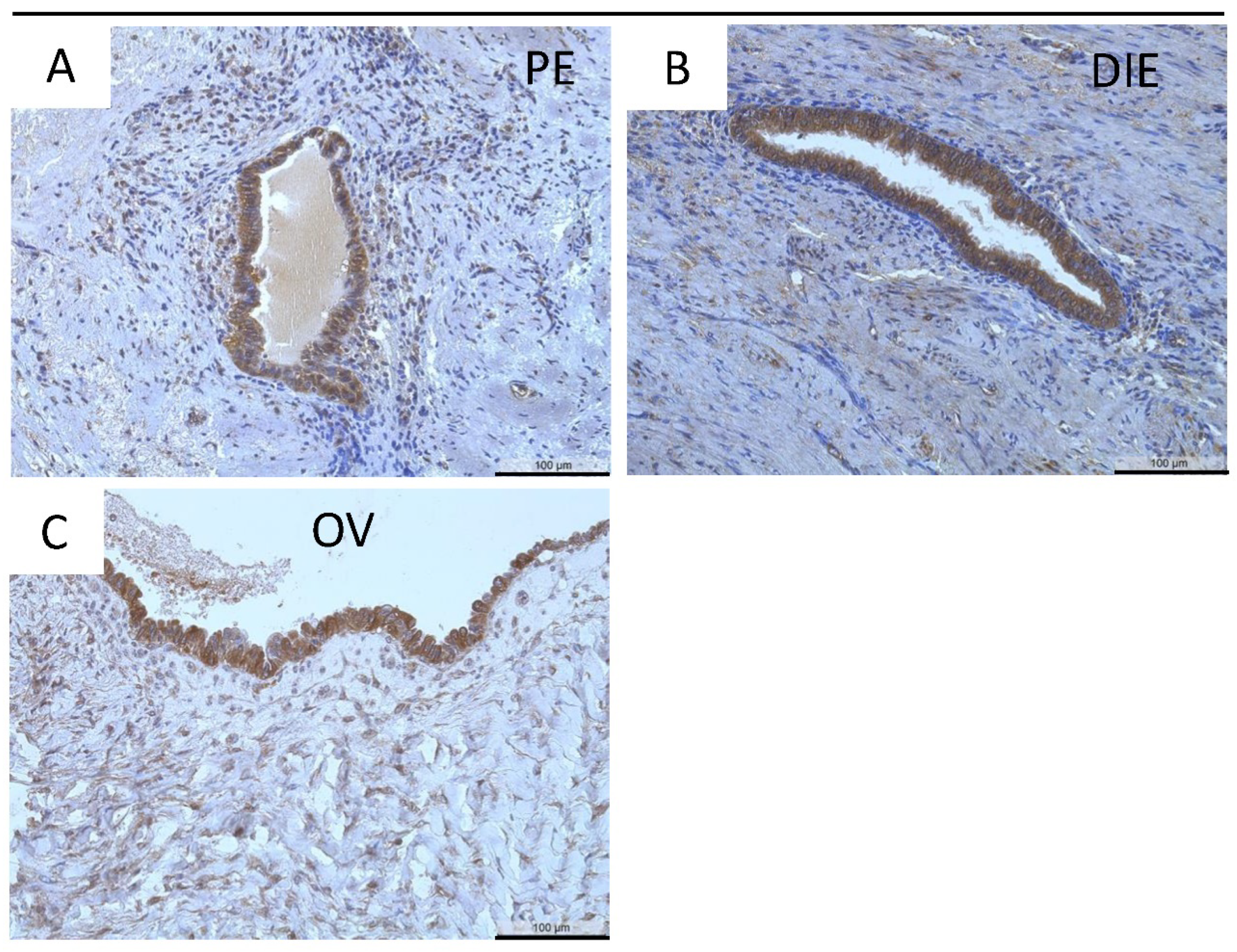
2.2. Immunohistochemical Analysis and Quantification
2.3. Statistics
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Giudice, L.C.; Kao, L.C. Endometriosis. Lancet 2004, 364, 1789–1799. [Google Scholar] [CrossRef]
- Clement, P.B. The pathology of endometriosis: A survey of the many faces of a common disease emphasizing diagnostic pitfalls and unusual and newly appreciated aspects. Adv. Anat. Pathol. 2007, 14, 241–260. [Google Scholar] [CrossRef] [PubMed]
- Colgrave, E.M.; Bittinger, S.; Healey, M.; Dior, U.P.; Rogers, P.A.W.; Keast, J.R.; Girling, J.E.; Holdsworth-Carson, S.J. Superficial peritoneal endometriotic lesions are histologically diverse and rarely demonstrate menstrual cycle synchronicity with matched eutopic endometrium. Hum. Reprod. 2020, 12, 2701–2714. [Google Scholar] [CrossRef] [PubMed]
- Donnez, D.J.; Nisolle, M.; Casanas-Roux, F. Three-dimensional architectures of peritoneal endometriosis. Fertil. Steril. 1992, 57, 980–983. [Google Scholar] [CrossRef]
- Saunders, P.T.K.; Horne, A.W. Endometriosis: Etiology, pathobiology, and therapeutic prospects. Cell 2021, 184, 2807–2824. [Google Scholar] [CrossRef]
- Sampson, J.A. Peritoneal endometriosis due to the menstrual dissemination of endometrial tissue into the peritoneal cavity. Am. J. Obstet. Gynecol. 1927, 14, 422–469. [Google Scholar] [CrossRef]
- Halme, J.; Hammond, M.G.; Hulka, J.F.; Raj, S.G.; Talbert, L.M. Retrograde menstruation in healthy women and in patients with endometriosis. Obstet. Gynecol. 1984, 64, 151–154. [Google Scholar] [PubMed]
- Liu, D.T.; Hitchcock, A. Endometriosis: Its association with retrograde menstruation, dysmenorrhoea and tubal pathology. Br. J. Obstet. Gynaecol. 1986, 93, 859–862. [Google Scholar] [CrossRef]
- Ghiasi, M.; Kulkarni, M.T.; Missmer, S.A. Is endometriosis more common and more severe than it was 30 years ago? J. Minim. Invasive Gynecol. 2020, 27, 452–461. [Google Scholar] [CrossRef]
- Young, V.J.; Brown, J.K.; Saunders, P.T.; Horne, A.W. The role of the peritoneum in the pathogenesis of endometriosis. Hum. Reprod. Update 2013, 19, 558–569. [Google Scholar] [CrossRef]
- Parasar, P.; Ozcan, P.; Terry, K.L. Endometriosis: Epidemiology, diagnosis and clinical management. Curr. Obstet. Gynecol. Rep. 2017, 6, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.W. The pathogenesis of adenomyosis vis-à-vis endometriosis. J. Clin. Med. 2020, 9, 485. [Google Scholar] [CrossRef] [PubMed]
