Clinical Manifestations, Pathogenesis, Diagnosis and Treatment of Peripheral Neuropathies in Connective Tissue Diseases: More Diverse and Frequent in Different Subtypes than Expected
Abstract
1. Introduction
2. Methods
- Classification
- 2.
- Prevalence and clinical manifestations
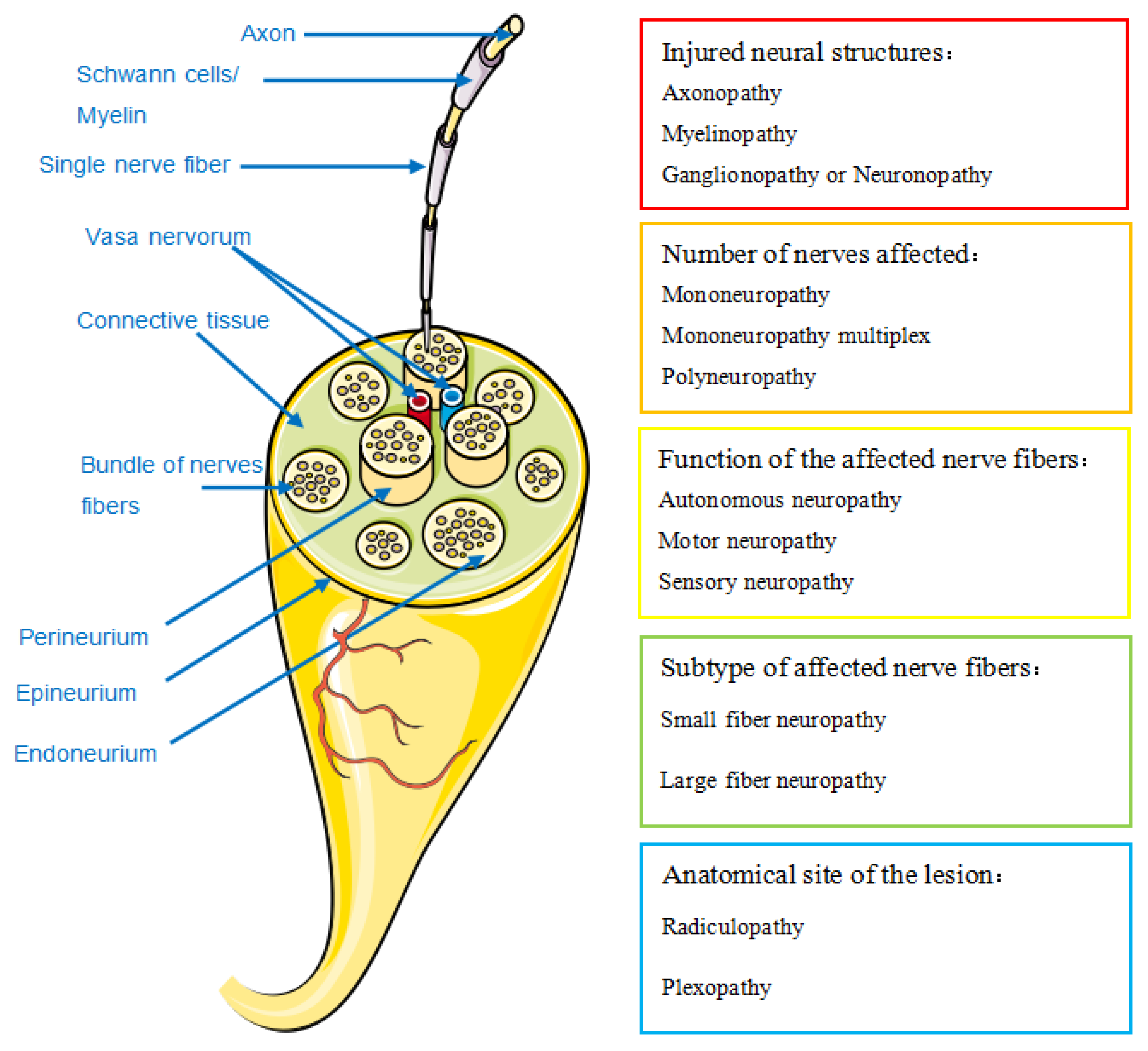
2.1. Systemic Lupus Erythematosus (SLE)
2.2. Sjögren’s Syndrome (SS)
2.3. Systemic Sclerosis (SSc) or Scleroderma
2.4. Polyarteritis Nodosa (PAN)
2.5. Antineutrophil Cytoplasmic Antibody (ANCA)-Associated Vasculitis
2.6. Rheumatoid Arthritis (RA)
2.7. Other CTDs
2.7.1. Giant Cell Arteritis (GCA) and Takayasu Arteritis (TAK)
2.7.2. Behçet Syndrome (BD)
2.7.3. Mixed Connective Tissue Disease (MCTD)
2.7.4. Dermatomyositis (DM) and Polymyositis (PM)
2.7.5. IgG4-Related Disease (IgG4-RD)
2.8. Peripheral Neuropathies Caused by Immunotherapy
| Authors | Prevalence/ Constituent Ratio (%) | Patients (N) | Type of Study | Main Electrodiagnostic Tests Pattern | Main Form of PN Manifestation |
|---|---|---|---|---|---|
| Systemic lupus erythematosus | |||||
| Xianbin et al. [8] | 1.5% | 4924 | Cross-sectional | Sensory (67.5%), motor (49.3%) | Polyneuropathy +++ Mononeuropathy ++ Cranial neuropathy ++ Myasthenia gravis ++ |
| Toledano et al. [10] | 17.7% | 524 | Cross-sectional | Sensory-motor (56%), axonal 80.3% | Polyneuropathy +++ Mononeuropathy ++ Cranial neuropathy + |
| Saigal et al. [11] | 36% | 50 | Cross-sectional | Sensory-motor, axonal | - |
| Bortoluzzi et al. [12] | 6.9% | 1224 | Cross-sectional | Sensory-motor (25%) | Polyneuropathy +++ Cranial neuropathy +++ Mononeuropathy ++ Mononeuritis multiplex + |
| Hanly et al. [13] | 7.6% | 1827 | Cohort | Sensory-motor (71%), sensory (16.1%) axonal (41.7%), demyelination (21.7%) | Polyneuropathy +++ Mononeuropathy ++ Cranial neuropathy ++ Mononeuritis multiplex ++ |
| Fargetti et al. [14] | 1.8% | 2074 | Cohort | Sensory-motor (68.4%), axonal (49.3%) | Polyneuropathy +++ Mononeuropathy ++ Polyradiculoneuropathy + Cranial neuropathy + |
| Sjögren’s syndrome | |||||
| Ye W et al. [17] | 19% pSS 31.1% sSS | 415 pSS 151 sSS | Cross-sectional | - | - |
| Seeliger et al. [18] | 44 SS + PNP | 108 PNP | Cross-sectional +case-control | Motor (100%), sensory (89%) axonal (36%), demyelinating (23%), both (41%) | - |
| Carvajal Alegria et al. [19] | 16% | 392 | Cohort | Sensory (57%), sensory-motor (33%) | Mononeuritis multiplex Polyneuropathy Cranial neuropathy |
| Przyńska-Mazan et al. [20] | 63.9% | 61 pSS | Cross-sectional | Sensory-motor axonal (47.5%), demyelination, both (5.1%) | Polyneuropathy +++ Mononeuropathy +++ Entrapment neuropathy ++ Mononeuritis multiplex ++ |
| Sireesha et al. [21] | - | 20 pSS 1 sSS | Cross-sectional | - | Mononeuritis multiplex +++ Ganglionopathy ++ Trigeminal neuropathy ++ |
| Jaskólska et al. [22] | 72% | 50 pSS | Cross-sectional | Sensory-motor axonal (22%) | Entrapment neuropathy +++ Mononeuropathy ++ Cranial neuropathy + |
| Jaskólska et al. [23] | 46% | 50 pSS | Cross-sectional | Sensory-motor (47%) | Mononeuropathy ++ Cranial neuropathy ++ |
| Systemic sclerosis (scleroderma) | |||||
| Raja et al. [27] | 36.6% | 60 | Cross-sectional | Sensory (65%), motor (53%) | Polyneuropathy +++ Mononeuropathy ++ Entrapment neuropathy ++ |
| Paik et al. [28] | 28% | 60 | Cross-sectional | Sensory-motor axonal, no demyelinating | - |
| * Yagci et al. [29] | 29.2% | 24 | Cross-sectional | - | Entrapment neuropathy Polyneuropathy |
| * Sriwong et al. [30] | 38% | 50 | Cohort | - | Median neuropathy at the wrist |
| Polyarteritis nodosa | |||||
| Sharma et al. [34] | 88.9% | 27 | Cross-sectional | Axonal sensory-motor (81.8%) | Mononeuritis multiplex |
| Eosinophilic granulomatosis with polyangiitis | |||||
