Is Ultrasound an Accurate Alternative for Mammography in Breast Cancer Screening in an Asian Population? A Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy and Selection Criteria
2.2. Inclusion Criteria
2.3. Data Extraction
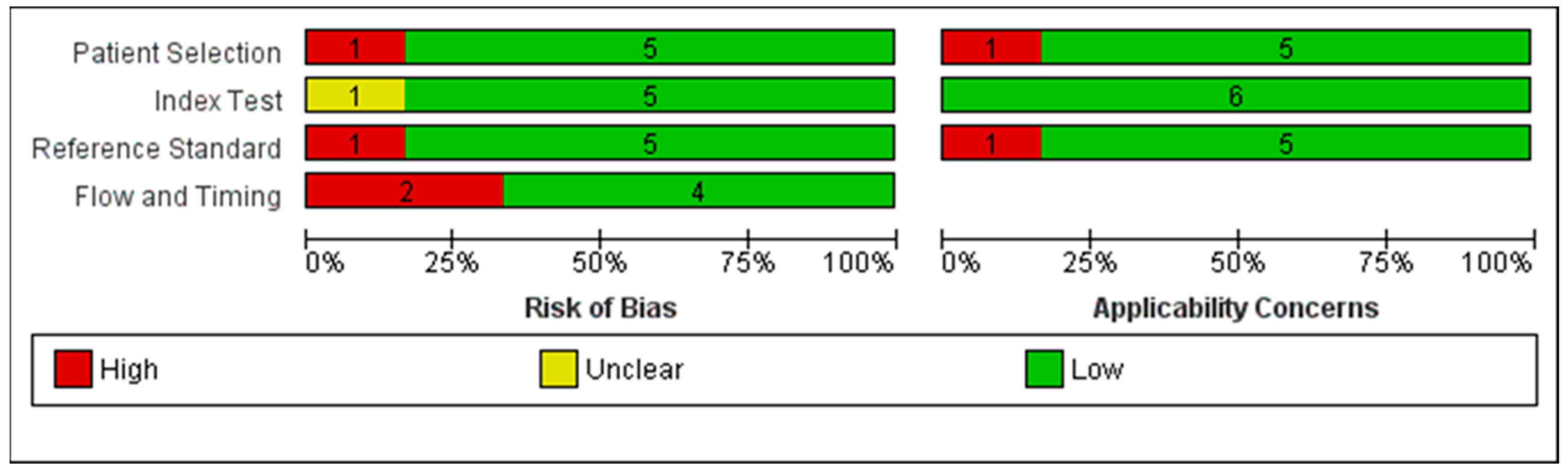
2.4. Quality Assessment
2.5. Data Analysis
3. Results
3.1. Studies Characteristics
3.2. Quality Assessment
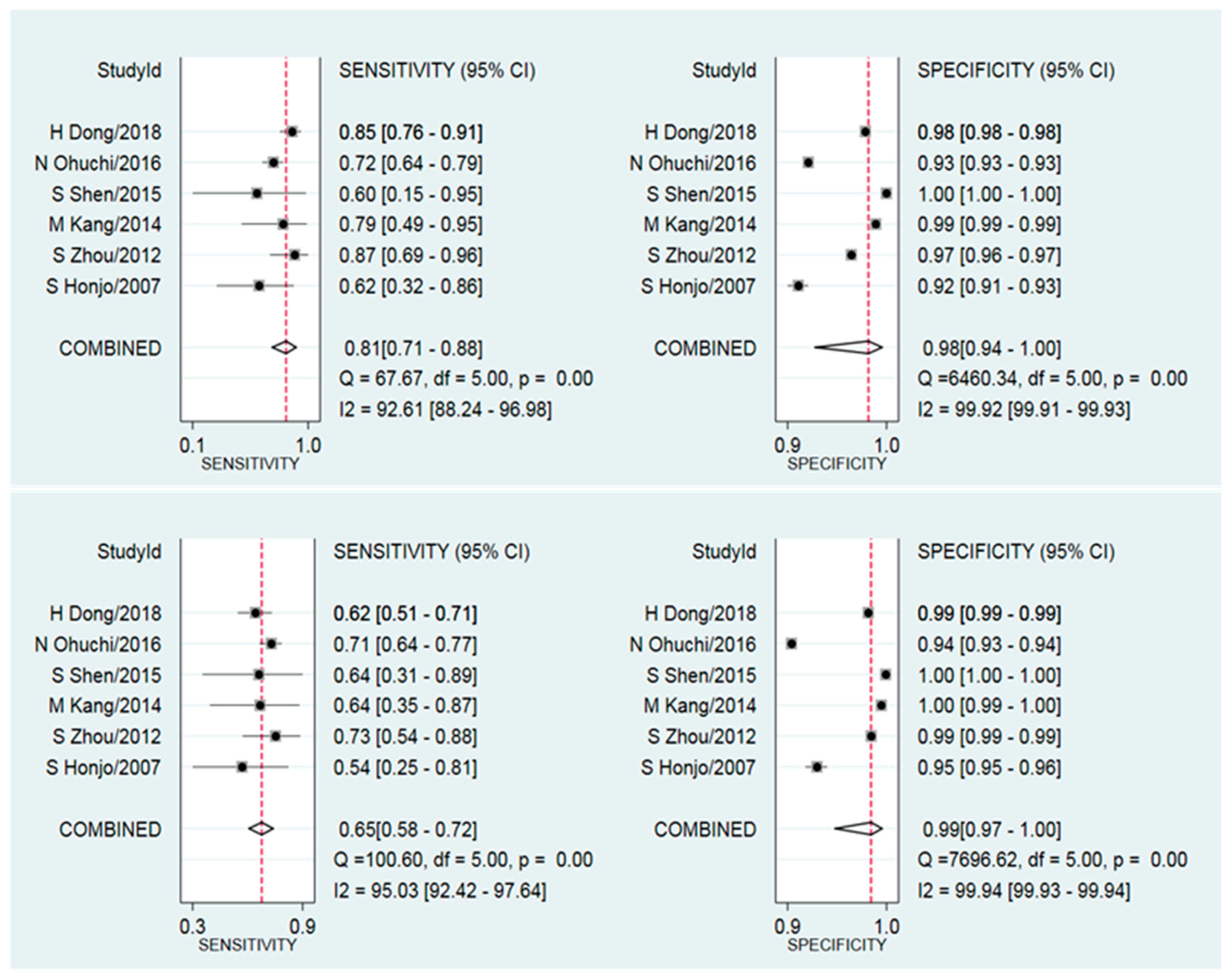
3.3. Synthesis Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- DeSantis, C.E.; Bray, F.; Ferlay, J.; Lortet-Tieulent, J.; Anderson, B.O.; Jemal, A. International Variation in Female Breast Cancer Incidence and Mortality Rates. Cancer Epidemiol. Biomark. Prev. 2015, 24, 1495–1506. [Google Scholar] [CrossRef]
- Fan, L.; Goss, P.E.; Strasser-Weippl, K.K. Current Status and Future Projections of Breast Cancer in Asia. Breast Care 2015, 10, 372–378. [Google Scholar] [CrossRef]
- Sung, H.; Rosenberg, P.S.; Chen, W.-Q.; Hartman, M.; Lim, W.-Y.; Chia, K.S.; Mang, O.W.-K.; Chiang, C.-J.; Kang, D.; Ngan, R.K.-C.; et al. Female Breast Cancer Incidence Among Asian and Western Populations: More Similar Than Expected. J. Natl. Cancer Inst. 2015, 107, djv107. [Google Scholar] [CrossRef]
- Moss, S.M.; Wale, C.; A Smith, R.; Evans, A.; Cuckle, H.; Duffy, S.W. Effect of mammographic screening from age 40 years on breast cancer mortality in the UK Age trial at 17 years’ follow-up: A randomised controlled trial. Lancet Oncol. 2015, 16, 1123–1132. [Google Scholar] [CrossRef]
- Tabár, L.; Yen, M.-F.; Vitak, B.; Chen, H.-H.T.; Smith, R.A.; Duffy, S.W. Mammography service screening and mortality in breast cancer patients: 20-year follow-up before and after introduction of screening. Lancet 2003, 361, 1405–1410. [Google Scholar] [CrossRef]
- De Munck, L.; Fracheboud, J.; De Bock, G.H.; Heeten, G.J.D.; Siesling, S.; Broeders, M.J.M. Is the incidence of advanced-stage breast cancer affected by whether women attend a steady-state screening program? Int. J. Cancer 2018, 143, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Gøtzsche, P.C.; Jørgensen, K.J. Screening for breast cancer with mammography. Cochrane Database Syst. Rev. 2013, 6, CD001877. [Google Scholar] [CrossRef] [PubMed]
- Marmot, M.; Altman, D.; Cameron, D.; Dewar, J.; Thompson, S.; Wilcox, M. The benefits and harms of breast cancer screening: An independent review. Br. J. Cancer 2013, 108, 2205–2240. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, L.L.; Helfand, M.; Chan, B.K.; Woolf, S.H. Breast Cancer Screening: A Summary of the Evidence for the U.S. Preventive Services Task Force. Ann. Intern. Med. 2002, 137, 347–360. [Google Scholar] [CrossRef]
