Development of an Automated Chemiluminescent Enzyme Immunoassay for Measuring Thrombopoietin in Human Plasma
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Construction of Rabbit Immune Antibody Library
2.3. Anti-TPO Antibody Screening Using a Phage Display
2.4. Conversion of Anti-TPO Rabbit Monoclonal IgG from Phage Antibodies
2.5. Preparation of Alkaline Phosphatase (ALP)-Labeled Anti-TPO Antibody
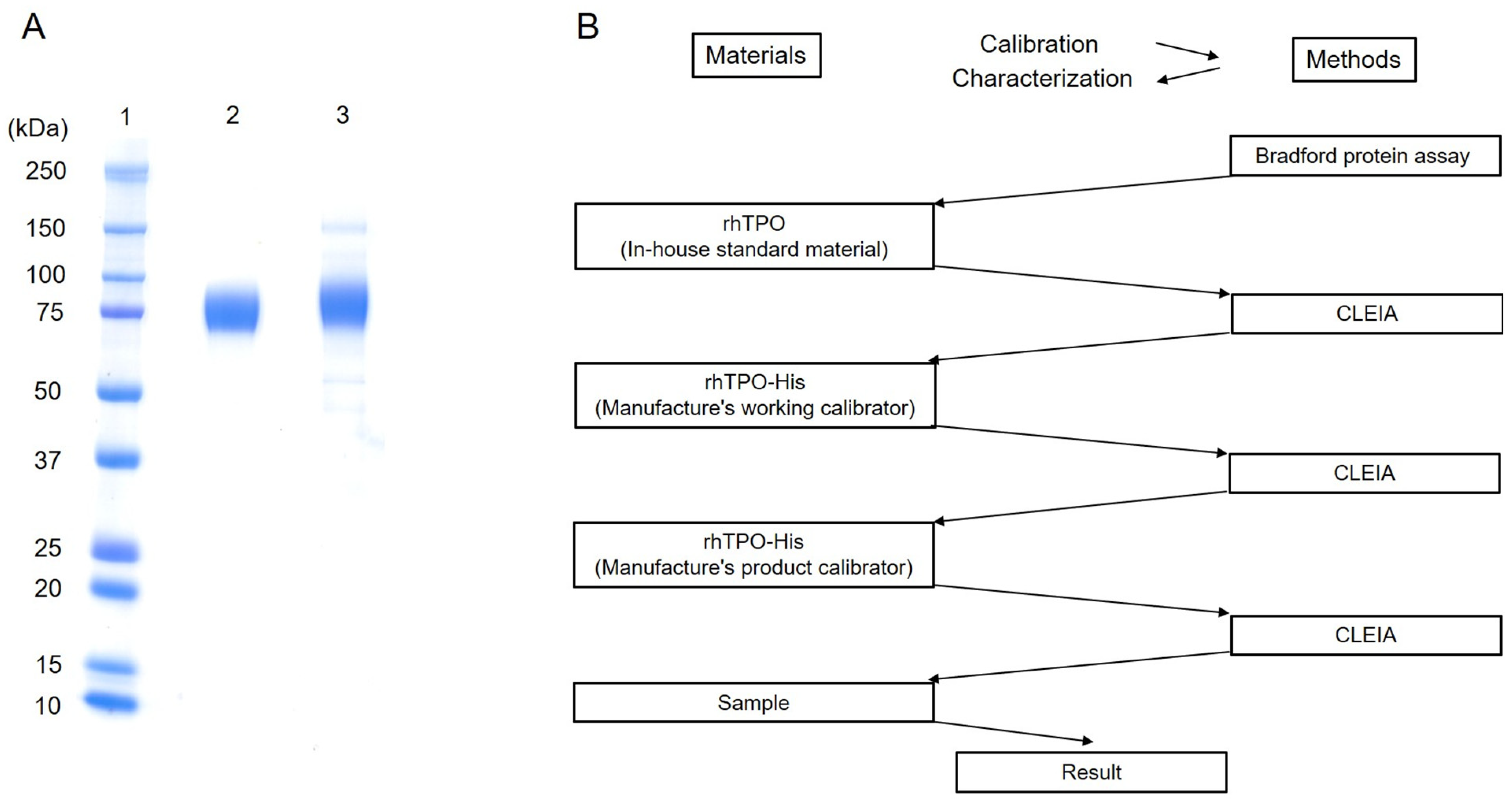
2.6. Recombinant Human TPO with a C-Terminal 6× His Tag (rhTPO-His) Antigen
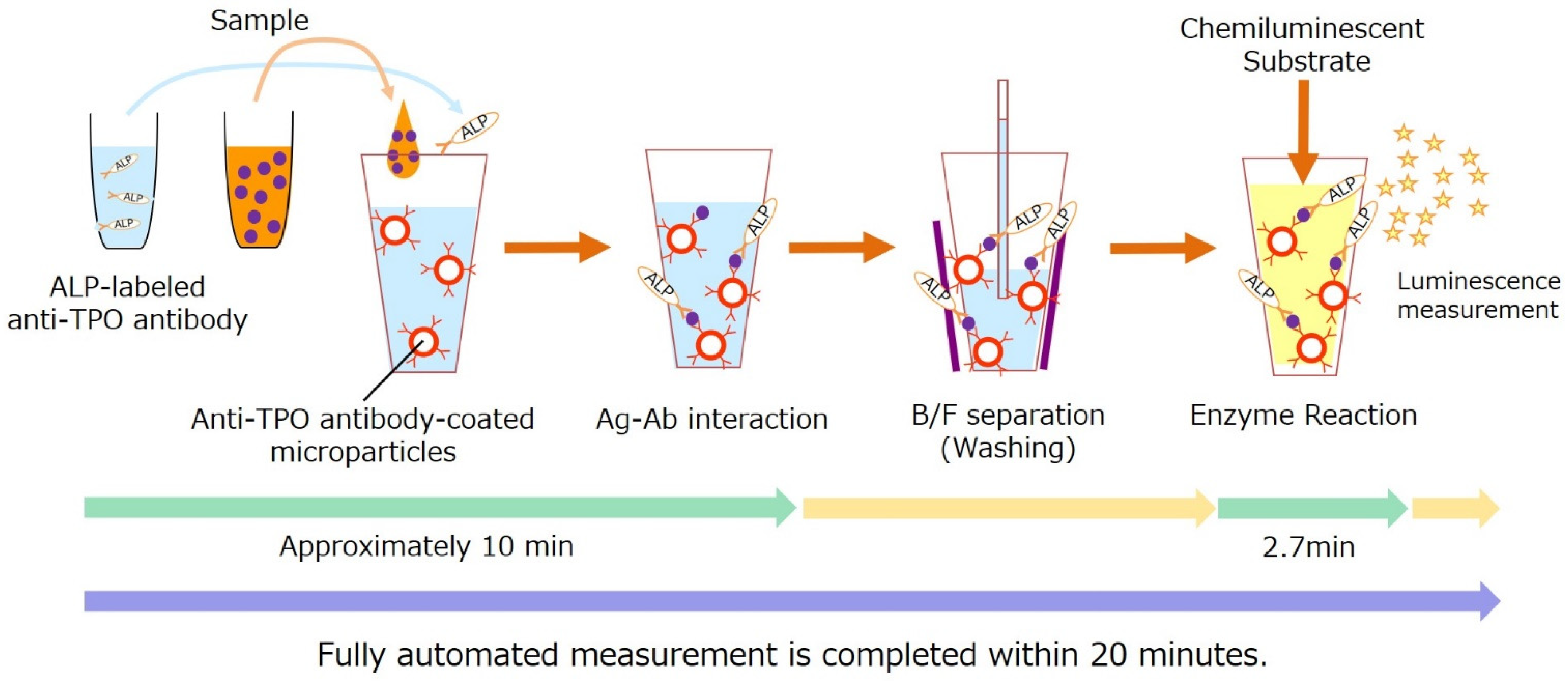
2.7. Measurement of Plasma TPO Concentrations
2.8. Characterization of In-House Standard Material and Concentration Assignment
2.9. Evaluation Methods for TPO-CLEIA
2.10. Statistical Analysis
3. Results
3.1. Characterization of Recombinant Human TPO Antigen
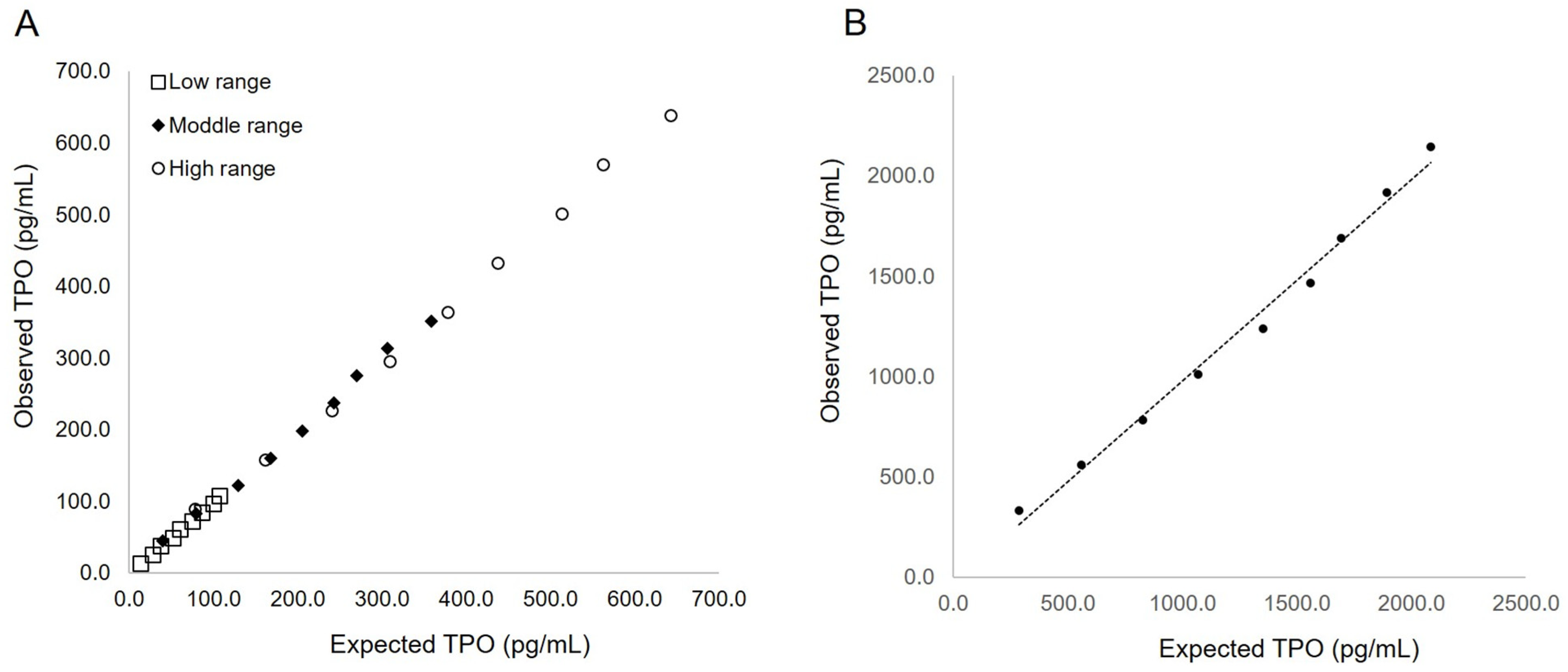
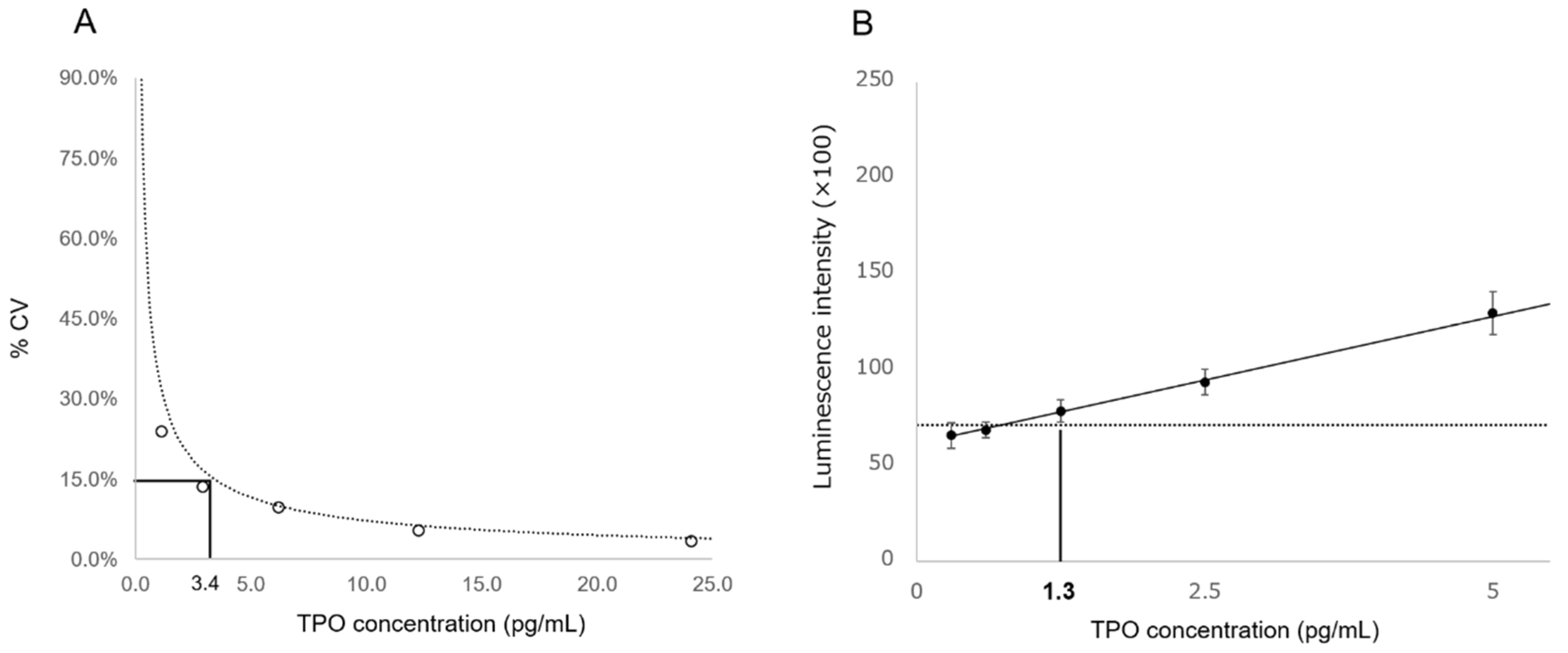
3.2. Analytical Performance of TPO-CLEIA
3.3. Clinical Validation in the Study Participants
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wendling, F.; Maraskovsky, E.; Debili, N.; Florindo, C.; Teepe, M.; Titeux, M.; Methia, N.; Breton-Gorius, J.; Cosman, D.; Vainchenker, W. cMpl Ligand Is a Humoral Regulator of Megakaryocytopoiesis. Nature 1994, 369, 571–574. [Google Scholar]
- Nagata, Y.; Shozaki, Y.; Nagahisa, H.; Nagasawa, T.; Abe, T.; Todokoro, K. Serum Thrombopoietin Level Is Not Regulated by Transcription but by the Total Counts of Both Megakaryocytes and Platelets During Thrombocytopenia and Thrombocytosis. Thromb. Haemost. 1997, 77, 808–814. [Google Scholar] [CrossRef]
- de Sauvage, F.J.; Carver-Moore, K.; Luoh, S.M.; Ryan, A.; Dowd, M.; Eaton, D.L.; Moore, M.W. Physiological Regulation of Early and Late Stages of Megakaryocytopoiesis by Thrombopoietin. J. Exp. Med. 1996, 183, 651–656. [Google Scholar] [CrossRef]
