Viscoelastic Biomarkers of Ex Vivo Liver Samples via Torsional Wave Elastography
Abstract
1. Introduction
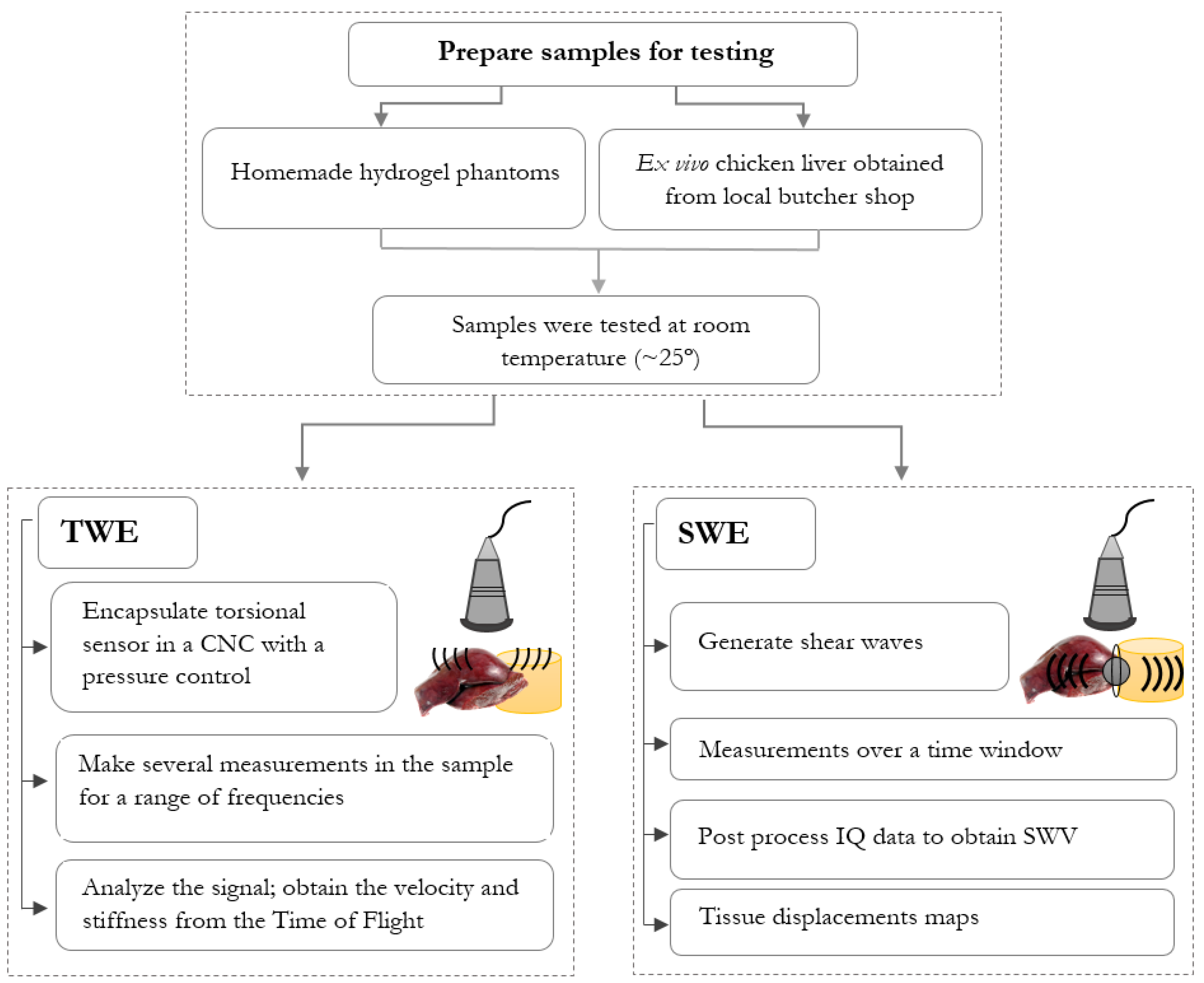
2. Materials and Methods
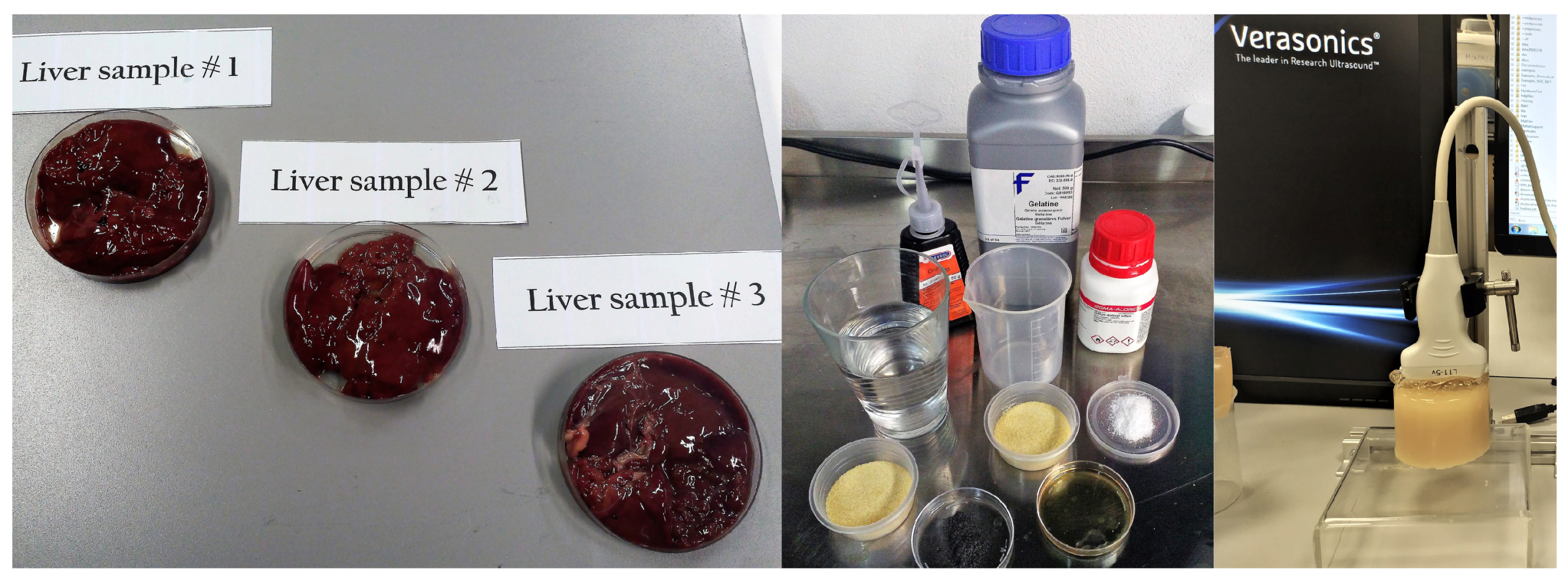
2.1. Hydrogel Phantoms and Ex Vivo Samples
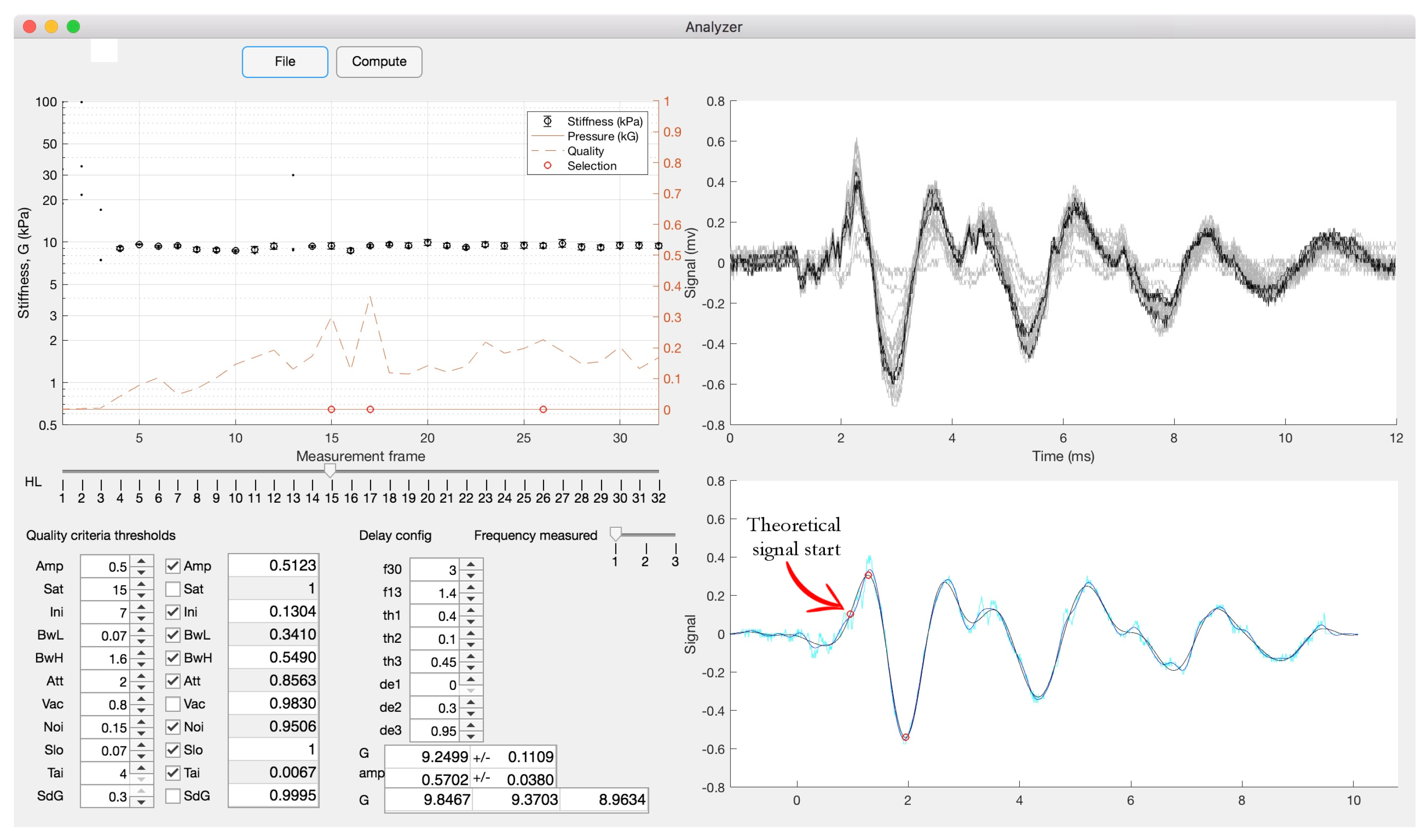
2.2. Torsional Wave Elastography
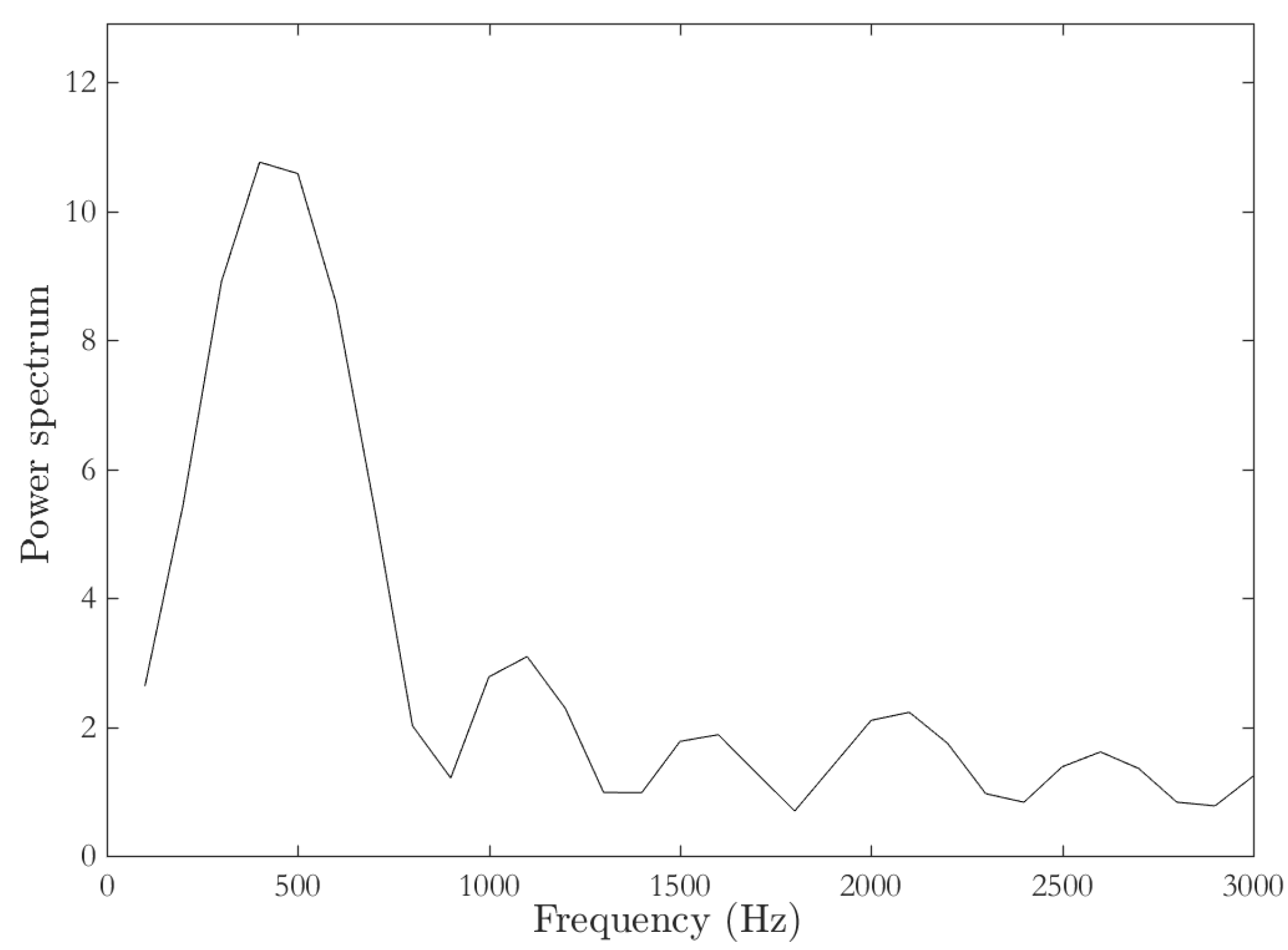
Time of Flight (TOF)- Signal Processing
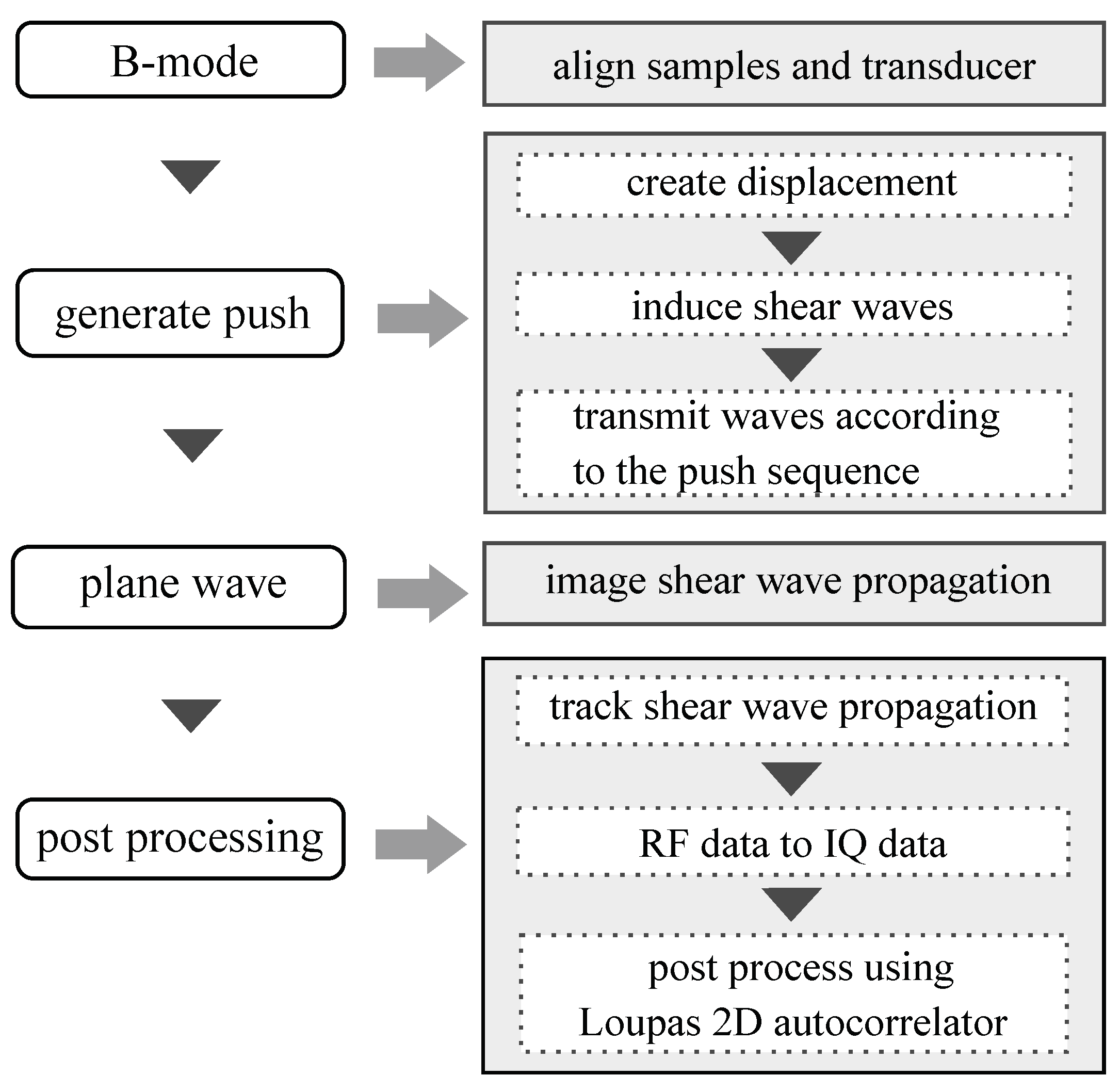
2.3. Shear Wave Elastography
2.3.1. Dispersion Velocity Calculation from Shear Wave Elastography Imaging (SWEI)
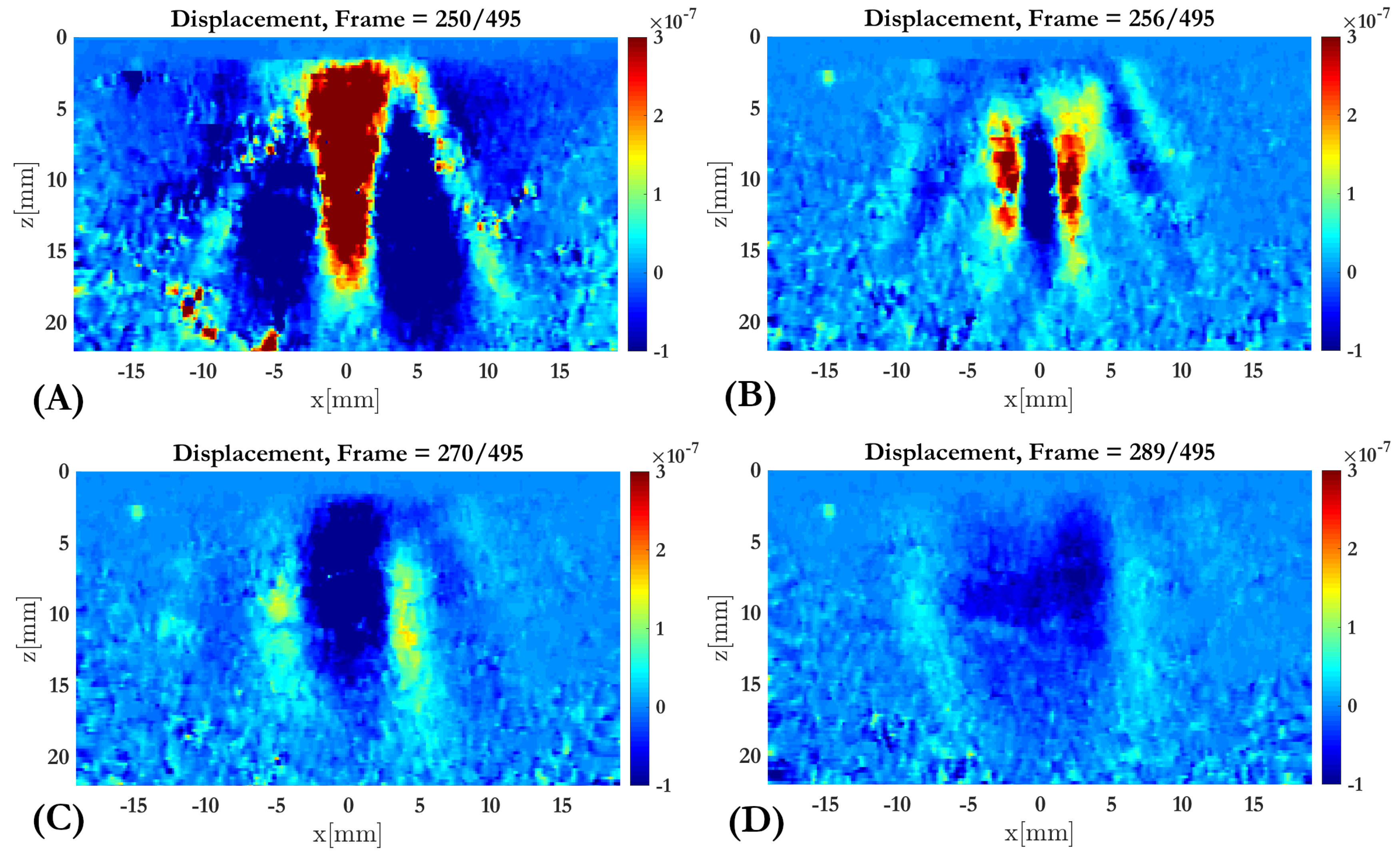
2.3.2. Tissue Motion Estimation
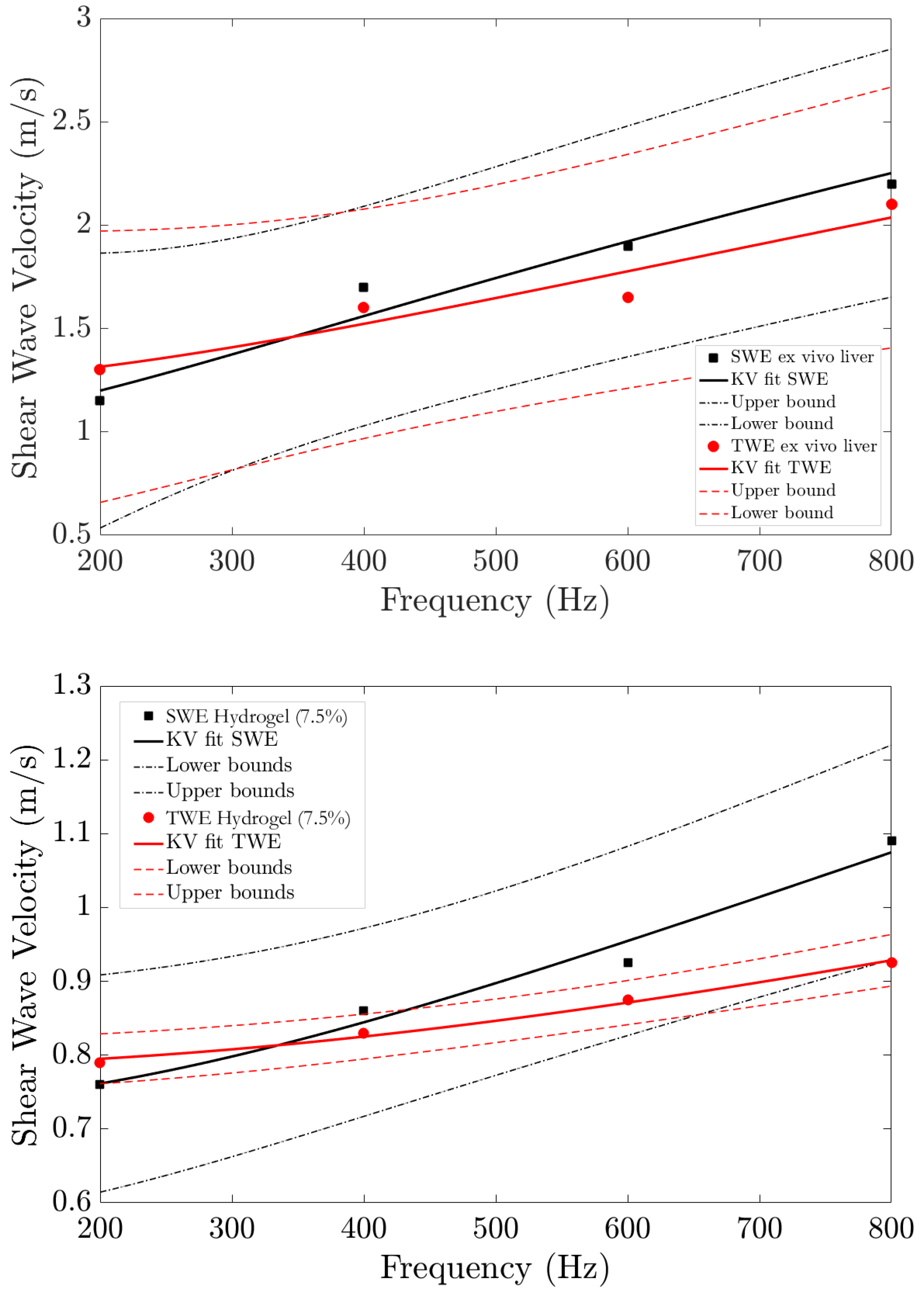
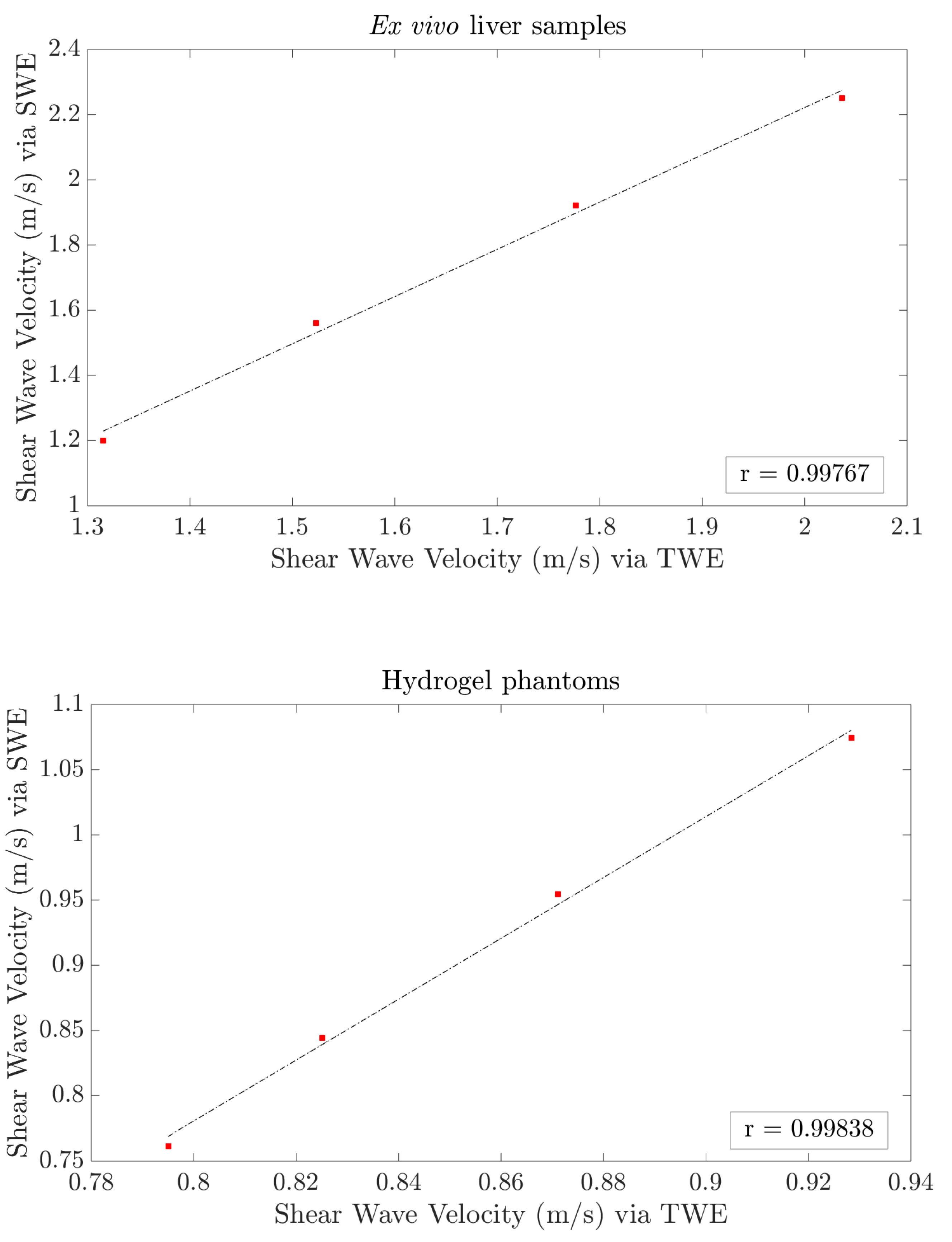
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| TWE | Torsional Wave Elastography |
| SWEI | Shear Wave Elastography Imaging |
| DE | Dynamic Elastography |
| ARFI | Acoustic Radiation Force Impulse |
| MRE | Magmatic Resonance Elastography |
| SWE | Shear Wave Elastography |
| KV | Kelvin–Voigt |
| M | Maxwell |
| PIP | Probabilistic Inverse Problem |
| FDTD | Finite Difference Time Domain |
| TOF | Time of Flight |
| ROI | Region of Interest |
| US | Ultrasonics |
| IQ | In-phase and Quadrature Data |
| CNC | Computer Numerical Control |
| SWV | Shear Wave Velocity |
References
- Tranquart, F.; Grenier, N.; Eder, V.; Pourcelot, L. Clinical use of ultrasound tissue harmonic imaging. Ultrasound Med. Biol. 1999, 25, 889–894. [Google Scholar] [CrossRef]
- Ophir, J.; Céspedes, I.; Ponnekanti, H.; Yazdi, Y.; Li, X. Elastography: A quantitative method for imaging the elasticity of biological tissues. Ultrasonic Imaging 1991, 13, 111–134. [Google Scholar] [CrossRef]
- luc Gennisson, J.; Deffieux, T.; Fink, M.; Tanter, M. Ultrasound elastography: Principles and techniques. Diagn. Interv. Imaging 2013, 94, 487–495. [Google Scholar] [CrossRef]
- Sarvazyan, A.P.; Rudenko, O.; Swanson, S.D.; Fowlkes, J.B.; Emelianov, S.Y. Shear wave elasticity imaging: A new ultrasonic technology of medical diagnostics. Ultrasound Med. Biol. 1998, 24, 1419–1435. [Google Scholar] [CrossRef]
- Urban, M.W.; Nenadic, I.Z.; Mitchell, S.A.; Chen, S.; Greenleaf, J.F. Generalized response of a sphere embedded in a viscoelastic medium excited by an ultrasonic radiation force. J. Acoust. Soc. Am. 2011, 130, 1133–1141. [Google Scholar] [CrossRef]
