Absolute and Relative Reliability of the Assessment of the Muscle Mechanical Properties of Pelvic Floor Muscles in Women with and without Urinary Incontinence
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
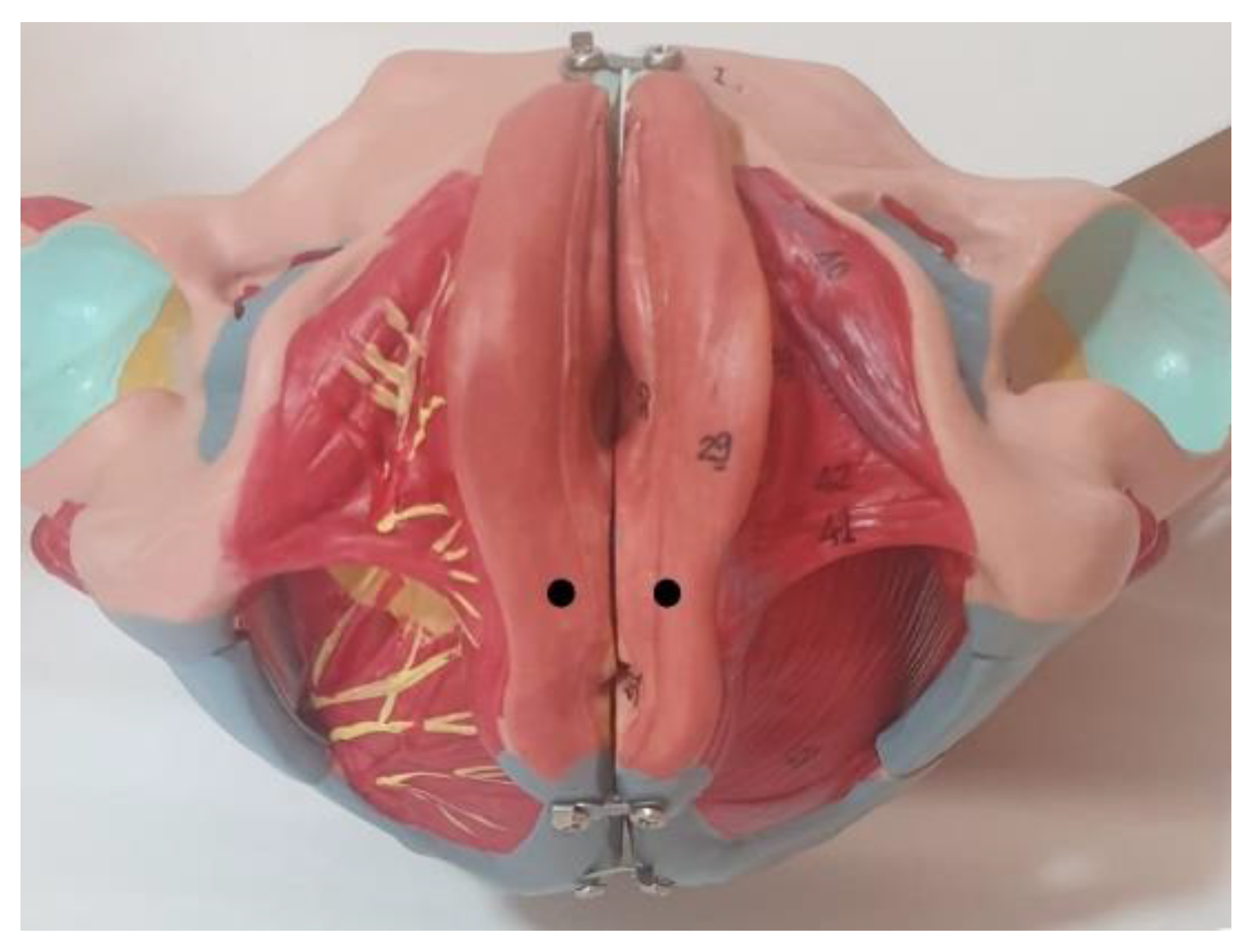
2.3. Assessments and Procedures
2.4. Statistical Analysis
3. Results
4. Discussion
4.1. Relative Reliability of MMPs of PFMs Assessment
4.2. Absolute Reliability of MMPs of PFMs Assessment
4.3. Clinical Consideration of Assessing the MMPs of PFMs in Women with and without UI
4.4. Strengths and Limitations of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Easley, D.C.; Abramowitch, S.D.; Moalli, P.A. Female Pelvic Floor Biomechanics: Bridging the Gap. Curr. Opin. Urol. 2017, 27, 262–267. [Google Scholar] [CrossRef]
- Eickmeyer, S.M. Anatomy and Physiology of the Pelvic Floor. Phys. Med. Rehabil. Clin. N. Am. 2017, 28, 455–460. [Google Scholar] [CrossRef]
- Shishido, K.; Peng, Q.; Jones, R.; Omata, S.; Constantinou, C.E. Influence of Pelvic Floor Muscle Contraction on the Profile of Vaginal Closure Pressure in Continent and Stress Urinary Incontinent Women. J. Urol. 2008, 179, 1917–1922. [Google Scholar] [CrossRef]
- Alves, J.O.; Da Luz, S.T.; Brandão, S.; Da Luz, C.M.; Jorge, R.N.; Da Roza, T. Urinary Incontinence in Physically Active Young Women: Prevalence and Related Factors. Int. J. Sports Med. 2017, 38, 937–941. [Google Scholar] [CrossRef]
- Almousa, S.; Bandin van Loon, A. The Prevalence of Urinary Incontinence in Nulliparous Adolescent and Middle-Aged Women and the Associated Risk Factors: A Systematic Review. Maturitas 2018, 107, 78–83. [Google Scholar] [CrossRef]
- Samuelsson, E.; Victor, A.; Svardsudd, K. Determinants of Urinary Incontinence in a Population of Young and Middle-Aged Women. Acta Obstet. Gynecol. Scand. 2000, 79, 208–215. [Google Scholar]
- Fozzatti, C.; Riccetto, C.; Herrmann, V.; Brancalion, M.F.; Raimondi, M.; Nascif, C.H.; Marques, L.R.; Palma, P.P. Prevalence Study of Stress Urinary Incontinence in Women Who Perform High-Impact Exercises. Int. Urogynecol. J. 2012, 23, 1687–1691. [Google Scholar] [CrossRef]
- Abrams, P.; Cardozo, L.; Fall, M.; Griffiths, D.; Rosier, P.; Ulmsten, U.; Van Kerrebroeck, P.; Victor, A.; Wein, A. The Standardisation of Terminology in Lower Urinary Tract Function: Report from the Standardisation Sub-Committee of the International Continence Society. Urology 2003, 61, 37–49. [Google Scholar] [CrossRef]
- Burnett, L.A.; Cook, M.; Shah, S.; Kado, D.M.; Alperin, M.; Sciences, R.; Surgery, R.; Diego, S.; States, U.; States, U.; et al. Age-Associated Changes in the Mechanical Properties of Human Cadaveric Pelvic Floor Muscles. J. Biomech. 2020, 98, 109436. [Google Scholar] [CrossRef] [PubMed]
