Lung Cancer Screening with Low-Dose CT in Smokers: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
3. Results
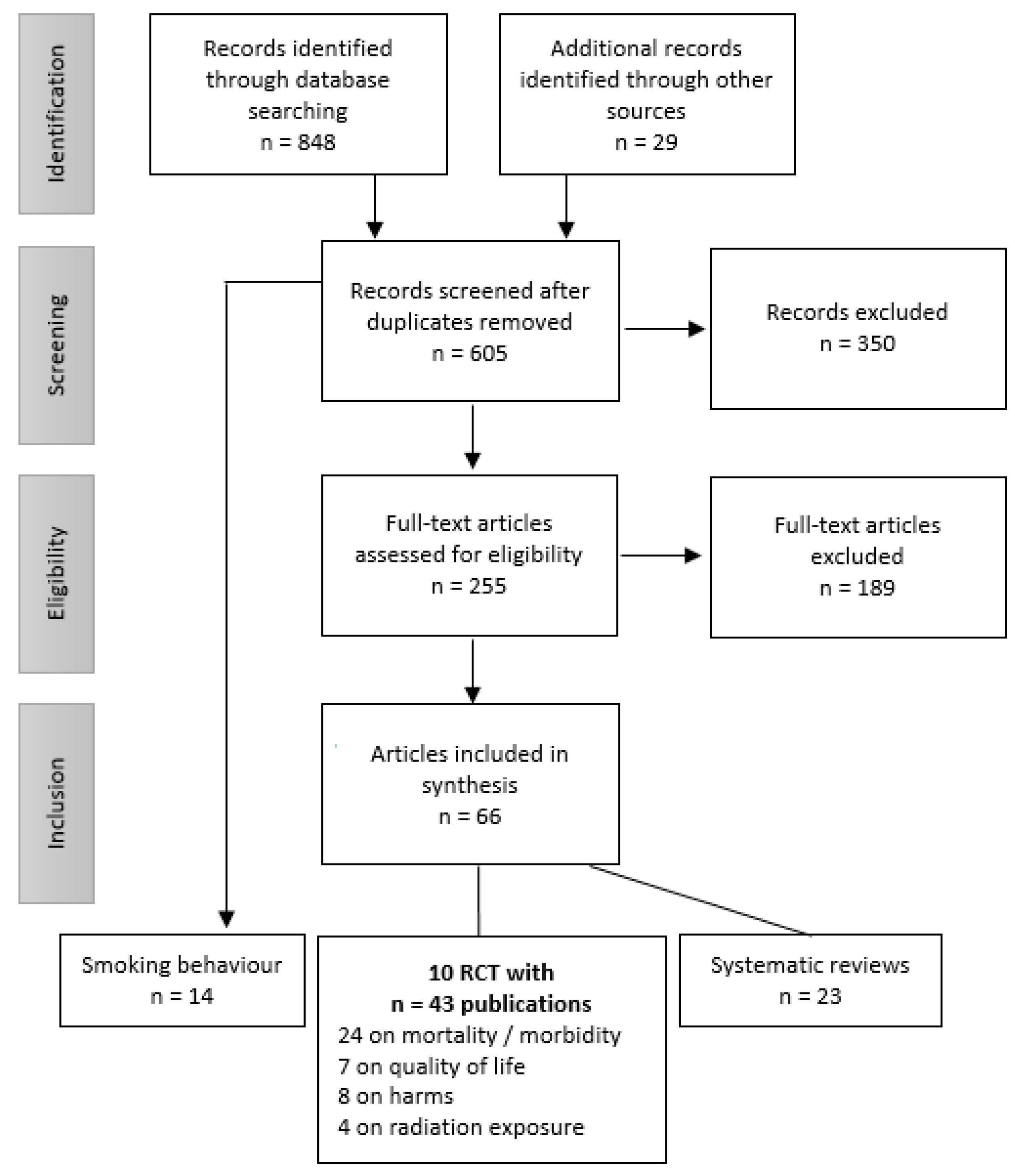
3.1. Search Results
3.2. Study Characteristics
3.3. CT Scanning and Diagnostic Evaluation Algorithms Used in the Considered RCTs
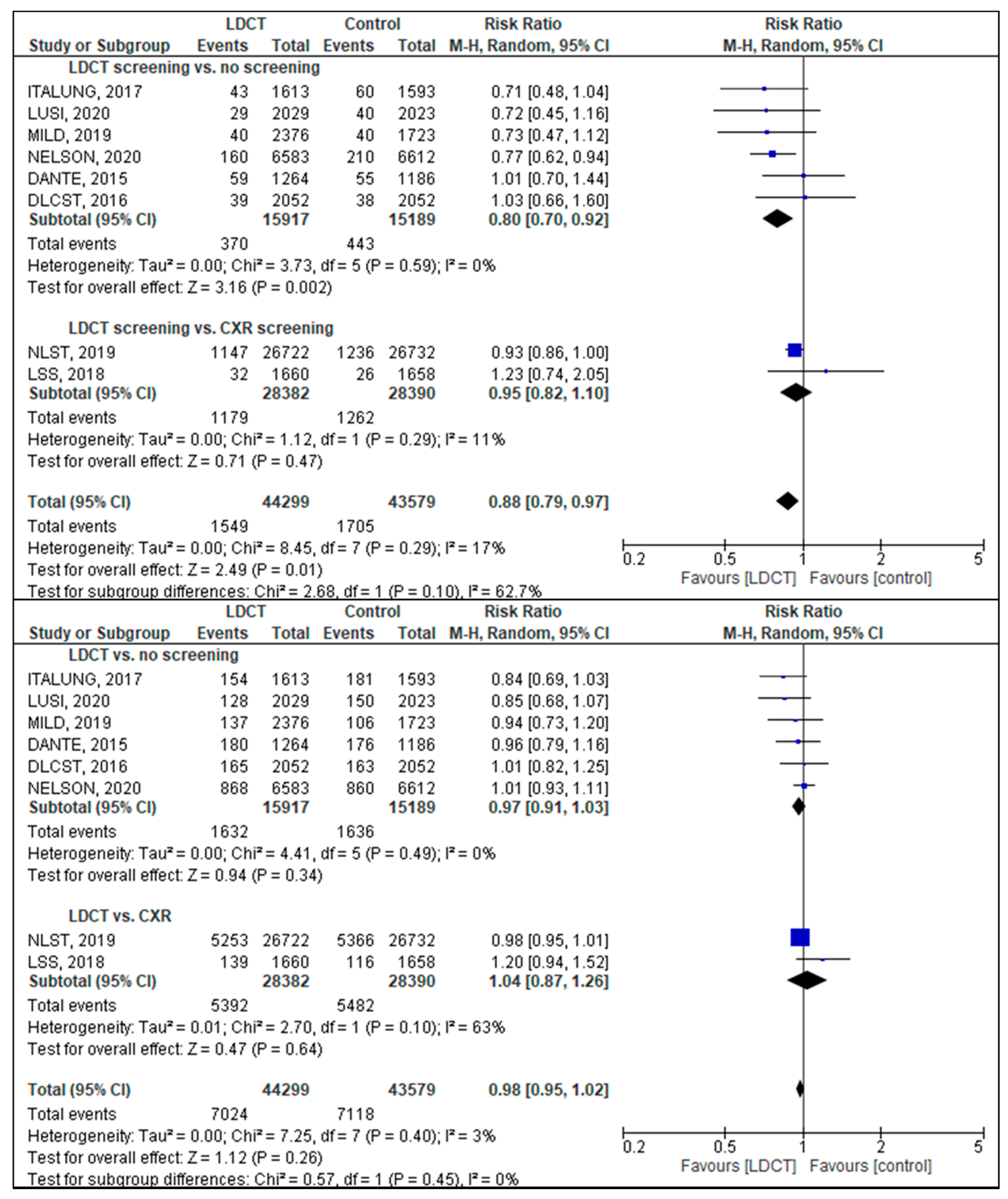
3.4. Lung Cancer and Overall Mortality
3.5. Lung Cancer Incidence
3.6. Consequences of Screening
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ballard, D.H.; Burton, K.R.; Lakomkin, N.; Kim, S.; Rajiah, P.; Patel, M.J.; Mazaheri, P.; Whitman, G.J. The Role of Imaging in Health Screening: Overview, Rationale of Screening, and Screening Economics. Acad. Radiol. 2021, 28, 540–547. [Google Scholar] [CrossRef]
- Lam, D.L.; Pandharipande, P.V.; Lee, J.M.; Lehman, C.D.; Lee, C.I. Imaging-Based Screening: Understanding the Controversies. AJR Am. J. Roentgenol. 2014, 203, 952–956. [Google Scholar] [CrossRef]
- International Atomic Energy Agency. Radiation Protection and Safety of Radiation Sources: International Basic Safety Standards, IAEA Safety Standards Series No. Gsr Part 3; International Atomic Energy Agency: Vienna, Austria, 2014. [Google Scholar]
- European Union. Council Directive 2013/59/Euratom of 5 December 2013 Laying Down Basic Safety Standards for Protection against the Dangers Arising from Exposure to Ionising Radiation, and Repealing Directives 89/618/Euratom, 90/641/Euratom, 96/29/Euratom, 97/43/Euratom and 2003/122/Euratom. Off. J. Eur. Union 2014, 17, L 13/1–L 13/73. [Google Scholar]
- International Agency for Research on Cancer. Cancer Screening in the European Union. Report on the Implementation of the Council Recommendation on Cancer Screening; International Agency for Research on Cancer: Lyon, France, 2017. [Google Scholar]
- Robert Koch Institut. Krebs in Deutschland 2015/2016; Robert Koch Institut: Berlin, Germany, 2019.
- Allemani, C.; Matsuda, T.; Di Carlo, V.; Harewood, R.; Matz, M.; Nikšić, M.; Bonaventure, A.; Valkov, M.; Johnson, C.J.; Estève, J.; et al. Global surveillance of trends in cancer survival 2000–14 (CONCORD-3): Analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet 2018, 391, 1023–1075. [Google Scholar] [CrossRef]
- Canadian Task Force on Preventive Health Care. Recommendations on Screening for Lung Cancer. CMAJ 2016, 188, 425–432. [Google Scholar] [CrossRef]
- Moyer, V.A. on behalf of the U.S. Preventive Services Task Force. Screening for Lung Cancer: U.S. Preventive Services Task Force Recommendation Statement. Ann. Intern. Med. 2014, 160, 330–338. [Google Scholar] [CrossRef]
- Oudkerk, M.; Devaraj, A.; Vliegenthart, R.; Henzler, T.; Prosch, H.; Heussel, C.P.; Bastarrika, G.; Sverzellati, N.; Mascalchi, M.; Delorme, S.; et al. European position statement on lung cancer screening. Lancet Oncol. 2017, 18, e754–e766. [Google Scholar] [CrossRef]
- Kauczor, H.-U.; Bonomo, L.; Gaga, M.; Nackaerts, K.; Peled, N.; Prokop, M.; Remy-Jardin, M.; von Stackelberg, O.; Sculier, J.-P. On behalf of the European Society of Radiology (ESR) and the European Respiratory Society (ERS). ESR/ERS White Paper on Lung Cancer Screening. Eur. Respir. J. 2015, 46, 28–39. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. For the PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Patz Jr, E.F.; Pinsky, P.; Gatsonis, C.; Sicks, J.D.; Kramer, B.S.; Tammemägi, M.C.; Chiles, C.; Black, W.C.; Aberle, D.R.; NLST Overdiagnosis Manuscript Writing Team. Overdiagnosis in Low-Dose Computed Tomography Screening for Lung Cancer. JAMA Intern. Med. 2014, 174, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Blanchon, T.; Bréchot, J.-M.; Grenier, P.A.; Ferretti, G.R.; Lemarié, E.; Milleron, B.; Chagué, D.; Laurent, F.; Martinet, Y.; Beigelman-Aubry, C.; et al. Baseline results of the Depiscan study: A French randomized pilot trial of lung cancer screening comparing low dose CT scan (LDCT) and chest X-ray (CXR). Lung Cancer 2007, 58, 50–58. [Google Scholar] [CrossRef]