| Bischof et al. [38] | 19% 23% 65% | 572 GPA 218 MPA 165 EPGA | Cross-sectional | Sensory-motor (32%), sensory (16%), motor (5%) | Mononeuritis multiplex +++ |
| Zhang et al. [39] | 46.4% | 110 EPGA | Retrospective cohort | - | Polyneuropathy +++ Mononeuritis multiplex ++ |
| Cho et al. [40] | 75% | 61 EPGA | Retrospective cohort | Sensory (44/46), motor (24/46) | Mononeuritis multiplex +++ Mononeuropathy ++ Polyneuropathy ++ |
| Nishi et al. [41] | - | 82 EPGA | Retrospective | Axonal | - |
| Rheumatoid Arthritis | |||||
| Kaeley et al. [49] | 75.28% | 89 | Cross-sectional | Asymmetrical sensorimotor axonal neuropathy, pure motor | Mononeuritis multiplex Entrapment neuropathy |
| Kumar et al. [50] | 34.4% (seropositive) 15.38% (seronegative) | 60 | Cross-sectional | - | - |
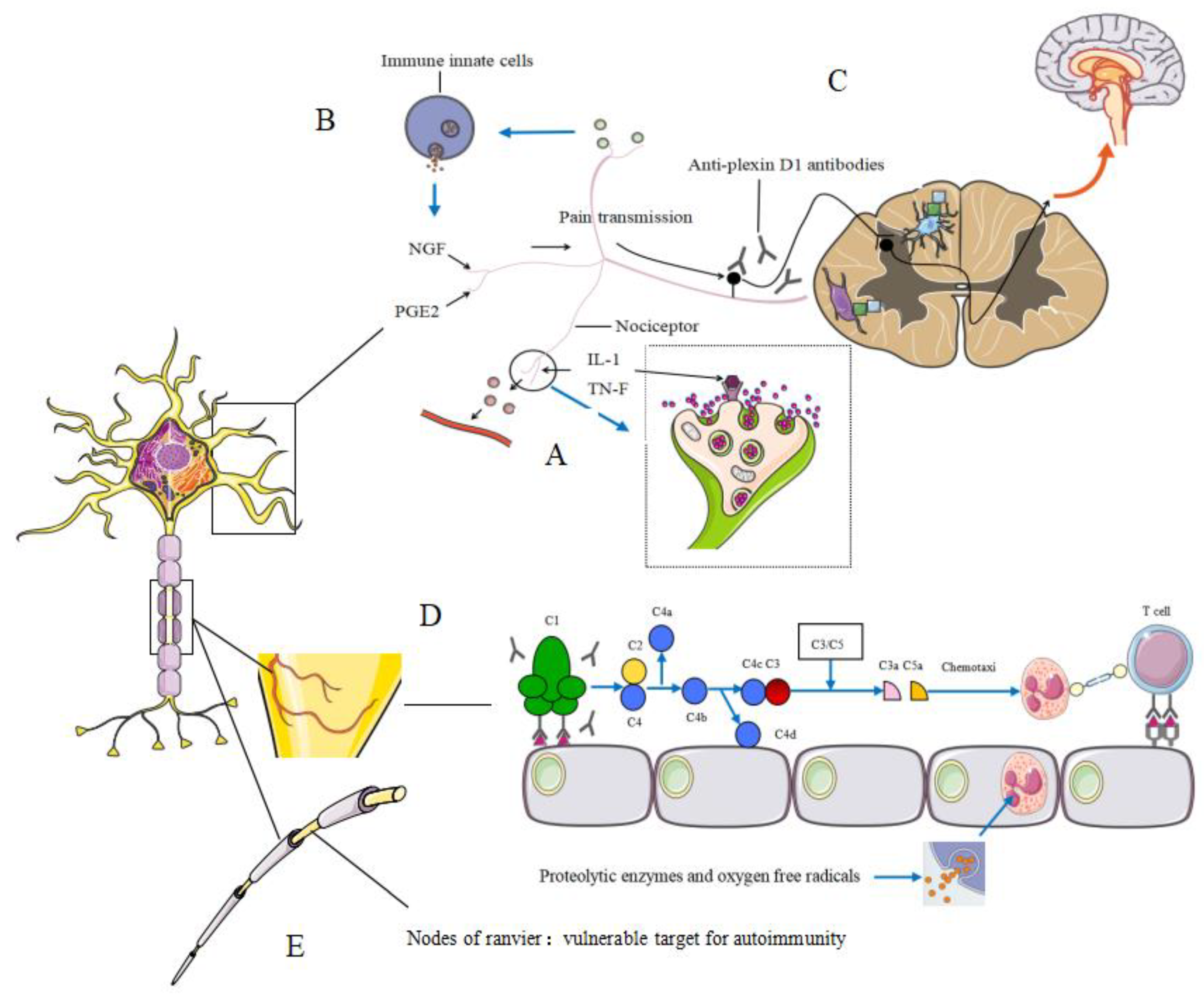
3. Pathogenesis
3.1. Peripheral Neuropathies Associated with Neurogenic Inflammation
3.2. Hypothetical Mechanisms Underlying Allergic Inflammation-Related Neuropathic Pain (NeP)
3.3. Vasculitic Neuropathy
3.4. Nodes of Ranvier and Autoantibodies
3.5. Metabolic Disorders
4. Diagnosis
4.1. Nerve Conduction Studies (NCS)
4.2. Laboratory Predictors
4.3. Histopathological Techniques
4.4. Diagnosis of Small Fibers Neuropathy
4.5. Modern Imaging Methods
5. New Treatments and Outcomes
5.1. Glucocorticoids with Immunosuppressants as Basic Therapy
5.2. Neuroactive Steroids in Neuropathic Pain
6. Conclusions
- The reported prevalence and clinical manifestation of PN in CTD varies widely;
- The main mechanism of PN is different for each CTD and the pathogenesis may also vary in different subtypes of the same disease;
- Neuroactive steroids may have protective effects against several PN features, especially neuropathic pain.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bougea, A.; Anagnostou, E.; Konstantinos, G.; George, P.; Triantafyllou, N.; Kararizou, E. A Systematic Review of Peripheral and Central Nervous System Involvement of Rheumatoid Arthritis, Systemic Lupus Erythematosus, Primary Sjögren’s Syndrome, and Associated Immunological Profiles. Int. J. Chronic Dis. 2015, 2015, 1–11. [Google Scholar] [CrossRef]
- Sommer, C.; Geber, C.; Young, P.; Forst, R.; Birklein, F.; Schoser, B. Polyneuropathies. Dtsch. Arztebl. Int. 2018, 115, 83–90. [Google Scholar] [CrossRef]
- Lerch, J.K.; Puga, D.A.; Bloom, O.; Popovich, P.G. Glucocorticoids and macrophage migration inhibitory factor (MIF) are neuroendocrine modulators of in-flammation and neuropathic pain after spinal cord injury. Semin. Immunol. 2014, 26, 409–414. [Google Scholar] [CrossRef]
- Falvo, E.; Diviccaro, S.; Melcangi, R.C.; Giatti, S. Physiopathological Role of Neuroactive Steroids in the Peripheral Nervous System. Int. J. Mol. Sci. 2020, 21, 9000. [Google Scholar] [CrossRef]
- Casteleyn, V.; Hahn, K.; Stenzel, W.; Siegert, E. Peripheral nerve involvement in rheumatic diseases. Zeitschrift fur Rheumatol. 2019, 78, 339–351. [Google Scholar] [CrossRef]
- Birnbaum, J.; Bingham, C.O., 3rd. Non-length-dependent and length-dependent small-fiber neuropathies associated with tumor necrosis factor (TNF)-inhibitor therapy in patients with rheumatoid arthritis: Expanding the spectrum of neurological disease associated with TNF-inhibitors. Semin. Arthritis Rheum. 2014, 43, 638–647. [Google Scholar] [CrossRef]
- Bortoluzzi, A.; Silvagni, E.; Furini, F.; Piga, M.; Govoni, M. Peripheral nervous system involvement in systemic lupus erythematosus: A review of the evidence. Clin. Exp. Rheumatol. 2018, 37, 146–155. [Google Scholar] [PubMed]
- Xianbin, W.; Mingyu, W.; Dong, X.; Huiying, L.; Yan, X.; Fengchun, Z.; Xiaofeng, Z. Peripheral Neuropathies Due to Systemic Lupus Erythematosus in China. Medicine 2015, 94, e625. [Google Scholar] [CrossRef] [PubMed]
- Shaban, A.; Leira, E.C. Neurological Complications in Patients with Systemic Lupus Erythematosus. Curr. Neurol. Neurosci. Rep. 2019, 19, 97. [Google Scholar] [CrossRef] [PubMed]
