A Spectral Comparison of Jarosites Using Techniques Relevant to the Robotic Exploration of Biosignatures on Mars
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.1.1. Biological Sample
2.1.2. Synthetic Sample
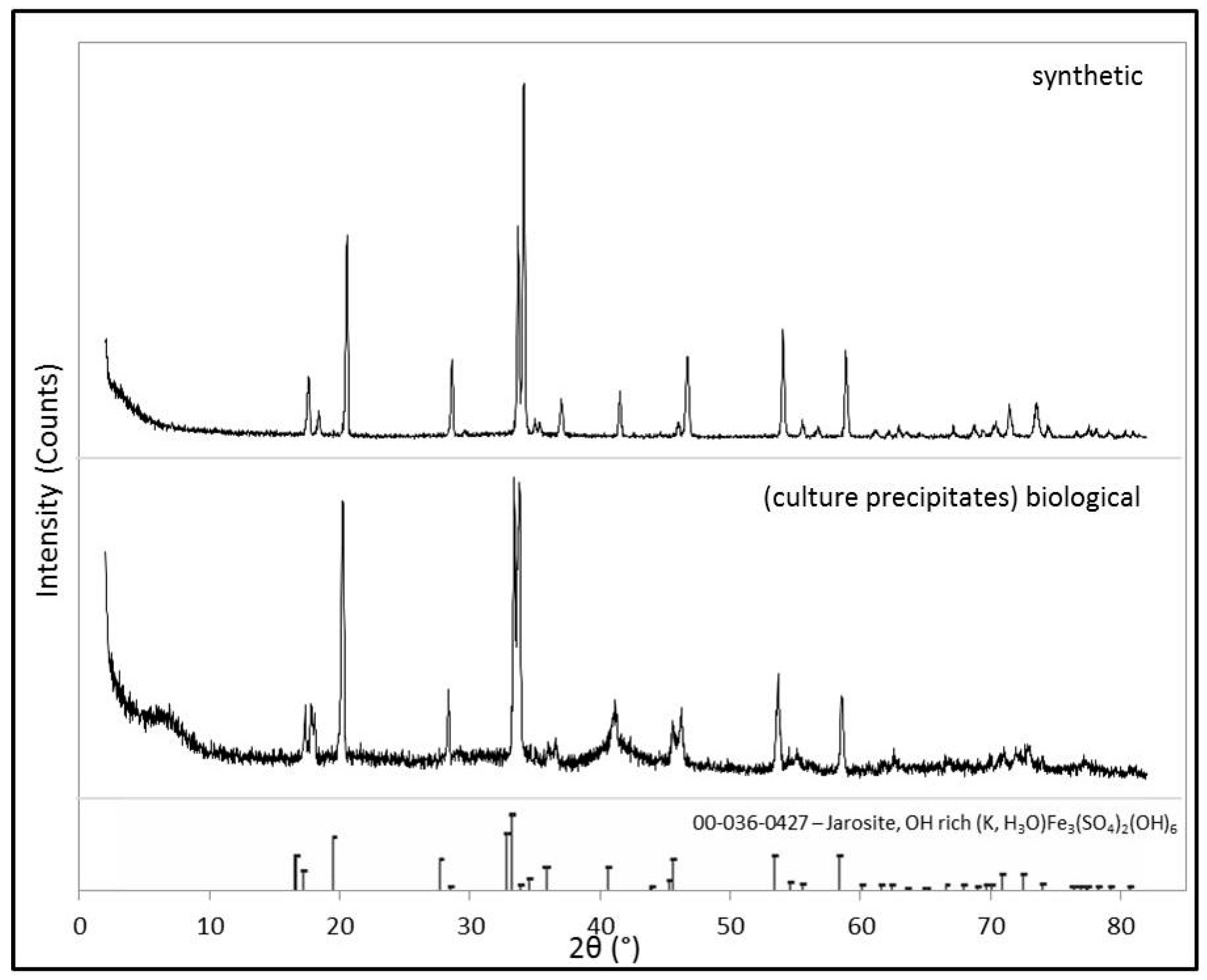
2.1.3. X-Ray Diffraction (XRD)
2.1.4. Scanning Electron Microscopy (SEM)
2.2. Spectroscopy of Jarosite Samples
2.2.1. Raman Spectroscopy
2.2.2. Mid-Infrared Spectroscopy (IR)
2.2.3. Visible Near-Infrared Reflectance Spectroscopy (VNIR)
2.2.4. Mössbauer Spectroscopy
3. Results
3.1. Samples
3.2. Spectroscopy
3.2.1. Raman Spectroscopy
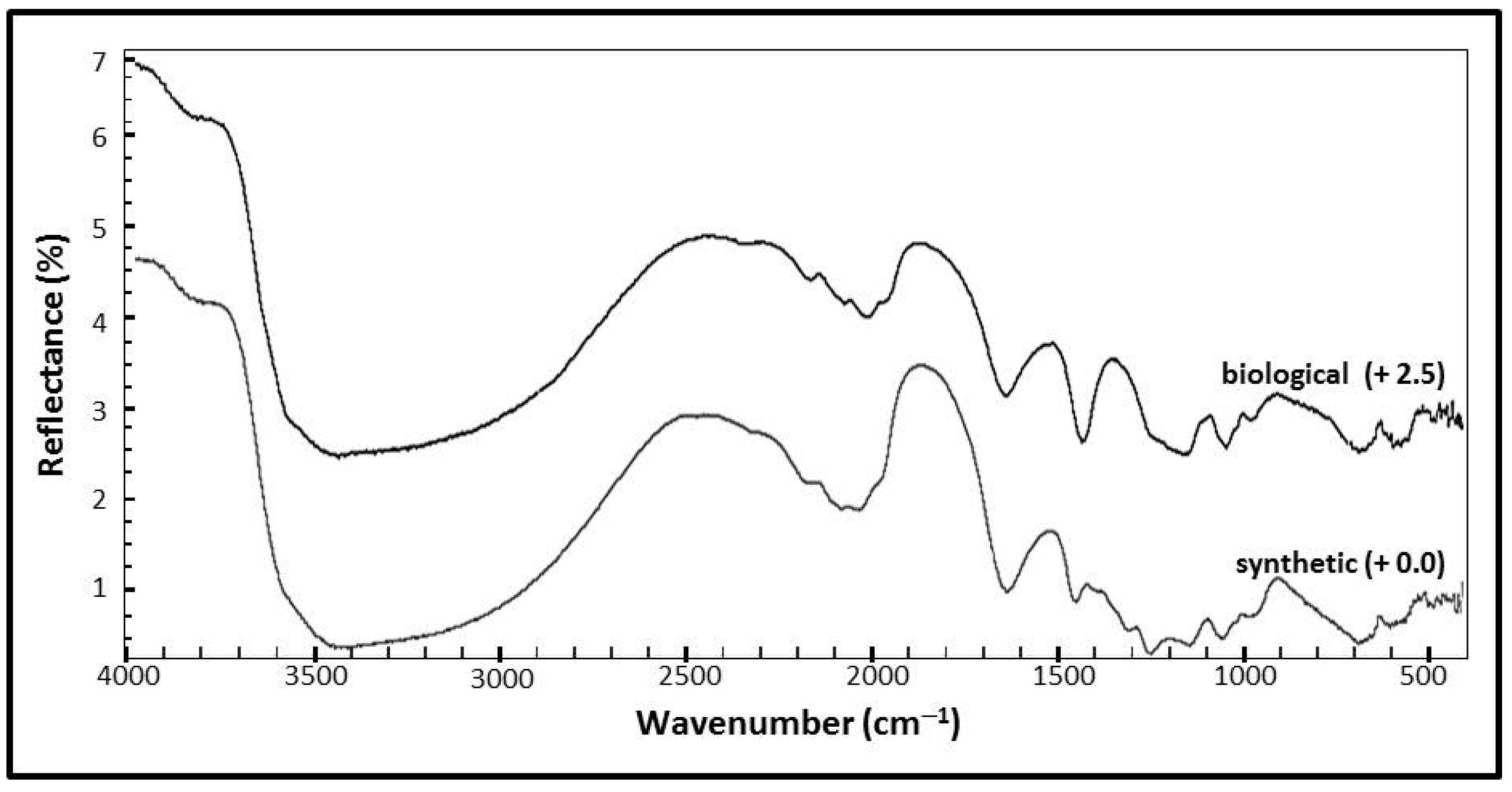
3.2.2. Mid-Infrared Spectroscopy (IR)
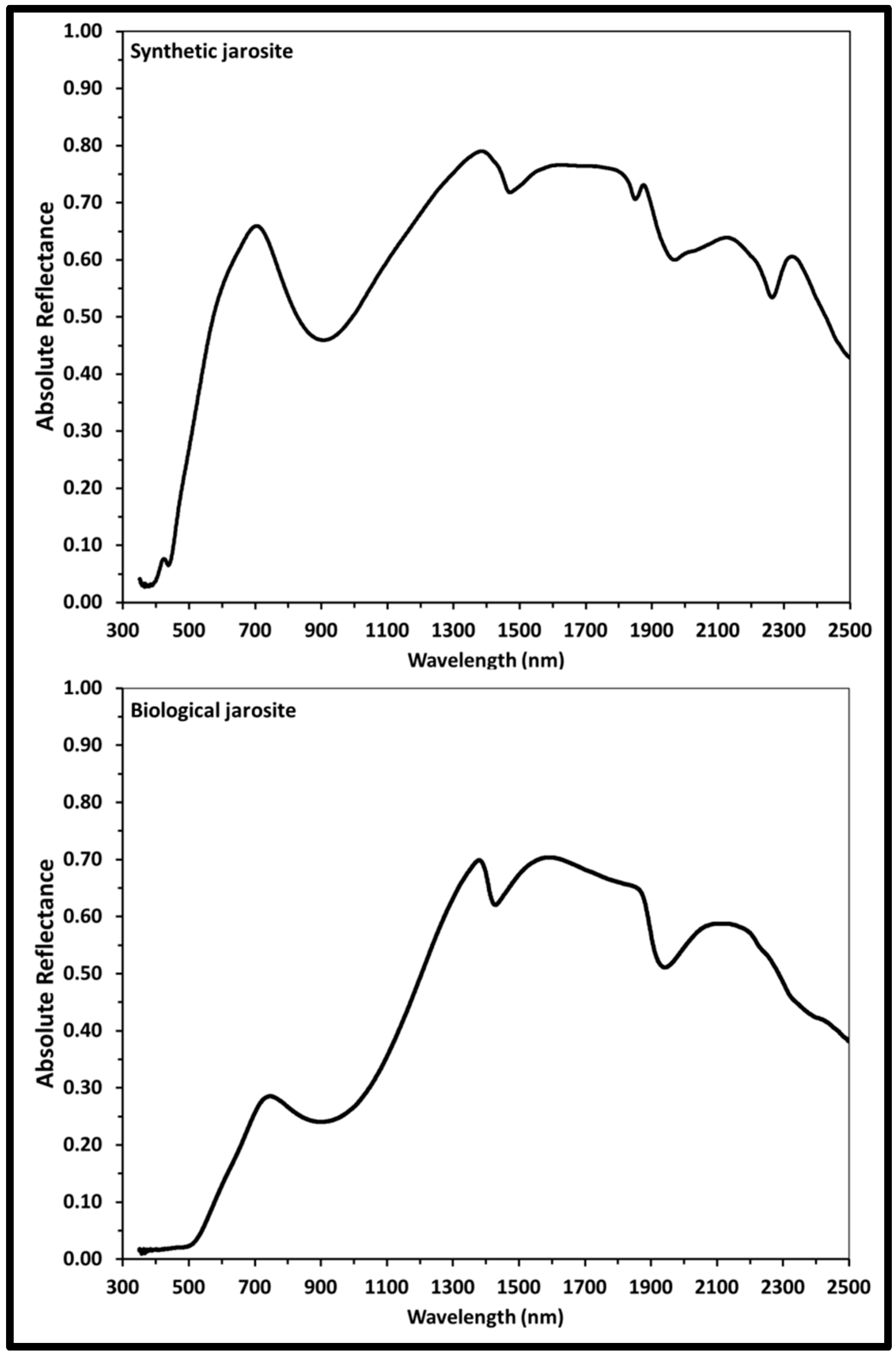
3.2.3. Visible Near-Infrared Reflectance Spectroscopy (VNIR)
3.2.4. Mössbauer Spectroscopy
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vago, J.L.; Westall, F.; Pasteur Instrument Teams, Landing Site Selection Working Group; Coates, A.J.; Jaumann, R.; Korablev, O.; Ciarletti, V.; Mitrofanov, I.; Josset, J.-L.; De Sanctis, M.C.; et al. Habitability on Early Mars and the Search for Biosignatures with the ExoMars Rover. Astrobiology 2017, 17, 471–510. [Google Scholar] [CrossRef]
- McSween, H.Y., Jr.; McLennan, S.M. 2.10—Mars. In Treatise on Geochemistry, 2nd ed.; Holland, H.D., Turekian, K.K., Eds.; Elsevier: Oxford, UK, 2014; pp. 251–300. ISBN 978-0-08-098300-4. [Google Scholar]
- Squyres, S.W.; Grotzinger, J.P.; Arvidson, R.E.; Bell, J.F.; Calvin, W.; Christensen, P.R.; Clark, B.C.; Crisp, J.A.; Farrand, W.H.; Herkenhoff, K.E.; et al. In Situ evidence for an ancient aqueous environment at Meridiani Planum, Mars. Science 2004, 306, 1709–1714. [Google Scholar] [CrossRef]
- King, P.L.; McLennan, S.M. Sulfur on Mars. Elements 2010, 6, 107–112. [Google Scholar] [CrossRef]
- Elwood Madden, M.E.; Bodnar, R.J.; Rimstidt, J.D. Jarosite as an indicator of water-limited chemical weathering on Mars. Nature 2004, 431, 821–823. [Google Scholar] [CrossRef]
- Klingelhöfer, G.; Morris, R.V.; Bernhardt, B.; Schröder, C.; Rodionov, D.S.; de Souza, P.A.; Yen, A.; Gellert, R.; Evlanov, E.N.; Zubkov, B.; et al. Jarosite and hematite at Meridiani Planum from Opportunity’s Mössbauer spectrometer. Science 2004, 306, 1740–1745. [Google Scholar] [CrossRef]
- Burns, R.G. Ferric sulfates on Mars. J. Geophys. Res. 1987, 92, E570–E574. [Google Scholar] [CrossRef]