- Hamashima, C.; Japanese Research Group for the Development of Breast Cancer Screening Guidelines; Hattori, M.; Honjo, S.; Kasahara, Y.; Katayama, T.; Nakai, M.; Nakayama, T.; Morita, T.; Ohta, K.; et al. The Japanese Guidelines for Breast Cancer Screening. Jpn. J. Clin. Oncol. 2016, 46, 482–492. [Google Scholar] [CrossRef]
- Lee, E.H.; Park, B.; Kim, N.-S.; Seo, H.-J.; Ko, K.L.; Min, J.; Shin, M.-H.; Lee, K.; Lee, S.; Choi, N.; et al. The Korean guideline for breast cancer screening. J. Korean Med Assoc. 2015, 58, 408–419. [Google Scholar] [CrossRef]
- Loy, E.Y.; Molinar, D.; Chow, K.Y.; Fock, C. National Breast Cancer Screening Programme, Singapore: Evaluation of participation and performance indicators. J. Med Screen. 2015, 22, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Leong, S.P.L.; Shen, Z.-Z.; Liu, T.-J.; Agarwal, G.; Tajima, T.; Paik, N.-S.; Sandelin, K.; Derossis, A.; Cody, H.; Foulkes, W.D. Is Breast Cancer the Same Disease in Asian and Western Countries? World J. Surg. 2010, 34, 2308–2324. [Google Scholar] [CrossRef] [PubMed]
- Maskarinec, G.; Meng, L.; Ursin, G. Ethnic differences in mammographic densities. Int. J. Epidemiol. 2001, 30, 959–965. [Google Scholar] [CrossRef] [PubMed]
- Rajaram, N.; Mariapun, S.; Eriksson, M.; Tapia, J.; Kwan, P.Y.; Ho, W.K.; Harun, F.; Rahmat, K.; Czene, K.; Taib, N.A.M.; et al. Differences in mammographic density between Asian and Caucasian populations: A comparative analysis. Breast Cancer Res. Treat. 2017, 161, 353–362. [Google Scholar] [CrossRef]
- Armstrong, K.; Moye, E.; Williams, S.; Berlin, J.A.; Reynolds, E.E. Screening Mammography in Women 40 to 49 Years of Age: A Systematic Review for the American College of Physicians. Ann. Intern. Med. 2007, 146, 516–526. [Google Scholar] [CrossRef]
- Rivera-Franco, M.M.; Leon-Rodriguez, E. Delays in Breast Cancer Detection and Treatment in Developing Countries. Breast Cancer Basic Clin. Res. 2018, 12, 1178223417752677. [Google Scholar] [CrossRef]
- Nothacker, M.; Duda, V.; Hahn, M.; Warm, M.; Degenhardt, F.; Madjar, H.; Weinbrenner, S.; Albert, U.-S. Early detection of breast cancer: Benefits and risks of supplemental breast ultrasound in asymptomatic women with mammographically dense breast tissue. A systematic review. BMC Cancer 2009, 9, 335. [Google Scholar] [CrossRef]
- Hwang, J.-Y.; Han, B.-K.; Ko, E.Y.; Shin, J.H.; Hahn, S.Y.; Nam, M.Y. Screening Ultrasound in Women with Negative Mammography: Outcome Analysis. Yonsei Med. J. 2015, 56, 1352–1358. [Google Scholar] [CrossRef]