- de Koning, H.J.; van der Aalst, C.M.; de Jong, P.A.; Scholten, E.T.; Nackaerts, K.; Heuvelmans, M.A.; Lammers, J.-W.J.; Weenink, C.; Yousaf-Khan, U.; Horeweg, N.; et al. Reduced Lung-Cancer Mortality with Volume CT Screening in a Randomized Trial. N. Engl. J. Med. 2020, 382, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Doroudi, M.; Pinsky, P.F.; Marcus, P.M. Lung Cancer Mortality in the Lung Screening Study Feasibility Trial. JNCI Cancer Spectr. 2018, 2, pky042. [Google Scholar] [CrossRef]
- Infante, M.; Cavuto, S.; Lutman, F.R.; Passera, E.; Chiarenza, M.; Chiesa, G.; Brambilla, G.; Angeli, E.; Aranzulla, G.; Chiti, A.; et al. Long-Term Follow-up Results of the DANTE Trial, a Randomized Study of Lung Cancer Screening with Spiral Computed Tomography. Am. J. Respir. Crit. Care Med. 2015, 191, 1166–1175. [Google Scholar] [CrossRef]
- National Lung Screening Trial Research Team. Lung Cancer Incidence and Mortality with Extended Follow-up in the National Lung Screening Trial. J. Thorac. Oncol. 2019, 14, 1732–1742. [Google Scholar] [CrossRef]
- Paci, E.; Puliti, D.; Lopes Pegna, A.; Carrozzi, L.; Picozzi, G.; Falaschi, F.; Pistelli, F.; Aquilini, F.; Ocello, C.; Zappa, M.; et al. Mortality, survival and incidence rates in the ITALUNG randomised lung cancer screening trial. Thorax 2017, 72, 825–831. [Google Scholar] [CrossRef]
- Pastorino, U.; Silva, M.; Sestini, S.; Sabia, F.; Boeri, M.; Cantarutti, A.; Sverzellati, N.; Sozzi, G.; Corrao, G.; Marchianò, A. Prolonged lung cancer screening reduced 10-year mortality in the MILD trial: New confirmation of lung cancer screening efficacy. Ann. Oncol. 2019, 30, 1162–1169. [Google Scholar] [CrossRef]
- Wille, M.M.W.; Dirksen, A.; Ashraf, H.; Saghir, Z.; Bach, K.S.; Brodersen, J.; Clementsen, P.F.; Hansen, H.; Larsen, K.R.; Mortensen, J.; et al. Results of the Randomized Danish Lung Cancer Screening Trial with Focus on High-Risk Profiling. Am. J. Respir. Crit. Care Med. 2016, 193, 542–551. [Google Scholar] [CrossRef]
- Becker, N.; Motsch, E.; Trotter, A.; Heussel, C.P.; Dienemann, H.; Schnabel, P.A.; Kauczor, H.-U.; Maldonado, S.G.; Miller, A.B.; Kaaks, R.; et al. Lung cancer mortality reduction by LDCT screening—Results from the randomized German LUSI trial. Int. J. Cancer 2020, 146, 1503–1513. [Google Scholar] [CrossRef]
- Field, J.K.; Duffy, S.W.; Baldwin, D.R.; Brain, K.E.; Devaraj, A.; Eisen, T.; Green, B.A.; Holemans, J.A.; Kavanagh, T.; Kerr, K.M.; et al. The UK Lung Cancer Screening Trial: A pilot randomised controlled trial of low-dose computed tomography screening for the early detection of lung cancer. Health Technol. Assess. 2016, 20, 1–146. [Google Scholar] [CrossRef]
- Aggestrup, L.M.; Hestbech, M.S.; Siersma, V.; Pedersen, J.H.; Brodersen, J. Psychosocial Consequences of Allocation to Lung Cancer Screening: A Randomised Controlled Trial. BMJ Open 2012, 2, e000663. [Google Scholar] [CrossRef]
- Becker, N.; Motsch, E.; Gross, M.-L.; Eigentopf, A.; Heussel, C.P.; Dienemann, H.; Schnabel, P.A.; Eichinger, M.; Optazaite, D.-E.; Puderbach, M.; et al. Randomized Study on Early Detection of Lung Cancer with MSCT in Germany: Results of the First 3 Years of Follow-up After Randomization. J. Thorac. Oncol. 2015, 10, 890–896. [Google Scholar] [CrossRef][Green Version]
