Performance of Seven SARS-CoV-2 Self-Tests Based on Saliva, Anterior Nasal and Nasopharyngeal Swabs Corrected for Infectiousness in Real-Life Conditions: A Cross-Sectional Test Accuracy Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patient Group and Sampling
2.2. Antigen Testing
2.3. qPCR and Virus Culture
2.4. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- ECDC. Options for the Use of Rapid Antigen Tests for COVID-19 in the EU/EEA and the UK; ECDC: Solna Municipality, Sweden, 2020.
- Brümmer, L.E.; Katzenschlager, S.; Gaeddert, M.; Erdmann, C.; Schmitz, S.; Bota, M.; Grilli, M.; Larmann, J.; Weigand, M.A.; Pollock, N.R.; et al. The accuracy of novel antigen rapid diagnostics for SARS-CoV-2: A living systematic review and meta-analysis. medRxiv 2021. [Google Scholar] [CrossRef]
- Dinnes, J.; Deeks, J.J.; Adriano, A.; Berhane, S.; Davenport, C.; Dittrich, S.; Emperador, D.; Takwoingi, Y.; Cunningham, J.; Beese, S.; et al. Rapid, point-of-care antigen and molecular-based tests for diagnosis of SARS-CoV-2 infection. Cochrane Database Syst. Rev. 2020, 8, CD013705. [Google Scholar] [CrossRef] [PubMed]
- Scheiblauer, H.; Filomena, A.; Nitsche, A.; Puyskens, A.; Corman, V.M.; Drosten, C.; Zwirglmaier, K.; Lange, C.; Emmerich, P.; Müller, M.; et al. Comparative sensitivity evaluation for 122 CE-marked SARS-CoV-2 antigen rapid tests. medRxiv 2021. [Google Scholar] [CrossRef]
- ECDC. ECDC Technical Report: Considerations Onthe Use of Self-Testsfor COVID-19 in the EU/EEA. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/Considerations-use-of-self-tests-for-COVID-19-in-the-EU-EEA-17-March2021-erratum.pdf (accessed on 6 June 2021).
- Bastos, M.L.; Perlman-Arrow, S.; Menzies, D.; Campbell, J.R. The sensitivity and costs of testing for SARS-CoV-2 infection with saliva versus nasopharyngeal swabs: A systematic review and meta-analysis. Ann. Intern. Med. 2021, 174, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Homza, M.; Zelena, H.; Janosek, J.; Tomaskova, H.; Jezo, E.; Kloudova, A.; Mrazek, J.; Svagera, Z.; Prymula, R. Five antigen tests for SARS-CoV-2: Virus viability matters. Viruses 2021, 13, 684. [Google Scholar] [CrossRef]