- Nightingale, K.; Soo, M.S.; Nightingale, R.W.; Trahey, G.E. Acoustic radiation force impulse imaging: In vivo demonstration of clinical feasibility. Ultrasound Med. Biol. 2002, 28, 227–235. [Google Scholar] [CrossRef]
- Bercoff, J.; Tanter, M.; Fink, M.K. Supersonic shear imaging: A new technique for soft tissue elasticity mapping. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2004, 51, 396–409. [Google Scholar] [CrossRef]
- Fahey, B.J.; Nightingale, K.; Nelson, R.C.; Palmeri, M.L.; Trahey, G.E. Acoustic radiation force impulse imaging of the abdomen: Demonstration of feasibility and utility. Ultrasound Med. Biol. 2005, 31, 1185–1198. [Google Scholar] [CrossRef]
- Hirsch, S.; Braun, J.; Sack, I. Magnetic Resonance Elastography: Physical Background and Medical Applications; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Asrani, S.K.; Devarbhavi, H.; Eaton, J.; Kamath, P.S. Burden of liver diseases in the world. J. Hepatol. 2019, 70, 151–171. [Google Scholar] [CrossRef]
- Marcellin, P.; Kutala, B.K. Liver diseases: A major, neglected global public health problem requiring urgent actions and large-scale screening. Liver Int. Off. J. Int. Assoc. Study Liver 2018, 38 (Suppl. 1), 2–6. [Google Scholar] [CrossRef]
- Sande, J.A.; Verjee, S.S.; Vinayak, S.; Amersi, F.; Ghesani, M.V. Ultrasound shear wave elastography and liver fibrosis: A Prospective Multicenter Study. World J. Hepatol. 2017, 9, 38–47. [Google Scholar] [CrossRef]
- Mancini, M.; Salomone Megna, A.; Ragucci, M.; De Luca, M.; Marino Marsilia, G.; Nardone, G.; Coccoli, P.; Prinster, A.; Mannelli, L.; Vergara, E.; et al. Reproducibility of shear wave elastography (SWE) in patients with chronic liver disease. PLoS ONE 2017, 12, e0185391. [Google Scholar] [CrossRef]
- Hoodeshenas, S.; Yin, M.; Venkatesh, S.K. Magnetic Resonance Elastography of Liver: Current Update. Top. Magn. Reson. Imaging 2018, 27, 319–333. [Google Scholar] [CrossRef]
- Huwart, L.; Sempoux, C.; Vicaut, E.; Salameh, N.; Annet, L.; Danse, E.; Peeters, F.; ter Beek, L.C.; Rahier, J.; Sinkus, R.; et al. Magnetic resonance elastography for the noninvasive staging of liver fibrosis. Gastroenterology 2008, 135, 32–40. [Google Scholar] [CrossRef]
