Heterocyst Thylakoid Bioenergetics
Abstract
1. Introduction
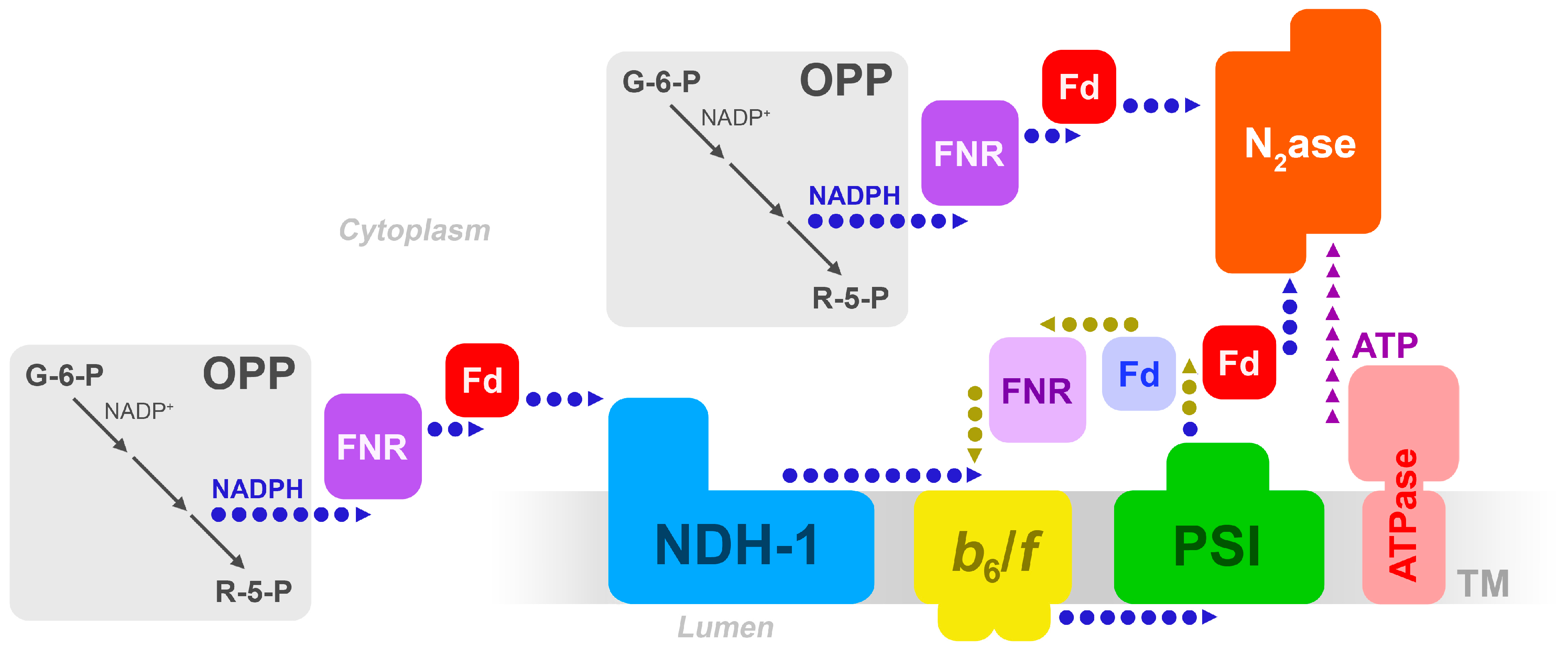
2. Enzymes in the Heterocyst Thylakoids
2.1. Photosystem II
2.2. Photosystem I
2.3. Cytochrome-b6/f
2.4. Ferredoxins
2.5. The Type-1 NADH Dehydrogenase
3. Balancing the Electron/Proton Budget
4. Heterocysts as Cell Factories
4.1. Biotechnological Uses of Heterocysts
4.1.1. Alcohol Production
4.1.2. H2 Production
4.2. Squeezing the Electron Budget
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Mullineaux, C.W. Co-existence of photosynthetic and respiratory activities in cyanobacterial thylakoid membranes. Biochim. Biophys. Acta 2014, 1837, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Fay, P. Oxygen Relations of Nitrogen-Fixation in Cyanobacteria. Microbiol. Rev. 1992, 56, 340–373. [Google Scholar] [PubMed]
- Gallon, J.R. Reconciling the incompatible N2 fixation and O2. New Phytol. 1992, 122, 571–609. [Google Scholar] [CrossRef]
- Cumino, A.C.; Marcozzi, C.; Barreiro, R.; Salerno, G.L. Carbon cycling in Anabaena sp PCC 7120. Sucrose synthesis in the heterocysts and possible role in nitrogen fixation. Plant Physiol. 2007, 143, 1385–1397. [Google Scholar] [CrossRef]
- Curatti, L.; Giarrocco, L.E.; Cumino, A.C.; Salerno, G.L. Sucrose synthase is involved in the conversion of sucrose to polysaccharides in filamentous nitrogen-fixing cyanobacteria. Planta 2008, 228, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Nurnberg, D.J.; Mariscal, V.; Bornikoel, J.; Nieves-Morion, M.; Krauss, N.; Herrero, A.; Maldener, I.; Flores, E.; Mullineaux, C.W. Intercellular diffusion of a fluorescent sucrose analog via the septal junctions in a filamentous cyanobacterium. mBio 2015, 6, e02109. [Google Scholar] [CrossRef] [PubMed]
- Magnuson, A.; Cardona, T. Thylakoid membrane function in heterocysts. BBA-Bioenergetics 2016, 1857, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.Y.; Ni, J.; Xu, P.; Tao, F. Enhancing Light-Driven 1,3-Propanediol Production by Using Natural Compartmentalization of Differentiated Cells. ACS Synth. Biol. 2018, 7, 2436–2446. [Google Scholar] [CrossRef]
- Avilan, L.; Roumezi, B.; Risoul, V.; Bernard, C.S.; Kpebe, A.; Belhadjhassine, M.; Rousset, M.; Brugna, M.; Latifi, A. Phototrophic hydrogen production from a clostridial FeFe hydrogenase expressed in the heterocysts of the cyanobacterium Nostoc PCC 7120. Appl. Microbiol. Biotechnol. 2018, 102, 5775–5783. [Google Scholar] [CrossRef]
- Meeks, J.C.; Campbell, E.L.; Summers, M.L.; Wong, F.C. Cellular differentiation in the cyanobacterium Nostoc punctiforme. Arch. Microbiol. 2002, 178, 395–403. [Google Scholar] [CrossRef]
- Golden, J.W.; Yoon, H.S. Heterocyst development in Anabaena. Curr. Opin. Microbiol. 2003, 6, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Flores, E.; Herrero, A. Compartmentalized function through cell differentiation in filamentous cyanobacteria. Nat. Rev. Microbiol. 2010, 8, 39–50. [Google Scholar] [CrossRef]