- Yamazaki, R.; Kuwana, M.; Mori, T.; Okazaki, Y.; Kawakami, Y.; Ikeda, Y.; Okamoto, S. Prolonged Thrombocytopenia After Allogeneic Hematopoietic Stem Cell Transplantation: Associations With Impaired Platelet Production and Increased Platelet Turnover. Bone Marrow Transplant. 2006, 38, 377–384. [Google Scholar] [CrossRef]
- Kuter, D.J.; Rosenberg, R.D. The Reciprocal Relationship of Thrombopoietin (c-Mpl Ligand) to Changes in the Platelet Mass During Busulfan-Induced Thrombocytopenia in the Rabbit. Blood 1995, 85, 2720–2730. [Google Scholar] [CrossRef] [PubMed]
- Kato, T.; Matsumoto, A.; Ogami, K.; Tahara, T.; Morita, H.; Miyazaki, H. Native Thrombopoietin: Structure and Function. Stem Cells 1998, 16, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Kaushansky, K. The Molecular Mechanisms That Control Thrombopoiesis. J. Clin. Investig. 2005, 115, 3339–3347. [Google Scholar] [CrossRef]
- Cines, D.B.; Blanchette, V.S. Immune Thrombocytopenic Purpura. N. Engl. J. Med. 2002, 346, 995–1008. [Google Scholar] [CrossRef] [PubMed]
- McMillan, R. The Pathogenesis of Chronic Immune Thrombocytopenic Purpura. Semin. Hematol. 2007, 44, S3–S11. [Google Scholar] [CrossRef]
- Kashiwagi, H.; Tomiyama, Y. Pathophysiology and Management of Primary Immune Thrombocytopenia. Int. J. Hematol. 2013, 98, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Marini, I.; Zlamal, J.; Pelzel, L.; Bethge, W.; Faul, C.; Holzer, U.; Althaus, K.; Bakchoul, T. Autoantibody-Mediated Desialylation Impairs Human Thrombopoiesis and Platelet Lifespan. Haematologica 2021, 106, 196–207. [Google Scholar] [CrossRef]
- Rodeghiero, F.; Stasi, R.; Gernsheimer, T.; Michel, M.; Provan, D.; Arnold, D.M.; Bussel, J.B.; Cines, D.B.; Chong, B.H.; Cooper, N.; et al. Standardization of Terminology, Definitions and Outcome Criteria in Immune Thrombocytopenic Purpura of Adults and Children: Report From an International Working Group. Blood 2009, 113, 2386–2393. [Google Scholar] [CrossRef]
