Laparoscopic Surgery in COVID-19 Era—Safety and Ethical Issues
Abstract
1. Introduction
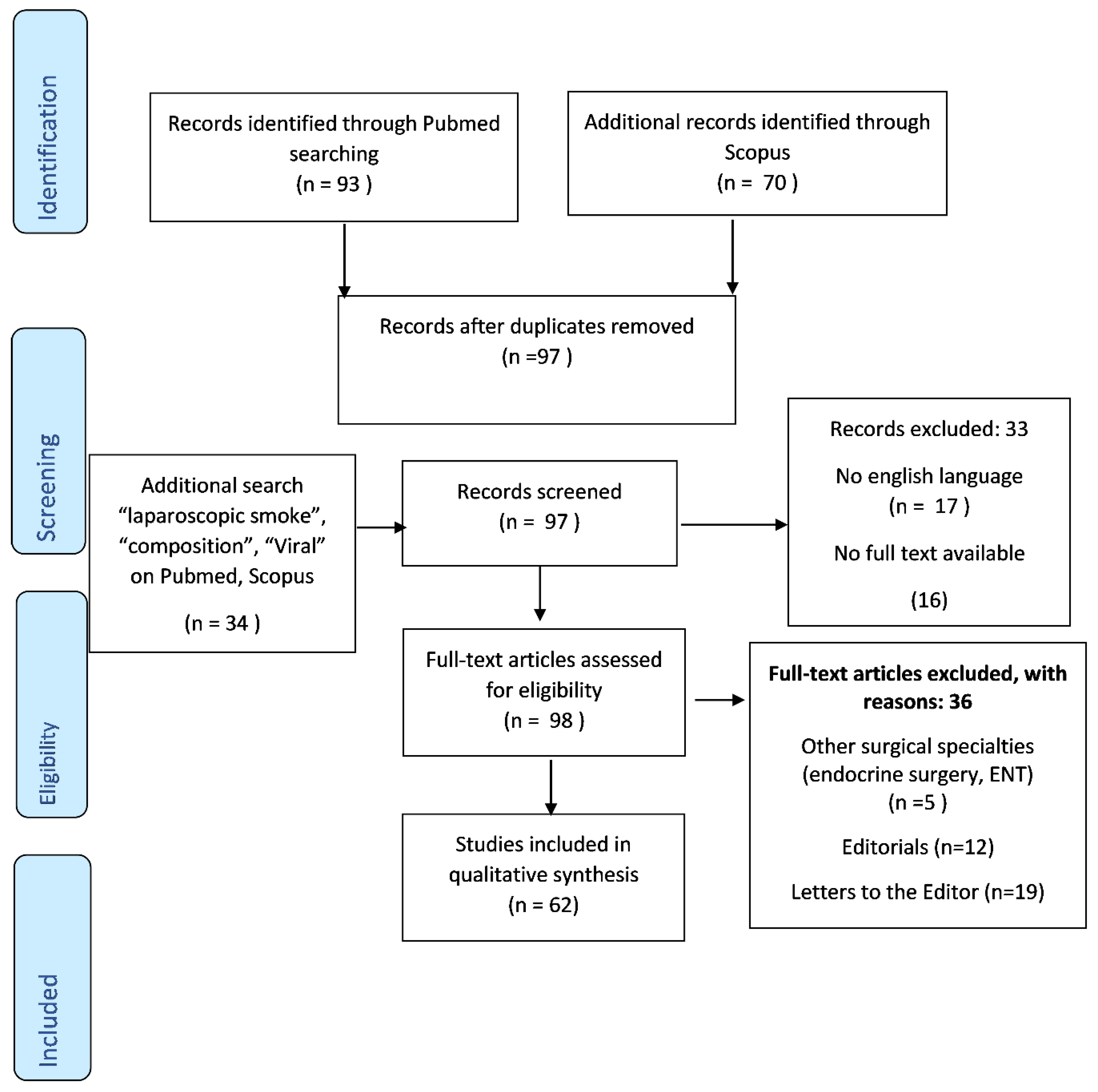
2. Materials and Methods
3. Results
3.1. Laparoscopy and Aerosolization
3.2. Health Risks Related to Surgical Smoke
3.3. Viral Transmission in Open Versus Laparoscopic Surgery
3.4. Clinical Evidences Regarding Laparoscopy in COVID-19 Positive Patients
3.5. Regulations for Increasing Safety in Laparoscopic Surgery
3.6. The Impact of the Novel Guidelines upon Surgical Practice
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dascalu, A.M.; Tudosie, M.S.; Smarandache, G.C.; Serban, D. Impact of the COVID-19 pandemic upon the ophthalmological clinical practice. Rom. J. Leg. Med. 2020, 28, 96–100. [Google Scholar] [CrossRef]
- Mallapay, S. How deadly is the coronavirus? Scientists are close to an answer. Nature 2020, 582, 467–468. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Huang, T.; Wang, Y.Q.; Wang, Z.P.; Liang, Y.; Huang, T.B.; Zhang, H.Y.; Sun, W.; Wang, Y. COVID-19 patients’ clinical characteristics, discharge rate and fatality rate of meta-analysis. J. Med. Virol. 2020, 92, 577–583. [Google Scholar] [CrossRef]
- Tran, K.; Cimon, K.; Severn, M.; Pessoa-Silva, C.L.; Conly, J. Aerosol generating procedures and risk of transmission of acute respiratory infections to healthcare workers: A systematic review. PLoS ONE 2012, 7, e35797. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.P.; Vunnam, S.R.; Patel, P.A.; Krill, K.L.; Korbitz, P.M.; Gallagher, J.P.; Suh, J.E.; Vunnam, R.R. Transmission of SARS-CoV-2: An update of current literature. Eur. J. Clin. Microbiol. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Shabbir, A.; Menon, R.K.; Somani, J.; So, J.B.; Ozman, M.; Chiu, P.W.; Lomanto, D. ELSA recommendations for minimally invasive surgery during a community spread pandemic: A centered approach in Asia from widespread to recovery phases. Surg. Endosc. 2020, 34, 3292–3297. [Google Scholar] [CrossRef]
- Francis, N.; Dort, J.; Cho, E.; Feldman, L.; Keller, D.; Lim, R.; Mikami, D.; Phillips, E.; Spaniolas, K.; Tsuda, S.; et al. SAGES and EAES recommendations for minimally invasive surgery during COVID-19 pandemic. Surg. Endosc. 2020, 34, 2327–2331. [Google Scholar] [CrossRef]
- Moletta, L.; Pierobon, E.S.; Capovilla, G.; Costantini, M.; Salvador, R.; Merigliano, S.; Valmasoni, M. International guidelines and recommendations for surgery during COVID-19 pandemic: A Systematic Review. Int. J. Surg. 2020, 79, 180–188. [Google Scholar] [CrossRef]
- Campanile, F.C.; Podda, M.; Arezzo, A.; Botteri, E.; Sartori, A.; Guerrieri, M.; Cassinotti, E.; Muttillo, I.; Pisano, M.; Contul, R.B.; et al. Acute cholecystitis during COVID-19 pandemic: A multisocietary position statement. World J. Emerg. Surg. 2020, 15, 38. [Google Scholar] [CrossRef]
