The Significance of VDR Genetic Polymorphisms in the Etiology of Preeclampsia in Pregnant Polish Women
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Genotyping
2.3. Statistical Analysis
3. Results
3.1. Baseline Demographics and Clinical Characteristics of Study Population
3.2. Associations between SNPs and Preeclampsia Risk
3.3. Association between VDR Gene Variants and Blood Pressure
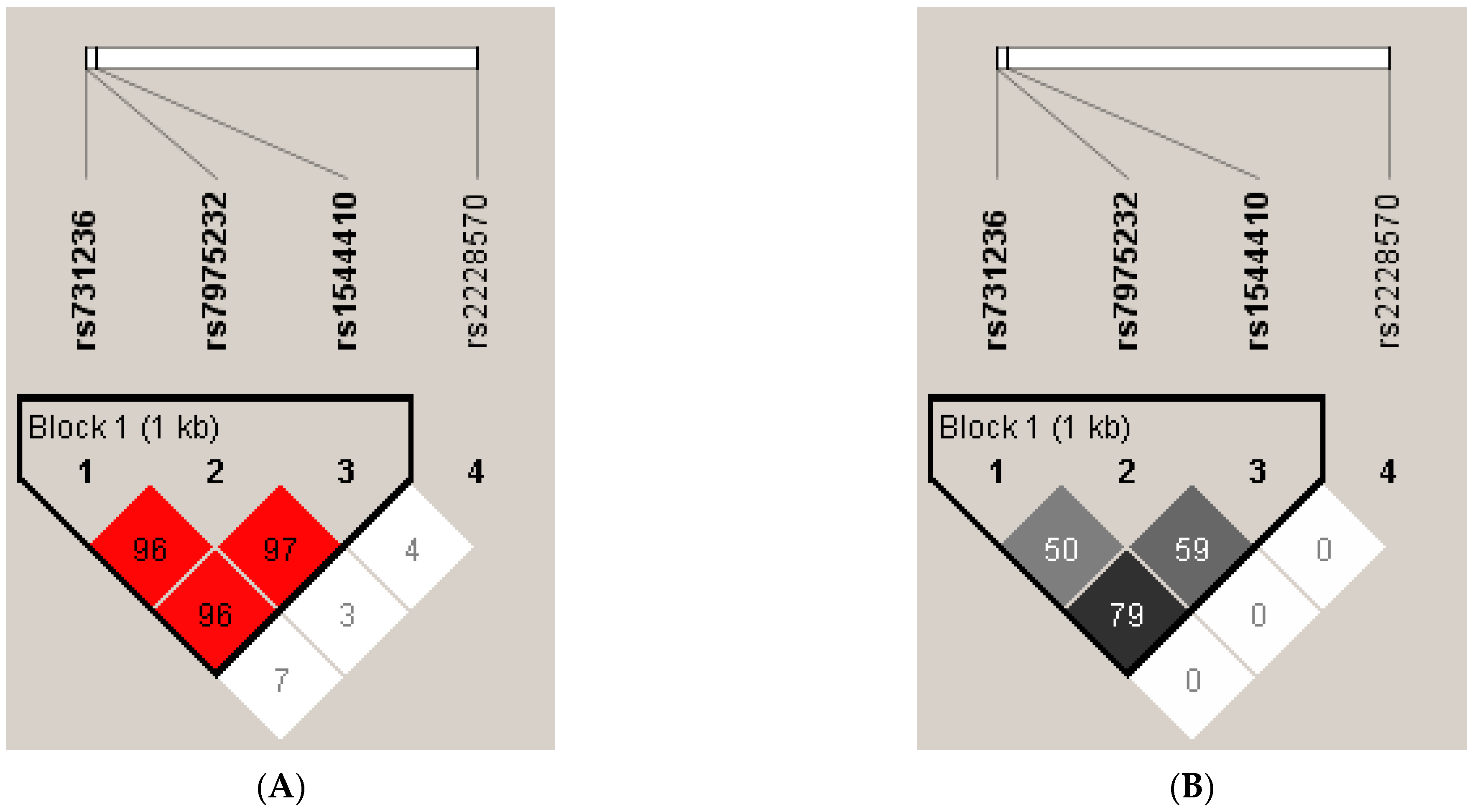
3.4. Linkage Disequilibrium and Haplotype Analysis of VDR SNPs
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Hutcheon, J.A.; Lisonkova, S.; Joseph, K.S. Epidemiology of pre-eclampsia and the other hypertensive disorders of pregnancy. Best Pract. Res. Clin. Obstet. Gynaecol. 2011, 25, 391–403. [Google Scholar] [CrossRef]
- Teliga-Czajkowska, J.; Czajkowski, K.; Januszewicz, A.; Sieradzki, J.; Więcek, A. Nadciśnienie tętnicze i diabetologia w pytaniach i odpowiedziach. In Nadciśnienie Tętnicze a Ciąża; Medycyna Praktyczna; Szczeklik, A., Gajewski, P., Eds.; Wyd. 1: Kraków, Poland, 2009; pp. 207–221. [Google Scholar]
- Szczepaniak-Chicheł, L.; Bręborowicz, G.; Tykarski, A. Leczenie nadciśnienia tętniczego u kobiet w ciąży. Nadciśnienie Tętnicze 2006, 10, 83–98. [Google Scholar]
- Bokslag, A.; van Weissenbruch, M.; Mol, B.W.; de Groot, C.J.M. Preeclampsia; short and long-term consequences for mother and neonate. Early Hum. Dev. 2016, 102, 47–50. [Google Scholar] [CrossRef]
- McKinney, D.; Boyd, H.; Langager, A.; Oswald, M.; Pfister, A.; Warshak, C.R. The impact of fetal growth restriction on latency in the setting of expectant management of preeclampsia. Am. J. Obstet. Gynecol. 2016, 214, 395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fox, R.; Kitt, J.; Leeson, P.; Aye, C.Y.L.; Lewandowski, A.J. Preeclampsia: Risk factors, diagnosis, management and the cardiovascular impact on the offspring. J. Clin. Med. 2019, 8, 1625. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ożarowski, M.; Karpiński, T.M.; Szulc, M.; Wielgus, K.; Kujawski, R.; Wolski, H.; Seremak-Mrozikiewicz, A. Plant phenolics and extracts in animal models of preeclampsia and clinical trials—Review of perspectives for novel therapies. Pharmaceuticals 2021, 14, 269. [Google Scholar] [CrossRef] [PubMed]
- Knabl, J.; Vattai, A.; Ye, Y.; Jueckstock, J.; Hutter, S.; Kainer, F.; Mahner, S.; Jeschke, U. Role of placental VDR expression and function in common late pregnancy disorders. Int. J. Mol. Sci. 2017, 18, 2340. [Google Scholar] [CrossRef] [Green Version]
- Rana, S.; Lemoine, E.; Granger, J.P.; Karumanchi, S.A. Preeclampsia: Pathophysiology, challenges, and perspectives. Circ Res. 2019, 124, 1094–1112. [Google Scholar] [CrossRef]
- Jena, M.K.; Sharma, N.R.; Petitt, M.; Maulik, D.; Nayak, N.R. Pathogenesis of preeclampsia and therapeutic approaches targeting the placenta. Biomolecules 2020, 10, 953. [Google Scholar] [CrossRef] [PubMed]
- Tsuprykov, O.; Chen, X.; Hocher, C.F.; Skoblo, R.; Lianghong, Y.; Hocher, B. Why should we measure free 25(OH) vitamin D? J. Steroid Biochem. Mol. Biol. 2018, 180, 87–104. [Google Scholar] [CrossRef]