- Rampe, E.B.; Ming, D.W.; Blake, D.F.; Bristow, T.F.; Chipera, S.J.; Grotzinger, J.P.; Morris, R.V.; Morrison, S.M.; Vaniman, D.T.; Yen, A.S.; et al. Mineralogy of an ancient lacustrine mudstone succession from the Murray formation, Gale crater, Mars. Earth Planet. Sci. Lett. 2017, 471, 172–185. [Google Scholar] [CrossRef]
- Grotzinger, J.P.; Crisp, J.; Vasavada, A.R.; Anderson, R.C.; Baker, C.J.; Barry, R.; Blake, D.F.; Conrad, P.; Edgett, K.S.; Ferdowski, B.; et al. Mars Science Laboratory mission and science investigation. Space Sci. Rev. 2012, 170, 5–56. [Google Scholar] [CrossRef]
- Farrand, W.H.; Glotch, T.D.; Rice, J.W.; Hurowitz, J.A.; Swayze, G.A. Discovery of jarosite within the Mawrth Vallis region of Mars: Implications for the geologic history of the region. Icarus 2009, 204, 478–488. [Google Scholar] [CrossRef]
- Knoll, A.H.; Carr, M.; Clark, B.; Des Marais, D.J.; Farmer, J.D.; Fischer, W.W.; Grotzinger, J.P.; McLennan, S.M.; Malin, M.; Schröder, C.; et al. An astrobiological perspective on Meridiani Planum. Earth Planet. Sci. Lett. 2005, 240, 179–189. [Google Scholar] [CrossRef]
- Blowes, D.W.; Ptacek, C.J.; Jambor, J.L.; Weisener, C.G. 9.05—The geochemistry of acid mine drainage. In Treatise on Geochemistry; Turekian, H.D.H.K., Ed.; Pergamon: Oxford, UK, 2003; pp. 149–204. ISBN 978-0-08-043751-4. [Google Scholar]
- Amils, R. Lessons learned from thirty years of geomicrobiological studies of Río Tinto. Res. Microbiol. 2016, 167, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Remolar, D.C.; Knoll, A.H. Fossilization potential of iron-bearing minerals in acidic environments of Rio Tinto, Spain: Implications for Mars exploration. Icarus 2008, 194, 72–85. [Google Scholar] [CrossRef]
- Fernández-Remolar, D.C.; Morris, R.V.; Gruener, J.E.; Amils, R.; Knoll, A.H. The Río Tinto Basin, Spain: Mineralogy, sedimentary geobiology, and implications for interpretation of outcrop rocks at Meridiani Planum, Mars. Earth Planet. Sci. Lett. 2005, 240, 149–167. [Google Scholar] [CrossRef]
- Fernández-Remolar, D.C.; Preston, L.J.; Sánchez-Román, M.; Izawa, M.R.M.; Huang, L.; Southam, G.; Banerjee, N.R.; Osinski, G.R.; Flemming, R.; Gómez-Ortíz, D.; et al. Carbonate precipitation under bulk acidic conditions as a potential biosignature for searching life on Mars. Earth Planet. Sci. Lett. 2012, 351–352, 13–26. [Google Scholar] [CrossRef]