- Porter, J.; Blau, E.; Gharagozloo, F.; Martino, M.; Cerfolio, R.; Duvvuri, U.; Caceres, A.; Badani, K.; Bhayani, S.; Collins, J.; et al. Society of Robotic Surgery Review: Recommendations Regarding the Risk of COVID-19 Transmission during Minimally Invasive Surgery. BJU Int. 2020, 126, 225–234. [Google Scholar] [CrossRef]
- Săftoiu, A.; Tomulescu, V.; Tanţău, M.; Gheorghe, C.; Dumitru, E.; Mateescu, B.; Negreanu, L.; Jinga, M.; Seicean, A.; Ciocîrlan, M. SRED-ARCE Recommendations for Minimally Invasive Interventions during the COVID-19 Pandemic in Romania. Chirurgia 2020, 115, 289–306. [Google Scholar] [CrossRef] [PubMed]
- DesCoteaux, J.G.; Picard, P.; Poulin, E.C.; Baril, M. Preliminary study of electrocautery smoke particles produced in vitro and during laparoscopic procedures. Surg. Endosc. 1996, 10, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Mowbray, N.G.; Ansell, J.; Horwood, J.; Cornish, J.; Rizkallah, P.; Parker, A.; Wall, P.; Spinelli, A.; Torkington, J. Safe management of surgical smoke in the age of COVID-19. Br. J. Surg. 2020. [Google Scholar] [CrossRef]
- Barrett, W.L.; Garber, S.M. Surgical smoke: A review of the literature. Is this just a lot of hot air? Surg. Endosc. 2003, 17, 979–987. [Google Scholar] [CrossRef]
- Dobrogowski, M.; Wesołowski, W.; Kucharska, M.; Sapota, A.; Pomorski, L.S. Chemical composition of surgical smoke formed in the abdominal cavity during laparoscopic cholecystectomy--assessment of the risk to the patient. Int. J. Occup. Med. Environ. Health 2014, 27, 314–325. [Google Scholar] [CrossRef]
- Dobrogowski, M.; Wesolowski, W.; Kucharska, M.; Paduszynska, K.; Dworzynska, A.; Szymczak, W.; Sapota, A.; Pomorski, L. Health risk to medical personnel of surgical smoke produced during laparoscopic surgery. Int. J. Occup. Med. Environ. Health 2015, 28, 831–840. [Google Scholar] [CrossRef]
- Fan, J.K.; Chan, F.S.; Chu, K.M. Surgical smoke. Asian J. Surg. 2009, 32, 253–257. [Google Scholar] [CrossRef]
- Baggish, M.S.; Elbakry, M. The effects of laser smoke on the lungs of rat. Am. J. Obstet. Gynecol. 1987, 156, 1260–1265. [Google Scholar] [CrossRef]
- Moot, A.R.; Ledingham, K.M.; Wilson, P.F.; Senthilmohan, S.T.; Lewis, D.R.; Roake, J.; Allardyce, R. Composition of volatile organic compounds in diathermy plume as detected by selected ion flow tube mass spectrometry. ANZ J. Surg. 2007, 77, 20–23. [Google Scholar] [CrossRef]
- Wenig, B.L.; Stenson, K.M.; Wenig, B.M.; Tracey, D. Effects of plume produced by the Nd:YAG laser and electrocautery on the respiratory syste. Lasers Surg. Med. 1993, 13, 242–245. [Google Scholar] [CrossRef]
- Capizzi, P.J.; Clay, R.P.; Battey, M.J. Microbiologic activity in laserresurfacing plume and debris. Lasers Surg. Med. 1998, 23, 172–174. [Google Scholar] [CrossRef]