- Hollis, B.W. Assessment of vitamin D status and definition of normal circulating range of 25-hydroxyvitamin D. Curr. Opin. Endocrinol. Diabetes. Obes. 2008, 15, 489–494. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D: A D-lightful health perspective. Nutr. Rev. 2008, 66, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Lensmeyer, G.; Poquette, M.; Wiebe, D.; Binkley, N. The C-3 epimer of 25-hydroxyvitamin D(3) is present in adult serum. J. Clin. Endocrinol. Metab. 2012, 97, 163–168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Specker, B. Vitamin D requirements during pregnancy. Am. J. Clin. Nutr. 2004, 80, 1740–1747. [Google Scholar] [CrossRef] [Green Version]
- Williams, A.F. Vitamin D In pregnancy: An old problem still to be solved? Arch. Dis. Child. 2007, 92, 740–741. [Google Scholar] [CrossRef]
- Hollis, B.W.; Wagner, C.L. Vitamin D and pregnancy: Skeletal effects, nonskeletal effects, and birth outcomes. Calcif. Tissue Int. 2013, 92, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Colonese, F.; Laganà, A.S.; Colonese, E.; Sofo, V.; Salmeri, F.M.; Granese, R.; Triolo, O. The pleiotropic effects of vitamin D in gynaecological and obstetric diseases: An overview on a hot topic. Biomed. Res. Int. 2015, 2015, 986281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagner, C.L.; Hollis, B.W. The Implications of Vitamin D Status during Pregnancy on Mother and her Developing Child. Front. Endocrinol. 2018, 9, 500. [Google Scholar] [CrossRef] [Green Version]
- Powe, C.E.; Levine, R.J.; Karumanchi, S.A. Preeclampsia, a disease of the maternal endothelium: The role of antiangiogenic factors and implications for later cardiovascular disease. Circulation 2011, 123, 2856–2869. [Google Scholar] [CrossRef]
- Widmer, R.J.; Lerman, A. Endothelial dysfunction and cardiovascular disease. Glob. Cardiol. Sci. Pract. 2014, 3, 291–308. [Google Scholar] [CrossRef] [PubMed]
- Konukoglu, D.; Uzun, H. Endothelial dysfunction and hypertension. Adv. Exp. Med. Biol. 2016, 956, 511–540. [Google Scholar]
- Liberis, A.; Stanulov, G.; Ali, E.C.; Hassan, A.; Pagalos, A.; Kontomanolis, E.N. Pre-eclampsia and the vascular endothelial growth factor: A new aspect. Clin. Exp. Obstet. Gynecol. 2016, 43, 9–13. [Google Scholar] [PubMed]
- Guney, G.; Taskin, M.I.; Tokmak, A. Increase of circulating inflammatory molecules in preeclampsia, an update. Eur. Cytokine Netw. 2020, 31, 18–31. [Google Scholar] [CrossRef]
- Harmon, A.C.; Cornelius, D.C.; Amaral, L.M.; Faulkner, J.L.; Cunningham, M.W.; Wallace, K.; LaMarca, B. The role of inflammation in the pathology of preeclampsia. Clin. Sci. 2016, 130, 409–419. [Google Scholar] [CrossRef] [Green Version]
- Barrera, D.; Díaz, L.; Noyola-Martínez, N.; Halhali, A. Vitamin D and inflammatory cytokines in healthy and preeclamptic pregnancies. Nutrients 2015, 7, 6465–6490. [Google Scholar] [CrossRef] [Green Version]
- Chan, S.Y.; Susarla, R.; Canovas, D.; Vasilopoulou, E.; Ohizua, O.; McCabe, C.J.; Hewison, M.; Kilby, M.D. Vitamin D promotes human extravillous trophoblast invasion in vitro. Placenta 2015, 36, 403–409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, H.; Daftary, G.S.; Lalwani, S.I.; Taylor, H.S. Direct regulation of HOXA10 by 1,25-(OH)2D3 in human myelomonocytic cells and human endometrial stromal cells. Mol. Endocrinol. 2005, 19, 2222–2233. [Google Scholar] [CrossRef] [Green Version]
- Boonstra, A.; Barrat, F.J.; Crain, C.; Heath, V.L.; Savelkoul, H.F.; O’Garra, A. 1alpha,25-Dihydroxyvitamin d3 has a direct effect on naive CD4(+) T cells to enhance the development of Th2 cells. J. Immunol. 2001, 167, 4974–4980. [Google Scholar] [CrossRef] [Green Version]