- Homza, M.; Zelena, H.; Janosek, J.; Tomaskova, H.; Jezo, E.; Kloudova, A.; Mrazek, J.; Svagera, Z.; Prymula, R. Covid-19 antigen testing: Better than we know? A test accuracy study. Infect. Dis. 2021, 53, 1–8. [Google Scholar] [CrossRef]
- WHO. Antigen-Detection in the Diagnosis of SARS-CoV-2 Infection Using Rapid Immunoassays: Interim Guidance, 11 September 2020; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Bullard, J.; Dust, K.; Funk, D.; Strong, J.E.; Alexander, D.; Garnett, L.; Boodman, C.; Bello, A.; Hedley, A.; Schiffman, Z.; et al. Predicting Infectious Severe Acute Respiratory Syndrome Coronavirus 2 From Diagnostic Samples. Clin. Infect. Dis. 2020, 71, 2663–2666. [Google Scholar] [CrossRef]
- La Scola, B.; Le Bideau, M.; Andreani, J.; Hoang, V.T.; Grimaldier, C.; Colson, P.; Gautret, P.; Raoult, D. Viral RNA load as determined by cell culture as a management tool for discharge of SARS-CoV-2 patients from infectious disease wards. Eur. J. Clin. Microbiol. Infect. Dis. Off. Publ. Eur. Soc. Clin. Microbiol. 2020, 39, 1059–1061. [Google Scholar] [CrossRef] [PubMed]
- Osmanodja, B.; Budde, K.; Zickler, D.; Naik, M.G.; Hofmann, J.; Gertler, M.; Hülso, C.; Rössig, H.; Horn, P.; Seybold, J.; et al. Accuracy of a novel SARS-CoV-2 antigen-detecting rapid diagnostic test from standardized self-collected anterior nasal swabs. J. Clin. Med. 2021, 10, 2099. [Google Scholar] [CrossRef] [PubMed]
- Nikolai, O.; Rohardt, C.; Tobian, F.; Junge, A.; Corman, V.M.; Jones, T.C.; Gaeddert, M.; Lainati, F.; Sacks, J.A.; Seybold, J.; et al. Anterior nasal versus nasal mid-turbinate sampling for a SARS-CoV-2 antigen-detecting rapid test: Does localisation or professional collection matter? medRxiv 2021. [Google Scholar] [CrossRef]
- Pollock, N.R.; Jacobs, J.R.; Tran, K.; Cranston, A.E.; Smith, S.; O’Kane, C.Y.; Roady, T.J.; Moran, A.; Scarry, A.; Carroll, M.; et al. Performance and implementation evaluation of the abbott binaxnow rapid antigen test in a high-throughput drive-through community testing site in massachusetts. J. Clin. Microbiol. 2021, 59, e00083-21. [Google Scholar] [CrossRef] [PubMed]