- Taffinder, N.J.; Cruaud, P.; Catheline, J.M.; Bron, M.; Champault, G. Bacterial contamination of pneumoperitoneum gas in peritonitis and controls: A prospective laparoscopic study. Acta Chir. Belg. 1997, 97, 215–216. [Google Scholar] [PubMed]
- Englehardt, R.K.; Nowak, B.M.; Seger, M.V.; Duperier, F.D. Contamination resulting from aerosolized fluid during laparoscopic surgery. JSLS 2014, 18. [Google Scholar] [CrossRef] [PubMed]
- Davies, C.G.; Khan, M.N.; Ghauri, A.S.; Ranaboldo, C.J. Blood and body fluid splashes during surgery—The need for eye protection and masks. Ann. R. Coll. Surg. Engl. 2007, 89, 770–772. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.K.; Robinson, W.S. Human immunodeficiency virus-1(HIV-1) in the vapors of surgical power instruments. J. Med. Virol. 1991, 33, 47–50. [Google Scholar] [CrossRef] [PubMed]
- Kwak, H.D.; Kim, S.-H.; Seo, Y.S.; Song, K.-J. Detecting hepatitis B virus in surgical smoke emitted during laparoscopic surgery. Occup. Environ. Med. 2016, 73, 857–863. [Google Scholar] [CrossRef] [PubMed]
- Alp, E.; Bijl, D.; Bleichrodt, R.P.; Hansson, B.; Voss, A. Surgical smoke and infection control. J. Hosp. Infect. 2006, 62, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Song, Y.; Hu, X.; Yan, L.; Zhu, X. Awareness of surgical smoke hazards and enhancement of surgical smoke prevention among the gynecologists. J. Cancer 2019, 10, 2788–2799. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.H.; Kwon, T.G.; Chung, S.K.; Kim, T.H. Surgical smoke may be a biohazard to surgeons performing laparoscopic surgery. Surg. Endosc. 2014, 28, 2374–2380. [Google Scholar] [CrossRef]
- Hallmo, P.; Naess, O. Laryngeal papillomatosis with human papillomavirus DNA contracted by a laser surgeon. Eur. Arch. Oto-Rhino-Laryngol. 1991, 248, 425–427. [Google Scholar] [CrossRef] [PubMed]
- Garden, J.M.; O’Banion, M.K.; Shelnitz, L.S.; Pinski, K.S.; Bakus, A.D.; E Reichmann, M.; Sundberg, J.P. Papillomavirus in the vapor of carbon dioxide laser-treated verrucae. JAMA 1988, 259, 1199–1202. [Google Scholar] [CrossRef] [PubMed]
- Garden, J.M.; O’Banion, M.K.; Bakus, A.D.; Olson, C. Viral disease transmitted by laser-generated plume (aerosol). Arch. Dermatol. 2002, 138, 1303–1307. [Google Scholar] [CrossRef]
- Fletcher, J.N.; Mew, D.; DesCoteaux, J.G. Dissemination of melanomacells within electrocautery plume. Am. J. Surg. 1999, 178, 57–59. [Google Scholar] [CrossRef]
- Zhou, Q.; Hu, X.; Zhou, J.; Zhao, M.; Zhu, X.; Zhu, X. Human papillomavirus DNA in surgical smoke during cervical loop electrosurgical excision procedures and its impact on the surgeon. Cancer Manag. Res. 2019, 11, 3643–3654. [Google Scholar] [CrossRef] [PubMed]
- CRSToday. Available online: https://crstoday.com/articles/2005-jan/0105_tanchel-html/ (accessed on 4 September 2020).