- Cardús, A.; Parisi, E.; Gallego, C.; Aldea, M.; Fernández, E.; Valdivielso, J.M. 1,25-Dihydroxyvitamin D3 stimulates vascular smooth muscle cell proliferation through a VEGF-mediated pathway. Kidney Int. 2006, 69, 1377–1384. [Google Scholar] [CrossRef] [Green Version]
- Barrera, D.; Noyola-Martínez, N.; Avila, E.; Halhali, A.; Larrea, F.; Díaz, L. Calcitriol inhibits interleukin-10 expression in cultured human trophoblasts under normal and inflammatory conditions. Cytokine 2012, 57, 316–321. [Google Scholar] [CrossRef]
- Xu, J.; Gu, Y.; Lewis, D.F.; Cooper, D.B.; McCathran, C.E.; Wang, Y. Downregulation of vitamin D receptor and miR-126-3p expression contributes to increased endothelial inflammatory response in preeclampsia. Am. J. Reprod. Immunol. 2019, 82, e13172. [Google Scholar] [CrossRef]
- Li, Y.C. Vitamin D regulation of the renin-angiotensin system. J. Cell Biochem. 2003, 88, 327–331. [Google Scholar] [CrossRef]
- Akbari, S.; Khodadadi, B.; Ahmadi, S.A.Y.; Abbaszadeh, S.; Shahsavar, F. Association of vitamin D level and vitamin D deficiency with risk of preeclampsia: A systematic review and updated meta-analysis. Taiwan J. Obstet. Gynecol. 2018, 57, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Lumbers, E.R.; Delforce, S.J.; Arthurs, A.L.; Pringle, K.G. Causes and Consequences of the Dysregulated Maternal Renin-Angiotensin System in Preeclampsia. Front. Endocrinol. 2019, 10, 563. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.R.; McDonnellm, D.P.; Hughes, M.; Crisp, T.M.; Mangelsdorf, D.J.; Haussler, M.R.; Pike, J.W.; Shine, J.; O’Malley, B.W. Cloning and expression of full-length cDNA encoding human vitamin D receptor. Proc. Natl. Acad. Sci. USA 1988, 85, 3294–3298. [Google Scholar] [CrossRef] [Green Version]
- Uitterlinden, A.G.; Fang, Y.; Van Meurs, J.B.J.; Pols, H.A.P.; Van Leeuwen, J.P. Genetics and biology of vitamin D receptor polymorphisms. Gene 2004, 338, 143–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arai, H.; Miyamoto, K.; Taketani, Y.; Yamamoto, H.; Iemori, Y.; Morita, K.; Tonai, T.; Nishisho, T.; Mori, S.; Takeda, E. A vitamin D receptor gene polymorphism in the translation initiation codon: Effect on protein activity and relation to bone mineral density in Japanese women. J. Bone Miner. Res. 1997, 12, 915–921. [Google Scholar] [CrossRef]
- ACOG. ACOG practice bulletin no. 202: Gestational hypertension and preeclampsia. Obs. Gynecol 2019, 133, e1–e25. [CrossRef]
- Farajian-Mashhadi, F.; Eskandari, F.; Rezaei, M.; Eskandari, F.; Najafi, D.; Teimoori, B.; Moradi-Sharbabak, M.; Salimi, S. The possible role of maternal and placental vitamin D receptor polymorphisms and haplotypes in pathogenesis of preeclampsia. Clin. Exp. Hypertens. 2020, 42, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Barchitta, M.; Maugeri, A.; La Rosa, M.C.; San Lio, R.M.; Favara, G.; Panella, M.; Cianci, A.; Agodi, A. The effect of vitamin D receptor gene (VDR) polymorphisms on adverse pregnancy outcomes—Including preterm birth (PTB and small for gestational age. Nutrients 2018, 10, 1172. [Google Scholar] [CrossRef] [Green Version]
- Pani, M.A.; Knapp, M.; Donner, H.; Braun, J.; Baur, M.P.; Usadel, K.H.; Badenhoop, K. Vitamin D receptor allele combinations influence genetic susceptibility to type 1 diabetes in Germans. Diabetes 2000, 49, 504–507. [Google Scholar] [CrossRef] [Green Version]