- Pilarowski, G.; Lebel, P.; Sunshine, S.; Liu, J.; Crawford, E.; Marquez, C.; Rubio, L.; Chamie, G.; Martinez, J.; Peng, J.; et al. Performance characteristics of a rapid severe acute respiratory syndrome coronavirus 2 antigen detection assay at a public plaza testing site in san francisco. J. Infect. Dis. 2021, 223, 1139–1144. [Google Scholar] [CrossRef] [PubMed]
- James, A.E.; Gulley, T.; Kothari, A.; Holder, K.; Garner, K.; Patil, N. Performance of the BinaxNOW coronavirus disease 2019 (COVID-19) antigen card test relative to the severe acute respiratory coronavirus virus 2 (SARS-CoV-2) real-time reverse transcriptase polymerase chain reaction (rRT-PCR) assay among symptomatic and asymptomatic healthcare employees. Infect. Control Hosp. Epidemiol. 2021, 1–3. [Google Scholar] [CrossRef]

| Relative to qPCR | Relative to qPCR Corrected for Viability | |
|---|---|---|
| True positives (TP) | PCR+/RAT+ | PCR+/RAT+; PCR− or viability+/RAT+ |
| True negatives (TN) | PCR−/RAT− | PCR−/AT− or PCR+/viability−/AT− |
| False positives (FP) | PCR−/RAT+ | PCR−/viability−/AT+ |
| False negatives (FN) | PCR+/RAT− | PCR+/viability+/AT− |
| Test | Sample Type | NPS | ANS 1 | ANS 2 | Saliva 1 | Saliva 2 | Saliva 3 | Saliva 4 |
|---|---|---|---|---|---|---|---|---|
| MDSe/Sp | >93/100 | >98/>99 | >95/>99 | >95/>98 | >92/>99 | >89/>96 | >98/>99 | |
| N of Patients | 480 | 488 | 406 | 217 | 191 | 407 | 98 | |
| Uncorrected for cell culture | Prevalence | 32.30% | 26.40% | 42.40% | 43.30% | 41.90% | 42.80% | 29.0% |
| (28.1–36.7%) | (22.60–30.60%) | (37.5–47.3%) | (36.6–50.2%) | (34.8–49.2%) | (37.9–47.7%) | (20.0–38.6%) | ||
| Sensitivity | 80.60% | 46.50% | 54.10% | 25.50% | 15.00% | 32.80% | 3.60% | |
| (73.5–86.5%) | (37.70–55.50%) | (46.3–61.7%) | (17.1–35.6%) | (8.0–24.7%) | (25.8–40.3%) | (0.10–18.3%) | ||
| Specificity | 98.50% | 99.40% | 97.40% | 98.40% | 89.20% | 89.30% | 97.10% | |
| (96.4–99.5%) | (98.00–99.90%) | (94.5–99.1%) | (94.2–99.8%) | (81.9–94.3%) | (84.6–92.9%) | (90.1–99.7%) | ||
| PPV | 96.20% | 96.80% | 93.90% | 92.30% | 50.00% | 69.50% | 33.30% | |
| (91.3–98.7%) | (88.80–99.60%) | (87.3–97.7%) | (74.9–99.1%) | (29.1–70.9%) | (58.4–79.2%) | (0.80–90.60%) | ||