- Venkatesh, S.K.; Wang, G.; Lim, S.G.; Wee, A. Magnetic resonance elastography for the detection and staging of liver fibrosis in chronic hepatitis B. Eur. Radiol. 2014, 24, 70–78. [Google Scholar] [CrossRef]
- Ouared, A.; Montagnon, E.; Cloutier, G. Generation of remote adaptive torsional shear waves with an octagonal phased array to enhance displacements and reduce variability of shear wave speeds: Comparison with quasi-plane shear wavefronts. Phys. Med. Biol. 2015, 60, 8161–8185. [Google Scholar] [CrossRef]
- Aglyamov, S.R.; Park, S.; Ilinskii, Y.A.; Emelianov, S.Y. Ultrasound imaging of soft tissue shear viscosity. In Proceedings of the IEEE Symposium on Ultrasonics, Honolulu, HI, USA, 5–8 October 2003; Volume 1, pp. 937–940. [Google Scholar]
- Guillermo, R.C.; Nicolás, B.; Juan Manuel, M.R.; Jesús, F.N.; Laura María, P.P. Transductor ultrasónico de torsión para diagnóstico tisular. Extensiones Internacionales PCT/ES2012/070380, 14 July 2011. [Google Scholar]
- Guillermo, R.C.; Alicia, V.M.; Elena, S.M.; Francisca Sonia, M.G. Dispositivo emisor de ondas ultrasónicas de torsión y transductor que lo comprende. Extensiones Internacionales PCT/ES2016/070540, 16 July 2015. [Google Scholar]
- Callejas, A.; Gomez, A.; Melchor, J.; Riveiro, M.; Massó, P.; Torres, J.; López-López, M.; Rus, G. Performance study of a torsional wave sensor and cervical tissue characterization. Sensors 2017, 17, 2078. [Google Scholar] [CrossRef]
- Callejas, A.; Gomez, A.; Faris, I.H.; Melchor, J.; Rus, G. Kelvin–Voigt Parameters Reconstruction of Cervical Tissue-Mimicking Phantoms Using Torsional Wave Elastography. Sensors 2019, 19, 3281. [Google Scholar] [CrossRef]
- Park, J.M.; Choi, S.M.; Kwon, S.J.; Jeong, M.K. Measurement of the shear modulus of an ultrasound tissue phantom. J. Acoust. Soc. Korea 2012, 31, 399–409. [Google Scholar] [CrossRef]
- Dunmire, B.L.; Kucewicz, J.C.; Mitchell, S.B.; Crum, L.A.; Sekins, K.M. Characterizing an agar/gelatin phantom for image guided dosing and feedback control of high-intensity focused ultrasound. Ultrasound Med. Biol. 2013, 39, 300–311. [Google Scholar] [CrossRef]
- Melchor, J.M.; Rus, G. Torsional ultrasonic transducer computational design optimization. Ultrasonics 2014, 54, 1950–1962. [Google Scholar] [CrossRef]