- Kosugi, S.; Kurata, Y.; Tomiyama, Y.; Tahara, T.; Kato, T.; Tadokoro, S.; Shiraga, M.; Honda, S.; Kanakura, Y.; Matsuzawa, Y. Circulating Thrombopoietin Level in Chronic Immune Thrombocytopenic Purpura. Br. J. Haematol. 1996, 93, 704–706. [Google Scholar] [CrossRef] [PubMed]
- Porcelijn, L.; Folman, C.C.; Bossers, B.; Huiskes, E.; Overbeeke, M.A.; van v d Schoot, C.E.; de Haas, M.; von dem Borne, A.E. The Diagnostic Value of Thrombopoietin Level Measurements in Thrombocytopenia. Thromb. Haemost. 1998, 79, 1101–1105. [Google Scholar] [CrossRef] [PubMed]
- Aledort, L.M.; Hayward, C.P.M.; Chen, M.G.; Nichol, J.L.; Bussel, J.; ITP Study Group. Prospective Screening of 205 Patients With ITP, Including Diagnosis, Serological Markers, and the Relationship Between Platelet Counts, Endogenous Thrombopoietin, and Circulating Antithrombopoietin Antibodies. Am. J. Hematol. 2004, 76, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Kuwana, M.; Okazaki, Y.; Satoh, T.; Asahi, A.; Kajihara, M.; Ikeda, Y. Initial Laboratory Findings Useful for Predicting the Diagnosis of Idiopathic Thrombocytopenic Purpura. Am. J. Med. 2005, 118, 1026–1033. [Google Scholar] [CrossRef]
- Kuwana, M.; Kurata, Y.; Fujimura, K.; Fujisawa, K.; Wada, H.; Nagasawa, T.; Nomura, S.; Kojima, T.; Yagi, H.; Ikeda, Y. Preliminary Laboratory Based Diagnostic Criteria for Immune Thrombocytopenic Purpura: Evaluation by Multi-Center Prospective Study. J. Thromb. Haemost. 2006, 4, 1936–1943. [Google Scholar] [CrossRef] [PubMed]
- Sakuragi, M.; Hayashi, S.; Maruyama, M.; Kabutomori, O.; Kiyokawa, T.; Nagamine, K.; Kato, H.; Kashiwagi, H.; Kanakura, Y.; Tomiyama, Y. Clinical Significance of IPF% or RP% Measurement in Distinguishing Primary Immune Thrombocytopenia From Aplastic Thrombocytopenic Disorders. Int. J. Hematol. 2015, 101, 369–375. [Google Scholar] [CrossRef]