| NPV | 91.4% | 83.80% | 74.30% | 63.40% | 59.30% | 64.00% | 71.60% | |
| (88.0–94.1%) | (80.00–87.20%) | (69.0–79.1%) | (56.1–70.2%) | (51.4–66.8%) | (58.5–69.2%) | (61.40–80.40%) | ||
| N of cell culture tested samples (viable virus present/absent) | 35 (11/24) | 69 (37/32) | 78 (28/40) | 72 (42/30) | 66 (39/27) | 141 (81/60) | 27 (16/11) | |
| Corrected for cell culture | Prevalence | 28.50% | 20.30% | 34.00% | 30.40% | 33.00% | 34.20% | 19.0% |
| (24.5–32.8%) | (16.80–24.10%) | (29.4–38.8%) | (24.4–37.0%) | (26.4–40.1%) | (29.6–39.0%) | (12.0–28.6%) | ||
| Sensitivity | 91.20% | 60.60% | 67.40% | 36.40% | 19.00% | 41.00% | 5.30% | |
| (85.2–95.4%) | (50.30–70.30%) | (58.9–75.1%) | (24.9–49.1%) | (10.2–30.9%) | (32.7–49.7%) | (0.1–26.0%) | ||
| Specificity | 98.50% | 99.50% | 97.80% | 98.70% | 90.60% | 90.70% | 97.50% | |
| (96.6–99.5%) | (98.20–99.90%) | (95.2–99.2%) | (95.3–99.8%) | (84.2–95.1%) | (86.5–93.9%) | (91.2–99.7%) | ||
| PPV | 96.20% | 96.80% | 93.90% | 92.30% | 50.00% | 69.50% | 33.30% | |
| (91.3–98.7%) | (88.80–99.60%) | (87.3–97.7%) | (74.9–99.1%) | (29.1–70.9%) | (58.4–79.2%) | (0.8–90.6%) | ||
| NPV | 96.60% | 90.80% | 85.30% | 78.00% | 69.50% | 74.80% | 81.1% | |
| (94.1–98.2%) | (87.70–93.40%) | (80.9–89.1%) | (71.5–83.7%) | (61.9–76.3%) | (69.7–79.4%) | (71.7–88.4%) | ||
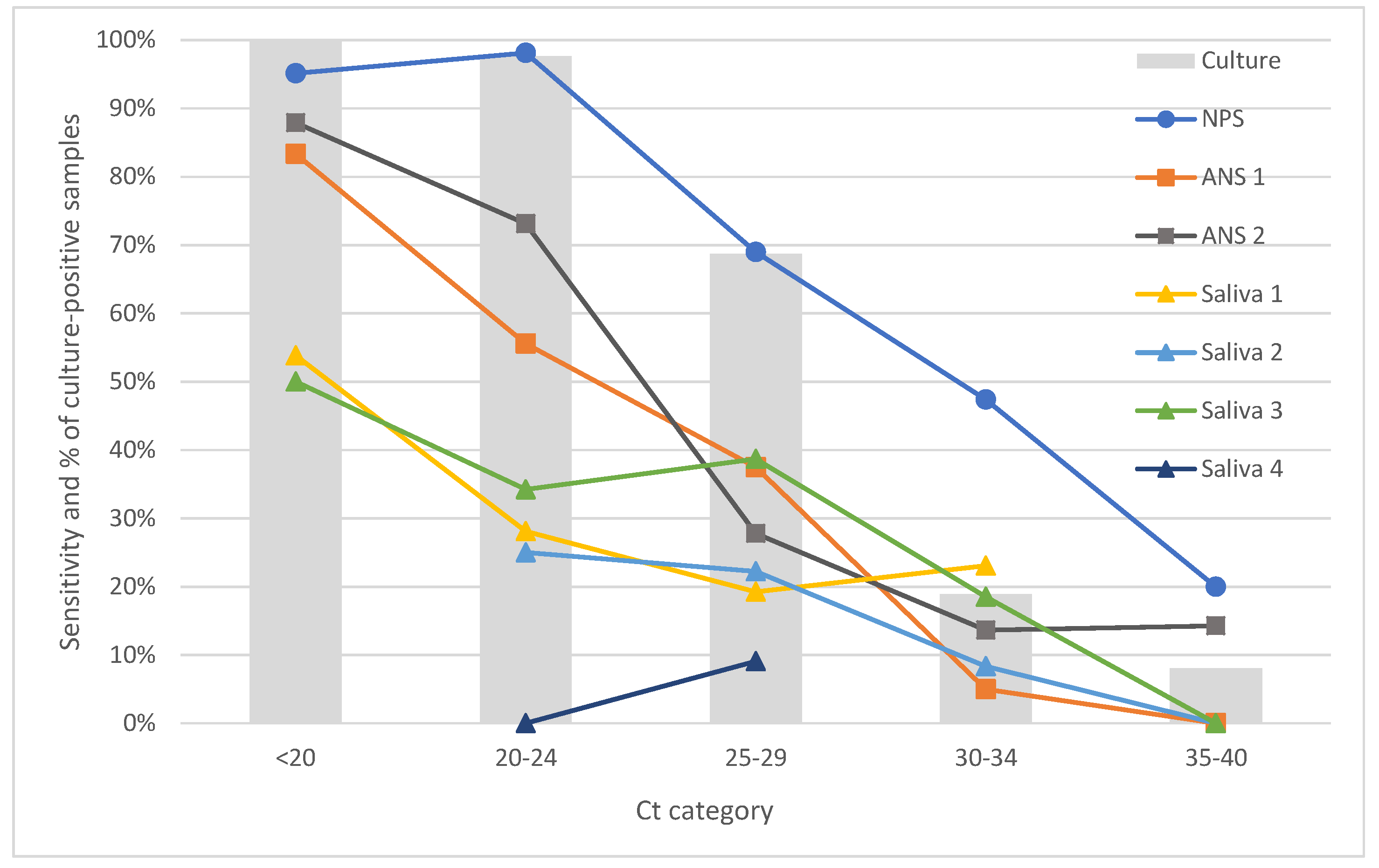
| 1. <20 | 2. 20–24 | 3. 25–29 | 4. 30–34 | 5. 35–40 | |
|---|---|---|---|---|---|
| NPS | 26.97 | 34.87 | 19.08 | 12.50 | 6.58 |
| ANS 1 | 18.60 | 41.86 | 18.60 | 15.50 | 5.43 |
| ANS 2 | 19.19 | 38.95 | 20.93 | 12.79 | 8.14 |
| Saliva 1 | 13.98 | 34.41 | 27.96 | 13.98 | 9.68 |
| Saliva 2 | 2.60 | 25.97 | 35.06 | 15.58 | 20.78 |
| Saliva 3 | 17.24 | 41.95 | 17.82 | 15.52 | 7.47 |
| Saliva 4 | 14.29 | 32.14 | 39.29 | 7.14 | 7.14 |
| Symptomatic | Asymptomatic | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Test (Sample Type) | N | Prevalence | Sensitivity | Specificity | PPV | NPV | N | Prevalence | Sensitivity | Specificity | PPV | NPV | |
| NPS | PCR | 237 | 49.0 (42.4–55.5) | 84.5 (76.6–90.5) | 99.2 (95.5–100) | 99.0 (94.5–100) | 87.0 (80.2–92.1) | 227 | 15.0 (10.6–20.3) | 64.7 (46.5–80.3) | 97.9 (94.8–99.4) | 84.6 (65.1–95.6) | 94 (89.8–96.9) |