- Preston, L.J.; Shuster, J.; Fernández-Remolar, D.; Banerjee, N.R.; Osinski, G.R.; Southam, G. The preservation and degradation of filamentous bacteria and biomolecules within iron oxide deposits at Rio Tinto, Spain: Preservation of biomolecules at Rio Tinto, Spain. Geobiology 2011, 9, 233–249. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Remolar, D.; Gómez-Elvira, J.; Gómez, F.; Sebastian, E.; Martíin, J.; Manfredi, J.A.; Torres, J.; González Kesler, C.; Amils, R. The Tinto River, an extreme acidic environment under control of iron, as an analog of the Terra Meridiani hematite site of Mars. Planet. Space Sci. 2004, 52, 239–248. [Google Scholar] [CrossRef]
- Preston, L.J.; Dartnell, L.R. Planetary habitability: Lessons learned from terrestrial analogues. Int. J. Astrobiol. 2014, 13, 81–98. [Google Scholar] [CrossRef]
- Amils, R.; Fernández-Remolar, D.; The IPBSL Team. Río Tinto: A geochemical and mineralogical terrestrial analogue of Mars. Life 2014, 4, 511–534. [Google Scholar] [CrossRef]
- Amils, R.; González-Toril, E.; Fernández-Remolar, D.; Gómez, F.; Aguilera, Á.; Rodríguez, N.; Malki, M.; García-Moyano, A.; Fairén, A.G.; de la Fuente, V.; et al. Extreme environments as Mars terrestrial analogs: The Rio Tinto case. Planet. Space Sci. 2007, 55, 370–381. [Google Scholar] [CrossRef]
- Marlow, J.J.; Martins, Z.; Sephton, M.A. Organic host analogues and the search for life on Mars. Int. J. Astrobiol. 2011, 10, 31–44. [Google Scholar] [CrossRef]
- Sobron, P.; Bishop, J.L.; Blake, D.F.; Chen, B.; Rull, F. Natural Fe-bearing oxides and sulfates from the Rio Tinto Mars analog site: Critical assessment of VNIR reflectance spectroscopy, laser Raman spectroscopy, and XRD as mineral identification tools. Am. Miner. 2014, 99, 1199–1205. [Google Scholar] [CrossRef]
- Navrotsky, A.; Forray, F.; Drouet, C. Jarosite stability on Mars. Icarus 2005, 176, 250–253. [Google Scholar] [CrossRef]
- Cloutis, E.A.; Craig, M.A.; Kruzelecky, R.V.; Jamroz, W.R.; Scott, A.; Hawthorne, F.C.; Mertzman, S.A. Spectral reflectance properties of minerals exposed to simulated Mars surface conditions. Icarus 2008, 195, 140–168. [Google Scholar] [CrossRef]
- Lewis, J.M.T.; Watson, J.S.; Najorka, J.; Luong, D.; Sephton, M.A. Sulfate minerals: A problem for the detection of organic compounds on Mars? Astrobiology 2015, 15, 247–258. [Google Scholar] [CrossRef] [PubMed]