- Moher, D.; PRISMA-P Group; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef]
- Deeks, J.; Wisniewski, S.; Davenport, C. Chapter 4: Guide to the contents of a Cochrane Diagnostic Test Accuracy Protocol. In Cochrane Handbook for Systematic Reviews of Diagnostic Test Accuracy; Version 1.0.0; Wiley: New York, NY, USA, 2013. [Google Scholar]
- Whiting, P.F.; Rutjes, A.W.; Westwood, M.E.; Mallett, S.; Deeks, J.J.; Reitsma, J.B.; Leeflang, M.M.; Sterne, J.A.; Bossuyt, P.M. QUADAS-2: A Revised Tool for the Quality Assessment of Diagnostic Accuracy Studies. Ann. Intern. Med. 2011, 155, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Macaskill, P.; Gatsonis, C.; Deeks, J.; Harbord, R.; Takwoingi, Y. Chapter 10: Analysing and Presenting Results. In Cochrane Handbook for Systematic Reviews of Diagnostic Test Accuracy; Version 1.0; Wiley: New York, NY, USA, 2010. [Google Scholar]
- Glas, A.S.; Lijmer, J.G.; Prins, M.H.; Bonsel, G.J.; Bossuyt, P.M.M. The diagnostic odds ratio: A single indicator of test performance. J. Clin. Epidemiol. 2003, 56, 1129–1135. [Google Scholar] [CrossRef]
- Leeflang, M. Systematic reviews and meta-analyses of diagnostic test accuracy. Clin. Microbiol. Infect. 2014, 20, 105–113. [Google Scholar] [CrossRef]
- Ohuchi, N.; Suzuki, A.; Sobue, T.; Kawai, M.; Yamamoto, S.; Zheng, Y.-F.; Shiono, Y.N.; Saito, H.; Kuriyama, S.; Tohno, E.; et al. Sensitivity and specificity of mammography and adjunctive ultrasonography to screen for breast cancer in the Japan Strategic Anti-cancer Randomized Trial (J-START): A randomised controlled trial. Lancet 2016, 387, 341–348. [Google Scholar] [CrossRef]
- Honjo, S.; Ando, J.; Tsukioka, T.; Morikubo, H.; Ichimura, M.; Sunagawa, M.; Hasegawa, T.; Watanabe, T.; Kodama, T.; Tominaga, K.; et al. Relative and Combined Performance of Mammography and Ultrasonography for Breast Cancer Screening in the General Population: A Pilot Study in Tochigi Prefecture, Japan. Jpn. J. Clin. Oncol. 2007, 37, 715–720. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.-C.; Fan, Y.-W.; Zeng, W.; Ding, J.-H.; Chen, M.; Wang, B.-H.; Qiu, Y.-F.; Gao, Y.; Zhang, X.; Chang, C. The first stage conclusion of breast cancer screening in Shanghai community: Missed and misdiagnostic cases analysis of mammography and ultrasonography. Shanghai Med. Imaging 2012, 21, 291–293, [In Chinese]. [Google Scholar]
- Kang, M.; Zhao, Y.; Huang, Y.; Li, J.; Liu, L.; Li, H. Accuracy and direct medical cost of different screening modalities for breast cancer among Chinese women. Zhonghua zhong liu za zhi 2014, 36, 236–240, [In Chinese]. [Google Scholar]