- Morrison, N.A.; Qi, J.C.; Tokita, A.; Kelly, P.J.; Crofts, L.; Nguyen, T.V.; Sambrook, P.N.; Eisman, J.A. Prediction of bone density from vitamin D receptor alleles. Nature 1994, 367, 284–287. [Google Scholar] [CrossRef]
- Harris, S.S.; Eccleshall, T.R.; Gross, C.; Dawson-Hughes, B.; Feldman, D. The vitamin D receptor start codon polymorphism (FokI) and bone mineral density in premenopausal American black and white women. J. Bone Miner. Res. 1997, 12, 1043–1048. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- González, J.R.; Armengol, L.; Solé, X.; Guinó, E.; Mercader, J.M.; Estivill, X.; Moreno, V. SNPassoc: An R package to perform whole genome association studies. Bioinformatics 2007, 23, 644–645. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Ma, J.; Manson, J.E.; Buring, J.E.; Gaziano, J.M.; Sesso, H.D. A prospective study of plasma vitamin D metabolites, vitamin D receptor gene polymorphisms, and risk of hypertension in men. Eur. J. Nutr. 2013, 52, 1771–1779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valdivielso, J.M.; Fernandez, E. Vitamin D receptor polymorphisms and diseases. Clin. Chim. Acta 2006, 371, 1–12. [Google Scholar] [CrossRef]
- Zhu, Y.B.; Li, Z.Q.; Ding, N.; Yi, H.L. The association between vitamin D receptor gene polymorphism and susceptibility to hypertension: A meta-analysis. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 9066–9074. [Google Scholar] [CrossRef]
- Liu, J.; Dai, Q.; Li, W.; Guo, Y.; Dai, A.; Wang, Y.; Deng, M.; Tang, Z.; She, L.; Chen, X.; et al. Association of vitamin D receptor gene polymorphisms with gestational diabetes mellitus-a case control study in Wuhan, China. BMC Pregnancy Childbirth 2021, 21, 142. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Wen, S.; Liu, M.; Zhang, S.; Jin, X.; Liu, A. Association between Gene Polymorphisms of Vitamin D Receptor and Gestational Diabetes Mellitus: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2020, 18, 205. [Google Scholar] [CrossRef]
- Apaydın, M.; Beysel, S.; Eyerci, N.; Pinarli, F.A.; Ulubay, M.; Kizilgul, M.; Ozdemir, O.; Caliskan, M.; Cakal, E. The VDR gene FokI polymorphism is associated with gestational diabetes mellitus in Turkish women. BMC Med. Genet. 2019, 20, 82. [Google Scholar] [CrossRef] [PubMed]
- Krpina, M.G.; Barišić, A.; Peterlin, A.; Tul, N.; Ostojić, S.; Peterlin, B.; Pereza, N. Vitamin D receptor polymorphisms in spontaneous preterm birth: A case-control study. Croat. Med. J. 2020, 61, 338–345. [Google Scholar] [CrossRef]
- Barišić, A.; Pereza, N.; Hodžić, A.; Krpina, M.G.; Ostojić, S.; Peterlin, B. Genetic variation in the maternal vitamin D receptor FokI gene as a risk factor for recurrent pregnancy loss. J. Matern. Fetal. Neonatal. Med. 2021, 34, 2221–2226. [Google Scholar] [CrossRef]
- Wolski, H.; Kurzawińska, G.; Ożarowski, M.; Mrozikiewicz, A.E.; Drews, K.; Karpiński, T.M.; Bogacz, A.; Seremak-Mrozikiewicz, A. Vitamin D receptor gene polymorphisms and haplotypes in the etiology of recurrent miscarriages. Sci. Rep. 2021, 11, 4646. [Google Scholar] [CrossRef] [PubMed]
- Wolski, H.; Kurzawinska, G.; Ozarowski, M.; Drews, K.; Barlik, M.; Piatek, K.; Malewski, Z.; Mrozikiewicz, A.E.; Magielda-Stola, J.; Kolanowska, D.; et al. FokI vitamin D receptor polymorphism as a protective factor in intrahepatic cholestasis of pregnancy. Ginekol. Pol. 2020, 91, 719–725. [Google Scholar] [CrossRef]