- Siegler, A.M.; Camilien, L. Adenomyosis. J. Reprod. Med. 1994, 39, 841–853. [Google Scholar]
- Puente, J.M.; Fabris, A.; Patel, J.; Patel, A.; Cerrillo, M.; Requena, A.; Garcia-Velasco, J.A. Adenomyosis in infertile women: Prevalence and the role of 3D ultrasound as a marker of severity of the disease. Reprod. Biol. Endocrinol. 2016, 14, 60. [Google Scholar] [CrossRef] [PubMed]
- Morassutto, C.; Monasta, L.; Ricci, G.; Barbone, F.; Ronfani, L. Incidence and estimated prevalence of endometriosis and adenomyosis in northeast Italy: A data linkage study. PLoS ONE 2016, 11, e0154227. [Google Scholar] [CrossRef]
- García-Solares, J.; Donnez, J.; Donnez, O.; Dolmans, M.M. Pathogenesis of uterine adenomyosis: Invagination or metaplasia? Fertil. Steril. 2018, 109, 371–379. [Google Scholar] [CrossRef]
- Ferraz, Z.; Nogueira-Martins, N.; Nogueira-Martins, F. Adenomyosis: Back to the future? Facts Views Vis. Obgyn. 2017, 9, 15–20. [Google Scholar]
- Graziano, A.; Lo Monte, G.; Piva, I.; Caserta, D.; Karner, M.; Engl, B.; Marci, R. Diagnostic findings in adenomyosis: A pictorial review on the major concerns. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 1146–1154. [Google Scholar]
- Maier, V.; Höll, M.; Dietze, R.; Mecha, E.O.; Omwandho, C.O.A.; Tinneberg, H.R.; Meinhold-Heerlein, I.; Konrad, L. Adenomyotic glands are highly related to endometrial glands. Reprod. Biomed. Online 2020, 40, 769–778. [Google Scholar] [CrossRef]
- Chevronnay, H.P.; Selvais, C.; Emonard, H.; Galant, C.; Marbaix, E.; Henriet, P. Regulation of matrix metalloproteinases activity studied in human endometrium as a paradigm of cyclic tissue breakdown and regeneration. Biochim. Biophys. Acta 2012, 1824, 146–156. [Google Scholar] [CrossRef]
- Bałkowiec, M.; Maksym, R.B.; Włodarskim, P.K. The bimodal role of matrix metalloproteinases and their inhibitors in etiology and pathogenesis of endometriosis. Mol. Med. Rep. 2018, 8, 3123–3136. [Google Scholar] [CrossRef]
- Di Nezza, L.A.; Misajon, A.; Zhang, J.; Jobling, T.; Quinn, M.A.; Östör, A.G.; Nie, G.; Lopata, A.; Salamonsen, L.A. Presence of active gelatinases in endometrial carcinoma and correlation of matrix metalloproteinase expression with increasing tumor grade and invasion. Cancer 2002, 94, 1466–1475. [Google Scholar] [CrossRef]
- Burney, R.O.; Giudice, L.C. Pathogenesis and pathophysiology of endometriosis. Fertil. Steril. 2012, 98, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Itoh, Y. Membrane-type matrix metalloproteinases: Their functions and regulations. Matrix Biol. 2015, 44, 207–223. [Google Scholar] [CrossRef]