- Melchor, J.; Muñoz, R.; Rus, G. Torsional ultrasound sensor optimization for soft tissue characterization. Sensors 2017, 17, 1402. [Google Scholar] [CrossRef]
- Rus, G.; Muñoz, R.; Melchor, J.; Molina, R.; Callejas, A.; Riveiro, M.; Massó, P.; Torres, J.; Moreu, G.; Molina, F.; et al. Torsion ultrasonic sensor for tissue mechanical characterization. In Proceedings of the 2016 IEEE International Ultrasonics Symposium (IUS), Tours, France, 18–21 September 2016; pp. 1–4. [Google Scholar]
- Nightingale, K.; Palmeri, M.L.; Nightingale, R.W.; Trahey, G.E. On the feasibility of remote palpation using acoustic radiation force. J. Acoust. Soc. Am. 2001, 110, 625–634. [Google Scholar] [CrossRef]
- Torr, G.R. The acoustic radiation force. Am. J. Phys. 1984, 52, 402–408. [Google Scholar] [CrossRef]
- Deng, Y.; Rouze, N.C.; Palmeri, M.L.; Nightingale, K.R. Ultrasonic shear wave elasticity imaging sequencing and data processing using a Verasonics research scanner. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2016, 64, 164–176. [Google Scholar] [CrossRef]
- Palmeri, M.L.; McAleavey, S.A.; Trahey, G.E.; Nightingale, K. Ultrasonic tracking of acoustic radiation force-induced displacements in homogeneous media. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2006, 53, 1300–1313. [Google Scholar] [CrossRef]
- Deffieux, T.; luc Gennisson, J.; Larrat, B.; Fink, M.; Tanter, M. The variance of quantitative estimates in shear wave imaging: Theory and experiments. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2012, 59, 2390–2410. [Google Scholar] [CrossRef]
- Carcione, J.M. (Ed.) Wave Fields in Real Media, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Peralta, L. Feasibility of Using Ultrasonic Shear Waves to Assess Cervical Remodelling During the Gestation Period. Ph.D. Thesis, Departamento de Mecánica Estructural e Ingeniería Hidráulica, University of Granada, Granada, Spain, 2015. [Google Scholar]
- Catheline, S.; luc Gennisson, J.; Delon, G.; Fink, M.; Sinkus, R.; Abouelkaram, S.; Culioli, J. Measuring of viscoelastic properties of homogeneous soft solid using transient elastography: An inverse problem approach. J. Acoust. Soc. Am. 2004, 116, 3734–3741. [Google Scholar] [CrossRef]
