Concurrent Validity and Reliability of an Inertial Measurement Unit for the Assessment of Craniocervical Range of Motion in Subjects with Cerebral Palsy
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
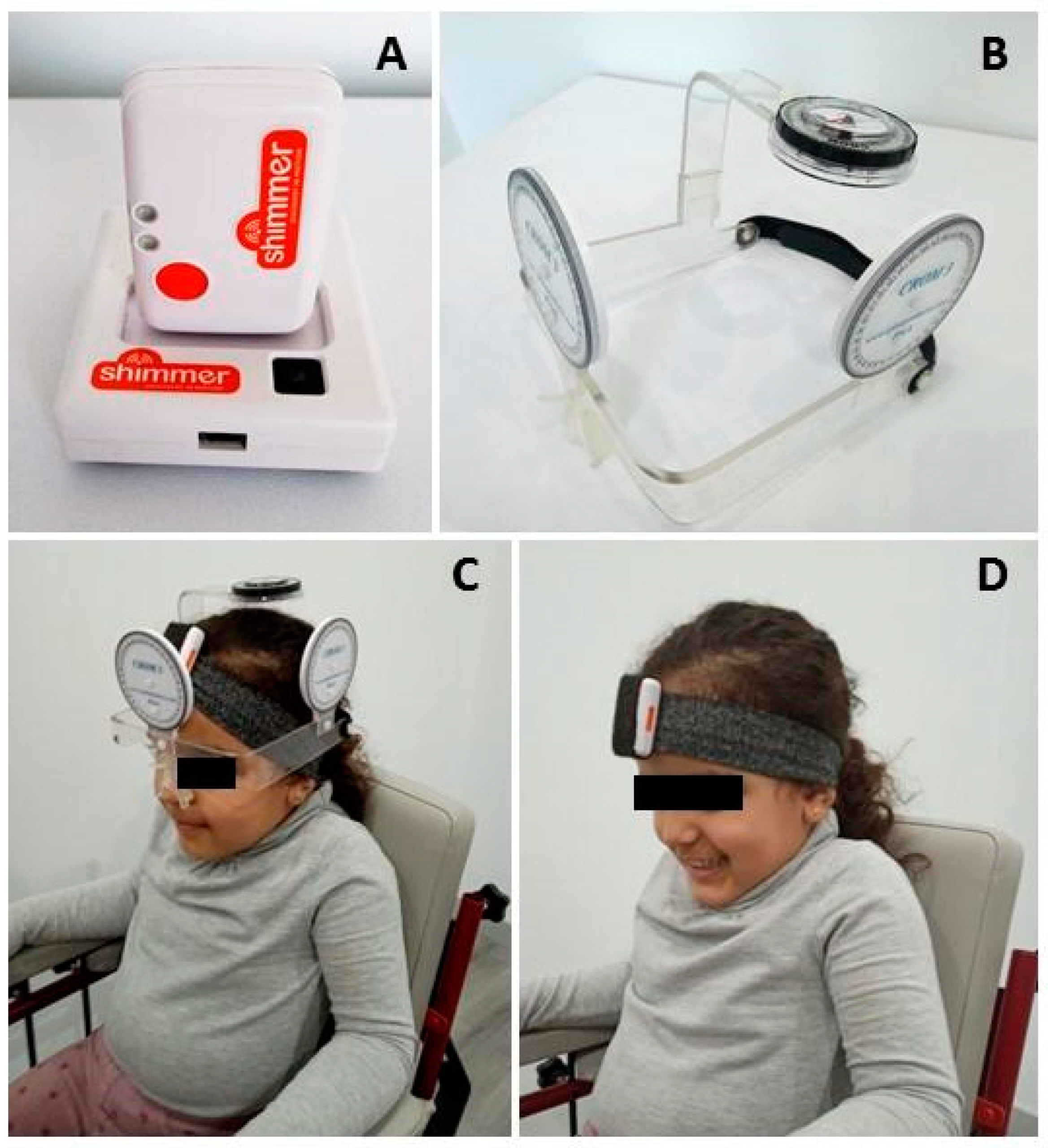
2.2. IMU Assessment
2.3. CROM Assessment
2.4. Muscle Tone Assessment
2.5. Procedures
2.6. Statistical Analysis
2.6.1. Concurrent Validity Analysis
2.6.2. Reliability Analysis
3. Results
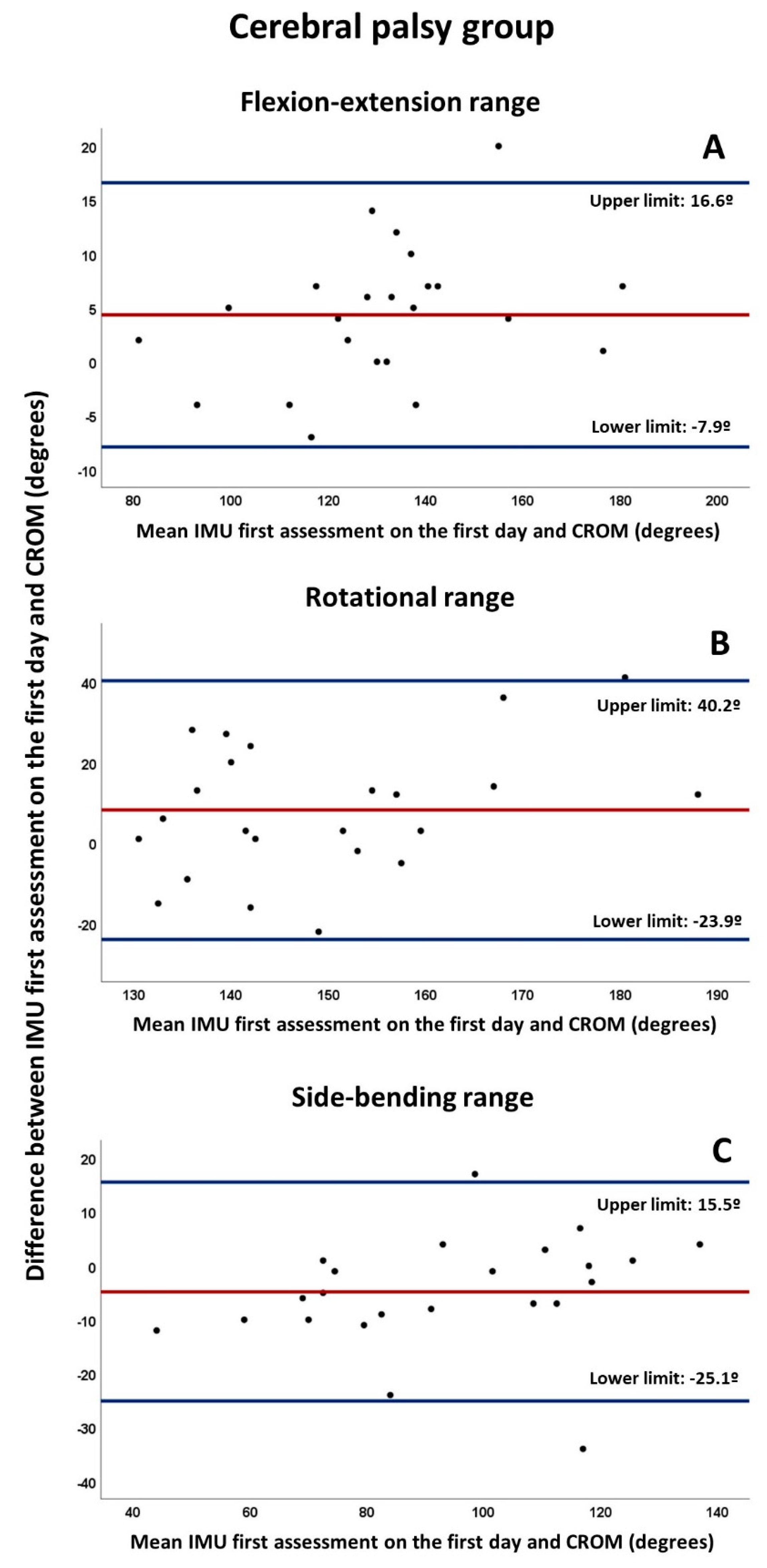
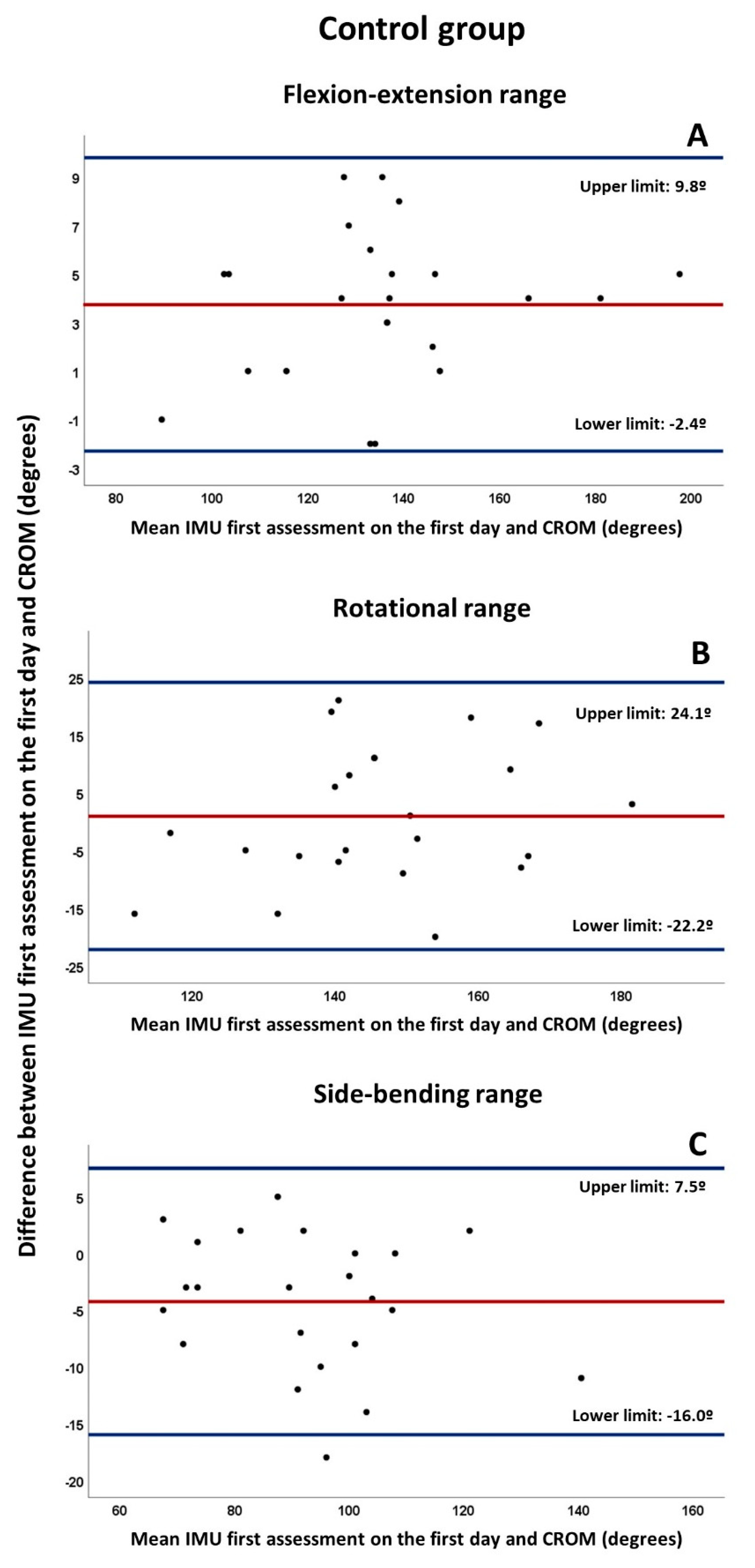
3.1. Concurrent Validity
3.2. Intra-Day and Inter-Day Reliability
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bax, M.; Frcp, D.M.; Rosenbaum, P.; Dan, B.; Universitaire, H.; Fabiola, R.; De Bruxelles, U.L.; Goldstein, M.; Pt, D.D.; Rosenbaum, P. Review Proposed definition and classification of cerebral palsy. Dev. Med. Child Neurol. 2005, 47, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Odding, E.; Roebroeck, M.E.; Stam, H.J. The epidemiology of cerebral palsy: Incidence, impairments and risk factors. Disabil. Rehabil. 2006, 28, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Zeitlin, J.; Mohangoo, A.; Delnord, M. European Perinatal Health Report. The health and care of pregnant women and babies in Europe in 2010, 1st ed.; Euro-Peristat: Paris, France, 2013; pp. 182–189. [Google Scholar]
- Winter, S.; Autry, A.; Boyle, C.; Yeargin-Allsopp, M. Trends in the prevalence of cerebral palsy in a population-based study. Pediatrics 2002, 110, 1220–1225. [Google Scholar] [CrossRef] [PubMed]
- Blair, E. Epidemiology of the cerebral palsies. Orthop. Clin. North Am. 2010, 41, 441–455. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Verma, I. Cerebral palsy in children: An overview. J. Clin. Orthop. Trauma 2012, 3, 77–81. [Google Scholar] [CrossRef]
- Papageorgiou, E.; Simon-Martinez, C.; Molenaers, G.; Ortibus, E.; Van Campenhout, A.; Desloovere, K. Are spasticity, weakness, selectivity, and passive range of motion related to gait deviations in children with spastic cerebral palsy? A statistical parametric mapping study. PLoS ONE 2019, 14, e0223363. [Google Scholar] [CrossRef]
- McDowell, B.C.; Salazar-Torres, J.J.; Kerr, C.; Cosgrove, A.P. Passive range of motion in a population-based sample of children with spastic cerebral palsy who walk. Phys. Occup. Ther. Pediatr. 2012, 32, 139–150. [Google Scholar] [CrossRef]
- Redstone, F.; West, J.F. The importance of postural control for feeding. Pediatr. Nurs. 2004, 30, 97–100. [Google Scholar]