- Chantereau, P.; Brieu, M.; Kammal, M.; Farthmann, J.; Gabriel, B.; Cosson, M. Mechanical Properties of Pelvic Soft Tissue of Young Women and Impact of Aging. Int. Urogynecol. J. 2014, 25, 1547–1553. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, S.; Watson, T.; Thompson, J.; Briffa, K. Stress Urinary Incontinence Is Highly Prevalent in Recreationally Active Women Attending Gyms or Exercise Classes. Int. Urogynecol. J. 2016, 27, 1175–1184. [Google Scholar] [CrossRef] [PubMed]
- Yip, C.; Kwok, E.; Sassani, F.; Jackson, R.; Cundiff, G. A Biomechanical Model to Assess the Contribution of Pelvic Musculature Weakness to the Development of Stress Urinary Incontinence. Comput. Methods Biomech. Biomed. Engin. 2014, 17, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Kruger, J.A.; Budgett, S.C.; Wong, V.; Nielsen, P.M.F.; Nash, M.P.; Smalldridge, J.; Hayward, L.M.; Tian, T.Y.; Taberner, A.J. Characterizing Levator-Ani Muscle Stiffness Pre- and Post-Childbirth in European and Polynesian Women in New Zealand: A Pilot Study. Acta Obstet. Gynecol. Scand. 2017, 96, 1234–1242. [Google Scholar] [CrossRef] [PubMed]
- Bernard, S.; Moffet, H.; Plante, M.; Ouellet, M.P.; Leblond, J.; Dumoulin, C. Pelvic-Floor Properties in Women Reporting Urinary Incontinence After Surgery and Radiotherapy for Endometrial Cancer. Phys. Ther. 2017, 97, 438–448. [Google Scholar] [CrossRef]
- Smith, M.D.; Coppieters, M.W.; Hodges, P.W. Postural Response of the Pelvic Floor and Abdominal Muscles in Women with and without Incontinence. Neurourol. Urodyn. 2007, 26, 377–385. [Google Scholar] [CrossRef]
- Davidson, M.J.; Bryant, A.L.; Bower, W.F.; Frawley, H.C. Myotonometry Reliably Measures Muscle Stiffness in the Thenar and Perineal Muscles. Physiother. Can. 2017, 69, 104–112. [Google Scholar] [CrossRef]
- Lucas, N.; Macaskill, P.; Irwig, L.; Moran, R.; Bogduk, N. Reliability of Physical Examination for Diagnosis of Myofascial Trigger Points: A Systematic Review of the Literature. Clin. J. Pain 2009, 25, 80–89. [Google Scholar] [CrossRef]
- Messelink, B.; Benson, T.; Berghmans, B.; Bø, K.; Corcos, J.; Fowler, C.; Laycock, J.; Lim, P.H.C.; Van Lunsen, R.; Lycklama, Á.; et al. Standardization of Terminology of Pelvic Floor Muscle Function and Dysfunction: Report from the Pelvic Floor Clinical Assessment Group of the International Continence Society. Neurourol. Urodyn. 2005, 24, 374–380. [Google Scholar] [CrossRef]
- Davidson, M.J.; Nielsen, P.M.F.; Taberner, A.J.; Kruger, J.A. Is It Time to Rethink Using Digital Palpation for Assessment of Muscle Stiffness? Neurourol. Urodyn. 2020, 39, 279–285. [Google Scholar] [CrossRef]
- Paolucci, T.; Bellomo, R.G.; Pezzi, L.; Frondaroli, F.; Frondaroli, S.; Santarelli, A.; Barbato, C.; Porreca, A.; Saggini, R. A Novel Rehabilitative Protocol in the Treatment of Mixed Urinary Incontinence in Women: The Effects of Focused Mechano-Acoustic Vibration. Biores. Open Access 2019, 8, 219–228. [Google Scholar] [CrossRef]