- Toledano, P.; Orueta, R.; Rodríguez-Pintó, I.; Valls-Solé, J.; Cervera, R.; Espinosa, G. Peripheral nervous system involvement in systemic lupus erythematosus: Prevalence, clinical and immunological characteristics, treatment and outcome of a large cohort from a single centre. Autoimmun. Rev. 2017, 16, 750–755. [Google Scholar] [CrossRef] [PubMed]
- Saigal, R.; Bhargav, R.; Goyal, L.; Agrawal, A.; Mital, P.; Wadhwani, D. Peripheral Neuropathy in Systemic Lupus Erythematosus: Clinical and Electrophysiological Properties and their Association with Disease Activity Parameters. J. Assoc. Physicians India 2015, 63, 15–19. [Google Scholar]
- Bortoluzzi, A.; Piga, M.; Silvagni, E.; Chessa, E.; Mathieu, A.; Govoni, M. Peripheral nervous system involvement in systemic lupus erythematosus: A retrospective study on prev-alence, associated factors and outcome. Lupus 2019, 28, 465–474. [Google Scholar] [CrossRef]
- Hanly, J.G.; Li, Q.; Su, L.; Urowitz, M.B.; Gordon, C.; Bae, S.C. Peripheral Nervous System Disease in Systemic Lupus Erythematosus: Results From an International In-ception Cohort Study. Arthritis Rheumatol. 2020, 72, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Fargetti, S.; Ugolini-Lopes, M.R.; Pasoto, S.G.; Seguro, L.P.; Shinjo, S.K.; Bonfa, E.; Borba, E.F. Short- and Long-Term Outcome of Systemic Lupus Erythematosus Peripheral Neuropathy: Bimodal Pattern of Onset and Treatment Response. J. Clin. Rheumatol. 2021, 27, S212–S216. [Google Scholar] [CrossRef]
- Zhang, N.; Cao, J.; Zhao, M.; Sun, L. The introspection on the diagnosis and treatment process of a case of Guillain-Barré syndrome (GBS) at-tributed to systemic lupus erythematosus (SLE): A case report. Medicine 2017, 96, e9037. [Google Scholar] [CrossRef]
- Patil, A.D.; Karnik, N.D.; Nadkar, M.Y.; Gupta, V.A.; Muralidhara, K.; Passidhi, S. Guillain Barré Syndrome, Systemic Lupus Erythematosus and Acute Intermittent Porphyria–A Deadly Trio. J. Assoc. Physicians India 2015, 63, 60–63. [Google Scholar] [PubMed]
- Ye, W.; Chen, S.; Huang, X.; Qin, W.; Zhang, T.; Zhu, X.; Zhu, X.; Lin, C.; Wang, X. Clinical features and risk factors of neurological involvement in Sjögren’s syndrome. BMC Neurosci. 2018, 19, 26. [Google Scholar] [CrossRef]
- Seeliger, T.; Prenzler, N.K.; Gingele, S.; Seeliger, B.; Körner, S.; Thiele, T. Neuro-Sjögren: Peripheral Neuropathy with Limb Weakness in Sjögren’s Syndrome. Front. Immunol. 2019, 10, 1600. [Google Scholar] [CrossRef]
- Alegria, G.C.; Guellec, D.; Mariette, X.; Gottenberg, J.-E.; Dernis, E.; Dubost, J.-J.; Trouvin, A.-P.; Hachulla, E.; Larroche, C.; Le Guern, V.; et al. Epidemiology of neurological manifestations in Sjögren’s syndrome: Data from the French ASSESS Cohort. RMD Open 2016, 2, e000179. [Google Scholar] [CrossRef] [PubMed]
- Perzyńska-Mazan, J.; Maślińska, M.; Gasik, R. Neurophysiological Features of Peripheral Nervous System Involvement and Immunological Profile of Patients with Primary Sjögren Syndrome. J. Rheumatol. 2020, 47, 1661–1667. [Google Scholar] [CrossRef] [PubMed]
- Kanikannan, M.A.; Sireesha, Y.; Pyal, A.; Sandeep, G.; Uppin, M.S.; Kandadai, R.M.; Jabeen, S.A.; Varaprasad, R.; Rajasekhar, L.; Neeharika, M.L.; et al. Patterns of peripheral neuropathy in Sjogren’s syndrome in a tertiary care hospital from South India. Neurol. India 2019, 67, 94–S99. [Google Scholar] [CrossRef] [PubMed]
- Jaskólska, M.; Chylińska, M.; Masiak, A.; Siemiński, M.; Ziętkiewicz, M.; Czuszyńska, Z. Neuro-Sjögren: Uncommon or underestimated problem? Brain Behav. 2020, 10, e01665. [Google Scholar] [CrossRef]
- Jaskólska, M.; Chylińska, M.; Masiak, A.; Nowicka-Sauer, K.; Siemiński, M.; Ziętkiewicz, M.; Czuszyńska, Z.; Zdrojewski, Z. Peripheral neuropathy and health-related quality of life in patients with primary Sjögren’s syndrome: A preliminary report. Rheumatol. Int. 2020, 40, 1267–1274. [Google Scholar] [CrossRef]