- Holt, K.G.; Ratcliffe, R.; Jeng, S. Head stability in walking in children with cerebral palsy and in children and adults without neurological impairment. Phys. Ther. 1999, 79, 1153–1162. [Google Scholar] [CrossRef]
- Velasco, M.A.; Raya, R.; Muzzioli, L.; Morelli, D.; Otero, A.; Iosa, M.; Cincotti, F.; Rocon, E. Evaluation of cervical posture improvement of children with cerebral palsy after physical therapy based on head movements and serious videogames. Biomed Eng Online 2017, 16, S74. [Google Scholar] [CrossRef]
- Gresty, M.A.; Halmagyi, G.M. Abnormal head movement. J. Neurol. Neurosurg. Psychiatry 1979, 42, 705–714. [Google Scholar]
- Velasco, M.A.; Raya, R.; Ceres, R.; Clemotte, A.; Ruiz Bedia, A.; Gonzalez Franco, T.; Rocon, E. Positive and negative motor signs of head motion in cerebral palsy: Assessment of impairment and task performance. IEEE Syst. J. 2016, 10, 967–973. [Google Scholar] [CrossRef]
- Palisano, R.; Rosenbaum, P.; Walter, S.; Russell, D.; Wood, E.; Galuppi, B. Development and reliability of a system to classify gross motor function in children with cerebral palsy. Dev Med Child Neurol 1997, 39, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Palisano, R.J.; Rosenbaum, P.; Bartlett, D.; Livingston, M.H. Content validity of the expanded and revised Gross Motor Function Classification System. Dev. Med. Child Neurol. 2008, 50, 744–750. [Google Scholar] [CrossRef]
- McDowell, B. The gross motor function classification system - Expanded and revised. Dev. Med. Child Neurol. 2008, 50, 725. [Google Scholar] [CrossRef]
- Ronen, G.M.; Fayed, N.; Rosenbaum, P.L. Outcomes in pediatric neurology: A review of conceptual issues and recommendationsThe 2010 Ronnie Mac Keith Lecture. Dev. Med. Child Neurol. 2011, 53, 305–312. [Google Scholar] [CrossRef]
- Cuesta-Vargas, A.I.; Galán-Mercant, A.; Williams, J.M. The use of inertial sensors system for human motion analysis. Phys Ther Rev 2010, 15, 462–473. [Google Scholar] [CrossRef]
- Kim, M.; Kim, B.H.; Jo, S. Quantitative evaluation of a low-cost noninvasive hybrid interface based on EEG and eye movement. IEEE Trans Neural Syst Rehabil Eng 2015, 23, 159–168. [Google Scholar] [CrossRef]
- Carcreff, L.; Gerber, C.N.; Paraschiv-Ionescu, A.; De Coulon, G.; Newman, C.J.; Armand, S.; Aminian, K. What is the best configuration of wearable sensors to measure spatiotemporal gait parameters in children with cerebral palsy? Sensors (Switzerland) 2018, 18, 394. [Google Scholar] [CrossRef]
- Li, X.; González Navas, C.; Garrido-Castro, J.L. Reliability and validity of cervical mobility analysis measurement using an inertial sensor in patients with axial spondyloarthritis. Rehabilitacion 2017, 51, 17–21. [Google Scholar] [CrossRef]