- Shen, S.; Zhou, Y.; Xu, Y.; Zhang, B.; Duan, X.; Huang, R.; Li, B.; Shi, Y.; Shao, Z.; Liao, H.; et al. A multi-centre randomised trial comparing ultrasound vs mammography for screening breast cancer in high-risk Chinese women. Br. J. Cancer 2015, 112, 998–1004. [Google Scholar] [CrossRef]
- Dong, H.; Huang, Y.; Song, F.; Dai, H.; Liu, P.; Zhu, Y.; Wang, P.; Han, J.; Hao, X.; Chen, K. Improved Performance of Adjunctive Ultrasonography After Mammography Screening for Breast Cancer Among Chinese Females. Clin. Breast Cancer 2018, 18, e353–e361. [Google Scholar] [CrossRef]
- Xu, J.; Wang, Q.; Ma, H.M.; Xia, J.H. Primary efficacy of physical examination combined with ultragraphy and complemented with mammography for breast cancer screening. Chin. J. Cancer Prev. Treat. 2013, 20, 1295–1299, [In Chinese]. [Google Scholar]
- Ma, D.; Wei, N.; Ni, Q. The primary investigation on the incidence of breast cancer in the institution of Guiyang city. Chin. J. Clin. Oncol. Rehab. 2014, 21, 774–778, [In Chinese]. [Google Scholar]
- Lee, E.H.; Kim, K.W.; Kim, Y.J.; Shin, D.-R.; Park, Y.M.; Lim, H.S.; Park, J.S.; Kim, H.-W.; Kim, Y.M.; Jun, J.K.; et al. Performance of Screening Mammography: A Report of the Alliance for Breast Cancer Screening in Korea. Korean J. Radiol. 2016, 17, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, A.; Kuriyama, S.; Kawai, M.; Amari, M.; Takeda, M.; Ishida, T.; Ohnuki, K.; Nishino, Y.; Tsuji, I.; Shibuya, D.; et al. Age-specific interval breast cancers in Japan: Estimation of the proper sensitivity of screening using a population-based cancer registry. Cancer Sci. 2008, 99, 2264–2267. [Google Scholar] [CrossRef]
- Fan, L.; Strasser-Weippl, K.; Li, J.-J.; Louis, J.S.; Finkelstein, D.M.; Yu, K.-D.; Chen, W.-Q.; Shao, Z.-M.; Goss, P.E. Breast cancer in China. Lancet Oncol. 2014, 15, e279–e289. [Google Scholar] [CrossRef]
- Najjar, H.; Easson, A. Age at diagnosis of breast cancer in Arab nations. Int. J. Surg. 2010, 8, 448–452. [Google Scholar] [CrossRef]
- Lee, S.K.; Society, K.B.C.; Kim, S.W.; Yu, J.-H.; Lee, J.E.; Kim, J.Y.; Woo, J.; Lee, S.; Kim, E.-K.; Moon, H.-G.; et al. Is the high proportion of young age at breast cancer onset a unique feature of Asian breast cancer? Breast Cancer Res. Treat. 2018, 173, 189–199. [Google Scholar] [CrossRef]
- Fan, L.; Zheng, Y.; Yu, K.-D.; Liu, G.-Y.; Wu, J.; Lu, J.-S.; Shen, K.-W.; Shen, Z.-Z.; Shao, Z.-M. Breast cancer in a transitional society over 18 years: Trends and present status in Shanghai, China. Breast Cancer Res. Treat. 2009, 117, 409–416. [Google Scholar] [CrossRef]