- Chung, H.W.; Lee, J.Y.; Moon, H.S.; Hur, S.E.; Park, M.H.; Wen, Y.; Polan, M.L. Matrix metalloproteinase-2, membranous type 1 matrix metalloproteinase, and tissue inhibitor of metalloproteinase-2 expression in ectopic and eutopic endometrium. Fertil. Steril. 2002, 78, 787–795. [Google Scholar] [CrossRef]
- Londero, A.P.; Calcagno, A.; Grassi, T.; Marzinotto, S.; Orsaria, M.; Beltrami, C.A.; Marchesoni, D.; Mariuzzi, L. Survivin, MMP-2, MT1-MMP, and TIMP-2: Their impact on survival, implantation, and proliferation of endometriotic tissues. Virchows Arch. 2012, 461, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Plaisier, M.; Koolwijk, P.; Hanemaaijer, R.; Verwey, R.A.; van der Weiden, R.M.; Risse, E.K.; Jungerius, C.; Helmerhorst, F.M.; van Hinsbergh, V.W. Membrane-type matrix metalloproteinases and vascularization in human endometrium during the menstrual cycle. Mol. Hum. Reprod. 2006, 12, 11–18. [Google Scholar] [CrossRef]
- Goffin, F.; Munaut, C.; Frankenne, F.; Perrier d’Hauterive, S.; Béliard, A.; Fridman, V.; Nervo, P.; Colige, A.; Foidart, J.M. Expression pattern of metalloproteinases and tissue inhibitors of matrix-metalloproteinases in cycling human endometrium. Biol. Reprod. 2003, 69, 976–984. [Google Scholar] [CrossRef]
- Zhang, J.; Hampton, A.L.; Nie, G.; Salamonsen, L.A. Progesterone inhibits activation of latent matrix metalloproteinase (MMP)-2 by membrane-type 1 MMP: Enzymes coordinately expressed in human endometrium. Biol. Reprod. 2000, 62, 85–94. [Google Scholar] [CrossRef]
- Gaetje, R.; Holtrich, U.; Engels, K.; Kourtis, K.; Cikrit, E.; Kissler, S.; Rody, A.; Karn, T.; Kaufmann, M. Expression of membrane-type 5 matrix metalloproteinase in human endometrium and endometriosis. Gynecol. Endocrinol. 2007, 23, 567–573. [Google Scholar] [CrossRef]
- Haas, D.; Chvatal, R.; Habelsberger, A.; Wurm, P.; Schimetta, W.; Oppelt, P. Comparison of revised American Fertility Society and ENZIAN staging: A critical evaluation of classifications of endometriosis on the basis of our patient population. Fertil. Steril. 2011, 95, 1574–1578. [Google Scholar] [CrossRef]
- Gordts, S.; Koninckx, P.; Brosens, I. Pathogenesis of deep endometriosis. Fertil. Steril. 2017, 108, 872–885. [Google Scholar] [CrossRef] [PubMed]