- Aranda Valera, I.C.; Mata Perdigón, F.J.; Martínez Sánchez, I.; González Navas, C.; Collantes Estévez, E.; Garrido Castro, J.L. Use of inertial sensors for the assessment of spinal mobility in axial spondyloarthritis patients. Rehabilitacion 2018, 52, 100–106. [Google Scholar] [CrossRef]
- Solomon, A.J.; Jacobs, J.V.; Lomond, K.V.; Henry, S.M. Detection of postural sway abnormalities by wireless inertial sensors in minimally disabled patients with multiple sclerosis: A case-control study. J. Neuroeng. Rehabil. 2015, 12, 74. [Google Scholar] [CrossRef] [PubMed]
- Spain, R.I.; Mancini, M.; Horak, F.B.; Bourdette, D. Body-worn sensors capture variability, but not decline, of gait and balance measures in multiple sclerosis over 18 months. Gait Posture 2014, 39, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Mancini, M.; Salarian, A.; Carlson-Kuhta, P.; Zampieri, C.; King, L.; Chiari, L.; Horak, F.B. ISway: A sensitive, valid and reliable measure of postural control. J. Neuroeng. Rehabil. 2012, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- Delrobaei, M.; Memar, S.; Pieterman, M.; Stratton, T.W.; McIsaac, K.; Jog, M. Towards remote monitoring of Parkinson’s disease tremor using wearable motion capture systems. J. Neurol. Sci. 2018, 384, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Paulis, W.D.; Horemans, H.L.D.; Brouwer, B.S.; Stam, H.J. Excellent test-retest and inter-rater reliability for Tardieu Scale measurements with inertial sensors in elbow flexors of stroke patients. Gait Posture 2011, 33, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Van den Noort, J.; Harlaar, J.; Scholtes Rehab Med, V. Inertial sensing improves clinical spasticity assessment. Gait Posture 2008, 28S, S47. [Google Scholar] [CrossRef]
- Raya, R.; Rocon, E.; Ceres, R.; Harlaar, J.; Geytenbeek, J. Characterizing head motor disorders to create novel interfaces for people with cerebral palsy: Creating an alternative communication channel by head motion. IEEE Int. Conf. Rehabil. Robot. 2011, 2011, 5975409. [Google Scholar]
- Van Den Noort, J.C.; Ferrari, A.; Cutti, A.G.; Becher, J.G.; Harlaar, J. Gait analysis in children with cerebral palsy via inertial and magnetic sensors. Med. Biol. Eng. Comput. 2013, 51, 377–386. [Google Scholar] [CrossRef]
- Hislop, H.; Avers, D.; Brown, M. Muscle Testing: Techniques of Manual Examination and Performance Testing, 9th ed.; Elsevier Inc.: St. Louis, MI, USA, 2014; pp. 1–6. [Google Scholar]
- Manikowska, F.; Chen, B.P.J.; Jówiak, M.; Lebiedowska, M.K. Validation of Manual Muscle Testing (MMT) in children and adolescents with cerebral palsy. NeuroRehabilitation 2018, 42, 1–7. [Google Scholar] [CrossRef]