- Herrero, A.; Picossi, S.; Flores, E. Gene expression during heterocyst differentiation. In Advances in Botanical Research, Genomics of Cyanobacteria; Chauvat, F., Cassier-Chauvat, C., Eds.; Academic Press: Cambridge, MA, USA, 2013; pp. 281–329. [Google Scholar]
- Santamaría-Gómez, J.; Mariscal, V.; Luque, I. Mechanisms for Protein Redistribution in Thylakoids of Anabaena during Cell Differentiation. Plant Cell Physiol. 2018, 59, 1860–1873. [Google Scholar] [CrossRef] [PubMed]
- Cardona, T.; Battchikova, N.; Zhang, P.P.; Stensjo, K.; Aro, E.M.; Lindblad, P.; Magnuson, A. Electron transfer protein complexes in the thylakoid membranes of heterocysts from the cyanobacterium Nostoc punctiforme. Biochim. Biophys. Acta 2009, 1787, 252–263. [Google Scholar] [CrossRef] [PubMed]
- Ow, S.Y.; Noirel, J.; Cardona, T.; Taton, A.; Lindblad, P.; Stensjo, K.; Wright, P.C. Quantitative overview of N2 fixation in Nostoc punctiforme ATCC 29133 through cellular enrichments and iTRAQ shotgun proteomics. J. Proteome Res. 2009, 8, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Valladares, A.; Maldener, I.; Muro-Pastor, A.M.; Flores, E.; Herrero, A. Heterocyst development and diazotrophic metabolism in terminal respiratory oxidase mutants of the cyanobacterium Anabaena sp strain PCC 7120. J. Bacteriol. 2007, 189, 4425–4430. [Google Scholar] [CrossRef]
- Kumazaki, S.; Akari, M.; Hasegawa, M. Transformation of Thylakoid Membranes during Differentiation from Vegetative Cell into Heterocyst Visualized by Microscopic Spectral Imaging. Plant Physiol. 2013, 161, 1321–1333. [Google Scholar] [CrossRef]
- Allahverdiyeva, Y.; Mustila, H.; Ermakova, M.; Bersanini, L.; Richaud, P.; Ajlani, G.; Battchikova, N.; Cournac, L.; Aro, E.M. Flavodiiron proteins Flv1 and Flv3 enable cyanobacterial growth and photosynthesis under fluctuating light. Proc. Natl. Acad. Sci. USA 2013, 110, 4111–4116. [Google Scholar] [CrossRef]
- Allahverdiyeva, Y.; Isojarvi, J.; Zhang, P.; Aro, E.M. Cyanobacterial oxygenic photosynthesis is protected by flavodiiron proteins. Life 2015, 5, 716–743. [Google Scholar] [CrossRef]
- Magnuson, A.; Cardona, T. Isolation of Intact Thylakoid Membranes from Heterocysts of Filamentous, Nitrogen-Fixing Cyanobacteria. Methods Mol. Biol. 2018, 1696, 137–145. [Google Scholar]
- Nozue, S.; Katayama, M.; Terazima, M.; Kumazaki, S. Comparative study of thylakoid membranes in terminal heterocysts and vegetative cells from two cyanobacteria, Rivularia M-261 and Anabaena variabilis, by fluorescence and absorption spectral microscopy. BBA-Bioenergetics 2017, 1858, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Plochinger, M.; Schwenkert, S.; von Sydow, L.; Schroder, W.P.; Meurer, J. Functional Update of the Auxiliary Proteins PsbW, PsbY, HCF136, PsbN, TerC and ALB3 in Maintenance and Assembly of PSII. Front. Plant Sci. 2016, 7, 13. [Google Scholar] [CrossRef] [PubMed]