- Mintz, Y.; Arezzo, A.; Boni, L.; Baldari, L.; Cassinotti, E.; Brodie, R.; Uranues, S.; Zheng, M.; Fingerhut, A. The risk of COVID-19 transmission by laparoscopic smoke may be lower than for laparotomy: A narrative review. Surg. Endosc. 2020, 34, 3298–3305. [Google Scholar] [CrossRef] [PubMed]
- Chadi, S.A.; Guidolin, K.; Caycedo-Marulanda, A.; Sharkawy, A.; Spinelli, A.; Quereshy, F.A.; Okrainec, A. Current Evidence for Minimally Invasive Surgery during the COVID-19 Pandemic and Risk Mitigation Strategies: A Narrative Review. Ann. Surg. 2020, 10. [Google Scholar] [CrossRef]
- de Leeuw, R.A.; Burger, N.B.; Ceccaroni, M.; Zhang, J.; Tuynman, J.B.; Mabrouk, M.; Soldevila, P.B.; Bonjer, H.J.; Ankum, P.; Huirne, J. COVID-19 and Laparoscopic Surgery: Scoping Review of Current Literature and Local Expertise. JMIR Public Health Surveill. 2020, 6, e18928. [Google Scholar] [CrossRef]
- Yeo, C.; Yeo, D.; Kaushal, S.; Ahmed, S. Is it too premature to recommend against laparoscopic emergency surgery in COVID-19 patients? Br. J. Surg. 2020, 107, e202. [Google Scholar] [CrossRef]
- Gupta, N.; Agrawal, H. COVID 19 and laparoscopic surgeons, the Indian scenario—Perspective. Int. J. Surg. 2020, 79, 165–167. [Google Scholar] [CrossRef]
- Savlovschi, C.; Serban, D.; Andreescu, C.; Dascalu, A.; Pantu, H. Economic analysis of medical management applied for left colostomy. Chirurgia 2013, 108, 666–669. [Google Scholar]
- Vourtzoumis, P.; Alkhamesi, N.; Elnahas, A.; Hawel, J.; Schlachta, C. Operating during COVID-19: Is there a risk of viral transmission from surgical smoke during surgery? Can. J. Surg. 2020, 63, E299–E301. [Google Scholar] [CrossRef] [PubMed]
- Safari, S.; Keyvani, H.; Alamdari, N.M.; Dehghanian, A.; Hashemi, M.R.; Honar, B.N.; Aminian, A. Abdominal Surgery in Patients with COVID-19: Detection of SARS-CoV-2 in Abdominal and Adipose Tissues. Ann. Surg. 2020. [Google Scholar] [CrossRef] [PubMed]
- Galvez, A.; King, K.; El Chaar, M.; Claros, L. Perforated Marginal Ulcer in a COVID-19 Patient. Laparoscopy in these Trying Times? Obes. Surg. 2020. [Google Scholar] [CrossRef] [PubMed]
- Ngaserin, S.H.; Koh, F.H.; Ong, B.C.; Chew, M.H. COVID-19 not detected in peritoneal fluid: A case of laparoscopic appendicectomy for acute appendicitis in a COVID-19-infected patient. Langenbeck’s Arch. Surg. 2020, 405, 353–355. [Google Scholar] [CrossRef]
- Pawar, T.; Pokharkar, A.; Gori, J.; Pandey, D.; Rohila, J.; Dsouza, A.; Saklani, A. The Technique and Justification for Minimally Invasive Surgery in COVID-19 Pandemic: Laparoscopic Anterior Resection for Near Obstructed Rectal Carcinoma. J. Laparoendosc. Adv. Surg. Tech. 2020, 30, 485–487. [Google Scholar] [CrossRef]
- Lovece, A.; Asti, E.; Bruni, B.; Bonavina, L. Subtotal laparoscopic cholecystectomy for gangrenous gallbladder during recovery from COVID-19 pneumonia. Int. J. Surg. Case Rep. 2020, 72, 335–338. [Google Scholar] [CrossRef]
- He, L.; Zhao, W.; Zhou, W.; Pang, P.; Liao, Y.; Liu, J. An Emergency Surgery in Severe Case Infected by COVID-19 With Perforated Duodenal Bulb Ulcer. Ann. Surg. 2020, 272, e35–e37. [Google Scholar] [CrossRef]
- Mattone, E.; Sofia, M.; Schembari, E.; Palumbo, V.; Bonaccorso, R.; Randazzo, V.; La Greca, G.; Iacobello, C.; Russello, D.; Latteri, S. Acute Acalculous Cholecystitis on a COVID-19 Patient: A Case Report, 15 July 2020; PREPRINT Available at Research Square; 2020. doi:10.21203/rs.3.rs-42230/v1. Available online: https://www.researchsquare.com/article/rs-42230/v1 (accessed on 4 September 2020).