- Becker, N.; Motsch, E.; Gross, M.-L.; Eigentopf, A.; Heussel, C.P.; Dienemann, H.; Schnabel, P.A.; Pilz, L.; Eichinger, M.; Optazaite, D.-E.; et al. Randomized study on early detection of lung cancer with MSCT in Germany: Study design and results of the first screening round. J. Cancer Res. Clin. Oncol. 2012, 138, 1475–1486. [Google Scholar] [CrossRef]
- Brain, K.; Lifford, K.J.; Carter, B.; Burke, O.; McRonald, F.; Devaraj, A.; Hansell, D.M.; Baldwin, D.; Duffy, S.W.; Field, J.K. Long-Term Psychosocial Outcomes of Low-Dose CT Screening: Results of the UK Lung Cancer Screening Randomised Controlled Trial. Thorax 2016, 71, 996–1005. [Google Scholar] [CrossRef]
- Gareen, I.F.; Duan, F.; Greco, E.M.; Snyder, B.S.; Boiselle, P.M.; Park, E.R.; Fryback, D.; Gatsonis, C. Impact of lung cancer screening results on participant health-related quality of life and state anxiety in the National Lung Screening Trial. Cancer 2014, 120, 3401–3409. [Google Scholar] [CrossRef]
- Gohagan, J.K.; Marcus, P.M.; Fagerstrom, R.M.; Pinsky, P.F.; Kramer, B.S.; Prorok, P.C.; Ascher, S.; Bailey, W.; Brewer, B.; Church, T.; et al. Final results of the Lung Screening Study, a randomized feasibility study of spiral CT versus chest X-ray screening for lung cancer. Lung Cancer 2005, 47, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Horeweg, N.; Scholten, E.T.; de Jong, P.A.; van der Aalst, C.M.; Weenink, C.; Lammers, J.-W.J.; Nackaerts, K.; Vliegenthart, R.; ten Haaf, K.; Yousaf-Khan, U.A.; et al. Detection of lung cancer through low-dose CT screening (NELSON): A prespecified analysis of screening test performance and interval cancers. Lancet Oncol. 2014, 15, 1342–1350. [Google Scholar] [CrossRef]
- Horeweg, N.; van der Aalst, C.M.; Vliegenthart, R.; Zhao, Y.; Xie, X.; Scholten, E.T.; Mali, W.; Thunnissen, E.; Weenink, C.; Groen, H.J.M.; et al. Volumetric computed tomography screening for lung cancer: Three rounds of the NELSON trial. Eur. Respir. J. 2013, 42, 1659–1667. [Google Scholar] [CrossRef]
- Infante, M.; Chiesa, G.; Solomon, D.; Morenghi, E.; Passera, E.; Lutman, F.R.; Bottoni, E.; Cariboni, U.; Errico, V.; Voulaz, E.; et al. Surgical Procedures in the DANTE Trial, a Randomized Study of Lung Cancer Early Detection with Spiral Computed Tomography: Comparative Analysis in the Screening and Control Arm. J. Thorac. Oncol. 2011, 6, 327–335. [Google Scholar] [CrossRef]
- Kaerlev, L.; Iachina, M.; Pedersen, J.H.; Green, A.; Norgard, B.M. CT-Screening for Lung Cancer Does Not Increase the Use of Anxiolytic or Antidepressant Medication. BMC Cancer 2012, 12, 188. [Google Scholar] [CrossRef] [PubMed]
- Larke, F.J.; Kruger, R.L.; Cagnon, C.H.; Flynn, M.J.; McNitt-Gray, M.M.; Wu, X.; Judy, P.F.; Cody, D.D. Estimated Radiation Dose Associated with Low-Dose Chest CT of Average-Size Participants in the National Lung Screening Trial. AJR Am. J. Roentgenol. 2011, 197, 1165–1169. [Google Scholar] [CrossRef]
- Lee, C.; Flynn, M.J.; Judy, P.F.; Cody, D.D.; Bolch, W.E.; Kruger, R.L. Body Size-Specific Organ and Effective Doses of Chest CT Screening Examinations of the National Lung Screening Trial. AJR Am. J. Roentgenol. 2017, 208, 1082–1088. [Google Scholar] [CrossRef] [PubMed]
- Lopes Pegna, A.; Picozzi, G.; Falaschi, F.; Carrozzi, L.; Falchini, M.; Carozzi, F.M.; Pistelli, F.; Comin, C.; Deliperi, A.; Grazzini, M.; et al. Four-Year Results of Low-Dose CT Screening and Nodule Management in the ITALUNG Trial. J. Thorac. Oncol. 2013, 8, 866–875. [Google Scholar] [CrossRef]
- Lopes Pegna, A.; Picozzi, G.; Mascalchi, M.; Carozzi, F.M.; Carrozzi, L.; Comin, C.; Spinelli, C.; Falaschi, F.; Grazzini, M.; Innocenti, F.; et al. Design, Recruitment and Baseline Results of the ITALUNG Trial for Lung Cancer Screening with Low-Dose CT. Lung Cancer 2009, 64, 34–40. [Google Scholar] [CrossRef]