- Loupas, T.; Powers, J.; Gill, R.W. An axial velocity estimator for ultrasound blood flow imaging, based on a full evaluation of the Doppler equation by means of a two-dimensional autocorrelation approach. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 1995, 42, 672–688. [Google Scholar] [CrossRef]
- Rodriguez-Molares, A.; Rindal, O.M.H.; Bernard, O.; Liebgott, H.; Austeng, A.; Lovstakken, L. The ultrasound toolbox. In Proceedings of the 2017 IEEE International Ultrasonics Symposium (IUS), Washington, DC, USA, 6–9 September 2017. [Google Scholar]
- Orescanin, M.B.; Qayyum, M.A.; Toohey, K.S.; Insana, M.F. Complex shear modulus of thermally-damaged liver. In Proceedings of the 2009 IEEE International Ultrasonics Symposium, Rome, Italy, 20–23 September 2009; pp. 127–130. [Google Scholar]
- Barry, C.T.; Mills, B.J.; Hah, Z.; Mooney, R.A.; Ryan, C.K.; Rubens, D.J.; Parker, K.J. Shear wave dispersion measures liver steatosis. Ultrasound Med. Biol. 2012, 38, 175–182. [Google Scholar] [CrossRef]
- Kim, S.O.; Lee, S.Y.; Jang, S.L.; jin Park, S.; Kwon, H.W.; Kim, S.H.; Lee, C.S.; Choi, E.S.; Cho, S.K.; Hong, S.H.; et al. Hepatic Stiffness Using Shear Wave Elastography and the Related Factors for a Fontan Circulation. Pediatr. Cardiol. 2017, 39, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.Z.; Dutta, D.; Mahmoud, A.M.; Tillman, B.W.; Leers, S.A.; Kim, K. An adaptive displacement estimation algorithm for improved reconstruction of thermal strain. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2015, 62, 138–151. [Google Scholar] [CrossRef] [PubMed]
- Pinton, G.; Dahl, J.J.; Trahey, G.E. Rapid tracking of small displacements with ultrasound. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2006, 53, 1103–1117. [Google Scholar] [CrossRef] [PubMed]
- Massó, P.; Callejas, A.; Melchor, J.; Molina, F.S.; Rus, G. In Vivo Measurement of Cervical Elasticity on Pregnant Women by Torsional Wave Technique: A Preliminary Study. Sensors 2019, 19, 3249. [Google Scholar] [CrossRef]
- Fink, M.; Sandrin, L.; Tanter, M.; Catheline, S.; Chaffai, S.; Bercoff, J.; Gennisson, J. Ultra high speed imaging of elasticity. In Proceedings of the 2002 IEEE Ultrasonics Symposium, Munich, Germany, 8–11 October 2002; Volume 2, pp. 1811–1820. [Google Scholar] [CrossRef]