- Barassi, G.; Bellomo, R.G.; Frondaroli, F.; Frondaroli, S.; Santarelli, A.; Di Felice, P.A.; Supplizi, M.; Palermo, T.; Saggini, T. Integrated Rehabilitation Approach with Manual and Mechanic-Acoustic Vibration Therapies for Urinary Incontinence. Adv. Exp. Med. Biol. 2019, 7, 41–50. [Google Scholar]
- Romero-Cullerés, G.; Peña-Pitarch, E.; Jané-Feixas, C.; Arnau, A.; Montesinos, J.; Abenoza-Guardiola, M. Intra-Rater Reliability and Diagnostic Accuracy of a New Vaginal Dynamometer to Measure Pelvic Floor Muscle Strength in Women with Urinary Incontinence. Neurourol. Urodyn. 2017, 36, 333–337. [Google Scholar] [CrossRef]
- Thibault-Gagnon, S.; Morin, M. Active and Passive Components of Pelvic Floor Muscle Tone in Women with Provoked Vestibulodynia: A Perspective Based on a Review of the Literature. J. Sex. Med. 2015, 12, 2178–2189. [Google Scholar] [CrossRef] [PubMed]
- Leonard, C.T.; Brown, J.S.; Price, T.R.; Queen, S.A.; Mikhailenok, E.L. Comparison of Surface Electromyography and Myotonometric Measurements during Voluntary Isometric Contractions. J. Electromyogr. Kinesiol. 2004, 14, 709–7014. [Google Scholar] [CrossRef] [PubMed]
- Drenth, H.; Zuidema, S.U.; Krijnen, W.P.; Bautmans, I.; van derSchans, C.; Hobbelen, H. Psychometric Properties of the MyotonPRO in Dementia Patients with Paratonia. Gerontology 2018, 64, 401–412. [Google Scholar] [CrossRef]
- Chuang, L.L.; Wu, C.Y.; Lin, K.C.; Lur, S.Y. Quantitative Mechanical Properties of the Relaxed Biceps and Triceps Brachii Muscles in Patients with Subacute Stroke: A Reliability Study of the Myoton-3 Myometer. Stroke Res. Treat. 2012, 2012, 617694. [Google Scholar] [CrossRef]
- Alcaraz-Clariana, S.; García-Luque, L.; Garrido-castro, J.L.; Carmona-Pérez, C.; Rodrigues-de-souza, D.P. Paravertebral Muscle Mechanical Properties and Spinal Range of Motion in Patients with Acute Neck or Low Back Pain: A Case-Control Study. Diagnostics 2021, 11, 352. [Google Scholar] [CrossRef]
- Young, F.C.; Cristi-Sánchez, I.; Danes-Daetz, C.; Monckeberg, J.E.; Aguirre, R.S. Patellar Tendon Stiffness in Elite Breakdancers Assessed by Myotonometric Measurement. J. Danc. Med. Sci. 2018, 22, 179–183. [Google Scholar] [CrossRef]
- Kottner, J.; Audigé, L.; Brorson, S.; Donner, A.; Gajewski, B.J.; Hróbjartsson, A.; Roberts, C.; Shoukri, M.; Streiner, D.L. Guidelines for Reporting Reliability and Agreement Studies (GRRAS) Were Proposed. J. Clin. Epidemiol. 2011, 64, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.S.; Bradley, C.S.; Subak, L.L.; Richter, H.E.; Kraus, S.R.; Brubaker, L.; Lin, F.; Vittinghoff, E.; Grady, D.; Diagnostic Aspects of Incontinence Study (DAISy) Research Group. The Sensitivity and Specificity of a Simple Test to Distinguish between Urge and Stress Urinary Incontinence. Ann. Intern. Med. 2006, 144, 715–723. [Google Scholar] [CrossRef]