- Alegria, G.C.; Guellec, D.; Devauchelle-Pensec, V.; Saraux, A. Is there specific neurological disorders of primary Sjögren’s syndrome? Jt. Bone Spine 2015, 82, 86–89. [Google Scholar] [CrossRef]
- Alunno, A.; Carubbi, F.; Bartoloni, E.; Cipriani, P.; Giacomelli, R.; Gerli, R. The kaleidoscope of neurological manifestations in primary Sjögren’s syndrome. Clin. Exp. Rheumatol. 2019, 118, 192–198. [Google Scholar]
- Sivadasan, A.; Muthusamy, K.; Patel, B.; Benjamin, R.; Prabhakar, A.T.; Mathew, V.; Aaron, S.; Alexander, M. Clinical Spectrum, Therapeutic Outcomes, and Prognostic Predictors in Sjogren’s Syndrome-associated Neuropathy. Ann. Indian Acad. Neurol. 2017, 20, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Raja, J.; Balaikerisnan, T.; Ramanaidu, L.P.; Goh, K.J. Large fiber peripheral neuropathy in systemic sclerosis: A prospective study using clinical and electrophysio-logical definition. Int. J. Rheum. Dis. 2021, 24, 347–354. [Google Scholar] [CrossRef]
- Paik, J.J.; Mammen, A.L.; Wigley, F.M.; Shah, A.A.; Hummers, L.K.; Polydefkis, M. Symptomatic and Electrodiagnostic Features of Peripheral Neuropathy in Scleroderma. Arthritis Rheum. 2015, 68, 1150–1157. [Google Scholar] [CrossRef] [PubMed]
- Yagci, I.; Kenis-Coskun, O.; Ozsoy, T.; Ozen, G.; Direskeneli, H. Increased stiffness of median nerve in systemic sclerosis. BMC Musculoskelet. Disord. 2017, 18, 434. [Google Scholar] [CrossRef]
- Sriwong, P.T.; Sirasaporn, P.; Foochareon, C.; Srichompoo, K. Median neuropathy at the wrist in patients with systemic sclerosis: Two-year follow-up study. Reumatologia 2018, 56, 294–300. [Google Scholar] [CrossRef]
- Karakulak, U.N.; Okutucu, S.; Şahiner, M.L.; Maharjan, N.; Aladag, E.; Akdogan, A.; Kilic, L.; Kaya, E.B.; Aytemir, K.; Tokgozoglu, L. Assessment of cardiac autonomic nervous system involvement in systemic sclerosis via exercise heart rate recovery. Med. Princ. Pr. 2014, 24, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Tadic, M.; Zlatanovic, M.; Cuspidi, C.; Stevanovic, A.; Celic, V.; Damjanov, N.; Kocijancic, V. Systemic sclerosis impacts right heart and cardiac autonomic nervous system. J. Clin. Ultrasound 2018, 46, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, G.D.; Tobaldini, E.; Bellocchi, C.; Santaniello, A.; Caronni, M.; Severino, A.; Froldi, M.; Beretta, L.; Soares, P.P.D.S.; Montano, N. Cardiac autonomic modulation at rest and during orthostatic stress among different systemic sclerosis subsets. Eur. J. Intern. Med. 2019, 66, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Pinto, B.; Dhooria, A.; Rathi, M.; Singhal, M.; Dhir, V.; Sharma, K.; Parkash, M.; Modi, M.; Vijayvergiya, R.; et al. Polyarteritis nodosa in north India: Clinical manifestations and outcomes. Int. J. Rheum. Dis. 2017, 20, 390–397. [Google Scholar] [CrossRef]
- de Boysson, H.; Guillevin, L. Polyarteritis Nodosa Neurologic Manifestations. Neurol. Clin. 2019, 37, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Imboden, J.B. Involvement of the Peripheral Nervous System in Polyarteritis Nodosa and Antineutrophil Cytoplasmic Anti-bodies-Associated Vasculitis. Rheum. Dis. Clin. North. Am. 2017, 43, 633–639. [Google Scholar] [CrossRef]
- James, J.; Jose, J.; Thulaseedharan, N.K. Acute Necrotizing Vasculitic Neuropathy due to Polyarteritis Nodosa. Oman Med. J. 2018, 33, 253–255. [Google Scholar] [CrossRef]
- Bischof, A.; Jaeger, V.K.; Hadden, R.D.; Luqmani, R.A.; Pröbstel, A.K.; Merkel, P.A. Peripheral neuropathy in antineutrophil cytoplasmic antibody-associated vasculitides: Insights from the DCVAS study. Neurol. Neuroimmunol. Neuroinflamm. 2019, 6, e615. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, S.; Guo, L.; Wang, L.; Wu, Q.; Zheng, W. Clinical Characteristics of Peripheral Neuropathy in Eosinophilic Granulomatosis with Polyangiitis: A Ret-rospective Single-Center Study in China. J. Immunol. Res. 2020, 2020, 3530768. [Google Scholar] [CrossRef]