- Sigrist, R.M.; Liau, J.; Kaffas, A.E.; Chammas, M.C.; Willmann, J.K. Ultrasound Elastography: Review of Techniques and Clinical Applications. Theranostics 2017, 7, 1303–1329. [Google Scholar] [CrossRef]
- Hong, E.K.; Choi, Y.H.; Cheon, J.E.; Kim, W.S.; Kim, I.O.; Kang, S.Y. Accurate measurements of liver stiffness using shear wave elastography in children and young adults and the role of the stability index. Ultrasonography 2018, 37, 226–232. [Google Scholar] [CrossRef]
- Samir, A.E.; Dhyani, M.; Vij, A.; Bhan, A.K.; Halpern, E.F.; Méndez-Navarro, J.; Corey, K.E.; Chung, R.T. Shear-wave elastography for the estimation of liver fibrosis in chronic liver disease: Determining accuracy and ideal site for measurement. Radiology 2015, 274, 888–896. [Google Scholar] [CrossRef]
- Mulabecirovic, A.; Mjelle, A.B.; Gilja, O.H.; Vesterhus, M.; Havre, R.F. Repeatability of shear wave elastography in liver fibrosis phantoms—Evaluation of five different systems. PLoS ONE 2018, 13, e0189671. [Google Scholar] [CrossRef]
- Dietrich, C.F.; Bamber, J.; Berzigotti, A.; Bota, S.; Cantisani, V.; Castera, L.; Cosgrove, D.; Ferraioli, G.; Friedrich-Rust, M.; Gilja, O.H.; et al. EFSUMB Guidelines and Recommendations on the Clinical Use of Liver Ultrasound Elastography, Update 2017 (Long Version). Ultraschall Med. 2017, 38, e48. [Google Scholar]
- Friedrich-Rust, M.; Poynard, T.; Castera, L. Critical comparison of elastography methods to assess chronic liver disease. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 402–411. [Google Scholar] [CrossRef]
- Deffieux, T.; Gennisson, J.L.; Bousquet, L.; Corouge, M.; Cosconea, S.; Amroun, D.; Tripon, S.; Terris, B.; Mallet, V.; Sogni, P.; et al. Investigating liver stiffness and viscosity for fibrosis, steatosis and activity staging using shear wave elastography. J. Hepatol. 2015, 62, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Salameh, N.; Larrat, B.; Abarca-Quinones, J.; Pallu, S.; Dorvillius, M.; Leclercq, I.; Fink, M.; Sinkus, R.; Van Beers, B.E. Early detection of steatohepatitis in fatty rat liver by using MR elastography. Radiology 2009, 253, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Muller, M.; Gennisson, J.L.; Deffieux, T.; Tanter, M.; Fink, M. Quantitative viscoelasticity mapping of human liver using