- Rezavand, N.; Tabarok, S.; Rahimi, Z.; Vaisi-Raygani, A.; Mohammadi, E.; Rahimi, Z. The effect of VDR gene polymorphisms and vitamin D level on blood pressure, risk of preeclampsia, gestational age, and body mass index. J. Cell. Biochem. 2019, 120, 6441–6448. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Liu, M.; You, Y.; Zhang, Y.; Wang, J.; Wang, X.; Liu, S.; Liu, X. Genetic variations in the vitamin-D receptor (VDR) gene in preeclampsia patients in the Chinese Han population. Hypertens. Res. 2015, 38, 513–517. [Google Scholar] [CrossRef]
- Baca, K.M.; Govil, M.; Zmuda, J.M.; Simhan, H.N.; Marazita, M.L.; Bodnar, L.M. Vitamin D metabolic loci and preeclampsia risk in multi-ethnic pregnant women. Physiol. Rep. 2018, 6, e13468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rezende, V.B.; Sandrim, V.C.; Palei, A.C.; Machado, L.; Cavalli, R.C.; Duarte, G.; Tanus-Santos, J.E. Vitamin D receptor polymorphisms in hypertensive disorders of pregnancy. Mol. Biol. Rep. 2012, 39, 10903–10906. [Google Scholar] [CrossRef] [Green Version]
- Ghorbani, Z.; Shakiba, M.; Rezavand, N.; Rahimi, Z.; Vaisi-Raygani, A.; Rahimi, Z.; Shakiba, E. Gene variants and haplotypes of Vitamin D biosynthesis, transport, and function in preeclampsia. Hypertens. Pregnancy 2021, 40, 1–8. [Google Scholar] [CrossRef]
- Caccamo, D.; Cannata, A.; Ricca, S.; Catalano, L.M.; Montalto, A.F.; Alibrandi, A.; Ercoli, A.; Granese, R. Role of Vitamin-D Receptor (VDR) single nucleotide polymorphisms in gestational hypertension development: A case-control study. PLoS ONE. 2020, 15, e0239407. [Google Scholar] [CrossRef]
| SNP | Chromosome Position * | Localization | Function | Alleles | Genomic Positions ** |
|---|---|---|---|---|---|
| rs2228570 (FokI) | chr12:47879112 | Exon 2 | Met-Thr | C>T | g.30920T>C |
| rs1544410 (BsmI) | chr12:47846052 | Intron 8 | Non-coding | G>A | g.63980G>A |
| rs7975232 (ApaI) | chr12:47845054 | Intron 8 | Non-coding | T>G | g.64978G>T |
| rs731236 (TaqI) | chr12:47844974 | Exon 9 | Ile-Ile | T>C | g.65058T>C |
| Variables | Preeclampsia n = 122 | Controls n = 184 | p |
|---|---|---|---|
| Maternal age (years), mean ± SD | 30.12 ± 5.52 | 30.58 ± 4.41 | 0.4507 |
| Gestational age (weeks), mean ± SD | 33.72 ± 3.52 | 39.01 ± 1.21 | <0.001 |
| Systolic blood pressure (mmHg), mean ± SD | 171.04 ± 19.27 | 102.31 ± 9.83 | <0.001 |
| Diastolic blood pressure (mmHg), mean ± SD | 106.61 ± 12.89 | 64.97 ± 7,69 | <0.001 |
| Pre-pregnancy BMI (kg/m2), mean ± SD | 24.56 ±.6.97 | 20.90 ± 1.97 | <0.001 |
| Post-pregnancy BMI (kg/m2), mean ± SD | 30.40 ± 5.30 | 26.24 ± 2.60 | <0.001 |
| Caesarean section, N (%) | 110 (90.16) | 61 (33.15) | <0.001 * |
| Primipara, N (%) | 75 (61.48) | 72 (39.13) | 0.0001 * |
| Infant birthweight (g), mean ± SD | 1887.95 ± 841.64 | 3417.66 ± 428.15 | <0.001 |
| 1-min Apgar score, median (IQR) | 8.00 (6.00–9.75) | 10.00 (10.00–10.00) | <0.001 # |
| 5-min Apgar score, median (IQR) | 9.00 (8.00–10.00) | 10.00 (10.00–10.00) | <0.001 # |
| Variables | EOPE n = 57 | LOPE n = 65 | p |
|---|---|---|---|
| Maternal age (years), mean ± SD | 29.70 ± 5.49 | 30.50 ± 5.57 | 0.4298 |
| Gestational age (weeks), mean ± SD | 30.60 ± 2.09 | 36.54 ± 1.69 | <0.001 |
| Systolic blood pressure (mmHg), mean ± SD | 175.65 ± 19.51 | 166.97 ± 18.26 | 0.0171 |
| Diastolic blood pressure (mmHg), mean ± SD | 107.44 ± 13.53 | 105.88 ± 12.37 | 0.5268 |