- Mascalchi, M.; Belli, G.; Zappa, M.; Picozzi, G.; Falchini, M.; Della Nave, R.; Allescia, G.; Masi, A.; Lopes Pegna, A.; Villari, N.; et al. Risk-Benefit Analysis of X-Ray Exposure Associated with Lung Cancer Screening in the Italung-CT Trial. AJR Am. J. Roentgenol. 2006, 187, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Mascalchi, M.; Mazzoni, L.N.; Falchini, M.; Belli, G.; Picozzi, G.; Merlini, V.; Vella, A.; Diciotti, S.; Falaschi, F.; Lopes Pegna, A.; et al. Dose exposure in the ITALUNG trial of lung cancer screening with low-dose CT. Br. J. Radiol. 2012, 85, 1134–1139. [Google Scholar] [CrossRef] [PubMed]
- National Lung Screening Trial Research Team; Aberle, D.R.; Adams, A.M.; Berg, C.D.; Black, W.C.; Clapp, J.D.; Fagerstrom, R.M.; Gareen, I.F.; Gatsonis, C.; Marcus, P.M.; et al. Reduced Lung-Cancer Mortality with Low-Dose Computed Tomographic Screening. N. Engl. J. Med. 2011, 365, 395–409. [Google Scholar] [CrossRef] [PubMed]
- National Lung Screening Trial Research Team; Church, T.R.; Black, W.C.; Aberle, D.R.; Berg, C.D.; Clingan, K.L.; Duan, F.; Fagerstrom, R.M.; Gareen, I.F.; Gierada, D.S.; et al. Results of Initial Low-Dose Computed Tomographic Screening for Lung Cancer. N. Engl. J. Med. 2013, 368, 1980–1991. [Google Scholar] [CrossRef]
- Pastorino, U.; Rossi, M.; Rosato, V.; Marchiano, A.; Sverzellati, N.; Morosi, C.; Fabbri, A.; Galeone, C.; Negri, E.; Sozzi, G.; et al. Annual or Biennial CT Screening Versus Observation in Heavy Smokers: 5-Year Results of the Mild Trial. Eur. J. Cancer Prev. 2012, 21, 308–315. [Google Scholar] [CrossRef]
- Pastorino, U.; Sverzellati, N.; Sestini, S.; Silva, M.; Sabia, F.; Boeri, M.; Cantarutti, A.; Sozzi, G.; Corrao, G.; Marchianò, A.; et al. Ten-Year Results of the Multicentric Italian Lung Detection Trial Demonstrate the Safety and Efficacy of Biennial Lung Cancer Screening. Eur. J. Cancer 2019, 118, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Pinsky, P.F.; Church, T.R.; Izmirlian, G.; Kramer, B.S. The National Lung Screening Trial: Results Stratified by Demographics, Smoking History, and Lung Cancer Histology. Cancer 2013, 119, 3976–3983. [Google Scholar] [CrossRef]
- Pinsky, P.F.; Marcus, P.M.; Kramer, B.S.; Freedman, M.; Nath, H.; Kvale, P.; Reding, D. Diagnostic Procedures after a Positive Spiral Computed Tomography Lung Carcinoma Screen. Cancer 2005, 103, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Pistelli, F.; Aquilini, F.; Falaschi, F.; Puliti, D.; Ocello, C.; Lopes Pegna, A.; Carozzi, F.M.; Picozzi, G.; Zappa, M.; Mascalchi, M.; et al. Smoking Cessation in the Italung Lung Cancer Screening: What Does “Teachable Moment” Mean? Nicotine Tob. Res. 2020, 22, 1484–1491. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, J.F.; Siersma, V.; Pedersen, J.H.; Brodersen, J. Psychosocial consequences in the Danish randomised controlled lung cancer screening trial (DLCST). Lung Cancer 2015, 87, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Saghir, Z.; Dirksen, A.; Ashraf, H.; Bach, K.S.; Brodersen, J.; Clementsen, P.F.; Døssing, M.; Hansen, H.; Kofoed, K.F.; Larsen, K.R.; et al. CT screening for lung cancer brings forward early disease. The randomised Danish Lung Cancer Screening Trial: Status after five annual screening rounds with low-dose CT. Thorax 2012, 67, 296–301. [Google Scholar] [CrossRef]
- Sverzellati, N.; Silva, M.; Calareso, G.; Galeone, C.; Marchianò, A.; Sestini, S.; Sozzi, G.; Pastorino, U. Low-dose computed tomography for lung cancer screening: Comparison of performance between annual and biennial screen. Eur. Radiol. 2016, 26, 3821–3829. [Google Scholar] [CrossRef]