- Coccolini, F.; Tartaglia, D.; Puglisi, A.; Giordano, C.; Pistello, M.; Lodato, M.; Chiarugi, M. SARS-CoV-2 is present in peritoneal fluid in COVID-19 patients. Ann. Surg. 2020, 272, e240–e242. [Google Scholar] [CrossRef]
- Tivey, D.R.; Davis, S.S.; Kovoor, J.G.; Babidge, W.J.; Tan, L.; Hugh, T.J.; Collinson, T.G.; Hewett, P.J.; Padbury, R.T.A.; Maddern, G.J. Safe surgery during the coronavirus disease 2019 crisis. ANZ J. Surg. 2020. [Google Scholar] [CrossRef]
- Zheng, M.H.; Boni, L.; Fingerhut, A. Minimally invasive surgery and the novel coronavirus outbreak: Lessons learned in China and Italy. Ann. Surg. 2020, 272, e5–e6. [Google Scholar] [CrossRef]
- Joseph, J.P.; Joseph, A.O.; Oomman, S.; Jayanthi, N.V.G. Laparoscopic versus open surgery: Aerosols and their implications for surgery during the COVID-19 pandemic. Eur. Surg. 2020, 52, 188–189. [Google Scholar] [CrossRef] [PubMed]
- Guraya, S.Y. Transforming laparoendoscopic surgical protocols during the COVID-19 pandemic; big data analytics, resource allocation and operational considerations. Int. J. Surg. 2020, 80, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Di Saverio, S.; Pata, F.; Khan, M.; Ietto, G.; Zani, E.; Carcano, G. Convert to open: The new paradigm for surgery during COVID-19? Br. J. Surg. 2020, 107, e194. [Google Scholar] [CrossRef] [PubMed]
- Ouzzane, A.; Colin, P. Cost-Effective Filtrating Suction to Evacuate Surgical Smoke in Laparoscopic and Robotic Surgery during the COVID-19 Pandemic. Surg. Laparosc. Endosc. Percutan. Tech. 2020. [Google Scholar] [CrossRef] [PubMed]
- Hanbali, N.; Herrod, P.; Patterson, J. A safe method to evacuate pneumoperitoneum during laparoscopic surgery in suspected COVID-19 patients. Ann. R. Coll. Surg. Engl. 2020, 102, 392–393. [Google Scholar] [CrossRef] [PubMed]
- Yeo, D.; Kaushal, S.; Ahmed, S. Clearing the air: An accessible system for suction and safe evacuation of pneumoperitoneum during laparoscopy in patients with COVID-19. Br. J. Surg. 2020, 107, e200. [Google Scholar] [CrossRef]
- Zago, M.; Uranues, S.; Chiarelli, M.E.; Grandi, S.; Fumagalli, L.; Tavola, M.; Chiarugi, M.; Mariani, D.; Wienerroither, V.; Kurihara, H.; et al. Enhancing safety of laparoscopic surgery in COVID-19 era: Clinical experience with low-cost filtration devices. Eur. J. Trauma Emerg. Surg. 2020. [Google Scholar] [CrossRef]
- Mintz, Y.; Arezzo, A.; Boni, L.; Chand, M.; Brodie, R.; Fingerhut, A.; The Technology Committee of the European Association for Endoscopic Surgery. A Low-cost, Safe and Effective Method for Smoke Evacuation in Laparoscopic Surgery for Suspected Coronavirus Patients. Ann. Surg. 2020, 272, e7–e8. [Google Scholar] [CrossRef]
- Wielogórska, N.L.; Ekwobi, C.C. COVID-19: What are the challenges for NHS surgery? Curr. Probl. Surg. 2020. [Google Scholar] [CrossRef]
- Javanmard-Emamghissi, H.; Boyd-Carson, H.; Hollyman, M.; Doleman, B.; Adiamah, A.; Lund, J.N.; Clifford, R.; Dickerson, L.; Richards, S.; Pearce, L.; et al. The management of adult appendicitis during the COVID-19 pandemic: An interim analysis of a UK cohort study. Tech. Coloproctol. 2020. [Google Scholar] [CrossRef]
- Lazaridis, I.I.; Kraljević, M.; Schneider, R.; Klasen, J.M.; Schizas, D.; Peterli, R.; Kow, L.; Delko, T. The Impact of the COVID-19 Pandemic on Bariatric Surgery: Results from a Worldwide Survey. Obes. Surg. 2020. [Google Scholar] [CrossRef]
- Manzia, T.M.; Angelico, R.; Parente, A.; Muiesan, P.; Tisone, G. MEGAVID (ManagEment of GAllstone disease during coVID-19 pandemic) Clinical Investigator Group. Global management of a common, underrated surgical task during the COVID-19 pandemic: Gallstone disease—An international survery. Ann. Med. Surg. 2020. [Google Scholar] [CrossRef] [PubMed]