- Mahaffy, P.R.; Webster, C.R.; Cabane, M.; Conrad, P.G.; Coll, P.; Atreya, S.K.; Arvey, R.; Barciniak, M.; Benna, M.; Bleacher, L.; et al. The Sample Analysis at Mars investigation and instrument suite. Space Sci. Rev. 2012, 170, 401–478. [Google Scholar] [CrossRef]
- Goesmann, F.; Brinckerhoff, W.B.; Raulin, F.; Goetz, W.; Danell, R.M.; Getty, S.A.; Siljeström, S.; Mißbach, H.; Steininger, H.; Arevalo, R.D.; et al. The Mars Organic Molecule Analyzer (MOMA) Instrument: Characterization of Organic Material in Martian Sediments. Astrobiology 2017, 17, 655–685. [Google Scholar] [CrossRef]
- Bauermeister, A.; Rettberg, P.; Flemming, H.-C. Growth of the acidophilic iron–sulfur bacterium acidithiobacillus ferrooxidans under Mars-like geochemical conditions. Planet. Space Sci. 2014, 98, 205–215. [Google Scholar] [CrossRef]
- Gómez, F.; Mateo-Martí, E.; Prieto-Ballesteros, O.; Martín-Gago, J.; Amils, R. Protection of chemolithoautotrophic bacteria exposed to simulated Mars environmental conditions. Icarus 2010, 209, 482–487. [Google Scholar] [CrossRef]
- Gordon, P.R.; Sephton, M.A. A method for choosing the best samples for Mars sample return. Astrobiology 2018. [Google Scholar] [CrossRef]
- Edwards, H.G.M.; Vandenabeele, P.; Jorge-Villar, S.E.; Carter, E.A.; Perez, F.R.; Hargreaves, M.D. The Rio Tinto Mars analogue site: An extremophilic Raman spectroscopic study. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2007, 68, 1133–1137. [Google Scholar] [CrossRef] [PubMed]
- Léveillé, R. Validation of astrobiology technologies and instrument operations in terrestrial analogue environments. C. R. Palevol. 2009, 8, 637–648. [Google Scholar] [CrossRef]
- Marshall, C.P.; Olcott Marshall, A. Raman hyperspectral imaging of microfossils: Potential pitfalls. Astrobiology 2013, 13, 920–931. [Google Scholar] [CrossRef] [PubMed]
- Rull, F.; Maurice, S.; Hutchinson, I.; Moral, A.; Perez, C.; Diaz, C.; Colombo, M.; Belenguer, T.; Lopez-Reyes, G.; Sansano, A.; et al. The Raman Laser Spectrometer for the ExoMars Rover Mission to Mars. Astrobiology 2017, 17, 627–654. [Google Scholar] [CrossRef]
- Korablev, O.I.; Dobrolensky, Y.; Evdokimova, N.; Fedorova, A.A.; Kuzmin, R.O.; Mantsevich, S.N.; Cloutis, E.A.; Carter, J.; Poulet, F.; Flahaut, J.; et al. Infrared Spectrometer for ExoMars: A Mast-Mounted Instrument for the Rover. Astrobiology 2017, 17, 542–564. [Google Scholar] [CrossRef] [PubMed]