- van den Bergh, K.A.M.; Essink-Bot, M.L.; Borsboom, G.J.J.M.; Scholten, E.T.; van Klaveren, R.J.; De Koning, H.J. Long-term effects of lung cancer computed tomography screening on health-related quality of life: The NELSON trial. Eur. Respir. J. 2011, 38, 154–161. [Google Scholar] [CrossRef]
- van den Bergh, K.A.M.; Essink-Bot, M.-L.; Borsboom, G.J.J.M.; Scholten, E.T.; Prokop, M.; de Koning, H.J.; van Klaveren, R.J. Short-term health-related quality of life consequences in a lung cancer CT screening trial (NELSON). Br. J. Cancer 2010, 102, 27–34. [Google Scholar] [CrossRef]
- van den Bergh, K.A.M.; Essink-Bot, M.-L.; Bunge, E.M.; Scholten, E.T.; Prokop, M.; van Iersel, C.A.; van Klaveren, R.J.; de Koning, H.J. Impact of computed tomography screening for lung cancer on participants in a randomized controlled trial (NELSON trial). Cancer 2008, 113, 396–404. [Google Scholar] [CrossRef]
- van Klaveren, R.J.; Oudkerk, M.; Prokop, M.; Scholten, E.T.; Nackaerts, K.; Vernhout, R.; van Iersel, C.A.; van den Bergh, K.A.M.; van’t Westeinde, S.; van der Aalst, C.; et al. Management of Lung Nodules Detected by Volume CT Scanning. N. Engl. J. Med. 2009, 361, 2221–2229. [Google Scholar] [CrossRef]
- van’t Westeinde, S.C.; Horeweg, N.; De Leyn, P.; Groen, H.J.M.; Lammers, J.-W.J.; Weenink, C.; Nackaerts, K.; van Klaveren, R.J. Complications following lung surgery in the Dutch-Belgian randomized lung cancer screening trial. Eur. J. Cardiothorac Surg. 2012, 42, 420–429. [Google Scholar] [CrossRef]
- Yousaf-Khan, U.; van der Aalst, C.; de Jong, P.A.; Heuvelmans, M.; Scholten, E.; Lammers, J.-W.; van Ooijen, P.; Nackaerts, K.; Weenink, C.; Groen, H.; et al. Final Screening Round of the Nelson Lung Cancer Screening Trial: The Effect of a 2.5-Year Screening Interval. Thorax 2017, 72, 48–56. [Google Scholar] [CrossRef]
- Ashraf, H.; Saghir, Z.; Dirksen, A.; Pedersen, J.H.; Thomsen, L.H.; Dossing, M.; Tonnesen, P. Smoking Habits in the Ran-domised Danish Lung Cancer Screening Trial with Low-Dose CT: Final Results after a 5-Year Screening Programme. Thorax 2014, 69, 574–579. [Google Scholar] [CrossRef]
- Ashraf, H.; Tonnesen, P.; Pedersen, J.H.; Dirksen, A.; Thorsen, H.; Dossing, M. Effect of CT screening on smoking habits at 1-year follow-up in the Danish Lung Cancer Screening Trial (DLCST). Thorax 2009, 64, 388–392. [Google Scholar] [CrossRef]
- Bade, M.; Bähr, V.; Brandt, U.; Eigentopf, A.; Brüchert, T.; Gross, M.-L.; Motsch, E.; Becker, N. Effect of smoking cessation counseling within a randomised study on early detection of lung cancer in Germany. J. Cancer Res. Clin. Oncol 2016, 142, 959–968. [Google Scholar] [CrossRef]
- Brain, K.; Carter, B.; Lifford, K.J.; Burke, O.; Devaraj, A.; Baldwin, D.R.; Duffy, S.; Field, J.K. Impact of Low-Dose CT Screening on Smoking Cessation among High-Risk Participants in the UK Lung Cancer Screening Trial. Thorax 2017, 72, 912–918. [Google Scholar] [CrossRef]
- Pozzi, P.; Munarini, E.; Bravi, F.; Rossi, M.; La Vecchia, C.; Boffi, R.; Pastorino, U. A Combined Smoking Cessation Intervention within a Lung Cancer Screening Trial: A Pilot Observational Study. Tumori 2015, 101, 306–311. [Google Scholar] [CrossRef] [PubMed]
- van der Aalst, C.M.; van den Bergh, K.A.M.; Willemsen, M.C.; de Koning, H.J.; van Klaveren, R.J. Lung Cancer Screening and Smoking Abstinence: 2 Year Follow-up Data from the Dutch-Belgian Randomised Controlled Lung Cancer Screening Trial. Thorax 2010, 65, 600–605. [Google Scholar] [CrossRef]
- Park, E.R.; Gareen, I.F.; Japuntich, S.; Lennes, I.; Hyland, K.; DeMello, S.; Sicks, J.D.; Rigotti, N.A. Primary Care Provider-Delivered Smoking Cessation Interventions and Smoking Cessation among Participants in the National Lung Screening Trial. JAMA Intern. Med. 2015, 175, 1509–1516. [Google Scholar] [CrossRef]