- Singhal, R. Internal Hernia in the Times of COVID-19: To Laparoscope or Not to Laparoscope? Obes. Surg. 2020, 30, 2812–2813. [Google Scholar] [CrossRef] [PubMed]
| Energy Source | Dimensions of Aerosols (μm) |
|---|---|
| Electrocautery | 0.007–0.420 |
| Ultrasonic scalpel | 0.35–6.5 |
| Laser | 0.1–0.8 |
| Article | Author | Year | Source | Procedure | Evidences |
|---|---|---|---|---|---|
| Preliminary study of electrocautery smoke particles produced in vitro and during laparoscopic procedures [12]. | DesCoteaux J.G. | 1996 | Surgical Endoscopy | Laparoscopic smoke | Smoke from 5 laparoscopic procedures was analyzed; 2 types of particles were identified: Large, irregular particles (2–25 μm): cellular fragments Small homogeneous spheres (0.1–0.5 μm) composed of sodium, magnesium, calcium and potassium salts. |
| Chemical composition of surgical smoke formed in the abdominal cavity during laparoscopic cholecystectomy-Assessment of the risk to the patient [15]. | Dobrogowski, M. | 2014 | International Journal of Occupational Medicine and Environmental Health | Laparoscopy | Vast array of chemical compounds, including aliphatic and aromatic hydrocarbons such as benzene and its alkyl derivatives, as well as aldehydes, nitriles, amines, polychlorinated dioxins and furans, including the highly toxic 2,3,7,8-TCDD are detected in the surgical smoke |
| Surgical smoke [17]. | Fan, J.K.-M. | 2009 | Asian Journal of Surgery | Electro-cautery, laparoscopy | Various chemicals (hydrogen cyanide, benzene, hydrocarbons, nitriles, fatty acids and phenols), viruses and bacteria, viable cells were identified in surgical smoke in reviewed studies N95 grade or equivalent respirator offers the best protection against surgical smoke HEPA filters or equiv. are necessary to remove smoke |
| Health risk to medical personnel of surgical smoke produced during laparoscopic surgery [16]. | Dobrogowski, M. | 2015 | International Journal of Occupational Medicine and Environmental Health | Laparoscopy | Toxic chemicals are present in the surgical smoke It is necessary to remove surgical smoke from the operating room in order to protect medical personnel |
| Detecting hepatitis B virus in surgical smoke emitted during laparoscopic surgery [26]. | Kwak H.D. | 2016 | Occupational and Environmental Medicine | Laparoscopy | HBV was detected in laparoscopically smoke in 10 of the 11 infected patients The infectious risk for surgeon was not assessed |
| Surgical smoke and infection control [27]. | Alp E. | 2006 | The Journal of Hospital Infection | Laser. Electrocautery laparoscopy | Bio-aerosols with viable and non-viable cellular material with possible risk of infection (human immunodeficiency virus, hepatitis B virus, human papillomavirus) Irritation to the lungs leading to acute and chronic inflammatory changes cytotoxic, genotoxic and mutagenic effects increased protection needed by |
| Activated carbon fiber filters could reduce the risk of surgical smoke exposure during laparoscopic surgery: application of volatile organic compounds [29]. | Choi S.H. | 2018 | Surgical Endoscopy | Laparoscopy | Activated carbon filters reduce by 85% the concentration of chemicals in evacuated pneumoperitoneum |
| Contamination resulting from aerosolized fluid during laparoscopic surgery [23]. | Engelhardt R. | 2014 | Journal of the Society of Laparoscopic & Robotic Surgeons | Laparoscopy | Consistent environmental contamination of blood and body fluid during rapid evacuation of the pneumoperitoneum. Using wrap-around style glasses/shields and protective masks prevent contamination of mucous membranes with viruses like HIV and VHC |