- Tahara, T.; Usuki, K.; Sato, H.; Ohashi, H.; Morita, H.; Tsumura, H.; Matsumoto, A.; Miyazaki, H.; Urabe, A.; Kato, T. A Sensitive Sandwich ELISA for Measuring Thrombopoietin in Human Serum: Serum Thrombopoietin Levels in Healthy Volunteers and in Patients With Haemopoietic Disorders. Br. J. Haematol. 1996, 93, 783–788. [Google Scholar] [CrossRef]
- Tahara, T.; Kuwaki, T.; Matsumoto, A.; Morita, H.; Watarai, H.; Inagaki, Y.; Ohashi, H.; Ogami, K.; Miyazaki, H.; Kato, T. Neutralization of Biological Activity and Inhibition of Receptor Binding by Antibodies Against Human Thrombopoietin. Stem Cells 1998, 16, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Feese, M.D.; Tamada, T.; Kato, Y.; Maeda, Y.; Hirose, M.; Matsukura, Y.; Shigematsu, H.; Muto, T.; Matsumoto, A.; Watarai, H.; et al. Structure of the Receptor-Binding Domain of Human Thrombopoietin Determined by Complexation With a Neutralizing Antibody Fragment. Proc. Natl. Acad. Sci. USA 2004, 101, 1816–1821. [Google Scholar] [CrossRef]
- Saito, C.; Ishiyama, K.; Yamazaki, H.; Zaimoku, Y.; Nakao, S. Hypomegakaryocytic Thrombocytopenia (HMT): An Immune-Mediated Bone Marrow Failure Characterized by an Increased Number of PNH-Phenotype Cells and High Plasma Thrombopoietin Levels. Br. J. Haematol. 2016, 175, 246–251. [Google Scholar] [CrossRef]
- Kurata, Y.; Hayashi, S.; Kiyoi, T.; Kosugi, S.; Kashiwagi, H.; Honda, S.; Tomiyama, Y. Diagnostic Value of Tests for Reticulated Platelets, Plasma Glycocalicin, and Thrombopoietin Levels for Discriminating Between Hyperdestructive and Hypoplastic Thrombocytopenia. Am. J. Clin. Pathol. 2001, 115, 656–664. [Google Scholar] [CrossRef]
- Satoh, T.; Miyazaki, K.; Shimohira, A.; Amano, N.; Okazaki, Y.; Nishimoto, T.; Akahoshi, T.; Munekata, S.; Kanoh, Y.; Ikeda, Y.; et al. Fcγ Receptor IIB Gene Polymorphism in Adult Japanese Patients With Primary Immune Thrombocytopenia. Blood 2013, 122, 1991–1992. [Google Scholar] [CrossRef]