- Treszezamsky, A.D.; Karp, D.; Dick-Biascoechea, M.; Ehsani, N.; Dancz, C.; Montoya, T.I.; Olivera, C.K.; Smith, A.L.; Cardenas, R.; Fashokun, T.; et al. Spanish Translation and Validation of Four Short Pelvic Floor Disorders Questionnaires. Int. Urogynecol. J. Pelvic Floor Dysfunct. 2013, 24, 655–670. [Google Scholar] [CrossRef]
- Barber, M.D.; Walters, M.D.; Bump, R.C. Short Forms of Two Condition-Specific Quality-of-Life Questionnaires for Women with Pelvic Floor Disorders (PFDI-20 and PFIQ-7). Am. J. Obstet. Gynecol. 2005, 193, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Sánchez, B.; Torres-Lacomba, M.; Yuste-Sánchez, M.J.; Navarro-Brazález, B.; Pacheco-da-Costa, S.; Gutiérrez-Ortega, C.; Zapico-Goñi, A. Cultural Adaptation and Validation of the Pelvic Floor Distress Inventory Short Form (PFDI-20) and Pelvic Floor Impact Questionnaire Short Form (PFIQ-7) Spanish Versions. Eur. J. Obs. Gynecol. Reprod. Biol. 2013, 170, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Zhu, Y.; Xu, W.; Liang, J.; Guan, Y.; Xu, X. Analysis of Biomechanical Properties of the Lumbar Extensor Myofascia in Elderly Patients with Chronic Low Back Pain and That in Healthy People. Biomed. Res. Int. 2020, 2020, 7649157. [Google Scholar] [CrossRef]
- Myoton, A.S. MyotonPro User Manual; Myoton AS: Tallinn, Estonia, 2012. [Google Scholar]
- Castro, J.L.G.; Valera, I.C.A.; Amaro, J.P.; Galisteo, A.M.; Navas, C.G.; De Souza, D.P.R.; Clariana, S.A.; Luque, L.G.; Sánchez, I.R.M.; Medina, C.L.; et al. Mechanical Properties of Lumbar and Cervical Paravertebral Muscles in Patients with Axial Spondyloarthritis: A Case - Control Study. Diagnostics 2021, 11, 1662. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Jiménez, M.; Alburquerque-Sendín, F.; Garrido-Castro, J.L.; Rodrigues-de-Sousa, D.P. Reliability of Cervical Muscles Strength Assessment with a Handheld Dynamometer in Individuals with Shoulder Pain. Physiother. Theory Pract. 2021, in press. [Google Scholar]
- Aird, L.; Samuel, D.; Stokes, M. Quadriceps Muscle Tone, Elasticity and Stiffness in Older Males: Reliability and Symmetry Using the MyotonPRO. Arch. Gerontol. Geriatr. 2012, 55, e31–e39. [Google Scholar] [CrossRef]
- Gavronski, G.; Veraksitš, A.; Vasar, E.; Maaroos, J. Evaluation of Viscoelastic Parameters of the Skeletal Muscles in Junior Triathletes. Physiol. Meas. 2007, 28, 625–637. [Google Scholar] [CrossRef]
- Agyapong-Badu, S.; Warner, M.B.; Samuel, D.; Koutra, V.; Stokes, M. Non-Invasive Biomarkers of Musculoskeletal Health with High Discriminant Ability for Age and Gender. J. Clin. Med. 2021, 10, 1352. [Google Scholar] [CrossRef]