- De Sanctis, M.C.; Altieri, F.; Ammannito, E.; Biondi, D.; De Angelis, S.; Meini, M.; Mondello, G.; Novi, S.; Paolinetti, R.; Soldani, M.; et al. Ma_MISS on ExoMars: Mineralogical Characterization of the Martian Subsurface. Astrobiology 2017, 17, 612–620. [Google Scholar] [CrossRef]
- Shuster, J.; Bolin, T.; MacLean, L.C.W.; Southam, G. The effect of iron-oxidising bacteria on the stability of gold (I) thiosulphate complex. Chem. Geol. 2014, 376, 52–60. [Google Scholar] [CrossRef]
- Shuster, J.; Reith, F.; Izawa, M.R.M.; Flemming, R.L.; Banerjee, N.R.; Southam, G. Biogeochemical cycling of silver in acidic, weathering environments. Minerals 2017, 7, 218. [Google Scholar] [CrossRef]
- Sandy Jones, F.; Bigham, J.M.; Gramp, J.P.; Tuovinen, O.H. Synthesis and properties of ternary (K, NH4, H3O)-jarosites precipitated from Acidithiobacillus ferrooxidans cultures in simulated bioleaching solutions. Mater. Sci. Eng. C 2014, 44, 391–399. [Google Scholar] [CrossRef]
- Sasaki, K.; Sakimoto, T.; Endo, M.; Konno, H. FE-SEM study of microbially formed jarosites by Acidithiobacillus ferrooxidans. Mater. Trans. 2006, 47, 1155–1162. [Google Scholar] [CrossRef]
- Sasaki, K.; Konno, H. Morphology of jarosite-group compounds precipitated from biologically and chemically oxidized Fe ions. Can. Miner. 2000, 38, 45–56. [Google Scholar] [CrossRef]
- Enders, M.S.; Knickerbocker, C.; Titley, S.R.; Southam, G. The role of bacteria in the supergene environment of the Morenci porphyry copper deposit, Greenlee County, Arizona. Econ. Geol. 2006, 101, 59–70. [Google Scholar] [CrossRef]
- Lazaroff, N.; Sigal, W.; Wasserman, A. Iron oxidation and precipitation of ferric hydroxysulfates by resting Thiobacillus ferrooxidans cells. Appl. Environ. Microbiol. 1982, 43, 924–938. [Google Scholar] [PubMed]
- Tuovinen, O.H.; Carlson, L. Jarosite in cultures of iron-oxidizing thiobacilli. Geomicrobiol. J. 1979, 1, 205–210. [Google Scholar] [CrossRef]
- Southam, G.; Beveridge, T.J. Examination of lipopolysaccharide (O-Antigen) populations of Thiobacillus ferrooxidans from two mine tailings. Appl. Environ. Microbiol. 1993, 59, 1283–1288. [Google Scholar] [PubMed]
- Dutrizac, J.E.; Kaiman, S. Synthesis and properties of jarosite-type compounds. Can. Miner. 1976, 14, 151–158. [Google Scholar]
- Bruker AXS. DIFFRACplus Evaluation Package Release 2005; Bruker AXS: Karlsruhe, Germany, 2005. [Google Scholar]