- Cho, H.-J.; Yune, S.; Seok, J.M.; Bin Cho, E.; Min, J.-H.; Seo, Y.L.; Lee, B.-J.; Kim, B.J.; Choi, D.-C. Clinical Characteristics and Treatment Response of Peripheral Neuropathy in the Presence of Eosinophilic Granulomatosis with Polyangiitis (Churg-Strauss Syndrome): Experience at a Single Tertiary Center. J. Clin. Neurol. 2017, 13, 77–83. [Google Scholar] [CrossRef]
- Nishi, R.; Koike, H.; Ohyama, K.; Fukami, Y.; Ikeda, S.; Kawagashira, Y.; Iijima, M.; Katsuno, M.; Sobue, G. Differential clinicopathologic features of EGPA-associated neuropathy with and without ANCA. Neurol. 2020, 94, e1726–e1737. [Google Scholar] [CrossRef]
- Moog, P.; Eren, O.; Witt, M.; Rauschel, V.; Kossegg, S.; Straube, A.; Grünke, M.; Schulze-Koops, H. Assessment of autonomic function in a cohort of patients with ANCA-associated vasculitis. Clin. Auton. Res. 2016, 26, 279–285. [Google Scholar] [CrossRef]
- Murata, K.; Endo, K.; Nishimura, H.; Tanaka, H.; Shishido, T.; Yamamoto, K. Eosinophilic granulomatosis with polyangiitis presenting as acute sciatic nerve neuropathy resembling lumbar disease. J. Orthop. Sci. 2015, 20, 224–228. [Google Scholar] [CrossRef]
- Camaralemarroy, C.R.; Infante-Valenzuela, A.; Villareal-Montemayor, H.J.; Soto-Rincon, C.A.; Davila-Olalde, J.A.; Villareal-Velazquez, H.J. Eosinophilic Granulomatosis with Polyangiitis Presenting as Acute Polyneuropathy Mimicking Guillain-Barre Syndrome. Case Rep. Neurol. Med. 2015, 2015, 1–3. [Google Scholar] [CrossRef]
- Wang, L.; Li, J.; Qian, M.; Zheng, W.; Wu, Q.; Zhang, W.; Tian, X.; Zhang, F. Clinical features of microscopic polyangiitis associated with peripheral neuropathy. Zhonghua Yi Xue Za Zhi 2015, 95, 2190–2193. [Google Scholar] [PubMed]
- Arienti, F.; Franco, G.; Monfrini, E.; Santaniello, A.; Bresolin, N.; Saetti, M.C.; Di Fonzo, A. Microscopic Polyangiitis With Selective Involvement of Central and Peripheral Nervous System: A Case Report. Front. Neurol. 2020, 11, 269. [Google Scholar] [CrossRef] [PubMed]
- Bougea, A.; Anagnostou, E.; Spandideas, N.; Triantafyllou, N.; Kararizou, E. An update of neurological manifestations of vasculitides and connective tissue diseases: A literature review. Einstein 2015, 13, 627–635. [Google Scholar] [CrossRef]
- Geier, C.; Steed, K. ‘Carpal tunnel syndrome’ and ‘tennis elbow’ as prodromes for granulomatosis with polyangiitis (for-merly Wegener’s granulomatosis). BMJ Case Rep. 2019, 12, bcr-2018. [Google Scholar] [CrossRef] [PubMed]
- Kaeley, N.; Ahmad, S.; Pathania, M.; Kakkar, R. Prevalence and patterns of peripheral neuropathy in patients of rheumatoid arthritis. J. Fam. Med. Prim. Care 2019, 8, 22–26. [Google Scholar] [CrossRef]
- Kumar, B.; Das, M.P.; Misra, A.K. A cross-sectional study of association of serostatus and extra-articular manifestations in patients with rheumatoid arthritis in a teaching hospital. J. Fam. Med. Prim. Care 2020, 9, 2789–2793. [Google Scholar] [CrossRef]
- Cojocaru, I.M.; Cojocaru, M.; Silosi, I.; Vrabie, C.D. Peripheral Nervous System Manifestations in Systemic Autoimmune Diseases. Maedica J. Clin. Med. 2014, 9, 289–294. [Google Scholar]
- Syngle, V.; Syngle, A.; Garg, N.; Krishan, P.; Verma, I. Predictors of autonomic neuropathy in rheumatoid arthritis. Auton. Neurosci. 2016, 201, 54–59. [Google Scholar] [CrossRef]
- DeQuattro, K.; Imboden, J.B. Neurologic Manifestations of Rheumatoid Arthritis. Rheum. Dis. Clin. North. Am. 2017, 43, 561–571. [Google Scholar] [CrossRef]
- Duval, F.; Lacoste, I.; Galli, G.; Chaumont, H.; Solé, G.; Léger, F.; Damon-Perrière, N.; Rouanet, M.; Le Masson, G.; Mathis, S. Acute Brachial Radiculoplexopathy and Giant Cell Arteritis. Neurologist 2018, 23, 23–28. [Google Scholar] [CrossRef]