- van der Aalst, C.M.; de Koning, H.J.; van den Bergh, K.A.M.; Willemsen, M.C.; van Klaveren, R.J. The Effectiveness of a Com-puter-Tailored Smoking Cessation Intervention for Participants in Lung Cancer Screening: A Randomised Controlled Trial. Lung Cancer 2012, 76, 204–210. [Google Scholar] [CrossRef]
- van der Aalst, C.M.; van Klaveren, R.J.; van den Bergh, K.A.M.; Willemsen, M.C.; de Koning, H.J. The impact of a lung cancer computed tomography screening result on smoking abstinence. Eur. Respir. J. 2011, 37, 1466–1473. [Google Scholar] [CrossRef]
- Clark, M.A.; Gorelick, J.J.; Sicks, J.D.; Park, E.R.; Graham, A.L.; Abrams, D.B.; Gareen, I.F. The Relations between False Positive and Negative Screens and Smoking Cessation and Relapse in the National Lung Screening Trial: Implications for Public Health. Nicotine Tob. Res. 2016, 18, 17–24. [Google Scholar] [CrossRef]
- Pistelli, F.; Aquilini, F.; Tavanti, L.; Cini, S.; Conti, B.; Falaschi, F.; Lopes Pegna, A.; Paci, E.; Carrozzi, L. Predictors of Smoking Cessation within a Lung Cancer CT Screening Trial. Eur. Respir. J. 2011, 38, p4248. [Google Scholar]
- Tammemägi, M.C.; Berg, C.D.; Riley, T.L.; Cunningham, C.R.; Taylor, K.L. Impact of Lung Cancer Screening Results on Smoking Cessation. J. Natl. Cancer Inst. 2014, 106, dju084. [Google Scholar] [CrossRef]
- Taylor, K.L.; Cox, L.S.; Zincke, N.; Mehta, L.; McGuire, C.; Gelmann, E. Lung cancer screening as a teachable moment for smoking cessation. Lung Cancer 2007, 56, 125–134. [Google Scholar] [CrossRef]
- Ebell, M.H.; Bentivegna, M.; Hulme, C. Cancer-Specific Mortality, All-Cause Mortality, and Overdiagnosis in Lung Cancer Screening Trials: A Meta-Analysis. Ann. Fam. Med. 2020, 18, 545–552. [Google Scholar] [CrossRef]
- Hoffman, R.M.; Atallah, R.P.; Struble, R.D.; Badgett, R.G. Lung Cancer Screening with Low-Dose CT: A Meta-Analysis. J. Gen. Intern. Med. 2020, 35, 3015–3025. [Google Scholar] [CrossRef]
- Mazzone, P.J.; Silvestri, G.A.; Patel, S.; Kanne, J.P.; Kinsinger, L.S.; Wiener, R.S.; Soo Hoo, G.; Detterbeck, F.C. Screening for Lung Cancer: CHEST Guideline and Expert Panel Report. Chest 2018, 153, 954–985. [Google Scholar] [CrossRef]
- Snowsill, T.; Yang, H.; Griffin, E.; Long, L.; Varley-Campbell, J.; Coelho, H.; Robinson, S.; Hyde, C. Low-dose computed tomography for lung cancer screening in high-risk populations: A systematic review and economic evaluation. Health Technol. Assess. 2018, 22, 1–276. [Google Scholar] [CrossRef]
- Oken, M.M.; Hocking, W.G.; Kvale, P.A.; Andriole, G.L.; Buys, S.S.; Church, T.R.; Crawford, E.D.; Fouad, M.N.; Isaacs, C.; Reding, D.J.; et al. Screening by Chest Radiograph and Lung Cancer Mortality: The Prostate, Lung, Colorectal, and Ovarian (PLCO) Randomized Trial. JAMA 2011, 306, 1865–1873. [Google Scholar] [CrossRef]
- Brodersen, J.; Voss, T.; Martiny, F.; Siersma, V.; Barratt, A.; Heleno, B. Overdiagnosis of Lung Cancer with Low-Dose Com-puted Tomography Screening: Meta-Analysis of the Randomised Clinical Trials. Breathe 2020, 16, 200013. [Google Scholar] [CrossRef]
- de Koning, H.J.; Meza, R.; Plevritis, S.K.; ten Haaf, K.; Munshi, V.N.; Jeon, J.; Erdogan, S.A.; Kong, C.Y.; Han, S.S.; van Rosmalen, J.; et al. Benefits and Harms of Computed Tomography Lung Cancer Screening Strategies: A Comparative Modeling Study for the U.S. Preventive Services Task Force. Ann. Intern. Med. 2014, 160, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Heuvelmans, M.A.; Oudkerk, M. Volume versus diameter assessment of small pulmonary nodules in CT lung cancer screening. Transl. Lung Cancer Res. 2017, 6, 52–61. [Google Scholar] [CrossRef]