- Dyar, M.D.; Schaefer, M.W.; Sklute, E.C.; Bishop, J.L. Mössbauer spectroscopy of phyllosilicates: Effects of fitting models on recoil-free fractions and redox ratios. Clay Miner. 2008, 43, 3–33. [Google Scholar] [CrossRef]
- Basciano, L.C.; Peterson, R.C. Jarosite hydronium jarosite solid-solution series with full iron site occupancy: Mineralogy and crystal chemistry. Am. Miner. 2007, 92, 1464–1473. [Google Scholar] [CrossRef]
- Frost, R.L.; Wills, R.-A.; Weier, M.L.; Martens, W. Comparison of the Raman spectra of natural and synthetic K- and Na-jarosites at 298 and 77 K. J. Raman Spectrosc. 2005, 36, 435–444. [Google Scholar] [CrossRef]
- Drouet, C.; Navrotsky, A. Synthesis, characterization, and thermochemistry of K-Na-H3O jarosites. Geochim. Cosmochim. Acta 2003, 67, 2063–2076. [Google Scholar] [CrossRef]
- Liu, J.; Li, B.; Zhong, D.; Xia, L.; Qiu, G. Preparation of jarosite by Acidithiobacillus ferrooxidans oxidation. J. Cent. South Univ. Technol. 2007, 14, 623–628. [Google Scholar] [CrossRef]
- Tourney, J.; Ngwenya, B.T. The role of bacterial extracellular polymeric substances in geomicrobiology. Chem. Geol. 2014, 386, 115–132. [Google Scholar] [CrossRef]
- Chio, C.H.; Sharma, S.K.; Ming, L.-C.; Muenow, D.W. Raman spectroscopic investigation on jarosite–yavapaiite stability. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 75, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Frost, R.L.; Weier, M.; Martinez-Frias, J.; Rull, F.; Jagannadha Reddy, B. Sulphate efflorescent minerals from El Jaroso Ravine, Sierra Almagrera—An SEM and Raman spectroscopic study. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2007, 66, 177–183. [Google Scholar] [CrossRef]
- Chio, C.H.; Sharma, S.K.; Muenow, D.W. Micro-Raman studies of hydrous ferrous sulfates and jarosites. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2005, 61, 2428–2433. [Google Scholar] [CrossRef]
- Sasaki, K.; Tanaike, O.; Konno, H. Distinction of jarosite-group compounds by Raman spectroscopy. Can. Miner. 1998, 36, 1225–1235. [Google Scholar]
- Serna, C.J.; Cortina, C.P.; Garcia Ramos, J.V. Infrared and Raman study of alunite—Jarosite compounds. Spectrochim. Acta Part A Mol. Spectrosc. 1986, 42, 729–734. [Google Scholar] [CrossRef]
- Lane, M.D.; Bishop, J.L.; Dyar, M.D.; Hiroi, T.; Mertzman, S.A.; Bish, D.L.; King, P.L.; Rogers, A.D. Mid-infrared emission spectroscopy and visible/near-infrared reflectance spectroscopy of Fe-sulfate minerals. Am. Miner. 2015, 100, 66–82. [Google Scholar] [CrossRef]