- Haik, M.N.; Alburquerque-Sendín, F.; Camargo, P.R. Reliability and minimal detectable change of 3-dimensional scapular orientation in individuals with and without shoulder impingement. J. Orthop. Sports Phys. Ther. 2014, 44, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Shrout, P.E.; Fleiss, J.L. Intraclass correlations: Uses in assessing rater reliability. Psychol. Bull. 1979, 86, 420–428. [Google Scholar] [CrossRef]
- Nakagawa, T.H.; Moriya, É.T.U.; Maciel, C.D.; Serrão, F.V. Test-retest reliability of three-dimensional kinematics using an electromagnetic tracking system during single-leg squat and stepping maneuver. Gait Posture 2014, 39, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Audette, I.; Dumas, J.P.; Côté, J.N.; De Serres, S.J. Validity and between-day reliability of the cervical range of motion (CROM) device. J. Orthop. Sports Phys. Ther. 2010, 40, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, J.P.; Bandy, W.D. Intrarater reliability of CROM measurement of cervical spine active range of motion in persons with and without neck pain. J. Orthop. Sports Phys. Ther. 2008, 38, 640–645. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, B. Preliminary trial of carisoprodol in multiple sclerosis. Practitioner 1964, 192, 540–542. [Google Scholar] [PubMed]
- Bohannon, R.W.; Smith, M.B. Interrater reliability of a modified ashworth scale of muscle spasticity. Class. Pap. Orthop. 1987, 67, 206–207. [Google Scholar] [CrossRef] [PubMed]
- Numanoǧlu, A.; Günel, M.K. Intraobserver reliability of modified Ashworth scale and modified Tardieu scale in the assessment of spasticity in children with cerebral palsy. Acta Orthop. Traumatol. Turc. 2012, 46, 196–200. [Google Scholar] [CrossRef]
- Bartlett, D.J.; Palisano, R.J. Factors influencing the acquisition of motor abilities of children with cerebral palsy: Implications for clinical reasoning. Phys Ther 2002, 82, 237–248. [Google Scholar] [CrossRef]
- Bartlett, D.J.; Palisano, R.J. A multivariate model of determinants of motor change for children with cerebral palsy. Phys Ther. 2000, 80, 598–614. [Google Scholar] [CrossRef]
- Van Den Noort, J.C.; Scholtes, V.A.; Harlaar, J. Evaluation of clinical spasticity assessment in Cerebral palsy using inertial sensors. Gait Posture 2009, 30, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Bieri, D.; Reeve, R.A.; Champion, D.; Addicoat, L.; Ziegler, J.B. The Faces Pain Scale for the self-assessment of the severity of pain experienced by children: Development, initial validation, and preliminary investigation for ratio scale properties. Pain 1990, 41, 139–150. [Google Scholar] [CrossRef]