| Pre-pregnancy BMI (kg/m2), mean ± SD | 24.46 ± 5.72 | 24.65 ± 7.93 | 0.8859 |
| Post-pregnancy BMI (kg/m2), mean ± SD | 30.02 ± 4.19 | 30.74 ± 6.12 | 0.4660 |
| Caesarean section, N (%) | 56 (98.25%) | 52 (80.00%) | 0.0171 ** |
| Primipara, N (%) | 35 (61.40%) | 40 (61.54%) | 0.9878 * |
| Infant birthweight (g), mean ± SD | 1336.11 ± 433.69 | 2403.61 ± 803.82 | <0.001 |
| 1-min Apgar score, median (IQR) | 7.00 (4.00–8.00) | 9.00 (8.00–10.00) | <0.001 # |
| 5-min Apgar score, median (IQR) | 8.00 (7.00–9.00) | 10.00 (9.00–10.00) | <0.001 # |
| ALT (IU/L), mean ± SD | 53.83 ± 58.51 | 23.08 ± 17.49 | 0.0411 |
| AST (IU/L), mean ± SD | 52.17 ± 52.50 | 25.50 ± 16.80 | 0.0492 |
| Urea (mg/dL), mean ± SD | 32.42 ± 13.65 | 29.59 ± 14.00 | 0.2740 |
| Uremic acid (mg/dL), mean ± SD | 6.74 ± 1.49 | 6.35 ± 1.16 | 0.1198 |
| Total protein (g/dL), mean ± SD | 5.47 ± 0.56 | 5.81± 0.78 | 0.0089 |
| Creatinine (mg/dL), mean ± SD | 0.81 ± 0.26 | 0.76 ± 0.30 | 0.5558 |
| Proteinuria (mg/dL), mean ± SD | 365.76 ± 185.91 | 195.07 ± 165.32 | <0.001 |
| Proteinuria (g/24 h), mean ± SD | 4.57 ± 2.92 | 2.92 ± 3.64 | 0.1680 |
| SNP | Alleles | Preeclampsia n = 244 | Controls n = 368 | p | p Corr. | ||
|---|---|---|---|---|---|---|---|
| MAF n (Frequency) | HWE p | MAF n (Frequency) | HWE p | ||||
| rs2228570 (FokI) | C>T | 118 (0.484) | 0.595 | 182 (0.495) | 0.140 | 0.791 | 1.000 |
| rs1544410 (BsmI) | G>A | 114 (0.467) | 0.021 | 130 (0.353) | 0.104 | 0.005 | 0.020 |
| rs7975232 (ApaI) | T>G | 107 (0.439) | 0.865 | 191 (0.519) | 0.448 | 0.051 | 0.204 |
| rs731236 (TaqI) | T>C | 101 (0.414) | 0.971 | 122 (0.332) | 0.554 | 0.038 | 0.152 |
| SNP | Genotypes/Models | Preeclampsia n = 122 n (%) | Controls n = 184 n (%) | OR (95%CI) | p | padj. |
|---|---|---|---|---|---|---|
| rs2228570 (FokI) | CC | 34 (27.9) | 42 (22.8) | 1.00 | 0.3897 | 0.5040 |
| CT | 58 (47.5) | 102 (55.4) | 1.42 (0.82–2.48) | |||
| TT | 30 (24.6) | 40 (21.7) | 1.08 (0.56–2.08) | |||
| Dominant | 88 (72.1) | 142 (77.2) | 1.31 (0.77–2.21) | 0.3194 | 0.5383 | |
| Recessive | 92 (75.4) | 144 (78.3) | 0.85 (0.50–1.46) | 0.5620 | 0.4498 | |
| rs1544410 (BsmI) | GG | 41 (33.6) | 82 (44.6) | 1.00 | 0.0263 | 0.0113 |
| GA | 48 (39.3) | 74 (40.2) | 0.77 (0.46–1.30) | |||
| AA | 33 (27.0) | 28 (15.2) | 0.42 (0.23–0.79) | |||
| Dominant | 81 (66.4) | 102 (55.4) | 0.63 (0.39–1.01) | 0.0545 | 0.0334 | |
| Recessive | 89 (73.0) | 156 (84.8) | 0.48 (0.27–0.85) | 0.0119 | 0.0048 | |
| rs7975232 (ApaI) | TT | 38 (31.1) | 40 (21.7) | 1.00 | 0.1304 | 0.0765 |
| TG | 61 (50.0) | 97 (52.7) | 1.51 (0.87–2.61) | |||
| GG | 23 (18.9) | 47 (25.5) | 1.94 (1.00–3.79) | |||
| Dominant | 84 (68.9) | 144 (78.3) | 1.63 (0.97–2.74) | 0.0660 | 0.0425 | |
| Recessive | 99 (81.1) | 137 (74.5) | 1.48 (0.84–2.59) | 0.1688 | 0.1064 | |
| rs731236 (TaqI) | TT | 42 (34.4) | 84 (45.7) | 1.00 | 0.1171 | 0.0830 |
| TC | 59 (48.4) | 78 (42.4) | 0.66 (0.40–1.09) | |||
| CC | 21 (17.2) | 22 (12.0) | 0.52 (0.26–1.06) | |||
| Dominant | 80 (65.6) | 100 (54.3) | 0.63 (0.39–1.00) | 0.0498 | 0.0375 | |
| Recessive | 101 (82.8) | 162 (88.0) | 0.65 (0.34–1.25) | 0.1987 | 0.1353 |
| SNP | Genotypes/ Models | Controls (n = 184) n (%) | EOPE (n = 57) n (%) | OR (95%CI) | p | padj. | LOPE (n = 65) n (%) | OR (95%CI) | p | padj. |
|---|---|---|---|---|---|---|---|---|---|---|
| rs2228570 (FokI) | CC | 42 (22.8) | 14 (24.6) | 1.00 | 0.5173 | 0.2164 | 20 (30.8) | 1.00 | 0.4240 | 0.4995 |