- Rosenberg, S.; Newman, L.A.; Partridge, A.H. Breast Cancer in Young Women. JAMA Oncol. 2015, 1, 877–878. [Google Scholar] [CrossRef]
- Del Carmen, M.G.; Halpern, E.F.; Kopans, D.B.; Moy, B.; Moore, R.H.; Goss, P.E.; Hughes, K.S. Mammographic Breast Density and Race. Am. J. Roentgenol. 2007, 188, 1147–1150. [Google Scholar] [CrossRef]
- Chinese Anti-Cancer Association. Chinese female breast cancer screening guideline. Chin. J. Clin. Oncol. 2019, 46, 429–431. [Google Scholar]
- Rebolj, M.; Assi, V.; Brentnall, A.; Parmar, D.; Duffy, S.W. Addition of ultrasound to mammography in the case of dense breast tissue: Systematic review and meta-analysis. Br. J. Cancer 2018, 118, 1559–1570. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Arao, R.F.; Sprague, B.L.; Kerlikowske, K.; Lehman, C.D.; Smith, R.A.; Henderson, L.M.; Rauscher, G.H.; Miglioretti, D.L. Performance of Screening Ultrasonography as an Adjunct to Screening Mammography in Women Across the Spectrum of Breast Cancer Risk. JAMA Intern. Med. 2019, 179, 658–667. [Google Scholar] [CrossRef] [PubMed]
- Lehman, C.D.; Arao, R.F.; Sprague, B.L.; Lee, J.M.; Buist, D.S.M.; Kerlikowske, K.; Henderson, L.M.; Onega, T.; Tosteson, A.N.A.; Rauscher, G.H.; et al. National Performance Benchmarks for Modern Screening Digital Mammography: Update from the Breast Cancer Surveillance Consortium. Radiology 2017, 283, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Nelson, H.D.; O’Meara, E.S.; Kerlikowske, K.; Balch, S.; Miglioretti, D. Factors Associated with Rates of False-Positive and False-Negative Results from Digital Mammography Screening: An Analysis of Registry Data. Ann. Intern. Med. 2016, 164, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Weigel, S.; Heindel, W.; Heidrich, J.; Heidinger, O.; Hense, H.W. Reduction of advanced breast cancer stages at subsequent participation in mammography screening. RöFo 2016, 13, 42–46. [Google Scholar] [CrossRef]
- Hofvind, S.; Geller, B.M.; Skelly, J.; Vacek, P.M. Sensitivity and specificity of mammographic screening as practised in Vermont and Norway. Br. J. Radiol. 2012, 85, e1226–e1232. [Google Scholar] [CrossRef] [PubMed]

| First Author [ref] | Year of Publication | Study Design | Country | Starting Age | Risk Level | Follow-Up (Year) |
|---|---|---|---|---|---|---|
| Ohuchi [26] | 2016 | RCT | Japan | 40 | High | 2.0 |
| Honjo [27] | 2007 | Cohort | Japan | 30 | Average | 1.0 |
| Zhou [28] | 2012 | Cohort | China | 35 | High | 0.5–0.8 |
| Kang [29] | 2014 | Cohort | China | 35 | Average | 1.0 |
| Shen [30] | 2015 | RCT | China | 30 | High | 1.0 |
| Dong [31] | 2018 | Cohort | China | 45 | Average | 1.0 |