- Raya, R.; Garcia-Carmona, R.; Sanchez, C.; Urendes, E.; Ramirez, O.; Martin, A.; Otero, A. An inexpensive and easy to use cervical range of motion measurement solution using inertial sensors. Sensors (Switzerland) 2018, 18, 2582. [Google Scholar] [CrossRef] [PubMed]
- Akoglu, H. User’s guide to correlation coefficients. Turk J Emerg Med. 2018, 18, 91–93. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Giavarina, D. Understanding Bland Altman analysis. Biochem. Medica 2015, 25, 141–151. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A Guideline of selecting and reporting Intraclass correlation coefficients for reliability research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef]
- Donoghue, D.; Murphy, A.; Jennings, A.; McAuliffe, A.; O’Neil, S.; Charthaigh, E.N.; Griffin, E.; Gilhooly, L.; Lyons, M.; Galvin, R.; et al. How much change is true change? The minimum detectable change of the Berg Balance Scale in elderly people. J. Rehabil. Med. 2009, 41, 343–346. [Google Scholar] [CrossRef]
- Lexell, J.E.; Downham, D.Y. How to assess the reliability of measurements in rehabilitation. Am. J. Phys. Med. Rehabil. 2005, 84, 719–723. [Google Scholar] [CrossRef]
- Weir, J.P. Quantifying test-retest reliability using the intraclass correlation coefficient and the SEM. J. Strength Cond. Res. 2005, 19, 231–240. [Google Scholar]
- Poitras, I.; Dupuis, F.; Bielmann, M.; Campeau-Lecours, A.; Mercier, C.; Bouyer, L.J.; Roy, J.S. Validity and reliability ofwearable sensors for joint angle estimation: A systematic review. Sensors (Switzerland) 2019, 19, 1555. [Google Scholar] [CrossRef]
- Chang, K.V.; Wu, W.T.; Chen, M.C.; Chiu, Y.C.; Han, D.S.; Chen, C.C. Smartphone application with virtual reality goggles for the reliable and valid measurement of active craniocervical range of motion. Diagnostics 2019, 9, 71. [Google Scholar] [CrossRef]
- Malmström, E.M.; Karlberg, M.; Fransson, P.A.; Melander, A.; Magnusson, M. Primary and coupled cervical movements: The effect of age, gender, and body mass index. A 3-dimensional movement analysis of a population without symptoms of neck disorders. Spine (Phila. Pa. 1976). 2006, 31, 44–50. [Google Scholar] [CrossRef]
- Guo, L.Y.; Lee, S.Y.; Lin, C.F.; Yang, C.H.; Hou, Y.Y.; Wu, W.L.; Lin, H.T. Three-dimensional characteristics of neck movements in subjects with mechanical neck disorder. J. Back Musculoskelet. Rehabil. 2012, 25, 47–53. [Google Scholar] [CrossRef]