- Burgess, B.K.; Lowe, D.J. Mechanism of molybdenum nitrogenase. Chem. Rev. 1996, 96, 2983–3011. [Google Scholar] [CrossRef]
- Almon, H.; Bohme, H. Photophosphorylation in isolated heterocysts from the blue-green-alga Nostoc muscorum. Biochim. Biophys. Acta 1982, 679, 279–286. [Google Scholar] [CrossRef]
- Janaki, S.; Wolk, C.P. Synthesis of nitrogenase by isolated heterocysts. Biochim. Biophys. Acta 1982, 696, 187–192. [Google Scholar] [CrossRef]
- Lindberg, P.; Schutz, K.; Happe, T.; Lindblad, P. A hydrogen-producing, hydrogenase-free mutant strain of Nostoc punctiforme ATCC 29133. Int. J. Hydrog. Energ. 2002, 27, 1291–1296. [Google Scholar] [CrossRef]
- Telor, E.; Stewart, W.D.P. Photosynthetic electron transport, ATP synthesis and nitrogenase activity in isolated heterocysts of Anabaena cylindrica. Biochim. Biophys. Acta 1976, 423, 189–195. [Google Scholar] [CrossRef]
- Ernst, A.; Bohme, H.; Boger, P. Phosphorylation and nitrogenase activity in isolated heterocysts from Anabaena variabilis (ATCC-29413). Biochim. Biophys. Acta 1983, 723, 83–90. [Google Scholar] [CrossRef]
- Ow, S.Y.; Cardona, T.; Taton, A.; Magnuson, A.; Lindblad, P.; Stensjo, K.; Wright, P.C. Quantitative shotgun proteomics of enriched heterocysts from Nostoc sp. PCC 7120 using 8-plex isobaric peptide tags. J. Proteome Res. 2008, 7, 1615–1628. [Google Scholar] [CrossRef]
- Munekage, Y.; Hashimoto, M.; Miyake, C.; Tomizawa, K.; Endo, T.; Tasaka, M.; Shikanai, T. Cyclic electron flow around Photosystem I is essential for photosynthesis. Nature 2004, 429, 579–582. [Google Scholar] [CrossRef]
- Johnson, G.N. Physiology of PSI cyclic electron transport in higher plants. BBA-Bioenergetics 2011, 1807, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Bottomley, P.J.; Stewart, W.D.P. ATP And Nitrogenase Activity In Nitrogen-Fixing Heterocystous Blue-Green-Algae. New Phytol. 1977, 79, 625–638. [Google Scholar] [CrossRef]
- Summers, M.L.; Wallis, J.G.; Campbell, E.L.; Meeks, J.C. Genetic evidence of a major role for glucose-6-phosphate-dehydrogenase in nitrogen-fixation and dark growth of the cyanobacterium Nostoc sp strain ATCC-29133. J. Bacteriol. 1995, 177, 6184–6194. [Google Scholar] [CrossRef] [PubMed]
- Ernst, A.; Bohme, H. Control of hydrogen-dependent nitrogenase activity by adenylates and electron flow in heterocysts of Anabaena variabilis (ATCC-29413). Biochim. Biophys. Acta 1984, 767, 362–368. [Google Scholar] [CrossRef]
- Kurisu, G.; Zhang, H.M.; Smith, J.L.; Cramer, W.A. Structure of the cytochrome b(6)f complex of oxygenic photosynthesis: Tuning the cavity. Science 2003, 302, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Peltier, G.; Aro, E.M.; Shikanai, T. NDH-1 and NDH-2 Plastoquinone Reductases in Oxygenic Photosynthesis. In Annual Review of Plant Biology, Volume 67; Merchant, S.S., Ed.; Annual Reviews: Palo Alto, CA, USA, 2016; pp. 55–80. [Google Scholar]