- White, A.; Abbott, H.; Masi, A.T.; Henderson, J.; Nair, K. Biomechanical Properties of Low Back Myofascial Tissue in Younger Adult Ankylosing Spondylitis Patients and Matched Healthy Control Subjects. Clin. Biomech. 2018, 57, 67–73. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Weir, J. Quantifying Test-Retest Reliabilty Using the Intraclass Correlation Coefficient and SEM. J. Strength Cond. Res. 2005, 19, 231–240. [Google Scholar] [PubMed]
- Haik, M.N.; Alburquerque-Sendín, F.; Camargo, P.R. Reliability and Minimal Detectable Change of 3-Dimensional Scapular Orientation in Individuals with and without Shoulder Impingement. J. Orthop. Sports Phys. Ther. 2014, 44, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Arouca, M.A.F.; Duarte, T.B.; Lott, D.A.M.; Magnani, P.S.; Nogueira, A.A.; Rosa-e-Silva, J.C.; Brito, L.G.O. Validation and Cultural Translation for Brazilian Portuguese Version of the Pelvic Floor Impact Questionnaire (PFIQ-7) and Pelvic Floor Distress Inventory (PFDI-20). Int. Urogynecol. J. 2016, 27, 1097–1106. [Google Scholar] [CrossRef]
- Cetin, S.Y.; Buyuk, A.; Ayan, A. Investigation of the Relationship between the Pelvic Floor and Sexual Dysfunction in Women with Sjögren’s Syndrome. Int. J. Rheum. Dis. 2020, 23, 1728–1733. [Google Scholar] [CrossRef]
- Gajdosik, R.L. Passive Extensibility of Skeletal Muscle: Review of the Literature with Clinical Implications. Clin. Biomech. 2001, 16, 87–101. [Google Scholar] [CrossRef]
- Chuang, L.L.; Wu, C.Y.; Lin, K.C. Reliability, Validity, and Responsiveness of Myotonometric Measurement of Muscle Tone, Elasticity, and Stiffness in Patients with Stroke. Arch. Phys. Med. Rehabil. 2012, 93, 532–540. [Google Scholar] [CrossRef]
- Hu, X.; Lei, D.; Li, L.; Leng, Y.; Yu, Q.; Wei, X.; Lo, W.L.A. Quantifying Paraspinal Muscle Tone and Stiffness in Young Adults with Chronic Low Back Pain: A Reliability Study. Sci. Rep. 2018, 8, 14343. [Google Scholar] [CrossRef] [PubMed]
- Van Deun, B.; Hobbelen, J.S.M.; Cagnie, B.; Van Eetvelde, B.; Van Den Noortgate, N.; Cambier, D. Reproducible Measurements of Muscle Characteristics Using the MyotonPRO Device: Comparison between Individuals with and Without Paratonia. J. Geriatr. Phys. Ther. 2018, 41, 194–203. [Google Scholar] [CrossRef]
- Czyrnyj, C.S.; Bérubé, M.È.; Varette, K.; McLean, L. The Impact of a Familiarization Session on the Magnitude and Stability of Active and Passive Pelvic Floor Muscle Forces Measured through Intravaginal Dynamometry. Neurourol. Urodyn. 2019, 38, 902–911. [Google Scholar] [CrossRef]
- Lemieux, J.; Beaton, D.E.; Hogg-Johnson, S.; Bordeleau, L.J.; Goodwin, P.J. Three Methods for Minimally Important Difference: No Relationship Was Found with the Net Proportion of Patients Improving. J. Clin. Epidemiol. 2007, 60, 448–455. [Google Scholar] [CrossRef]