- Song, H.; Zhai, X.; Gao, Z.; Lu, T.; Tian, Q.; Li, H.; He, X. Reliability and validity of a Coda Motion 3-D Analysis system for measuring cervical range of motion in healthy subjects. J. Electromyogr. Kinesiol. 2018, 38, 56–66. [Google Scholar] [CrossRef]
- Choi, S.; Shin, Y.B.; Kim, S.Y.; Kim, J. A novel sensor-based assessment of lower limb spasticity in children with cerebral palsy. J. Neuroeng. Rehabil. 2018, 15, 45. [Google Scholar] [CrossRef]
- Gerber, C.N.; Carcreff, L.; Paraschiv-Ionescu, A.; Armand, S.; Newman, C.J. Reliability of single-day walking performance and physical activity measures using inertial sensors in children with cerebral palsy. Ann. Phys. Rehabil. Med. 2019, (in press). [CrossRef]
- Bar-On, L.; Molenaers, G.; Aertbeliën, E.; Van Campenhout, A.; Feys, H.; Nuttin, B.; Desloovere, K. Spasticity and its contribution to hypertonia in cerebral palsy. Biomed Res. Int. 2015, 2015, 317047. [Google Scholar] [CrossRef]
- Sarathy, K.; Doshi, C.; Aroojis, A. Clinical examination of children with cerebral palsy. Indian, J. Orthop. 2019, 53, 35–44. [Google Scholar]
- Pascoe, J.; Thomason, P.; Graham, H.K.; Reddihough, D.; Sabin, M.A. Body mass index in ambulatory children with cerebral palsy: A cohort study. J. Paediatr. Child Health 2016, 52, 417–421. [Google Scholar] [CrossRef]
- Rimmer, J.H.; Yamaki, K.; Lowry, D.M.D.; Wang, E.; Vogel, L.C. Obesity and obesity-related secondary conditions in adolescents with intellectual/developmental disabilities. J. Intellect. Disabil. Res. 2010, 54, 787–794. [Google Scholar] [CrossRef]
- Budelmann, K.; Von Piekartz, H.; Hall, T. Is there a difference in head posture and cervical spine movement in children with and without pediatric headache? Eur. J. Pediatr. 2013, 172, 1349–1356. [Google Scholar] [CrossRef]
- Murgia, M.; Venditto, T.; Paoloni, M.; Hodo, B.; Alcuri, R.; Bernetti, A.; Santilli, V.; Mangone, M. Assessing the cervical range of motion in infants with positional plagiocephaly. J. Craniofac. Surg. 2016, 27, 1060–1064. [Google Scholar] [CrossRef][Green Version]
- Lee, J.Y.; Koh, S.E.; Lee, I.S.; Jung, H.; Lee, J.; Kang, J.-I.; Bang, H. The cervical range of motion as a factor affecting outcome in patients with congenital muscular torticollis. Ann. Rehabil. Med. 2013, 37, 183–190. [Google Scholar] [CrossRef]
- Rihar, A.; Sgandurra, G.; Beani, E.; Cecchi, F.; Pašič, J.; Cioni, G.; Dario, P.; Mihelj, M.; Munih, M. CareToy: Stimulation and Assessment of Preterm Infant’s Activity Using a Novel Sensorized System. Ann. Biomed. Eng. 2016, 44, 3593–3605. [Google Scholar] [CrossRef]