- Bohme, H.; Schrautemeier, B. Comparative characterization of ferredoxins from heterocysts and vegetative cells of Anabaena variabilis. Biochim. Biophys. Acta 1987, 891, 1–7. [Google Scholar] [CrossRef]
- Hurley, J.K.; Weber-Main, A.M.; Stankovich, M.T.; Benning, M.M.; Thoden, J.B.; Vanhooke, J.L.; Holden, H.M.; Chae, Y.K.; Xia, B.; Cheng, H.; et al. Structure-function relationships in Anabaena ferredoxin: Correlations between X-ray crystal structures, reduction potentials, and rate constants of electron transfer to ferredoxin:NADP+ reductase for site-specific ferredoxin mutants. Biochemistry 1997, 36, 11100–11117. [Google Scholar] [CrossRef]
- Schmitz, S.; Schrautemeier, B.; Bohme, H. Evidence from directed mutagenesis that positively charged amino-acids are necessary for interaction of nitrogenase with the 2Fe-2S heterocyst ferredoxin (FdxH) from the cyanobacterium Anabaena sp, PCC7120. Mol. Gen. Genet. 1993, 240, 455–460. [Google Scholar]
- Hurley, J.K.; Schmeits, J.L.; Genzor, C.; GomezMoreno, C.; Tollin, G. Charge reversal mutations in a conserved acidic patch in Anabaena ferredoxin can attenuate or enhance electron transfer to ferredoxin:NADP(+) reductase by altering protein/protein orientation within the intermediate complex. Arch. Biochem. Biophys. 1996, 333, 243–250. [Google Scholar] [CrossRef]
- Masepohl, B.; Scholisch, K.; Gorlitz, K.; Kutzki, C.; Bohme, H. The heterocyst-specific fdxH gene product of the cyanobacterium Anabaena sp. PCC 7120 is important but not essential for nitrogen fixation. Mol. Gen. Genet. 1997, 253, 770–776. [Google Scholar] [CrossRef]
- Schrautemeier, B.; Bohme, H.; Boger, P. In vitro studies on pathways and regulation of electron-transport to nitrogenase with a cell-free-extract from heterocysts of Anabaena variabilis. Arch. Microbiol. 1984, 137, 14–20. [Google Scholar] [CrossRef]
- Schrautemeier, B.; Bohme, H.; Boger, P. Reconstitution of a light-dependent nitrogen-fixing and transhydrogenase system with heterocyst thylakoids. Biochim. Biophys. Acta 1985, 807, 147–154. [Google Scholar] [CrossRef]
- Valladares, A.; Muro-Pastor, A.M.; Fillat, M.F.; Herrero, A.; Flores, E. Constitutive and nitrogen-regulated promoters of the petH gene encoding ferredoxin:NADP+ reductase in the heterocyst-forming cyanobacterium Anabaena sp. FEBS Lett. 1999, 449, 159–164. [Google Scholar] [CrossRef]
- Omairi-Nasser, A.; de Gracia, A.G.; Ajlani, G. A larger transcript is required for the synthesis of the smaller isoform of ferredoxin: NADP oxidoreductase. Mol. Microbiol. 2011, 81, 1178–1189. [Google Scholar] [CrossRef] [PubMed]