| PCR+viab. | 44.3 (37.9–50.9) | 93.3 (86.7–97.3) | 99.2 (95.9–100) | 99.0 (94.5–100) | 94.9 (89.8–97.9) | 11.9 (8.0–16.8) | 81.5 (61.9–93.7) | 98.0 (95.0–99.5) | 84.6 (65.1–95.6) | 97.5 (94.3–99.2) | |||
| ANS 1 | PCR | 194 | 48.5 (41.2–55.7) | 52.1 (41.6–62.5) | 99.0 (94.6–100) | 98.0 (89.4–100) | 68.8 (60.5–76.2) | 288 | 11.5 (8–15.7) | 30.3 (15.6–48.7) | 99.6 (97.8–100) | 90.9 (58.7–99.8) | 91.7 (87.8–94.7) |
| PCR+viab. | 37.6 (30.8–44.9) | 67.1 (55.1–77.7) | 99.2 (95.5–100) | 98.0 (89.4–99.9) | 83.3 (76.2–89.0) | 8.3 (5.4–12.1) | 41.7 (22.1–63.4) | 99.6 (97.9–1) | 90.9 (58.7–99.8) | 94.9 (91.7–97.2) | |||
| ANS 2 | PCR | 243 | 60.5 (54–66.7) | 56.5 (48.1–64.6) | 96.9 (91.1–99.4) | 96.5 (90.1–99.3) | 59.2 (51.1–67) | 158 | 14.6 (9.5–21.0) | 43.5 (23.2–65.5) | 97.8 (93.6–99.5) | 76.9 (46.2–95) | 91 (85.2–95.1) |
| PCR+viab. | 50.6 (44.2–57.1) | 67.5 (58.4–75.6) | 97.5 (92.9–99.5) | 96.5 (90.1–99.3) | 74.5 (67.0–81.1) | 9.5 (5.4–15.2) | 66.7 (38.4–88.2) | 97.9 (94.0–99.6) | 76.9 (46.2–95.0) | 96.6 (92.1–98.9) | |||
| Saliva 1 | PCR | 127 | 55.1 (46.0–64.0) | 27.1 (17.2–39.1) | 96.5 (87.9–99.6) | 90.5 (69.6–98.8) | 51.9 (42–61.7) | 86 | 25.0 (16.2–35.6) | 23.8 (8.2–47.2) | 100 (94.3–100) | 100 (47.8–100) | 79.8 (69.2–88) |
| PCR+viab. | 38.6 (30.1–47.6) | 38.8 (25.2–53.8) | 97.4 (91.0–99.7) | 90.5 (69.6–98.8) | 71.7 (62.1–80.0) | 16.7 (9.4–26.4) | 35.7 (12.8–64.9) | 100 (94.9–100) | 100 (47.8–100) | 88.6 (79.5–94.7) | |||
| Saliva 2 | PCR | 116 | 60.3 (50.8–69.3) | 15.7 (8.1–26.4) | 84.8 (71.1–93.7) | 61.1 (35.8–82.7) | 39.8 (30–50.2) | 75 | 13.3 (6.6–23.2) | 10.0 (0.3–44.5) | 92.3 (83–97.5) | 16.7 (0.4–64.1) | 87 (76.7–93.9) |
| PCR+viab. | 49.1 (39.7–58.6) | 19.3 (10.0–31.9) | 88.1 (77.1–95.1) | 61.1 (35.7–82.7) | 53.1 (42.7–63.2) | 8.0 (3.0–16.6) | 16.7 (0.4–64.1) | 92.8 (83.9–97.6) | 16.7 (0.4–64.1) | 92.8 (83.9–97.6) | |||
| Saliva 3 | PCR | 219 | 60.7 (53.9–67.2) | 36.1 (28–44.9) | 93.0 (85.4–97.4) | 88.9 (77.4–95.8) | 48.5 (40.6–56.4) | 182 | 20.3 (14.7–26.9) | 21.6 (9.8–38.2) | 87.6 (81.1–92.5) | 30.8 (14.3–51.8) | 81.4 (74.4–87.2) |