- Calle-Lopez, Y.; Fernandez-Ramirez, A.F.; Franco-Dager, E.; Gomez-Lopera, J.G.; Vanegas-Garcia, A.L. «Man-in-the-barrel» syndrome: Atypical manifestation of giant cell arteritis. Rev. Neurol. 2018, 66, 373–376. [Google Scholar]
- Conway, R.; Kinsella, J.A.; Molloy, E.S. Peroneal neuropathy in giant cell arteritis. Rheumatol. 2016, 56, 169–170. [Google Scholar] [CrossRef][Green Version]
- Kim, D.; Roche-Nagle, G. Axillary artery aneurysm combined with brachial plexus palsy due to Takayasu arteritis. BMJ Case Rep. 2018, 2018. [Google Scholar] [CrossRef]
- Lee, J.; Cho, S.; Zheng, Z.; Park, H.; Bang, D. Carpal Tunnel Syndrome in Behçet’s Disease. Yonsei Med. J. 2015, 56, 1015–1020. [Google Scholar] [CrossRef]
- Hao, Y.; Feng, L.; Teng, Y.; Cheng, Y.; Feng, J. Management of multiple neurological complications in mixed connective tissue disease: A case report. Medicine 2018, 97, e11360. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.J.; Li, Y.H.; Zhang, X.W.; Chen, S.; Liu, Y.Y. Clinical analysis of 12 cases of mixed connective tissue disease-associated trigeminal neuropathy. Zhonghua Yi Xue Za Zhi 2020, 100, 938–941. [Google Scholar] [PubMed]
- Jasińska, D.; Boczon, J. Melkersson–Rosenthal syndrome as an early manifestation of mixed connective tissue disease. Eur. J. Med. Res. 2015, 20, 1–3. [Google Scholar] [CrossRef]
- Naqvi, S.; Talib, V.; Aijaz, R.; Ali, Z.; Bashir, S.; Ahmad, S.M.; Naveed, S. Autoamputation and Polyneuropathy in Mixed Connective Tissue Disorder: A Case Report. Cureus 2017, 9, e1313. [Google Scholar] [CrossRef]
- Irie, T.; Shigeto, H.; Koge, J.; Yamaguchi, H.; Murai, H.; Kira, J.-I. Dermatomyositis complicated with asymmetric peripheral neuritis on exacerbation: A case report and literature review. Clin. Exp. Neuroimmunol. 2016, 7, 373–380. [Google Scholar] [CrossRef]
- AbdelRazek, A.M.; Venna, N.; Stone, J.H. IgG4-related disease of the central and peripheral nervous systems. Lancet Neurol. 2018, 17, 183–192. [Google Scholar] [CrossRef]
- Zhan, L.; Fan, M.; Cai, N.; Cai, B. Combination of autoimmune pancreatitis and peripheral neuropathy on an IgG4-related disease patient with 4 years following-up. J. Neuroimmunol. 2020, 348, 577378. [Google Scholar] [CrossRef]
- Lin, Y.-J.; Anzaghe, M.; Schülke, S. Update on the Pathomechanism, Diagnosis, and Treatment Options for Rheumatoid Arthritis. Cells 2020, 9, 880. [Google Scholar] [CrossRef] [PubMed]
- Pardanani, A.; Laborde, R.R.; Lasho, T.L.; Finke, C.; Begna, K.; Al-Kali, A.; Hogan, W.J.; Litzow, M.R.; Leontovich, A.; Kowalski, M.; et al. Safety and efficacy of CYT387, a JAK1 and JAK2 inhibitor, in myelofibrosis. Leukemia 2013, 27, 1322–1327. [Google Scholar] [CrossRef] [PubMed]
- Etminan, M.; Sodhi, M.; Samii, A.; Carleton, B.; Kezouh, A.; Avina-Zubieta, J.A. Tumor necrosis factor inhibitors and risk of peripheral neuropathy in patients with rheumatic diseases. Semin. Arthritis Rheum. 2018, 48, 1083–1086. [Google Scholar] [CrossRef] [PubMed]
- Tsouni, P.; Bill, O.; Truffert, A.; Liaudat, C.; Ochsner, F.; Steck, A.J.; Kuntzer, T. Anti-TNF alpha medications and neuropathy. J. Peripher. Nerv. Syst. 2015, 20, 397–402. [Google Scholar] [CrossRef]
- Zinebi, A.; Akhouad, Y.; Rkiouak, A.; Reggad, A.; Kasmy, Z.; Boudlal, M. Peripheral neuropathy during Infliximab therapy: A case study. Pan Afr. Med. J. 2016, 24, 271. [Google Scholar]
- Fernández-Menéndez, S.; Nafría, N.G.; Redondo-Robles, L.; Sierra-Ausín, M.; García-Santiago, R.; Saponaro-González, A. Multifocal-motor-neuropathy-like disease associated with Infliximab treatment in a patient with Crohn’s disease. J. Neurol. Sci. 2015, 349, 246–248. [Google Scholar] [CrossRef] [PubMed]