- Morin, M.; Denis Gravel, D.; Bourbonnais, C.; Dumoulin, S.O. Reliability of Dynamometric Passive Properties of the Pelvic Floor Muscles in Postmenopausal Women with Stress Urinary Incontinence. Neurourol. Urodyn. 2008, 27, 819–825. [Google Scholar] [CrossRef]
- Bérubé, M.È.; Czyrnyj, C.S.; McLean, L. An Automated Intravaginal Dynamometer: Reliability Metrics and the Impact of Testing Protocol on Active and Passive Forces Measured from the Pelvic Floor Muscles. Neurourol. Urodyn. 2018, 37, 1875–1888. [Google Scholar] [CrossRef]
- Brækken, I.H.; Stuge, B.; Tveter, A.T.; Bø, K. Reliability, Validity and Responsiveness of Pelvic Floor Muscle Surface Electromyography and Manometry. Int. Urogynecol. J. 2021, 32, 3267–3274. [Google Scholar] [CrossRef]
- Cuesta-Vargas, A.I.; Galán-Mercant, A.; Williams, J.M. The Use of Inertial Sensors System for Human Motion Analysis. Phys. Ther. Rev. 2010, 15, 462–473. [Google Scholar] [CrossRef] [PubMed]
- Carmona-Pérez, C.; Garrido-Castro, J.L.; Vidal, F.T.; Alcaraz-clariana, S.; García-Luque, L.; Alburquerque-Sendín, F. Concurrent Validity and Reliability of an Inertial Measurement Unit for the Assessment of Craniocervical Range of Motion in Subjects with Cerebral Palsy. Diagnostics 2020, 10, 80. [Google Scholar] [CrossRef]
- Pool-Goudzwaard, A.; Hoek Van Dijke, G.; Van Gurp, M.; Mulder, P.; Snijders, C.; Stoeckart, R. Contribution of Pelvic Floor Muscles to Stiffness of the Pelvic Ring. Clin. Biomech. 2004, 19, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Epstein, L.B.; Graham, C.A.; Heit, M.H. Systemic and Vaginal Biomechanical Properties of Women with Normal Vaginal Support and Pelvic Organ Prolapse. Am. J. Obstet. Gynecol. 2007, 197, 165.e1–165.e6. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Lee, J.H.; Wen, Y.; Constantinou, C.; Yoshinobu, M.; Omata, S.; Chen, B. Biomechanical Properties and Associated Collagen Composition in Vaginal Tissue of Women with Pelvic Organ Prolapse. J. Urol. 2012, 188, 875–880. [Google Scholar] [CrossRef]

| UI Group (n = 38) | Healthy Group (n = 40) | p-Value | |
|---|---|---|---|
| Age (years) | 22.3 ± 2.4 | 22.6 ± 2.7 | 0.652 |
| BMI (kg/m2) | 23.5 ± 3.5 | 21.4 ± 1.7 | 0.272 |
| Types of UI | SUI: 23; UUI: 8; MUI: 7 | ||
| Time suffering UI (years) | 3.2 ± 5.3 | ||
| PFDI-20 | 32.9 ± 30.0 | 18.6 ± 17.1 | 0.010 |
| PFIQ-7 | 18.9 ± 33.0 | 8.0 ± 10.8 | 0.048 |
| Session | ICC2,1 (95% CI) | SEM (SEM%) | MDC90 | Session | ICC2,1 (95% CI) | SEM (SEM%) | MDC90 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| UI Group | Healthy Group | ||||||||||||
| 1st session (n = 33) | 2nd session (n = 33) | 1st session (n = 36) | 2nd session (n = 36) | ||||||||||