supersonic shear imaging: Preliminary in vivo feasibility study. Ultrasound Med. Biol. 2009, 35, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhang, C.; Li, J.; Huo, H.; Song, D. Diagnostic Accuracy of Real-Time Shear Wave Elastography for Staging of Liver Fibrosis: A Meta-Analysis. Med Sci. Monit. Int. Med J. Exp. Clin. Res. 2016, 22, 1349–1359. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cassinotto, C.; Lapuyade, B.; Mouries, A.; Hiriart, J.B.; Vergniol, J.; Gaye, D.; Castain, C.; Le Bail, B.; Chermak, F.; Foucher, J.; et al. Non-invasive assessment of liver fibrosis with impulse elastography: Comparison of Supersonic Shear Imaging with ARFI and FibroScan®. J. Hepatol. 2014, 61, 550–557. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Sanchez, W.; Callstrom, M.R.; Gorman, B.; Lewis, J.T.; Sanderson, S.O.; Greenleaf, J.F.; Xie, H.; Shi, Y.; Pashley, M.; et al. Assessment of liver viscoelasticity by using shear waves induced by ultrasound radiation force. Radiology 2013, 266, 964–970. [Google Scholar] [CrossRef]
- Deffieux, T.; Montaldo, G.; Tanter, M.; Fink, M. Shear wave spectroscopy for in vivo quantification of human soft tissues visco-elasticity. IEEE Trans. Med Imaging 2008, 28, 313–322. [Google Scholar] [CrossRef]
- Bercoff, J.; Tanter, M.; Muller, M.; Fink, M. The role of viscosity in the impulse diffraction field of elastic waves induced by the acoustic radiation force. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2004, 51, 1523–1536. [Google Scholar] [CrossRef]
- Barry, C.T.; Hah, Z.; Partin, A.; Mooney, R.A.; Chuang, K.H.; Augustine, A.; Almudevar, A.; Cao, W.; Rubens, D.J.; Parker, K.J. Mouse liver dispersion for the diagnosis of early-stage fatty liver disease: A 70-sample study. Ultrasound Med. Biol. 2014, 40, 704–713. [Google Scholar] [CrossRef]
- Zafra, C.; Manuel, A. Feasibility of Using Torsional Waves to Assess Viscoelasticity of Cervical Tissue; Universidad de Granada: Granada, Spain, 2019. [Google Scholar]



| Measurements Acquisition Parameters | Value |
|---|---|
| Sampling frequency | 80 Hz (Decimated 10 × after 800 Hz) |
| Ring-disc radius | 3 mm |
| Frequency | 200–800 Hz |
| Averaging | 10 × |
| Excitation power | 20 V |
| Property | Value |
|---|---|
| Number of Elements | 128 |