- Omairi-Nasser, A.; Galmozzi, C.V.; Latifi, A.; Muro-Pastor, M.I.; Ajlani, G. NtcA is responsible for accumulation of the small isoform of ferredoxin:NADP oxidoreductase. Microbiology 2014, 160, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Razquin, P.; Fillat, M.F.; Schmitz, S.; Stricker, O.; Bohme, H.; Gomez-Moreno, C.; Peleato, M.L. Expression of ferredoxin-NADP+ reductase in heterocysts from Anabaena sp. Biochem. J. 1996, 316, 157–160. [Google Scholar] [CrossRef] [PubMed]
- Alcantara-Sanchez, F.; Leyva-Castillo, L.E.; Chagolla-Lopez, A.; de la Vara, L.G.; Gomez-Lojero, C. Distribution of isoforms of ferredoxin-NADP(+) reductase (FNR) in cyanobacteria in two growth conditions. Int. J. Biochem. Cell Biol. 2017, 85, 123–134. [Google Scholar] [CrossRef]
- Meng, B.Y.; Matsubayashi, T.; Wakasugi, T.; Shinozaki, K.; Sugiura, M.; Hirai, A.; Mikami, T.; Kishima, Y.; Kinoshita, T. Ubiquity of the genes for components of a NADH dehydrogenase in higher-plant chloroplast genomes. Plant Sci. 1986, 47, 181–184. [Google Scholar] [CrossRef]
- Ogawa, T. A gene homologous to the subunit-2 gene of nadh dehydrogenase is essential to inorganic carbon transport of synechocystis PCC6803. Proc. Natl. Acad. Sci. USA 1991, 88, 4275–4279. [Google Scholar] [CrossRef]
- Arteni, A.A.; Zhang, P.P.; Battchikova, N.; Ogawa, T.; Aro, E.M.; Boekema, E.J. Structural characterization of NDH-1 complexes of Thermosynechococcus elongatus by single particle electron microscopy. BBA-Bioenergetics 2006, 1757, 1469–1475. [Google Scholar] [CrossRef]
- Baradaran, R.; Berrisford, J.M.; Minhas, G.S.; Sazanov, L.A. Crystal structure of the entire respiratory complex I. Nature 2013, 494, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, D.; Schubert, H.; Georg, J.; Hess, W.R.; Hagemann, M. The Gene sml0013 of Synechocystis Species Strain PCC 6803 Encodes for a Novel Subunit of the NAD(P)H Oxidoreductase or Complex I That Is Ubiquitously Distributed among Cyanobacteria. Plant Physiol. 2013, 163, 1191–1202. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.M.; Deng, Y.; Ogawa, T.; Mi, H.L. Active NDH-1 complexes from the cyanobacterium Synechocystis sp strain PCC 6803. Plant Cell Physiol 2006, 47, 1432–1436. [Google Scholar] [CrossRef] [PubMed]
- Mi, H.L.; Endo, T.; Ogawa, T.; Asada, K. Thylakoid membrane-bound, NADPH-specific pyridine-nucleotide dehydrogenase complex mediates cyclic electron transport in the cyanobacterium Synechocystis sp PCC-6803. Plant Cell Physiol. 1995, 36, 661–668. [Google Scholar]
- Peng, L.W.; Shikanai, T. Supercomplex Formation with Photosystem I Is Required for the Stabilization of the Chloroplast NADH Dehydrogenase-Like Complex in Arabidopsis. Plant Physiol. 2011, 155, 1629–1639. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.; Peng, L.W.; Fukao, Y.; Shikanai, T. An Src Homology 3 Domain-Like Fold Protein Forms a Ferredoxin Binding Site for the Chloroplast NADH Dehydrogenase-Like Complex in Arabidopsis. Plant Cell 2011, 23, 1480–1493. [Google Scholar] [CrossRef]