- Psarelis, S.; Hajineocli, A.P.; Hadjicosta, E.; Elliott HS, A.; Johnson, P. Is secukinumab a safe alternative treatment for ankylosing spondylitis with Guillain Barré syndrome after anti-TNF-α treatment? Case report and literature review. Clin. Rheumatol. 2017, 36, 1197–1199. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Yamasaki, R.; Kira, J.-I. Novel Neuropathic Pain Mechanisms Associated With Allergic Inflammation. Front. Neurol. 2019, 10, 1337. [Google Scholar] [CrossRef]
- Hawke, S.H.B.; Davies, L.; Pamphlett, R.; Guo, Y.P.; Pollard, J.D.; McLeod, J.G. Vasculitic neuropathy. Acta Reumatol. Port. 2011, 36, 102–109. [Google Scholar] [CrossRef]
- Spirin, N.; Bulanova, V.; Pizova, N.V.; Shilkina, N.P. Peripheral nervous system lesion syndromes and the mechanisms of their formation in connective tissue diseases. Neurosci. Behav. Physiol. 2007, 37, 1–6. [Google Scholar] [CrossRef]
- Garg, N.; Park, S.B.; Yiannikas, C.; Vucic, S.; Howells, J.; Noto, Y.-I.; Mathey, E.K.; Pollard, J.D.; Kiernan, M.C. Neurofascin-155 IGG4 Neuropathy: Pathophysiological Insights, Spectrum of Clinical Severity and Response To treatment. Muscle Nerve 2018, 57, 848–851. [Google Scholar] [CrossRef]
- Karam, C. Peripheral Neuropathies Associated With Vasculitis and Autoimmune Connective Tissue Disease. Contin. Lifelong Learn. Neurol. 2020, 26, 1257–1279. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, L.; Zhang, Z.; Li, H.; Jiang, L.; Wang, L.; Li, Z. Clinical characteristics of rheumatoid arthritis patients with peripheral neuropathy and potential related risk factors. Clin. Rheumatol. 2019, 38, 2099–2107. [Google Scholar] [CrossRef]
- Ohyama, K.; Koike, H.; Takahashi, M.; Kawagashira, Y.; Iijima, M.; Watanabe, H.; Sobue, G. Immunoglobulin G4-related pathologic features in inflammatory neuropathies. Neurology 2015, 85, 1400–1407. [Google Scholar] [CrossRef] [PubMed]
- Yesil, H.; Sungur, U.; Akdeniz, S.; Gurer, G.; Yalcın, B.; Dundar, U. Association between serum vitamin D levels and neuropathic pain in rheumatoid arthritis patients: A cross-sectional study. Int. J. Rheum. Dis. 2018, 21, 431–439. [Google Scholar] [CrossRef]
- Provitera, V.; Gibbons, C.H.; Wendelschafer-Crabb, G.; Donadio, V.; Vitale, D.F.; Loavenbruck, A.; Stancanelli, A.; Caporaso, G.; Liguori, R.; Wang, N.; et al. The role of skin biopsy in differentiating small-fiber neuropathy from ganglionopathy. Eur. J. Neurol. 2018, 25, 848–853. [Google Scholar] [CrossRef]
- Sommer, C. Nerve and skin biopsy in neuropathies. Curr. Opin. Neurol. 2018, 31, 534–540. [Google Scholar] [CrossRef]
- Nolano, M.; Tozza, S.; Caporaso, G.; Provitera, V. Contribution of Skin Biopsy in Peripheral Neuropathies. Brain Sci. 2020, 10, 989. [Google Scholar] [CrossRef]
- Terkelsen, A.J.; Karlsson, P.; Lauria, G.; Freeman, R.; Finnerup, N.; Jensen, T.S. The diagnostic challenge of small fibre neuropathy: Clinical presentations, evaluations, and causes. Lancet Neurol. 2017, 16, 934–944. [Google Scholar] [CrossRef]
- Lauria, G.; Hsieh, S.-T.; Johansson, O.; Kennedy, W.R.; Leger, J.M.; Mellgren, S.I.; Nolano, M.; Merkies, I.S.J.; Polydefkis, M.; Smith, A.G.; et al. European Federation of Neurological Societies/Peripheral Nerve Society Guideline on the use of skin biopsy in the diagnosis of small fiber neuropathy. Report of a joint task force of the European Fe-deration of Neurological Societies and the Peripheral Nerve Society. Eur. J. Neurol. 2010, 17, 903-e49. [Google Scholar] [CrossRef] [PubMed]
- Devigili, G.; Rinaldo, S.; Lombardi, R.; Cazzato, D.; Marchi, M.; Salvi, E.; Eleopra, R.; Lauria, G. Diagnostic criteria for small fibre neuropathy in clinical practice and research. Brain 2019, 142, 3728–3736. [Google Scholar] [CrossRef] [PubMed]
- Lefaucheur, J.-P.; Wahab, A.; Planté-Bordeneuve, V.; Sène, D.; Ménard-Lefaucheur, I.; Rouie, D.; Tebbal, D.; Salhi, H.; Créange, A.; Zouari, H.; et al. Diagnosis of small fiber neuropathy: A comparative study of five neurophysiological tests. Neurophysiol. Clin. Neurophysiol. 2015, 45, 445–455. [Google Scholar] [CrossRef]