- Neunert, C.; Terrell, D.R.; Arnold, D.M.; Buchanan, G.; Cines, D.B.; Cooper, N.; Cuker, A.; Despotovic, J.M.; George, J.N.; Grace, R.F.; et al. American Society of Hematology 2019 Guidelines for Immune Thrombocytopenia. Blood Adv. 2019, 3, 3829–3866. [Google Scholar] [CrossRef] [PubMed]
- Friedberg, J.W. Relapsed/Refractory Diffuse Large B-Cell Lymphoma. Hematol. Am. Soc. Hematol. Educ. Program 2011, 2011, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Morino, K.; Katsumi, H.; Akahori, Y.; Iba, Y.; Shinohara, M.; Ukai, Y.; Kohara, Y.; Kurosawa, Y. Antibody Fusions With Fluorescent Proteins: A Versatile Reagent for Profiling Protein Expression. J. Immunol. Methods 2001, 257, 175–184. [Google Scholar] [CrossRef]
- Yakushiji, H.; Kobayashi, K.; Takenaka, F.; Kishi, Y.; Shinohara, M.; Akehi, M.; Sasaki, T.; Ohno, E.; Matsuura, E. Novel Single-Chain Variant of Antibody Against Mesothelin Established by Phage Library. Cancer Sci. 2019, 110, 2722–2733. [Google Scholar] [CrossRef] [PubMed]
- Higo-Moriguchi, K.; Akahori, Y.; Iba, Y.; Kurosawa, Y.; Taniguchi, K. Isolation of Human Monoclonal Antibodies That Neutralize Human Rotavirus. J. Virol. 2004, 78, 3325–3332. [Google Scholar] [CrossRef][Green Version]
- Ishikawa, E.; Imagawa, M.; Hashida, S.; Yoshitake, S.; Hamaguchi, Y.; Ueno, T. Enzyme-Labeling of Antibodies and Their Fragments for Enzyme Immunoassay and Immunohistochemical Staining. J. Immunoass. 1983, 4, 209–327. [Google Scholar] [CrossRef]
- Panteghini, M. Traceability as a Unique Tool to Improve Standardization in Laboratory Medicine. Clin. Biochem. 2009, 42, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Panteghini, M.; Forest, J.C. Standardization in laboratory medicine: New challenges. Clin. Chim. Acta 2005, 355, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Klee, G.G. Clinical Interpretation of Reference Intervals and Reference Limits. A Plea for Assay Harmonization. Clin. Chem. Lab. Med. 2004, 42, 752–757. [Google Scholar] [CrossRef] [PubMed]