| Right side | Frequency (Hz) | 15.03 ± 1.23 | 15.07 ± 1.02 | 0.760 | 0.89 (0.77, 0.95) | 0.37 (2.46) | 0.86 | 15.89 ± 1.92 | 15.45 ± 1.70 | 0.077 | 0.81 (0.63, 0.90) | 0.79 (5.05) | 1.84 |
| Stiffness (N/m) | 222.50 ± 31.90 | 219.20 ± 29.66 | 0.461 | 0.82 (0.62, 0.91) | 12.98 (5.88) | 30.01 | 246.39 ± 60.49 | 237.94 ± 49.13 | 0.370 | 0.83 (0.61, 0.93) | 22.57 (9.32) | 52.34 | |
| Decrement | 1.02 ± 0.09 | 0.99 ± 0.12 | 0.066 | 0.78 (0.54, 0.90) | 0.05 (4.90) | 0.11 | 1.09 ± 0.19 | 1.02 ± 0.14 | 0.015 * | 0.75 (0.43, 0.90) | 0.08 (8.19) | 0.20 | |
| Relaxation (ms) | 18.12 ± 1.94 | 18.46 ± 1.83 | 0.047 | 0.94 (0.87, 0.97) | 0.46 (2.51) | 1.02 | 16.84 ± 2.26 | 17.77 ± 2.70 | 0.051 | 0.70 (0.40, 0.87) | 1.53 (8.84) | 3.55 | |
| Creep (De) | 1.00 ± 0.10 | 1.02 ± 0.09 | 0.230 | 0.85 (0.68, 0.93) | 0.04 (3.77) | 0.09 | 0.93 ± 0.10 | 0.99 ± 0.14 | 0.016 * | 0.63 (0.36, 0.81) | 0.07 (6.95) | 0.15 | |
| Left side | Frequency (Hz) | 15.23 ± 1.54 | 14.49 ± 1.34 | 0.010 * | 0.86 (0.70, 0.93) | 0.55 (3.73) | 1.28 | 16.02 ± 1.57 | 16.03 ± 1.40 | 0.954 | 0.76 (0.42, 0.89) | 0.55 (3.44) | 1.28 |
| Stiffness (N/m) | 228.00 ± 28.40 | 215.60 ± 27.81 | 0.012 * | 0.82 (0.61, 0.91) | 12.31 (5.55) | 28.55 | 250.28 ± 46.54 | 250.61 ± 39.25 | 0.966 | 0.86 (0.63, 0.95) | 22.56 (9.01) | 52.33 | |
| Decrement | 1.04 ± 0.13 | 1.00 ± 0.12 | 0.051 | 0.77 (0.50, 0.90) | 0.06 (5.92) | 0.14 | 1.05 ± 0.19 | 1.05 ± 0.12 | 0.848 | 0.75 (0.41, 0.89) | 0.09 (8.79) | 0.21 | |
| Relaxation (ms) | 17.69 ± 1.87 | 18.35 ± 1.78 | 0.004 * | 0.88 (0.76, 0.95) | 0.64 (3.53) | 1.48 | 16.43 ± 1.70 | 16.99 ± 1.89 | 0.045 * | 0.85 (0.63, 0.96) | 1.20 (7.20) | 2.79 | |
| Creep (De) | 0.98 ± 0.09 | 0.98 ± 0.07 | 0.953 | 0.68 (0.40, 0.82) | 0.05 (4.71) | 0.11 | 0.91 ± 0.07 | 0.96 ± 0.09 | 0.002 * | 0.66 (0.38, 0.83) | 0.07 (7.64) | 0.16 | |
| Rater | ICC2,2 (95% CI) | SEM (SEM%) | MDC90 | Rater | p-Value | ICC2,2 (95% CI) | SEM (SEM%) | MDC90 | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| UI Group | Healthy Group | ||||||||||||
| Rater 1 (n = 38) | Rater 2 (n = 38) | Rater 1 (n = 40) | Rater 2 (n = 40) | ||||||||||
| Right side | Frequency (Hz) | 15.02 ± 2.79 | 15.04 ± 1.29 | 0.406 | 0.83 (0.65, 0.95) | 0.89 (5.93) | 2.07 | 15.78 ± 1.93 | 15.39 ± 1.27 | 0.090 | 0.89 (0.69, 0.98) | 0.53 (3.43) | 1.24 |
| Stiffness (N/m) | 224.18 ± 55.96 | 220.27 ± 38.01 | 0.265 | 0.80 (0.63, 0.91) | 20.13 (9.06) | 46.68 | 241.74 ± 59.59 | 232.32 ± 38.97 | 0.288 | 0.82 (0.56, 0.95) | 19.78 (8.34) | 45.88 | |
| Decrement | 1.07 ± 0.17 | 1.04 ± 0.09 | 0.326 | 0.78 (0.48, 0.92) | 0.07 (6.17) | 0.15 | 1.08 ± 0.19 | 1.03 ± 0.15 | 0.054 | 0.87 (0.65, 0.98) | 0.06 (5.87) | 0.14 | |
| Relaxation (ms) | 17.45 ± 2.84 | 17.47 ± 1.22 | 0.953 | 0.78 (0.47, 0.92) | 1.02 (5.84) | 2.36 | 16.99 ± 2.30 | 17.79 ± 2.03 | 0.035 * | 0.73 (0.40, 0.91) | 1.12 (6.46) | 2.61 | |