| Blood and body fluid splashes during surgery—The need for eye protection and masks [24]. | Davies C.G. | 2007 | Annals of the Royal College of Surgeons of England | Laparoscopy | 50% risk of contamination with aerosolized blood in laparoscopic surgery Most exposure to projectile blood and body fluid occurs towards the end of a case when ports are removed and pneumoperitoneum is released via the port sites |
| Composition of volatile organic compounds in diathermy plume as detected by selected ion flow tube mass spectrometry [19]. | Moot A.R. | 2007 | ANZ Journal of Surgery | laparoscopy | 1 g tissue burned by electrocautery = same carcinogens as in 6 cigarettes smoked/day Smoke filtration is needed |
| Awareness of surgical smoke hazards and enhancement of surgical smoke prevention among the gynecologists [28]. | Liu Y. | 2019 | Journal of Cancer | Various laser gynecological procedures | HPV contamination lead to laryngeal papilloma and HPV16 tonsillar squamous cell cancer in 4 reported cases Surgical smoke contains chemicals, blood and tissue particles, bacteria and viruses, which has been shown to exhibit potential risks for surgeons, nurses, anesthesiologists |
| Microbiologic activity in laser resurfacing plume and debris [21]. | Capizzi P.J. | 1998 | Lasers in Surgery and Medicine | CO2 laser resurfacing | Smoke from laser surfacing was analyzed for 13 patients; in 38% of cases coagulase-negative Staphylococcus was present; one of these cases associated Corynebacterium, one Neisseria no viral positive culture |
| Laryngeal papillomatosis with human papillomavirus DNA contracted by a laser surgeon [30]. | Hallmo P. | 1991 | European Archives of Oto-Rhino-Laryngology | Laser, not specified | Laryngeal papillomatosis secondary to HPV types 6 and 11 in a laser surgeon probably due to transmission via surgical smoke from genitals papilloma |
| Papillomavirus in the vapor of carbon dioxide laser-treated verrucae [31]. | Garden J.M. | 1988 | JAMA | CO2 laser | Vapor produced by the carbon dioxide laser during the vaporization of papillomavirus-infected verrucae showed intact viral DNA content. Two models were used for evaluation: an in vitro cutaneous bovine fibropapilloma and an in vivo human verruca model. |
| Viral disease trans-mitted by laser-generated plume (aerosol) [32]. | Garden J.M. | 2002 | Archives of Dermatology | CO2 laser | Bovine papillomavirus–induced cutaneous fibropapillomas were exposed to the carbon dioxide laser. The laser plume was suctioned reinoculated onto the skin of calves, producing HPV infection |
| Human immunodeficiency virus-1(HIV-1) in the vapors of surgical power instruments [25]. | Johnson G.K. | 1991 | Journal of Medical Virology | CO2 laser | HIV-1 can remain viable in cool aerosols generated by certain surgical power tools |
| Dissemination of melanoma cells within electrocautery plume [33]. | Fletcher J.N. | 1999 | Americal Journal of Surgery | CO2 laser | Pellets of B16-F0 mouse melanoma cells were cauterized and the plume collected into culture medium; viable melanoma cells were collected and grown in culture |
| Human papillomavirus DNA in surgical smoke during cervical loop electrosurgical excision procedures and its impact on the surgeon [34]. | Zhou Q. | 2019 | Cancer Management and Research | Loop electrosurgical excision procedures (LEEPs) | HPV DNA in surgical smoke produced by LEEP 1 The nasal epithelial cells of two surgeons were positive for HPV DNA, the same type as those of resected lesion |