| CT | 102 (55.4) | 27 (47.4) | 1.26 (0.60–2.64) | 31 (47.7) | 1.57 (0.80–3.05) | |||||
| TT | 40 (21.7) | 16 (28.1) | 0.83 (0.36–1.93) | 14 (21.5) | 1.36 (0.61–3.05) | |||||
| Dominant | 142 (77.2) | 43 (75.4) | 1.10 (0.55–2.20) | 0.7873 | 0.6852 | 45 (69.2) | 1.50 (0.80–2.82) | 0.2097 | 0.2387 | |
| Recessive | 144 (78.3) | 41 (71.9) | 0.71 (0.36–1.40) | 0.3299 | 0.0815 | 51 (78.5) | 1.01 (0.51–2.01) | 0.9731 | 0.7277 | |
| rs1544410 (BsmI) | GG | 82 (44.6) | 17 (29.8) | 1.00 | 0.0913 | 0.0464 | 24 (36.9) | 1.00 | 0.0563 | 0.0835 |
| GA | 74 (40.2) | 26 (45.6) | 0.59 (0.30–1.17) | 22 (33.8) | 0.98 (0.51–1.90) | |||||
| AA | 28 (15.2) | 14 (24.6) | 0.41 (0.18–0.95) | 19 (29.2) | 0.43 (0.21–0.90) | |||||
| Dominant | 102 (55.4) | 40 (70.2) | 0.53 (0.28–1.00) | 0.0451 | 0.0498 | 41 (63.1) | 0.73 (0.41–1.30) | 0.2819 | 0.2465 | |
| Recessive | 89 (73.0) | 43 (75.4) | 0.55 (0.27–1.14) | 0.1150 | 0.0305 | 46 (70.8) | 0.43 (0.22–0.85) | 0.0165 | 0.0263 | |
| rs7975232 (ApaI) | TT | 40 (21.7) | 17 (29.8) | 1.00 | 0.0724 | 0.0356 | 21 (32.3) | 1.00 | 0.2232 | 0.2956 |
| TG | 97 (52.7) | 33 (57.9) | 1.25 (0.63–2.49) | 28 (43.1) | 1.82 (0.93–3.57) | |||||
| GG | 47 (25.5) | 7 (12.3) | 2.85 (1.08–7.57) | 16 (24.6) | 1.54 (0.71–3.35) | |||||
| Dominant | 144 (78.3) | 40 (70.2) | 1.53 (0.79–2.98) | 0.2176 | 0.0911 | 44 (67.7) | 1.72 (0.92–3.22) | 0.0906 | 0.1258 | |
| Recessive | 137 (74.5) | 50 (87.7) | 2.45 (1.04–5.78) | 0.0275 | 0.0185 | 49 (75.4) | 1.05 (0.54–2.02) | 0.8820 | 0.7935 | |
| rs731236 (TaqI) | TT | 84 (45.7) | 18 (31.6) | 1.00 | 0.1613 | 0.1940 | 24 (36.9) | 1.00 | 0.2311 | 0.2636 |
| TC | 78 (42.4) | 31 (54.4) | 0.54 (0.28–1.04) | 28 (43.1) | 0.80 (0.43–1.49) | |||||
| CC | 22 (12.0) | 8 (14.0) | 0.59 (0.23–1.53) | 13 (20.0) | 0.48 (0.21–1.10) | |||||
| Dominant | 100 (54.3) | 39 (68.4) | 0.55 (0.29–1.03) | 0.0574 | 0.0843 | 41 (63.1) | 0.70 (0.39–1.25) | 0.2199 | 0.2094 | |
| Recessive | 162 (88.0) | 49 (86.0) | 0.83 (0.35–1.99) | 0.6815 | 0.2832 | 52 (80.0) | 0.54 (0.26–1.15) | 0.1200 | 0.1492 |
| Mean ± SD (mmHg) | Codominant | Dominant | Recessive | ||||||
|---|---|---|---|---|---|---|---|---|---|
| AA | AB | BB | p | padj. | p | padj. | p | padj. | |
| rs2228570 (FokI) | |||||||||
| SBP | 131.27 ± 35.25 | 124.91 ± 34.93 | 132.15 ± 39.84 | 0.2736 | 0.3156 | 0.3920 | 0.6805 | 0.3070 | 0.2073 |
| DBP | 81.87 ± 22.63 | 79.49 ± 22.15 | 81.90 ± 23.44 | 0.6620 | 0.6926 | 0.5860 | 0.9268 | 0.6048 | 0.4344 |
| rs1544410 (BsmI) | |||||||||
| SBP | 122.58 ± 33.55 | 127.69 ± 35.04 | 140.08 ± 41.00 | 0.0091 | 0.0033 | 0.0303 | 0.0172 | 0.0041 | 0.0015 |
| DBP | 76.74 ± 21.47 | 81.03 ± 21.51 | 87.57 ± 25.00 | 0.0094 | 0.0026 | 0.0148 | 0.0073 | 0.0074 | 0.0020 |
| rs7975232 (ApaI) | |||||||||
| SBP | 136.84 ± 40.02 | 126.79 ± 34.90 | 121.54 ± 33.16 | 0.0319 | 0.0121 | 0.0151 | 0.0066 | 0.0855 | 0.0436 |
| DBP | 85.34 ± 24.21 | 80.12 ± 22.00 | 76.54 ± 21.06 | 0.0591 | 0.0196 | 0.0345 | 0.0127 | 0.0870 | 0.0431 |
| rs731236 (TaqI) | |||||||||
| SBP | 122.56 ± 33.78 | 130.91 ± 36.45 | 135.71 ± 40.54 | 0.0641 | 0.0432 | 0.0263 | 0.0169 | 0.1451 | 0.1229 |
| DBP | 76.90 ± 21.93 | 82.54 ± 21.82 | 85.45 ± 25.15 | 0.0448 | 0.0263 | 0.0171 | 0.0104 | 0.1352 | 0.0936 |
| rs731236 | rs7975232 | rs1544410 | rs2228570 | Frequency (Overall) | Frequency (PE, Control) | χ2 | p Value | * p Value |