| First Author [ref] | Method | Sample Size | Crude Prevalence‰ (95%CI) | TP (%) | FP (%) | FN (%) | TN (%) | PPV (%) | NPV (%) |
|---|---|---|---|---|---|---|---|---|---|
| Ohuchi [26] | US | 36,049 | 5.6 (4.9–6.4) | 143 (0.4) | 2289 (6.3) | 59 (0.2) | 33,558 (93.1) | 5.9 | 99.8 |
| MG | 35,130 | 4.3 (3.7–5.1) | 109 (0.3) | 2576 (7.3) | 43 (0.1) | 32,402 (92.2) | 4.1 | 99.9 | |
| Honjo [27] | US | 3453 | 3.8 (2.0–6.4) | 7 (0.2) | 158 (4.6) | 6 (0.2) | 3282 (95.0) | 4.2 | 99.8 |
| MG | 3453 | 3.8 (2.0–6.4) | 8 (0.2) | 271 (7.8) | 5 (0.2) | 3169 (91.8) | 2.9 | 99.8 | |
| Zhou [28] | US | 7017 | 4.3 (2.9–6.1) | 22 (0.3) | 72 (1.0) | 8 (0.1) | 6915 (98.6) | 23.4 | 99.9 |
| MG | 7020 | 4.3 (2.9–6.1) | 26 (0.4) | 220 (3.1) | 4 (0.1) | 6770 (96.4) | 10.6 | 99.9 | |
| Kang [29] | US | 2471 | 5.7 (3.1–9.5) | 9 (0.4) | 9 (0.4) | 5 (0.2) | 2448 (99.0) | 50.0 | 99.8 |
| MG | 2471 | 5.7 (3.1–9.5) | 11 (0.5) | 24 (1.0) | 3 (0.1) | 2433 (98.4) | 31.4 | 99.9 | |
| Shen [30] | US | 4214 | 2.6 (1.3–4.7) | 7 (0.2) | 3 (0.1) | 4 (0.1) | 4200 (99.6) | 70.0 | 99.9 |
| MG | 4170 | 1.2 (0.4–2.8) | 3 (0.1) | 1 (0.0) | 2 (0.1) | 4164 (99.8) | 75.0 | 99.9 | |
| Dong [31] | US | 31,918 | 3.1 (2.5–3.8) | 61 (0.2) | 389 (1.2) | 38 (0.1) | 31,430 (98.5) | 13.6 | 99.9 |
| MG | 31,918 | 3.1 (2.5–3.8) | 84 (0.3) | 604 (1.9) | 15 (0.1) | 31,215 (97.7) | 12.2 | 99.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Zheng, S.; Ding, L.; Liang, X.; Wang, Y.; Greuter, M.J.W.; de Bock, G.H.; Lu, W. Is Ultrasound an Accurate Alternative for Mammography in Breast Cancer Screening in an Asian Population? A Meta-Analysis. Diagnostics 2020, 10, 985. https://doi.org/10.3390/diagnostics10110985
Wang J, Zheng S, Ding L, Liang X, Wang Y, Greuter MJW, de Bock GH, Lu W. Is Ultrasound an Accurate Alternative for Mammography in Breast Cancer Screening in an Asian Population? A Meta-Analysis. Diagnostics. 2020; 10(11):985. https://doi.org/10.3390/diagnostics10110985
Chicago/Turabian StyleWang, Jing, Senshuang Zheng, Lanjun Ding, Xuan Liang, Yuan Wang, Marcel J.W. Greuter, Geertruida H. de Bock, and Wenli Lu. 2020. "Is Ultrasound an Accurate Alternative for Mammography in Breast Cancer Screening in an Asian Population? A Meta-Analysis" Diagnostics 10, no. 11: 985. https://doi.org/10.3390/diagnostics10110985
APA StyleWang, J., Zheng, S., Ding, L., Liang, X., Wang, Y., Greuter, M. J. W., de Bock, G. H., & Lu, W. (2020). Is Ultrasound an Accurate Alternative for Mammography in Breast Cancer Screening in an Asian Population? A Meta-Analysis. Diagnostics, 10(11), 985. https://doi.org/10.3390/diagnostics10110985