| Pitch (mm) | 0.3 |
| Elevation focus (mm) | 18 |
| Sensitivity (dB) | −52 ± 3 |
| Parameter | Value for the Transducer |
|---|---|
| Push frequency (MHz) | 4.8 |
| Track frequency (MHz) | 7.81 |
| Push duration (cycles) | 1000 |
| Pulse repetition interval | 100 |
| Impulse duration (cycles/) | 1000, 128 |
| Impulse focus (mm) | 16 for ex vivo liver and 12 for hydrogel phantoms |
| Beam focus configuration | Plane wave, fully open |
| IQ data beam forming sampling frequency | 0.25 |
| Excitation voltage (V) | 40 for ex vivo liver and 28 for hydrogel phantoms |
| Sampling frequency(Hz) | 3000 |
| Number of transmission channels | 128 |
| Number of reception channels | 128 |
| Elastic Parameter: Shear Moduli in kPa | ||||
|---|---|---|---|---|
| Ex Vivo Liver Samples | Hydrogel Phantoms | |||
| Frequency (Hz) | ||||
| 200 | ||||
| 400 | ||||
| 600 | ||||
| 800 | ||||
| Sample | Fit | Viscous Parameters | The Goodness of Fit R-square | ||||
|---|---|---|---|---|---|---|---|
| TWE | SWEI | TWE | SWEI | ||||
| Ex vivo liver | Kelvin–Voigt (KV) | kPa | Pa·s | kPa | Pa·s | 0.9198 | 0.9572 |
| Maxwell (M) | kPa | Pa·s | kPa | Pa·s | 0.835 | 0.9861 | |
| Hydrogel phantom | Kelvin–Voigt (KV) | kPa | Pa·s | kPa | Pa·s | 0.9926 | 0.9764 |
| Maxwell (M) | kPa | Pa·s | kPa | Pa·s | 0.7879 | 0.8237 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faris, I.H.; Melchor, J.; Callejas, A.; Torres, J.; Rus, G. Viscoelastic Biomarkers of Ex Vivo Liver Samples via Torsional Wave Elastography. Diagnostics 2020, 10, 111. https://doi.org/10.3390/diagnostics10020111
Faris IH, Melchor J, Callejas A, Torres J, Rus G. Viscoelastic Biomarkers of Ex Vivo Liver Samples via Torsional Wave Elastography. Diagnostics. 2020; 10(2):111. https://doi.org/10.3390/diagnostics10020111
Chicago/Turabian StyleFaris, Inas H., Juan Melchor, Antonio Callejas, Jorge Torres, and Guillermo Rus. 2020. "Viscoelastic Biomarkers of Ex Vivo Liver Samples via Torsional Wave Elastography" Diagnostics 10, no. 2: 111. https://doi.org/10.3390/diagnostics10020111
APA StyleFaris, I. H., Melchor, J., Callejas, A., Torres, J., & Rus, G. (2020). Viscoelastic Biomarkers of Ex Vivo Liver Samples via Torsional Wave Elastography. Diagnostics, 10(2), 111. https://doi.org/10.3390/diagnostics10020111