- Seiki, Y.; Sasaki, Y.; Hosokawa, K.; Saito, C.; Sugimori, N.; Yamazaki, H.; Takami, A.; Nakao, S. Increased Plasma Thrombopoietin Levels in Patients with Myelodysplastic Syndrome: A Reliable Marker for a Benign Subset of Bone Marrow Failure. Haematologica 2013, 98, 901–907. [Google Scholar] [CrossRef] [PubMed]
- Cinquanta, L.; Fontana, D.E.; Bizzaro, N. Chemiluminescent Immunoassay Technology: What Does it Change in Autoantibody Detection? Auto Immun. Highlights 2017, 8, 9. [Google Scholar] [CrossRef]
- Kawasaki, T.; Takeshita, A.; Souda, K.; Kobayashi, Y.; Kikuyama, M.; Suzuki, F.; Kageyama, F.; Sasada, Y.; Shimizu, E.; Murohisa, G.; et al. Serum Thrombopoietin Levels in Patients With Chronic Hepatitis and Liver Cirrhosis. Am. J. Gastroenterol. 1999, 94, 1918–1922. [Google Scholar] [CrossRef]

| High-Titer TPO Plasma Samples from AA Patients | TPO-CLEIA Value (pg/mL) | Multiplied by the Coefficient * | TPO-ELISA Value (pg/mL) | % Difference |
|---|---|---|---|---|
| No. 1 | 294.8 | 1263.7 | 1242.9 | 2.0 |
| No. 2 | 200.1 | 857.7 | 896.1 | −0.4 |
| No. 3 | 446.3 | 1912.8 | 2083.6 | −0.8 |
| No. 4 | 479.6 | 2055.4 | 2155.3 | −0.5 |
| No. 5 | 503.6 | 2158.5 | 2142.6 | 1.0 |
| 10-fold dilution of No. 5 | 52.7 | 225.9 | 240.1 | −5.9 |
| 20-fold dilution of No. 5 | 26.6 | 114.0 | 120.0 | −5.0 |
| 40-fold dilution of No. 5 | 12.8 | 54.6 | 58.6 | −6.7 |
| 80-fold dilution of No. 5 | 6.2 | 26.4 | 18.5 | 42.7 |
| TPO Level of Samples | Precision | Intra-Assay Precision | Interassay Precision | ||||
|---|---|---|---|---|---|---|---|
| Reagent Lot | Lot.1 | Lot.2 | Lot.3 | Lot.1 | Lot.2 | Lot.3 | |
| Low level | Mean (pg/mL) | 52.2 | 50.7 | 52.2 | 46.8 | 45.9 | 47.1 |
| SD (pg/mL) | 1.3 | 1.3 | 0.9 | 1.3 | 1.5 | 1.1 | |