- Saiz, B.M.; Maris, E.; Mussin, P.; Lopesino, R.A.; Martı, I.; Callejo, S.H.; Lo, R.R.; Lara, S.L. Short-term effects of an intervention program through an inertial sensor (ENLAZA) for the improving of head control in children with cerebral palsy. Gait Posture 2016, 49, S227. [Google Scholar]
- Porciuncula, F.; Roto, A.V.; Kumar, D.; Davis, I.; Roy, S.; Walsh, C.J.; Awad, L.N. Wearable Movement Sensors for Rehabilitation: A Focused Review of Technological and Clinical Advances. PM R 2018, 10, S220–S232. [Google Scholar] [CrossRef]
- Walmsley, C.P.; Williams, S.A.; Grisbrook, T.; Elliott, C.; Imms, C.; Campbell, A. Measurement of Upper Limb Range of Motion Using Wearable Sensors: A Systematic Review. Sport. Med. Open 2018, 4, 53. [Google Scholar] [CrossRef]
- Theobald, P.S.; Jones, M.D.; Williams, J.M. Do inertial sensors represent a viable method to reliably measure cervical spine range of motion? Man. Ther. 2012, 17, 92–96. [Google Scholar] [CrossRef]
| CP Group (n = 23) | Control Group (n = 23) | p-Value | |
|---|---|---|---|
| Age (years) | 9.2 (3.2) | 8.7 (3.3) | 0.594 |
| Sex (women/men) | 14/9 | 14/9 | |
| Weight (kg) | 28.3 (12.7) | 34.6 (16.8) | 0.161 |
| Height (m) | 1.32 (0.20) | 1.36 (0.20) | 0.503 |
| BMI (kg/m2) | 15.5 (3.4) | 17.5 (3.5) | 0.049* |
| GMFCS level (frequency) | I: 11; II: 4; III: 1; IV: 7 | - | - |
| Flexor muscles tone level (frequency) | 0: 10; 1: 7; 1+: 6 | - | - |
| Extensor muscles tone level (frequency) | 0: 10; 1: 7; 1+: 6 | - | - |
| Right sternocleidomastoid muscles tone level (frequency) | 0: 9; 1: 8; 1+: 6 | - | - |
| Left sternocleidomastoid muscles tone level (frequency) | 0: 9; 1: 8; 1+: 6 | - | - |
| Spatial Plane | IMU First Assessment on the First Day (Standard Deviation) | CROM Assessment (Standard Deviation) | Pearson r (p-Value) | Student’s t-test (p-Value) |
|---|---|---|---|---|
| CP group (n = 23) | ||||
| Flexion-Extension | 133.3 (24.6) | 129.0 (22.4) | 0.969 (<0.001) | −3.333 (0.003) |
| Rotational | 153.5 (19.9) | 145.3 (14.7) | 0.601 (0.003) | −2.396 (0.026) |
| Side-bending | 91.3 (25.7) | 96.1 (23.3) | 0.916 (<0.001) | 2.236 (0.036) |
| Control group (n = 23) | ||||
| Flexion-Extension | 137.0 (24.5) | 133.3 (23.9) | 0.992 (<0.001) | −5.771 (<0.001) |
| Rotational | 147.0 (19.0) | 146.1 (16.1) | 0.786 (<0.001) | −0.371 (0.714) |
| Side-bending | 90.7 (17.5) | 94.9 (18.9) | 0.949 (<0.001) | 3.413 (0.002) |
| Intra-Day Reliability | ||||
| Spatial Plane | IMU Second Assessment on the First Day (Standard Deviation) | ICC (95%CI) | SEM (º) | MDC90 (º) |
| CP group (n = 23) | ||||
| Flexion-Extension | 138.8 (26.5) | 0.900 (0.762, 0.958) | 8.0 | 18.6 |