| Paper | Surgery | Patient’s Status | Sample RT-PCR Test for SARS-CoV-2 | Result | Prevention Intraoperatory Measures |
|---|---|---|---|---|---|
| Ngaserin S.H. [45] | Laparoscopic appendicectomy | Asymptomatic, COVID-19+ | Peritoneal fluid | − | PPE 1 |
| Safari S. [43] | 1. Laparoscopic cholecystectomy 2. Perforated ileum. Chron disease–laparotomy 3. Open appendectomy 4. Peptic ulcer repair-laparotomy | COVID-19+, symptomatic | Peritoneal fluid Digestive wall Feces Abdominal fat, omentum | − + + − | PPE |
| Galvez A. [44] | Perforated ulcer repair | COVID-19 pneumonia | Gastric wall | + | PPE Laparoscopic HEPA 2 filter |
| He L. [48] | Perforated ulcer repair | COVID-19 pneumonia | none | − | PPE Negative pressure in OR 3 (−5 kPa) Skilled laparoscopic expert to minimize risk |
| Pawar T. [46] | Laparoscopic Anterior Resection for Rectal Carcinoma | COVID-19 suspects (not tested) | none | PPE Air seal (CONMED, Utica, NY) and high-efficiency particulate air (HEPA) filter was utilized for safe gas evacuation | |
| Lovece A. [47] | Subtotal cholecystectomy | COVID-19 pneumonia | none | PPE Low Pneumoperitoneum pressure (9 mmHg) Low energy power Pneumoperitoneum was evacuated by the suction device before trocar removal and specimen extraction |
| Precautions | SAGES/EAES | ELSA | ALSGBI | ESGE |
|---|---|---|---|---|
| Sizing the incisions | Incision ports as small as possible | Purse-string suture or disposable trocar with skin blocking system should be used | Balloon/self-sealing trocars | + |
| Creating pneumoperitoneum using the most familiar technique | + | |||
| Low CO2 insufflation pressure (10–12 mmHg) | + | + | + | + |
| Minimal use of electrocautery | + | + | + | |
| Low power setting of electrocautery | + | Prevent plume formation by low energy, keeping instruments clean, limited dissection, frequent suction | ||
| Ultra-filtration of smoke | HEPA/ULPA filters are strongly recommended | Passive or active filtration systems of pneumoperitoneum | + | + |
| Disposable instruments to prevent viral contamination | + | |||
| Drain only if necessary | + | |||
| Attach a CO2 filter to one of the ports for smoke evacuation if needed, do not open the tap of any ports unless they are attached to a CO2 filter or being used to deliver the gas | + | |||
| Minimize introduction and removal of instruments through the ports as much as possible | + | |||
| All pneumoperitoneum safely evacuated via filtration system before closure, trocar removal, specimen extraction or conversion to open | + | + | + | + |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serban, D.; Smarandache, C.G.; Tudor, C.; Duta, L.N.; Dascalu, A.M.; Aliuș, C. Laparoscopic Surgery in COVID-19 Era—Safety and Ethical Issues. Diagnostics 2020, 10, 673. https://doi.org/10.3390/diagnostics10090673
Serban D, Smarandache CG, Tudor C, Duta LN, Dascalu AM, Aliuș C. Laparoscopic Surgery in COVID-19 Era—Safety and Ethical Issues. Diagnostics. 2020; 10(9):673. https://doi.org/10.3390/diagnostics10090673
Chicago/Turabian StyleSerban, Dragos, Catalin Gabriel Smarandache, Corneliu Tudor, Lucian Nicolae Duta, Ana Maria Dascalu, and Cătălin Aliuș. 2020. "Laparoscopic Surgery in COVID-19 Era—Safety and Ethical Issues" Diagnostics 10, no. 9: 673. https://doi.org/10.3390/diagnostics10090673
APA StyleSerban, D., Smarandache, C. G., Tudor, C., Duta, L. N., Dascalu, A. M., & Aliuș, C. (2020). Laparoscopic Surgery in COVID-19 Era—Safety and Ethical Issues. Diagnostics, 10(9), 673. https://doi.org/10.3390/diagnostics10090673