- Katki, H.A.; Kovalchik, S.A.; Petito, L.; Cheung, L.C.; Jacobs, E.; Jemal, A.; Berg, C.D.; Chaturvedi, A.K. Implications of Nine Risk Prediction Models for Selecting Ever-Smokers for Computed Tomography Lung Cancer Screening. Ann. Intern. Med. 2018, 169, 10–19. [Google Scholar] [CrossRef] [PubMed]
- ten Haaf, K.; Bastani, M.; Cao, P.; Jeon, J.; Toumazis, I.; Han, S.S.; Plevritis, S.K.; Blom, E.F.; Kong, C.Y.; Tammemägi, M.C.; et al. A Comparative Modeling Analysis of Risk-Based Lung Cancer Screening Strategies. J. Natl. Cancer Inst. 2020, 112, 466–479. [Google Scholar] [CrossRef] [PubMed]
- Tammemägi, M.C.; Katki, H.A.; Hocking, W.G.; Church, T.R.; Caporaso, N.; Kvale, P.A.; Chaturvedi, A.K.; Silvestri, G.A.; Riley, T.L.; Commins, J.; et al. Selection Criteria for Lung-Cancer Screening. N. Engl. J. Med. 2013, 368, 728–736. [Google Scholar] [CrossRef] [PubMed]
- Yousaf-Khan, U.; van der Aalst, C.; de Jong, P.A.; Heuvelmans, M.; Scholten, E.; Walter, J.; Nackaerts, K.; Groen, H.; Vliegenthart, R.; ten Haaf, K.; et al. Risk Stratification Based on Screening History: The NELSON Lung Cancer Screening Study. Thorax 2017, 72, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Godtfredsen, N.S.; Prescott, E.; Osler, M. Effect of Smoking Reduction on Lung Cancer Risk. JAMA 2005, 294, 1505–1510. [Google Scholar] [CrossRef] [PubMed]
- Lemmens, V.; Oenema, A.; Knut, I.K.; Brug, J. Effectiveness of smoking cessation interventions among adults: A systematic review of reviews. Eur. J. Cancer Prev. 2008, 17, 535–544. [Google Scholar] [CrossRef]
- Stang, A.; Schuler, M.; Kowall, B.; Darwiche, K.; Kühl, H.; Jöckel, K.-H. Lung Cancer Screening Using Low Dose CT Scanning in Germany. Dtsch. Aerzteblatt Int 2015, 112, 637–644. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Brix, G.; Nekolla, E.A.; Griebel, J. Early Detection of Diseases by Radiological Imaging: New Legal Situation and Evaluation of Service Offers using CT Examinations as an Example. RöFo—Fortschritte auf dem Gebiet der Röntgenstrahlen und der bildgebenden Verfahren 2020, 192, 139–149. [Google Scholar] [CrossRef] [PubMed]

| Study Country, Recruitment Period | Study Sites | Age Range (Years) | Smoking History | Smoking Abstinence (Years) | Screening Interval (Years) | Screening Visits | Control Group | Follow-Up (Years) |
|---|---|---|---|---|---|---|---|---|
| DANTE Italy, 2001–2006 | 3 | 60–74 | ≥20 PY | <10 | 1 | 5 | no screening | 8.35 |
| DEPISCAN France, 2002–2004 | 14 | 50–75 | ≥15 cig/day for 20 yrs | <15 | 1 | 3 | CXR | not reported |
| DLCST Denmark, 2004–2006 | 1 | 50–70 | ≥20 PY | <10 | 1 | 5 | no screening | 9.8 |
| ITALUNG Italy, 2004–2006 | 3 | 55–69 | ≥20 PY | 1 | 4 | no screening | 9.3 | |
| LSS USA, 2000 | 6 | 55–74 | ≥30 PY | <10 | 1 | 2 | CXR | 1 |
| LUSI Germany, 2007–2011 | 1 | 50–69 | ≥15 cig/day for 25 yrs OR ≥10 cig/day for 30 yrs | <10 | 1 | 5 | no screening | 8.9 |
| MILD Italy, 2005–2011 | 1 | 49–75 | ≥20 PY | <10 | 1/2 | 7/4 | no screening | 10 |
| NELSON Belgium/The Netherlands, 2003–2005 | 4 | 50–75 | ≥15 cig/day for ≥25 yrs OR ≥10 cig/day for ≥30 yrs | <10 | 1/2/2.5 | 4 | no screening | 8.16 |
| NLST USA, 2002–2004 | 33 | 55–75 | ≥30 PY | <15 | 1 | 3 | CXR | 12.3 |
| Subjects Randomized | Subjects in Screening Arm | Male Participants (%) | Age (Years), Mean or Median | Pack-Years, Mean or Median | Current Smokers in Screening vs. Control Group (%) | |
|---|---|---|---|---|---|---|
| DANTE | 2450 | 1264 | 100 | 64 | 47 | 56.5 vs. 57.4 |
| DEPISCAN | 765 | 385 | 71 | 56 | 32 | 65 vs. 64 |
| DLCST | 4104 | 2052 | 56 | 58 | 36 | 75.3 vs. 76.9 |
| ITALUNG | 3206 | 1613 | 64.7 | 61 | 40 | 66.5 vs. 63.1 |
| LSS | 3318 | 1660 | 59 | 68% younger than 64 | 54 | 57.9 vs. 57.1 |
| LUSI | 4052 | 2029 | 64.7 | 55 | - | 50.2 vs. 49.8 |