| CV | 2.5% | 2.5% | 1.8% | 2.9% | 3.2% | 2.3% | |
| Middle level | Mean (pg/mL) | 127.3 | 121.2 | 126.1 | 108.2 | 106.7 | 106.8 |
| SD (pg/mL) | 3.1 | 5.7 | 1.0 | 4.6 | 2.0 | 1.4 | |
| CV | 2.4% | 4.7% | 0.8% | 4.3% | 1.9% | 1.3% | |
| High level | Mean (pg/mL) | 326.0 | 314.3 | 331.5 | 309.2 | 288.6 | 286.7 |
| SD (pg/mL) | 6.3 | 12.3 | 8.2 | 5.6 | 6.7 | 16.9 | |
| CV | 1.9% | 3.9% | 2.5% | 1.8% | 2.3% | 5.9% | |
| TPO Level of Samples | Unspiked Samples (pg/mL) | Spiked TPO (pg/mL) | Observed (pg/mL) | Recovery |
|---|---|---|---|---|
| Low level | 41.8 | 49.3 | 87.9 | 93.5% |
| 137 | 181 | 101.6% | ||
| 333.9 | 349.6 | 92.2% | ||
| Middle level | 148.1 | 49.3 | 194.9 | 95.0% |
| 137 | 282.6 | 98.2% | ||
| 333.9 | 437.9 | 86.8% | ||
| High level | 398.4 | 49.3 | 450.6 | 106.0% |
| 137 | 539.3 | 102.9% | ||
| 333.9 | 719.4 | 96.2% |
| Interfering Substances | Concentration | % Difference from Control | ||
|---|---|---|---|---|
| TPO Level of Samples | ||||
| Low | Middle | High | ||
| Hemoglobin | 500 mg/dL | 2.3 | 0.7 | 4.0 |
| Free bilirubin | 20 mg/dL | −2.1 | 2.1 | −8.0 |
| Conjugated bilirubin | 20 mg/dL | −2.3 | −0.4 | −2.6 |
| Chyle material | 1360 FTU | −2.7 | 0.7 | −0.9 |
| Rheumatoid factor | 500 IU/mL | 1.2 | 0.4 | −2.4 |
| High concentration of IgG | 500 mg/dL | −6.5 | 0.2 | −6.9 |
| Sample Type | % Difference from Control | Average Difference, % | |
|---|---|---|---|
| Spiked TPO (pg/mL) | |||
| 50 pg/mL, % | 200 pg/mL, % | ||
| EDTA plasma-1 | 2.3 | −6.0 | −0.9 |
| EDTA plasma-2 | 11.6 | −3.3 | |
| EDTA plasma-3 | −1.6 | −5.9 | |
| EDTA plasma-4 | 4.4 | −1.1 | |
| EDTA plasma-5 | −1.4 | −7.7 | |
| Heparin plasma-1 | 10.5 | −1.1 | 2.2 |
| Heparin plasma-2 | 11.7 | 1.6 | |
| Heparin plasma-3 | −6.8 | −6.1 | |
| Heparin plasma-4 | 8.7 | 0.7 | |
| Heparin plasma-5 | 6.1 | −3.1 | |
| Citrate plasma-1 | 1.1 | −8.0 | −0.7 |