- Konrad, L.; Kortum, J.; Nabham, R.; Gronbach, J.; Dietze, R.; Oehmke, F.; Berkes, E.; Tinneberg, H.R. Composition of the stroma in the human endometrium and endometriosis. Reprod. Sci. 2018, 25, 1106–1115. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, N.; Ung, L.; Otani, T.; Wilkinson, D.; Lopata, A. Uterine cavity matrix metalloproteinases and cytokines in patients with leiomyoma, adenomyosis or endometrial polyp. Eur. J. Obstet. Gynecol. Reprod. Biol. 2003, 111, 197–203. [Google Scholar] [CrossRef]
- Tokyol, C.; Aktepe, F.; Dilek, F.H.; Sahin, O.; Arioz, D.T. Expression of cyclooxygenase-2 and matrix metalloproteinase-2 in adenomyosis and endometrial polyps and its correlation with angiogenesis. Int. J. Gynecol. Pathol. 2009, 28, 148–156. [Google Scholar] [CrossRef]
- Wenzl, R.J.; Heinzl, H. Localization of matrix metalloproteinase-2 in uterine endometrium and ectopic implants. Gynecol. Obstet. Investig. 1998, 45, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Ito, E.; Yana, I.; Fujita, C.; Irifune, A.; Takeda, M.; Madachi, A.; Mori, S.; Hamada, Y.; Matsuura, N. The role of MT2-MMP in cancer progression. Biochem. Biophys. Res. Commun. 2010, 393, 222–227. [Google Scholar] [CrossRef]
- Chen, L.; Zhou, Q.; Xu, B.; Liu, J.; Shi, L.; Zhu, D.; Wu, C.; Jiang, J. MT2-MMP expression associates with tumor progression and angiogenesis in human lung cancer. Int. J. Clin. Exp. Pathol. 2014, 7, 3469–3477. [Google Scholar]
- Chen, Y.; Lu, H.; Tao, D.; Fan, M.; Zhuang, Q.; Xing, Z.; Chen, Z.; He, X. Membrane type-2 matrix metalloproteinases improve the progression of renal cell cancer. Int. J. Clin. Exp. Pathol. 2017, 10, 10618–10626. [Google Scholar]
- Xu, X.; Chen, L.; Xu, B.; Xie, Q.; Sun, M.; Deng, X.; Wu, C.; Jiang, J. Increased MT2-MMP expression in gastric cancer patients is associated with poor prognosis. Int. J. Clin. Exp. Pathol. 2015, 8, 1985–1990. [Google Scholar]
- Liu, Y.; Sun, X.; Feng, J.; Deng, L.L.; Liu, Y.; Li, B.; Zhu, M.; Lu, C.; Zhou, L. MT2-MMP induces proteolysis and leads to EMT in carcinomas. Oncotarget 2016, 7, 48193–48205. [Google Scholar] [CrossRef] [PubMed]
- Lafleur, M.A.; Handsley, M.M.; Knäuper, V.; Murphy, G.; Edwards, D.R. Endothelial tubulogenesis within fibrin gels specifically requires the activity of membrane-type-matrix metalloproteinases (MT-MMPs). J. Cell Sci. 2002, 115, 3427–3438. [Google Scholar] [CrossRef]
- Bjørn, S.F.; Hastrup, N.; Larsen, J.F.; Lund, L.R.; Pyke, C. Messenger RNA for membrane-type 2 matrix metalloproteinase, MT2-MMP, is expressed in human placenta of first trimester. Placenta 2000, 21, 170–176. [Google Scholar] [CrossRef]