- Battchikova, N.; Wei, L.Z.; Du, L.Y.; Bersanini, L.; Aro, E.M.; Ma, W.M. Identification of Novel Ssl0352 Protein (NdhS), Essential for Efficient Operation of Cyclic Electron Transport around Photosystem I, in NADPH:plastoquinone Oxidoreductase (NDH-1) Complexes of Synechocystis sp. PCC 6803. J. Biol. Chem. 2011, 286, 36992–37001. [Google Scholar] [CrossRef] [PubMed]
- He, Z.H.; Zheng, F.F.; Wu, Y.Z.; Li, Q.H.; Lv, J.; Fu, P.C.; Mi, H.L. NDH-1L interacts with ferredoxin via the subunit NdhS in Thermosynechococcus elongatus. Photosynth. Res. 2015, 126, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Lanzilotta, W.N.; Ryle, M.J.; Seefeldt, L.C. Nucleotide hydrolysis and protein conformational-changes in azotobacter-vinelandii nitrogenase iron protein—Defining the function of aspartate-129. Biochemistry 1995, 34, 10713–10723. [Google Scholar] [CrossRef]
- Jasniewski, A.J.; Sickerman, N.S.; Hu, Y.L.; Ribbe, M.W. The Fe Protein: An Unsung Hero of Nitrogenase. Inorganics 2018, 6, 14. [Google Scholar] [CrossRef]
- Muller, V.; Hess, V. The Minimum Biological Energy Quantum. Front. Microbiol. 2017, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Pogoryelov, D.; Klyszejko, A.L.; Krasnoselska, G.O.; Heller, E.M.; Leone, V.; Langerd, J.D.; Vonck, J.; Muller, D.J.; Faraldo-Gomez, J.D.; Meier, T. Engineering rotor ring stoichiometries in the ATP synthase. Proc. Natl. Acad. Sci. USA 2012, 109, E1599–E1608. [Google Scholar] [CrossRef] [PubMed]
- Srirangan, K.; Pyne, M.E.; Chou, C.P. Biochemical and genetic engineering strategies to enhance hydrogen production in photosynthetic algae and cyanobacteria. Bioresour. Technol. 2011, 102, 8589–8604. [Google Scholar] [CrossRef] [PubMed]
- Wegelius, A.; Li, X.; Turco, F.; Stensjo, K. Design and characterization of a synthetic minimal promoter for heterocyst-specific expression in filamentous cyanobacteria. PLoS ONE 2018, 13, e0203898. [Google Scholar] [CrossRef] [PubMed]
- Angermayr, S.A.; Rovira, A.G.; Hellingwerf, K.J. Metabolic engineering of cyanobacteria for the synthesis of commodity products. Trends Biotechnol. 2015, 33, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Dexter, J.; Armshaw, P.; Sheahan, C.; Pembroke, J.T. The state of autotrophic ethanol production in Cyanobacteria. J. Appl. Microbiol. 2015, 119, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Wijffels, R.H.; Kruse, O.; Hellingwerf, K.J. Potential of industrial biotechnology with cyanobacteria and eukaryotic microalgae. Curr. Opin. Biotechnol. 2013, 24, 405–413. [Google Scholar] [CrossRef]
- Anderson, S.L.; McIntosh, L. Light-activated heterotrophic growth of the cyanobacterium synechocystis sp strain PCC-6803—A blue-light-requiring PROCESS. J. Bacteriol. 1991, 173, 2761–2767. [Google Scholar] [CrossRef]
- Rippka, R.; Deruelles, J.; Waterbury, J.B.; Herdman, M.; Stanier, R.Y. Generic assignments, strain histories and properties of pure cultures of Cyanobacteria. J. Gen. Microbiol. 1979, 111, 1–61. [Google Scholar] [CrossRef]