| PCR+viab. | 50.7 (43.9–57.5) | 43.2 (33.9–53.0) | 94.4 (88.3–97.9) | 88.9 (77.4–95.8) | 61.8 (53.9–69.3) | 13.2 (8.6–19.0) | 33.3 (15.6–55.3) | 88.6 (82.6–93.1) | 30.8 (14.3–51.8) | 89.7 (83.9–94.0) | |||
| Saliva 4 | PCR | 37 | 46.0 (29.5–63.1) | 5.9 (0.2–28.7) | 100 (83.2–100) | 100 (2.5–100) | 55.6 (38.1–72.1) | 57 | 17.5 (8.8–29.9) | 0 (0–30.9) | 95.7 (85.5–99.5) | 0 (0–84.2) | 81.8 (69.1–90.9) |
| PCR+viab. | 32.0 (18.0–49.8) | 8.3 (0.2–38.5) | 100 (86.3–100) | 100 (2.5–100) | 69.4 (51.9–83.7) | 11.0 (4.0–21.5) | 0.0 (0.0–45.9) | 96.1 (86.5–99.5) | 0.0 (0.0–84.2) | 89.1 (77.8–95.9) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Homza, M.; Zelena, H.; Janosek, J.; Tomaskova, H.; Jezo, E.; Kloudova, A.; Mrazek, J.; Murinova, V.; Madar, R. Performance of Seven SARS-CoV-2 Self-Tests Based on Saliva, Anterior Nasal and Nasopharyngeal Swabs Corrected for Infectiousness in Real-Life Conditions: A Cross-Sectional Test Accuracy Study. Diagnostics 2021, 11, 1567. https://doi.org/10.3390/diagnostics11091567
Homza M, Zelena H, Janosek J, Tomaskova H, Jezo E, Kloudova A, Mrazek J, Murinova V, Madar R. Performance of Seven SARS-CoV-2 Self-Tests Based on Saliva, Anterior Nasal and Nasopharyngeal Swabs Corrected for Infectiousness in Real-Life Conditions: A Cross-Sectional Test Accuracy Study. Diagnostics. 2021; 11(9):1567. https://doi.org/10.3390/diagnostics11091567
Chicago/Turabian StyleHomza, Miroslav, Hana Zelena, Jaroslav Janosek, Hana Tomaskova, Eduard Jezo, Alena Kloudova, Jakub Mrazek, Vera Murinova, and Rastislav Madar. 2021. "Performance of Seven SARS-CoV-2 Self-Tests Based on Saliva, Anterior Nasal and Nasopharyngeal Swabs Corrected for Infectiousness in Real-Life Conditions: A Cross-Sectional Test Accuracy Study" Diagnostics 11, no. 9: 1567. https://doi.org/10.3390/diagnostics11091567
APA StyleHomza, M., Zelena, H., Janosek, J., Tomaskova, H., Jezo, E., Kloudova, A., Mrazek, J., Murinova, V., & Madar, R. (2021). Performance of Seven SARS-CoV-2 Self-Tests Based on Saliva, Anterior Nasal and Nasopharyngeal Swabs Corrected for Infectiousness in Real-Life Conditions: A Cross-Sectional Test Accuracy Study. Diagnostics, 11(9), 1567. https://doi.org/10.3390/diagnostics11091567