- Majzlan, J.; Alpers, C.N.; Koch, C.B.; McCleskey, R.B.; Myneni, S.C.B.; Neil, J.M. Vibrational, X-ray absorption, and Mössbauer spectra of sulfate minerals from the weathered massive sulfide deposit at Iron Mountain, California. Chem. Geol. 2011, 284, 296–305. [Google Scholar] [CrossRef]
- Cloutis, E.; Hawthorne, F.; Mertzman, S.; Krenn, K.; Craig, M.; Marcino, D.; Methot, M.; Strong, J.; Mustard, J.; Blaney, D. Detection and discrimination of sulfate minerals using reflectance spectroscopy. Icarus 2006, 184, 121–157. [Google Scholar] [CrossRef]
- Lane, M.D. Mid-infrared emission spectroscopy of sulfate and sulfate-bearing minerals. Am. Miner. 2007, 92, 1–18. [Google Scholar] [CrossRef]
- Bishop, J.L.; Murad, E. The visible and infrared spectral properties of jarosite and alunite. Am. Miner. 2005, 90, 1100–1107. [Google Scholar] [CrossRef]
- Powers, D.A.; Rossman, G.R.; Schugar, H.J.; Gray, H.B. Magnetic behavior and infrared spectra of jarosite, basic iron sulfate, and their chromate analogs. J. Solid State Chem. 1975, 13, 1–13. [Google Scholar] [CrossRef]
- Crowley, J.K.; Williams, D.E.; Hammarstrom, J.M.; Piatak, N.; Chou, I.-M.; Mars, J.C. Spectral reflectance properties (0.4–2.5 μm) of secondary Fe-oxide, Fe-hydroxide, and Fe-sulphate-hydrate minerals associated with sulphide-bearing mine wastes. Geochem. Explor. Environ. Anal. 2003, 3, 219–228. [Google Scholar] [CrossRef]
- Hunt, G.R.; Ashley, R.P. Spectra of altered rocks in the visible and near infrared. Econ. Geol. 1979, 74, 1613–1629. [Google Scholar] [CrossRef]
- Frost, R.L.; Wills, R.-A.; Weier, M.L.; Martens, W.; Mills, S. A Raman spectroscopic study of selected natural jarosites. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2006, 63, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Bowen, B.B.; Seo, J.-H.; Michalski, G. Laboratory and field characterization of visible to near-infrared spectral reflectance of nitrate minerals from the Atacama Desert, Chile, and implications for Mars. Am. Miner. 2018, 103, 197–206. [Google Scholar] [CrossRef]
- Bishop, J.; Murad, E. Schwertmannite on Mars? Spectroscopic analyses of schwertmannite, its relationship to other ferric minerals, and its possible presence in the surface material on Mars. In Mineral Spectroscopy: A Tribute to Roger G. Burns; Geochemical Society: New York, NY, USA, 1996; pp. 337–358. [Google Scholar]