- Gao, Z.X.; Zhao, H.; Li, Z.M.; Tan, X.M.; Lu, X.F. Photosynthetic production of ethanol from carbon dioxide in genetically engineered cyanobacteria. Energ. Environ. Sci. 2012, 5, 9857–9865. [Google Scholar] [CrossRef]
- Vidal, R. Alcohol dehydrogenase AdhA plays a role in ethanol tolerance in model cyanobacterium Synechocystis sp PCC 6803. Appl. Microbiol. Biotechnol. 2017, 101, 3473–3482. [Google Scholar] [CrossRef]
- Ehira, S.; Takeuchi, T.; Higo, A. Spatial separation of photosynthesis and ethanol production by cell type-specific metabolic engineering of filamentous cyanobacteria. Appl. Microbiol. Biotechnol. 2018, 102, 1523–1531. [Google Scholar] [CrossRef] [PubMed]
- Masukawa, H.; Mochimaru, M.; Sakurai, H. Disruption of the uptake hydrogenase gene, but not of the bidirectional hydrogenase gene, leads to enhanced photobiological hydrogen production by the nitrogen-fixing cyanobacterium Anabaena sp PCC 7120. Appl. Microbiol. Biotechnol. 2002, 58, 618–624. [Google Scholar] [CrossRef]
- Gartner, K.; Lechno-Yossef, S.; Cornish, A.J.; Wolk, C.P.; Hegg, E.L. Expression of Shewanella oneidensis MR-1 FeFe -Hydrogenase Genes in Anabaena sp Strain PCC 7120. Appl. Environ. Microbiol. 2012, 78, 8579–8586. [Google Scholar] [CrossRef] [PubMed]
- Khamees, H.S.; Gallon, J.R.; Chaplin, A.E. The pattern of acetylene-reduction by cyanobacteria grown under alternating light and darkness. Br. Phycol. J. 1987, 22, 55–60. [Google Scholar] [CrossRef]
- Raleiras, P.; Khanna, N.; Miranda, H.; Meszaros, L.S.; Krassen, H.; Ho, F.; Battchikova, N.; Aro, E.M.; Magnuson, A.; Lindblad, P.; et al. Turning around the electron flow in an uptake hydrogenase. EPR spectroscopy and in vivo activity of a designed mutant in HupSL from Nostoc punctiforme. Energ. Environ. Sci. 2016, 9, 581–594. [Google Scholar] [CrossRef]
- Liang, F.Y.; Englund, E.; Lindberg, P.; Lindblad, P. Engineered cyanobacteria with enhanced growth show increased ethanol production and higher biofuel to biomass ratio. Metab. Eng. 2018, 46, 51–59. [Google Scholar] [CrossRef]
- Watt, I.N.; Montgomery, M.G.; Runswick, M.J.; Leslie, A.G.W.; Walker, J.E. Bioenergetic cost of making an adenosine triphosphate molecule in animal mitochondria. Proc. Natl. Acad. Sci. USA 2010, 107, 16823–16827. [Google Scholar] [CrossRef]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magnuson, A. Heterocyst Thylakoid Bioenergetics. Life 2019, 9, 13. https://doi.org/10.3390/life9010013
Magnuson A. Heterocyst Thylakoid Bioenergetics. Life. 2019; 9(1):13. https://doi.org/10.3390/life9010013
Chicago/Turabian StyleMagnuson, Ann. 2019. "Heterocyst Thylakoid Bioenergetics" Life 9, no. 1: 13. https://doi.org/10.3390/life9010013
APA StyleMagnuson, A. (2019). Heterocyst Thylakoid Bioenergetics. Life, 9(1), 13. https://doi.org/10.3390/life9010013