- Bar-Or, A.; Nuttall, R.K.; Duddy, M.; Alter, A.; Kim, H.J.; Ifergan, I.; Pennington, C.J.; Bourgoin, P.; Edwards, D.R.; Yong, V.W. Analyses of all matrix metalloproteinase members in leukocytes emphasize monocytes as major inflammatory mediators in multiple sclerosis. Brain 2003, 126, 2738–2749. [Google Scholar] [CrossRef]
- Roth, L.; Kalev-Altman, R.; Monsonego-Ornan, E.; Sela-Donenfeld, D. A new role of the membrane-type matrix metalloproteinase 16 (MMP16/MT3-MMP) in neural crest cell migration. Int. J. Dev. Biol. 2017, 61, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Tatti, O.; Gucciardo, E.; Pekkonen, P.; Holopainen, T.; Louhimo, R.; Repo, P.; Maliniemi, P.; Lohi, J.; Rantanen, V.; Hautaniemi, S.; et al. MMP16 mediates a proteolytic switch to promote cell–cell adhesion, collagen alignment, and lymphatic invasion in melanoma. Cancer Res. 2015, 75, 2083–2094. [Google Scholar] [CrossRef] [PubMed]
- Nakada, M.; Nakamura, H.; Ikeda, E.; Fujimoto, N.; Yamashita, J.; Sato, H.; Seiki, M.; Okada, Y. Expression and tissue localization of membrane-type 1, 2, and 3 matrix metalloproteinases in human astrocytic tumors. Am. J. Pathol. 1999, 154, 417–428. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Yoshihara, K.; Suda, K.; Nakaoka, H.; Yachida, N.; Ueda, H.; Sugino, K.; Mori, Y.; Yamawaki, K.; Tamura, R.; et al. Three-dimensional understanding of the morphological complexity of the human uterine endometrium. iScience 2021, 24, 102258. [Google Scholar] [CrossRef]
- Konrad, L.; Dietze, R.; Riaz, M.A.; Scheiner-Bobis, G.; Behnke, J.; Horné, F.; Hoerscher, A.; Reising, C.; Meinhold-Heerlein, I. Epithelial-mesenchymal transition in endometriosis—When does it happen? J. Clin. Med. 2020, 9, 1915. [Google Scholar] [CrossRef]



| Tissues | EM | Ov | PE | DIE |
|---|---|---|---|---|
| All samples | n = 56 | n = 23 | n = 26 (27) | n= 28 (29) |
| Median age ± SD | 36 ± 5.4 | 36 ± 5.4 | 33 ± 4.6 | 32 ± 5.0 |
| Leiomyoma | n = 36 | |||
| Adenomyosis | n = 41 | |||
| Adenomyosis only | n = 17 | |||
| Proliferative | n = 25 | |||
| Secretory | n = 31 | |||
| Bladder | 3 | 2 | ||
| Uterosacral lig | 3 | 3 | ||
| Ovarian fossa | 3 | |||
| Pouch of douglas | 2 | |||
| Round lig of uterus | 2 | |||
| Pelvic wall | 2 | |||
| Rectum | 2 | 11 | ||
| Rectosigmoid | 3 | |||
| Rectovaginal septum | 3 | 5 | ||
| Paraurethral/rectal | 1 | 1 | ||
| Sigmoid colon | 2 | 2 | ||
| Vagina | 1 | |||