|---|---|---|---|---|---|---|---|---|
| T | G | G | 0.476 | 0.425, 0.510 | 4.325 | 0.0376 | 0.1705 | |
| C | T | A | 0.356 | 0.410, 0.320 | 5.090 | 0.0241 | 0.0940 | |
| T | T | G | 0.117 | 0.104, 0.125 | 0.654 | 0.4185 | 0.9505 | |
| T | T | A | 0.038 | 0.045, 0.033 | 0.657 | 0.4178 | 0.9503 | |
| G | G | C | 0.256 | 0.238, 0.269 | 0.734 | 0.3917 | 0.9553 | |
| G | G | T | 0.225 | 0.188, 0.250 | 3.225 | 0.0725 | 0.3043 | |
| T | A | C | 0.205 | 0.229, 0.190 | 1.347 | 0.2458 | 0.7768 | |
| T | A | T | 0.188 | 0.226, 0.163 | 3.812 | 0.0509 | 0.2231 | |
| T | G | T | 0.075 | 0.064, 0.082 | 0.708 | 0.4000 | 0.9594 | |
| T | G | C | 0.045 | 0.043, 0.046 | 0.028 | 0.8660 | 1.0000 | |
| T | G | G | C | 0.252 | 0.236, 0.262 | 0.502 | 0.4789 | 0.9959 |
| T | G | G | T | 0.225 | 0.188, 0.249 | 3.091 | 0.0787 | 0.3742 |
| C | T | A | C | 0.190 | 0.220, 0.170 | 2.419 | 0.1199 | 0.5402 |
| C | T | A | T | 0.166 | 0.189, 0.150 | 1.598 | 0.2062 | 0.8185 |
| T | T | G | T | 0.073 | 0.062, 0.080 | 0.673 | 0.4122 | 0.9892 |
| T | T | G | C | 0.044 | 0.042, 0.045 | 0.046 | 0.8299 | 1.0000 |
| T | T | A | T | 0.022 | 0.036, 0.013 | 3.618 | 0.0572 | 0.2777 |
| T | T | A | C | 0.016 | 0.009, 0.020 | 1.015 | 0.3138 | 0.9720 |
| 4-Marker | SBP | DBP | |||||
|---|---|---|---|---|---|---|---|
| Haplotype | Frequency | Difference | 95%CI | p-Value | Difference | 95%CI | p-Value |
| TGGC | 0.253 | 122.07 | Ref. | 76.31 | Ref. | ||
| CTAC | 0.195 | 7.94 | (−2.24–18.13) | 0.1264 | 6.10 | (−0.37–12.57) | 0.0648 |
| CTAT | 0.162 | 8.12 | (−1.55–17.79) | 0.0998 | 4.57 | (−1.42–10.56) | 0.1348 |
| TTGT | 0.073 | 2.63 | (−10.61–15.86) | 0.6970 | 0.90 | (−7.42–9.21) | 0.8324 |
| TGGT | 0.222 | -2.17 | (−11.79–7.45) | 0.6587 | −0.51 | (−6.72–5.71) | 0.8728 |
| rare | 0.094 | 4.99 | (−6.38–16.37) | 0.3897 | 3.00 | (−4.29–10.28) | 0.4201 |
| 3-marker | |||||||
| TGG | 0.475 | 120.21 | Ref. | 75.93 | Ref. | ||
| CTA | 0.357 | 8.84 | (2.36–15.33) | 0.0075 | 5.53 | (1.50–9.56) | 0.0072 |
| TTG | 0.114 | 3.59 | (−6.10–13.27) | 0.4677 | 1.38 | (−4.64–7.40) | 0.6529 |
| rare | 0.053 | 7.76 | (−4.23–19.75) | 0.2046 | 4.12 | (−3.33–11.58) | 0.2780 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magiełda-Stola, J.; Kurzawińska, G.; Ożarowski, M.; Karpiński, T.M.; Drews, K.; Seremak-Mrozikiewicz, A. The Significance of VDR Genetic Polymorphisms in the Etiology of Preeclampsia in Pregnant Polish Women. Diagnostics 2021, 11, 1698. https://doi.org/10.3390/diagnostics11091698
Magiełda-Stola J, Kurzawińska G, Ożarowski M, Karpiński TM, Drews K, Seremak-Mrozikiewicz A. The Significance of VDR Genetic Polymorphisms in the Etiology of Preeclampsia in Pregnant Polish Women. Diagnostics. 2021; 11(9):1698. https://doi.org/10.3390/diagnostics11091698
Chicago/Turabian StyleMagiełda-Stola, Justyna, Grażyna Kurzawińska, Marcin Ożarowski, Tomasz M. Karpiński, Krzysztof Drews, and Agnieszka Seremak-Mrozikiewicz. 2021. "The Significance of VDR Genetic Polymorphisms in the Etiology of Preeclampsia in Pregnant Polish Women" Diagnostics 11, no. 9: 1698. https://doi.org/10.3390/diagnostics11091698
APA StyleMagiełda-Stola, J., Kurzawińska, G., Ożarowski, M., Karpiński, T. M., Drews, K., & Seremak-Mrozikiewicz, A. (2021). The Significance of VDR Genetic Polymorphisms in the Etiology of Preeclampsia in Pregnant Polish Women. Diagnostics, 11(9), 1698. https://doi.org/10.3390/diagnostics11091698