| Rotational | 151.3 (20.1) | 0.821 (0.600, 0.920) | 8.3 | 19.4 |
| Side-bending | 91.3 (23.7) | 0.925 (0.822, 0.968) | 6.7 | 15.5 |
| Control group (n = 23) | ||||
| Flexion-Extension | 135.0 (24.7) | 0.893 (0.750, 0.955) | 7.9 | 18.4 |
| Rotational | 147.6 (20.3) | 0.902 (0.750, 0.961) | 5.4 | 12.6 |
| Side-bending | 86.3 (16.1) | 0.913 (0.772, 0.965) | 5.0 | 11.4 |
| Inter-day reliability | ||||
| IMU assessment on the second day (standard deviation) | ||||
| CP group (n = 23) | ||||
| Flexion-Extension | 140.8 (26.4) | 0.873 (0.680, 0.947) | 9.0 | 21.1 |
| Rotational | 156.2 (19.5) | 0.533 (0.117, 0.803) | 13.3 | 30.9 |
| Side-bending | 94.4 (24.8) | 0.890 (0.743, 0.953) | 8.3 | 19.3 |
| Control group (n = 23) | ||||
| Flexion-Extension | 140.2 (29.5) | 0.831 (0.602, 0.928) | 11.0 | 25.6 |
| Rotational | 153.2 (19.1) | 0.846 (0.601, 0.935) | 6.5 | 15.1 |
| Side-bending | 93.1 (16.5) | 0.864 (0.653, 0.946) | 6.1 | 14.2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carmona-Pérez, C.; Garrido-Castro, J.L.; Torres Vidal, F.; Alcaraz-Clariana, S.; García-Luque, L.; Alburquerque-Sendín, F.; Rodrigues-de-Souza, D.P. Concurrent Validity and Reliability of an Inertial Measurement Unit for the Assessment of Craniocervical Range of Motion in Subjects with Cerebral Palsy. Diagnostics 2020, 10, 80. https://doi.org/10.3390/diagnostics10020080
Carmona-Pérez C, Garrido-Castro JL, Torres Vidal F, Alcaraz-Clariana S, García-Luque L, Alburquerque-Sendín F, Rodrigues-de-Souza DP. Concurrent Validity and Reliability of an Inertial Measurement Unit for the Assessment of Craniocervical Range of Motion in Subjects with Cerebral Palsy. Diagnostics. 2020; 10(2):80. https://doi.org/10.3390/diagnostics10020080
Chicago/Turabian StyleCarmona-Pérez, Cristina, Juan Luis Garrido-Castro, Francisco Torres Vidal, Sandra Alcaraz-Clariana, Lourdes García-Luque, Francisco Alburquerque-Sendín, and Daiana Priscila Rodrigues-de-Souza. 2020. "Concurrent Validity and Reliability of an Inertial Measurement Unit for the Assessment of Craniocervical Range of Motion in Subjects with Cerebral Palsy" Diagnostics 10, no. 2: 80. https://doi.org/10.3390/diagnostics10020080
APA StyleCarmona-Pérez, C., Garrido-Castro, J. L., Torres Vidal, F., Alcaraz-Clariana, S., García-Luque, L., Alburquerque-Sendín, F., & Rodrigues-de-Souza, D. P. (2020). Concurrent Validity and Reliability of an Inertial Measurement Unit for the Assessment of Craniocervical Range of Motion in Subjects with Cerebral Palsy. Diagnostics, 10(2), 80. https://doi.org/10.3390/diagnostics10020080