| MILD | 4099 | annual: 1190 biennial: 1186 | 68.4 | 58 | 39 | 68.9 vs. 89.7 |
| NELSON | 15,822 | 7915 | 84 | 59 | 42 | 55.5 vs. 54.8 |
| NLST | 53,452 | 26,722 | 59 | 43% younger than 59; 26% older than 65 | - | 48.1 vs. 48.1 |
| CT Detector Rows | Use of Volumetry Software | Tube Voltage (kV) | Effective Tube Current (mA) or Current-Time-Product (mAs) | Effective Dose Per LDCT Scan (mSv) | Minimal Nodule Size or Growth for Recall/Follow-Up | |
|---|---|---|---|---|---|---|
| DANTE | 1–16 | no | 140 | 40 mA | n. r. | All solid, non-smooth |
| DEPISCAN | n. r. | no | 100–140 | 20–100 mA | n. r. | D > 5 mm |
| DLCST | 16 | yes | 120 | 40 mA | around 1 | D ≥ 5 mm |
| ITALUNG | 1–64 | no | 120–140 | 20–43 mA | 1.2–1.4 | D > 5 mm |
| LSS | n. r. | no | 120–140 | 60 mA | n. r. | Baseline: D > 3 mm; other rounds: D > 4 mm |
| LUSI | 16 and 128 | yes | n. r. | n. r. | < 1.6–2.0 | D ≥ 5 mm or VDT = 400–600 days and D < 7.5 mm |
| MILD | 16 | yes | 120 | 30 mAs | n. r. | V ≥ 60 mm3 or D ≥ 5 mm |
| NELSON | 16 and 64 | yes | 80–140 | n. r. | 0.4–1.6 | D > 5 mm or V > 50 mm3 or VDT = 400–600 days |
| NLST | 4–64 | no | 120 | 20–40 mAs | 1.6–2.4 | D > 4 mm |
| Number of Diagnosed Lung Cancers (%) | Number of Non-Screening Detected Lung Cancers in the Screening Group (%) | ||
|---|---|---|---|
| Screening | Control | ||
| DANTE | 104 (8.2) | 72 (6.1) | 38 (37) |
| DEPISCAN | 8 (2.1) | 1 (0.3) | not reported |
| DLCST | 100 (4.9) | 53 (2.6) | not reported |
| ITALUNG | 67 (4.2) | 71 (4.4) | 25 (37) |
| LSS | 40 (2.4) | 20 (1.2) | 2 (5) |
| LUSI | 85 (4.2) | 67 (3.3) | 6 (7) |
| MILD | 98 (4.1) | 60 (3.5) | 27 (28) |
| NELSON | 344 (4.3) | 304 (3.8) | 141 (41) |
| NLST | 1701 (6.4) | 1681 (6.3) | 44 (3) |
| Number of Screening-LDCTs Performed | Positive or Indeterminate LDCT Findings N (%) | Recall Rate (%) | Lung Cancer Detection Rate (%) | |
|---|---|---|---|---|
| DANTE | 6482 | - | Tt: 28.1 | Tt: 5.3 |
| DEPISCAN | 336 | 81 (24.1) | Tt: 24 | Tt: 2.4 |
| DLCST | 9800 | 512 (5.2) | T0: 7.6 | T0: 0.83 |
| Tt: 0.70 | ||||
| ITALUNG | 5333 | 1044 (19.6) | T0: 30.3 | T0: 1.5 |
| Tt: 52.7 | Tt: 0.5 | |||
| LSS | 2984 | 655 (22.0) | T0: 25.8 | T0: 1.9 |
| Tt: 34.5 | T1: 0.57 | |||
| LUSI | 9405 | Positive: 174 (1.9) | T0: 22.2 | T0: 1.1 |
| Indeterminate: 642 (6.8) | ||||
| MILD | ||||
| annual | 7369 | Positive: 91 (1.2) | T0: 14.8 | T0: 0.96 |
| Indeterminate: 177 (2.4) | Tt: 5.81 | Tt: 0.56 | ||
| biennial | 5006 | Positive: 59 (1.2) | T0: 13.7 | T0: 0.52 |
| Indeterminate: 158 (3.1) | Tt: 6.97 | Tt: 0.56 | ||
| NELSON * | 22,600 | Positive: 467 (2.1) | T0: 20.4 | T0: 0.9 |
| Indeterminate: 2069 (9.2) | Tt: 3.2 | |||
| NLST | 75,126 | 18,146 (24.2) | T0: 27.3 | T0: 1.1 |
| Tt: 24.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hunger, T.; Wanka-Pail, E.; Brix, G.; Griebel, J. Lung Cancer Screening with Low-Dose CT in Smokers: A Systematic Review and Meta-Analysis. Diagnostics 2021, 11, 1040. https://doi.org/10.3390/diagnostics11061040
Hunger T, Wanka-Pail E, Brix G, Griebel J. Lung Cancer Screening with Low-Dose CT in Smokers: A Systematic Review and Meta-Analysis. Diagnostics. 2021; 11(6):1040. https://doi.org/10.3390/diagnostics11061040
Chicago/Turabian StyleHunger, Theresa, Eva Wanka-Pail, Gunnar Brix, and Jürgen Griebel. 2021. "Lung Cancer Screening with Low-Dose CT in Smokers: A Systematic Review and Meta-Analysis" Diagnostics 11, no. 6: 1040. https://doi.org/10.3390/diagnostics11061040
APA StyleHunger, T., Wanka-Pail, E., Brix, G., & Griebel, J. (2021). Lung Cancer Screening with Low-Dose CT in Smokers: A Systematic Review and Meta-Analysis. Diagnostics, 11(6), 1040. https://doi.org/10.3390/diagnostics11061040