- Choi, S.-J.; Ahn, J.H.; Ryu, D.S.; Kang, C.H.; Jung, S.M.; Park, M.S.; Shin, D.-R. Ultrasonography for nerve compression syndromes of the upper extremity. Ultrasonography 2015, 34, 275–291. [Google Scholar] [CrossRef] [PubMed]
- Bignotti, B.; Ghio, M.; Panico, N.; Tagliafico, G.; Martinoli, C.; Tagliafico, A. High-resolution ultrasound of peripheral nerves in systemic sclerosis: A pilot study of computer-aided quantitative assessment of nerve density. Skelet. Radiol. 2015, 44, 1761–1767. [Google Scholar] [CrossRef]
- Anno, S.; Okano, T.; Mamoto, K.; Sugioka, Y.; Takeda, S.; Hashimoto, A.; Yamashita, E.; Morinaka, R.; Ueda, H.; Inui, K.; et al. Comparison of median nerve stiffness with and without rheumatoid arthritis by ultrasound real-time tissue elastography: A propensity score matching study. Mod. Rheumatol. 2019, 30, 481–488. [Google Scholar] [CrossRef]
- Bao, H.; Wang, S.; Wang, G.; Yang, L.; Hasan, M.-U.; Yao, B.; Wu, C.; Zhang, X.; Chen, W.; Chan, Q.; et al. Diffusion-weighted MR neurography of median and ulnar nerves in the wrist and palm. Eur. Radiol. 2017, 27, 2359–2366. [Google Scholar] [CrossRef] [PubMed]
- Collins, M.P.; Dyck, P.J.B.; Gronseth, G.S.; Guillevin, L.; Hadden, R.D.; Heuss, D. Peripheral Nerve Society Guideline on the classification, diagnosis, investigation, and immunosuppres-sive therapy of non-systemic vasculitic neuropathy: Executive summary. J. Peripher. Nerv. Syst. 2010, 15, 176–184. [Google Scholar] [CrossRef]
- Ding, Y.Q.; Zhang, L.; Zeng, H.L.; Liu, S.Y. Clinical analysis of 46 rheumatoid arthritis patients with peripheral neuropathy. Zhonghua Nei Ke Za Zhi 2021, 60, 222–226. [Google Scholar] [PubMed]
- Govoni, M.; Bortoluzzi, A.; Padovan, M.; Silvagni, E.; Borrelli, M.; Donelli, F.; Ceruti, S.; Trotta, F. The diagnosis and clinical management of the neuropsychiatric manifestations of lupus. J. Autoimmun. 2016, 74, 41–72. [Google Scholar] [CrossRef] [PubMed]
| Associated Factors |
|---|
| Systemic lupus erythematosus |
| Older age at SLE diagnosis [10,12,13] |
| Higher SLE Disease Activity Index 2000 scores [11,12,13,14] |
| Cutaneous vasculitis [12,14] |
| Sjögren’s syndrome |
| Salivary gland enlargement [22,26] |
| Hypocomplementemia [17,22] |
| Eosinophilic granulomatosis with polyangiitis |
| Myeloperoxidase-ANCA positivity [38,39] |
| Skin, musculoskeletal, cardiovascular involvement [38] |
| Rheumatoid arthritis |
| DAS-28 [49] |
| Inflammatory markers of disease activity [49,78] |
| Increased platelet numbers [78] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, L.; Liu, Y. Clinical Manifestations, Pathogenesis, Diagnosis and Treatment of Peripheral Neuropathies in Connective Tissue Diseases: More Diverse and Frequent in Different Subtypes than Expected. Diagnostics 2021, 11, 1956. https://doi.org/10.3390/diagnostics11111956
Jin L, Liu Y. Clinical Manifestations, Pathogenesis, Diagnosis and Treatment of Peripheral Neuropathies in Connective Tissue Diseases: More Diverse and Frequent in Different Subtypes than Expected. Diagnostics. 2021; 11(11):1956. https://doi.org/10.3390/diagnostics11111956
Chicago/Turabian StyleJin, Lei, and Yu Liu. 2021. "Clinical Manifestations, Pathogenesis, Diagnosis and Treatment of Peripheral Neuropathies in Connective Tissue Diseases: More Diverse and Frequent in Different Subtypes than Expected" Diagnostics 11, no. 11: 1956. https://doi.org/10.3390/diagnostics11111956
APA StyleJin, L., & Liu, Y. (2021). Clinical Manifestations, Pathogenesis, Diagnosis and Treatment of Peripheral Neuropathies in Connective Tissue Diseases: More Diverse and Frequent in Different Subtypes than Expected. Diagnostics, 11(11), 1956. https://doi.org/10.3390/diagnostics11111956