| Citrate plasma-2 | 7.9 | −4.2 | |
| Citrate plasma-3 | 1.8 | −5.9 | |
| Citrate plasma-4 | 10.5 | −0.9 | |
| Citrate plasma-5 | −2.1 | −6.7 | |
| Principle of Measurement | CLEIA | ELISA |
|---|---|---|
| Trade name | Under consideration | Human Thrombopoietin Quantikine ELISA Kit |
| Manufacturer | Medical & Biological Laboratories Co., Ltd. | R & D Systems |
| Capture antibody | Monoclonal | Monoclonal |
| Detection antibody | Monoclonal | Polyclonal |
| Operation | Fully automated | Hand method |
| Measurement time | 20 min | 4.5 h |
| Sample volume | 40 μL | 200 μL |
| LoQ | 14.6 * (3.4) pg/mL | 40.7 pg/mL |
| LoD | 5.6 * (1.3) pg/mL | 21.4 pg/mL |
| Upper limit of measurement | 3000 * (700) pg/mL | 2000 pg/mL |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nishikawa, Y.; Nishida, S.; Kuroda, K.; Kashiwagi, H.; Tomiyama, Y.; Kuwana, M. Development of an Automated Chemiluminescent Enzyme Immunoassay for Measuring Thrombopoietin in Human Plasma. Diagnostics 2022, 12, 313. https://doi.org/10.3390/diagnostics12020313
Nishikawa Y, Nishida S, Kuroda K, Kashiwagi H, Tomiyama Y, Kuwana M. Development of an Automated Chemiluminescent Enzyme Immunoassay for Measuring Thrombopoietin in Human Plasma. Diagnostics. 2022; 12(2):313. https://doi.org/10.3390/diagnostics12020313
Chicago/Turabian StyleNishikawa, Yukihiro, Shiyo Nishida, Keiko Kuroda, Hirokazu Kashiwagi, Yoshiaki Tomiyama, and Masataka Kuwana. 2022. "Development of an Automated Chemiluminescent Enzyme Immunoassay for Measuring Thrombopoietin in Human Plasma" Diagnostics 12, no. 2: 313. https://doi.org/10.3390/diagnostics12020313
APA StyleNishikawa, Y., Nishida, S., Kuroda, K., Kashiwagi, H., Tomiyama, Y., & Kuwana, M. (2022). Development of an Automated Chemiluminescent Enzyme Immunoassay for Measuring Thrombopoietin in Human Plasma. Diagnostics, 12(2), 313. https://doi.org/10.3390/diagnostics12020313