| Creep (De) | 0.97 ± 0.14 | 0.96 ± 0.08 | 0.682 | 0.42 (0.15, 0.75) | 0.09 (8.85) | 0.20 | 0.93 ± 0.10 | 0.98 ± 0.10 | 0.014 * | 0.40 (0.11, 0.73) | 0.08 (8.14) | 0.18 | |
| Left side | Frequency (Hz) | 15.44 ± 1.62 | 14.70 ± 1.55 | 0.040 * | 0.85 (0.66, 0.95) | 0.63 (4.15) | 1.45 | 15.99 ± 1.53 | 15.97 ± 1.16 | 0.892 | 0.86 (0.64, 0.98) | 0.50 (3.12) | 1.15 |
| Stiffness (N/m) | 242.18 ± 47.97 | 222.27 ± 36.26 | 0.050 | 0.83 (0.64, 0.93) | 17.88 (7.70) | 41.48 | 249.21 ± 55.18 | 247.79 ± 42.15 | 0.853 | 0.81 (0.53, 0.95) | 20.98 (8.45) | 48.67 | |
| Decrement | 1.00 ± 0.17 | 1.00 ± 0.14 | 0.124 | 0.80 (0.50, 0.93) | 0.07 (6.95) | 0.16 | 1.04 ± 0.19 | 1.05 ± 0.17 | 0.535 | 0.92 (0.84, 0.96) | 0.05 (4.73) | 0.12 | |
| Relaxation (ms) | 17.51 ± 1.86 | 18.52 ± 1.71 | 0.011 * | 0.75 (0.49, 0.89) | 0.92 (5.12) | 2.14 | 16.49 ± 1.67 | 17.22 ± 1.82 | 0.014 * | 0.77 (0.46, 0.92) | 0.84 (4.98) | 1.95 | |
| Creep (De) | 0.95 ± 0.09 | 0.99 ± 0.08 | 0.026 * | 0.44 (0.20, 0.77) | 0.07 (6.77) | 0.15 | 0.91 ± 0.07 | 0.98 ± 0.09 | <0.001 * | 0.46 (0.20, 0.76) | 0.06 (6.63) | 0.15 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodrigues-de-Souza, D.P.; Alcaraz-Clariana, S.; García-Luque, L.; Carmona-Pérez, C.; Garrido-Castro, J.L.; Cruz-Medel, I.; Camargo, P.R.; Alburquerque-Sendín, F. Absolute and Relative Reliability of the Assessment of the Muscle Mechanical Properties of Pelvic Floor Muscles in Women with and without Urinary Incontinence. Diagnostics 2021, 11, 2315. https://doi.org/10.3390/diagnostics11122315
Rodrigues-de-Souza DP, Alcaraz-Clariana S, García-Luque L, Carmona-Pérez C, Garrido-Castro JL, Cruz-Medel I, Camargo PR, Alburquerque-Sendín F. Absolute and Relative Reliability of the Assessment of the Muscle Mechanical Properties of Pelvic Floor Muscles in Women with and without Urinary Incontinence. Diagnostics. 2021; 11(12):2315. https://doi.org/10.3390/diagnostics11122315
Chicago/Turabian StyleRodrigues-de-Souza, Daiana Priscila, Sandra Alcaraz-Clariana, Lourdes García-Luque, Cristina Carmona-Pérez, Juan Luis Garrido-Castro, Inés Cruz-Medel, Paula R. Camargo, and Francisco Alburquerque-Sendín. 2021. "Absolute and Relative Reliability of the Assessment of the Muscle Mechanical Properties of Pelvic Floor Muscles in Women with and without Urinary Incontinence" Diagnostics 11, no. 12: 2315. https://doi.org/10.3390/diagnostics11122315
APA StyleRodrigues-de-Souza, D. P., Alcaraz-Clariana, S., García-Luque, L., Carmona-Pérez, C., Garrido-Castro, J. L., Cruz-Medel, I., Camargo, P. R., & Alburquerque-Sendín, F. (2021). Absolute and Relative Reliability of the Assessment of the Muscle Mechanical Properties of Pelvic Floor Muscles in Women with and without Urinary Incontinence. Diagnostics, 11(12), 2315. https://doi.org/10.3390/diagnostics11122315