| Intestine | 1 | |||
| Scar/Symphysis | 2 | |||
| Lig latum uteri | 1 | |||
| Mesovarium | 1 |
| Tissues | EM | Ov | PE | DIE |
|---|---|---|---|---|
| All samples | n = 47 | n = 18 | n = 17 (18) | n= 15 (17) |
| Median age ± SD | 43 ± 7.9 | 35.3 ± 4.0 | 31 ± 3.7 | 33 ± 4.6 |
| Leiomyoma | n = 30 | |||
| Adenomyosis | n = 31 | |||
| Adenomyosis only | n = 13 | |||
| Proliferative | n = 21 | |||
| Secretory | n = 26 | |||
| Bladder | 1 | 1 | ||
| Uterosacral lig | 3 | |||
| Ovarian fossa | 2 | |||
| Pouch of douglas | 3 | |||
| Round lig of uterus | 2 | |||
| Pelvic wall | 2 | |||
| Rectum | 4 | |||
| Rectosigmoid | 1 | |||
| Rectovaginal septum | 1 | 5 | ||
| Paraurethral/rectal | 2 | 1 | ||
| Sigmoid colon | 1 | 2 | ||
| Scar/Symphysis | 2 | |||
| Lig latum uteri | 1 | |||
| Mesovarium | 1 |
| Ctrl (Healthy) | Em-En | Em-Ad | All | Ad | |||||
|---|---|---|---|---|---|---|---|---|---|
| MT2-MMP | P | S | P | S | P | S | All | P | S |
| Mean | 67 | 59 | 61 | 56 | 67 | 70 | 64 | 90 | 83 |
| SEM | 4.6 | 8.1 | 8.2 | 9.3 | 11.7 | 6.8 | 3.5 | 6.8 | 5.2 |
| p-Value | n.s. | n.s. | n.s. | n.s | n.s. | n.s. | 0.0001 | n.s. | n.s. |
| N | 6 | 8 | 8 | 10 | 10 | 14 | 56 | 19 | 22 |
| Age | 35 ± 13 | 41 ± 5.3 | 33 ± 6.7 | 41 ± 6.3 | 43.5 ± 5.5 | 45 ± 5.9 | 42 ± 7.6 | 44 ± 5.4 | 45 ± 5.8 |
| MT3-MMP | P | S | P | S | P | S | All | P | S |
| Mean | 191 | 136 | 168 | 128 | 182 | 150 | 157 | 161 | 146 |
| SEM | 21.6 | 19.5 | 21.8 | 9.5 | 14 | 14.6 | 7.0 | 13.9 | 16.3 |
| p-Value | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | 0.0001 | n.s. | n.s. |
| N | 6 | 6 | 6 | 11 | 9 | 9 | 47 | 15 | 16 |
| Age | 41 ± 13 | 43 ± 6.0 | 32.5 ± 5.8 | 42 ± 6.0 | 45 ± 5.0 | 45 ± 6.8 | 43 ± 8.0 | 45 ± 5.1 | 43.5 ± 6.7 |
| Ctrl (Healthy) | Em-En | Em-Ad | All | Ad | |||||
|---|---|---|---|---|---|---|---|---|---|
| MT2-MMP | P | S | P | S | P | S | All | P | S |
| Mean | 57 | 65 | 62 | 59 | 70 | 69 | 65 | 84 | 81 |
| SEM | 7.4 | 7.9 | 7.2 | 9.1 | 9.1 | 5.7 | 3.2 | 5.0 | 4.6 |
| p-Value | n.s. | n.s. | n.s | n.s | n.s. | n.s. | 0.0001 | n.s | n.s |
| N | 6 | 8 | 8 | 10 | 10 | 14 | 56 | 19 | 22 |
| Age | 35 ± 13 | 41 ± 5.3 | 33 ± 6.7 | 41 ± 6.3 | 43.5 ± 5.5 | 45 ± 5.9 | 42 ± 7.6 | 44 ± 5.4 | 45 ± 5.8 |
| MT3-MMP | P | S | P | S | P | S | All | P | S |
| Mean | 99 | 89 | 98 | 99 | 100 | 98 | 98 | 95 | 95 |
| SEM | 0.7 | 7.4 | 1.9 | 0.4 | 0.0 | 1.2 | 1.0 | 3.6 | 3.2 |
| p-Value | n.s. | n.s | n.s. | n.s. | n.s. | n.s. | 0.0001 | n.s | n.s. |
| N | 6 | 6 | 6 | 11 | 9 | 9 | 47 | 15 | 16 |
| Age | 41 ± 13 | 43 ± 6.0 | 32.5 ± 5.8 | 42 ± 6.0 | 45 ± 5.0 | 45 ± 6.8 | 43 ± 8.0 | 45 ± 5.1 | 43.5 ± 6.7 |
| EM (a) | Ov (b) | DIE (c) | PE (d) | |
|---|---|---|---|---|
| MT2-MMP | ||||
| Mean | 64 | 33 | 45 | 38 |
| SEM | 3.5 | 5.9 | 5.8 | 5.9 |
| P | n.s. | a, b < 0.001 | a, c ≤ 0.05 | a, d < 0.01 |
| N | 56 | 23 | 29 | 27 |
| Age | 42 ± 7.6 | 36 ± 5.4 | 32 ± 5.0 | 33 ± 4.6 |
| MT3-MMP | ||||
| Mean | 157 | 120 | 101 | 96 |
| SEM | 7.0 | 5.5 | 10.1 | 6.4 |
| P | n.s. | n.s. | a, c < 0.001 | a, d < 0.001 |
| N | 47 | 18 | 17 | 18 |
| age | 43 ± 8.0 | 35.5 ± 4.4 | 32 ± 5.2 | 31 ± 3.7 |
| EM (a) | Ov (b) | DIE (c) | PE (d) | |
|---|---|---|---|---|
| MT2-MMP | ||||
| Mean | 65 | 33 | 42 | 36 |
| SEM | 3.2 | 5.8 | 5.1 | 5.7 |
| P | n.s. | a, b < 0.001 | a, c < 0.01 | a, d ˂ 0.001 |
| N | 56 | 23 | 29 | 27 |
| Age | 42 ± 7.6 | 36 ± 5.4 | 32 ± 5.0 | 33 ± 4.6 |
| MT3-MMP | ||||
| Mean | 98 | 96 | 83 | 85 |
| SEM | 1.0 | 2.6 | 4.6 | 4.2 |
| P | n.s. n.s. | n.s. n.s. | a, c ≤ 0.05 b, c ≤ 0.05 | n.s. n.s. |
| N | 47 | 18 | 17 | 18 |
| age | 43 ± 8.0 | 35.5 ± 4.4 | 32 ± 5.2 | 31 ± 3.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maoga, J.B.; Riaz, M.A.; Mwaura, A.N.; Scheiner-Bobis, G.; Mecha, E.; Omwandho, C.O.A.; Meinhold-Heerlein, I.; Konrad, L. Impaired Expression of Membrane Type-2 and Type-3 Matrix Metalloproteinases in Endometriosis but Not in Adenomyosis. Diagnostics 2022, 12, 779. https://doi.org/10.3390/diagnostics12040779
Maoga JB, Riaz MA, Mwaura AN, Scheiner-Bobis G, Mecha E, Omwandho COA, Meinhold-Heerlein I, Konrad L. Impaired Expression of Membrane Type-2 and Type-3 Matrix Metalloproteinases in Endometriosis but Not in Adenomyosis. Diagnostics. 2022; 12(4):779. https://doi.org/10.3390/diagnostics12040779
Chicago/Turabian StyleMaoga, Jane B., Muhammad A. Riaz, Agnes N. Mwaura, Georgios Scheiner-Bobis, Ezekiel Mecha, Charles O. A. Omwandho, Ivo Meinhold-Heerlein, and Lutz Konrad. 2022. "Impaired Expression of Membrane Type-2 and Type-3 Matrix Metalloproteinases in Endometriosis but Not in Adenomyosis" Diagnostics 12, no. 4: 779. https://doi.org/10.3390/diagnostics12040779
APA StyleMaoga, J. B., Riaz, M. A., Mwaura, A. N., Scheiner-Bobis, G., Mecha, E., Omwandho, C. O. A., Meinhold-Heerlein, I., & Konrad, L. (2022). Impaired Expression of Membrane Type-2 and Type-3 Matrix Metalloproteinases in Endometriosis but Not in Adenomyosis. Diagnostics, 12(4), 779. https://doi.org/10.3390/diagnostics12040779