- Hyde, B.C.; King, P.L.; Dyar, M.D.; Spilde, M.N.; Ali, A.-M.S. Methods to analyze metastable and microparticulate hydrated and hydrous iron sulfate minerals. Am. Miner. 2011, 96, 1856–1869. [Google Scholar] [CrossRef]
- Dyar, M.D.; Agresti, D.G.; Schaefer, M.W.; Grant, C.A.; Sklute, E.C. Mössbauer spectroscopy of Earth and planetary materials. Annu. Rev. Earth Planet. Sci. 2006, 34, 83–125. [Google Scholar] [CrossRef]
- Bigham, J.M.; Carlson, L.; Murad, E. Schwertmannite, a new iron oxyhydroxysulphate from Pyhasalmi, Finland, and other localities. Miner. Mag. 1994, 58, 641–648. [Google Scholar] [CrossRef]
- Bigham, J.M.; Schwertmann, U.; Carlson, L.; Murad, E. A poorly crystallized oxyhydroxysulfate of iron formed by bacterial oxidation of Fe(II) in acid mine waters. Geochim. Cosmochim. Acta 1990, 54, 2743–2758. [Google Scholar] [CrossRef]
- Dyar, M.D.; Breves, E.; Jawin, E.; Marchand, G.; Nelms, M.; O’Connor, V.; Peel, S.; Rothstein, Y.; Sklute, E.C.; Lane, M.D.; et al. Mössbauer parameters of iron in sulfate minerals. Am. Miner. 2013, 98, 1943–1965. [Google Scholar] [CrossRef]
- Schwertmann, U.; Cornell, R.M. Iron Oxides in the Laboratory: Preparation and Characterization, 2nd ed.; Wiley-VCH: Weinheim, Germany; New York, NY, USA, 2000; ISBN 978-3-527-29669-9. [Google Scholar]
- Dyar, M.D.; Jawin, E.R.; Breves, E.; Marchand, G.; Nelms, M.; Lane, M.D.; Mertzman, S.A.; Bish, D.L.; Bishop, J.L. Mossbauer parameters of iron in phosphate minerals: Implications for interpretation of martian data. Am. Miner. 2014, 99, 914–942. [Google Scholar] [CrossRef]


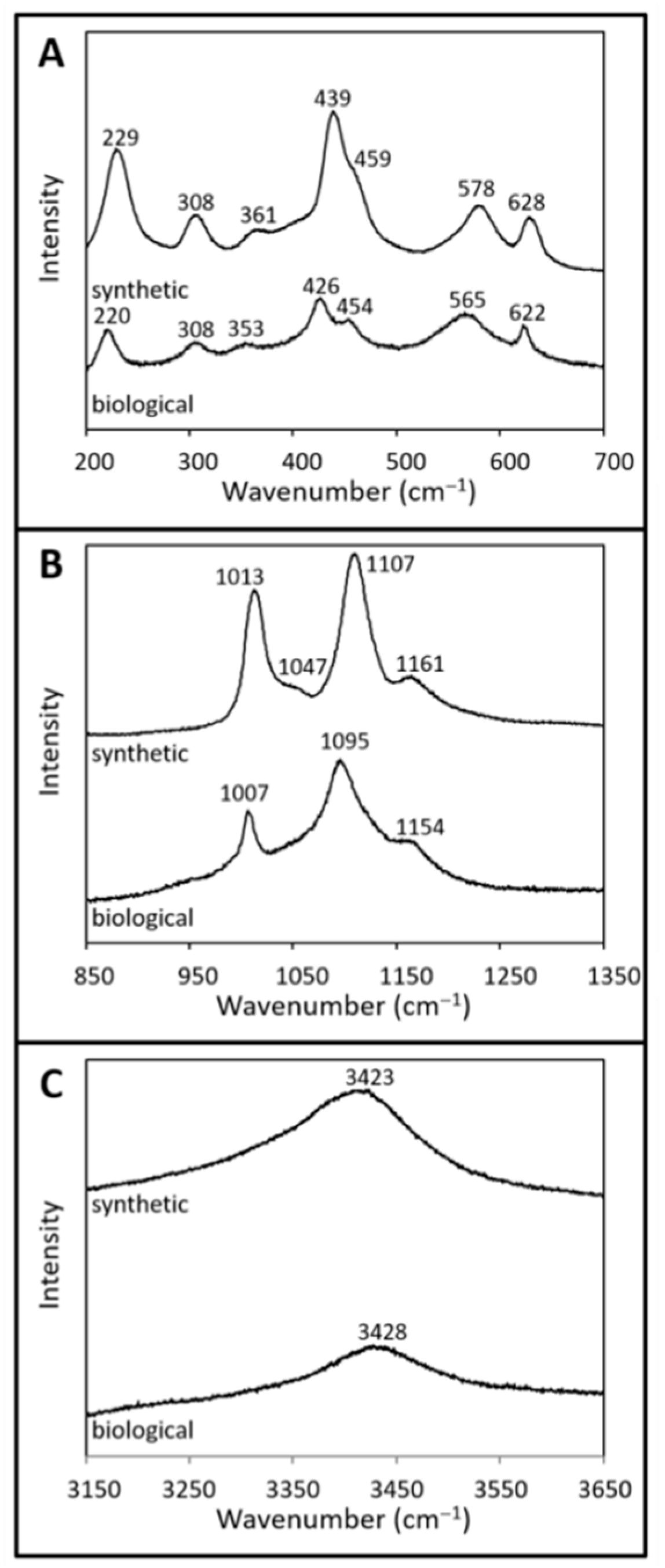
| Sample | νOH | ν3 | ν1 (NO3) | ν1 | ν4 | γ(OH) | ν2 | Fe−O |
|---|---|---|---|---|---|---|---|---|
| Synthetic jarosite | 3423 | 1161 | 1047 | 1013 | 628 | 578 | 459 | 439 |
| 1107 | 361 | |||||||
| 308 | ||||||||
| 229 | ||||||||
| Biological jarosite | 3428 | 1154 | 1007 | 622 | 565 | 454 | 426 | |
| 1095 | 353 | |||||||
| 308 | ||||||||
| 220 |
| Reflectance Spectra (all bands in cm−1) | ||
|---|---|---|
| Synthetic | Biological | Assignment |
| 3956 | 3ν3 (SO4)2−, OH OT/C | |
| 3835 | 3ν3 (SO4)2−, OH OT/C | |
| 3405 | 3406 | ν (OH) |
| 2460 | ||
| 2322 | 2ν3 (SO4)2−, 2 δ (OH) | |
| 2175 | 2163 | 2ν3 (SO4)2−, 2 δ (OH) |
| 2075 | 2069 | 2ν3 (SO4)2−, 2 δ (OH) |
| 2031 | 2025 | 2ν3 (SO4)2−, 2 δ (OH) |
| 1968 | 1945 | 2ν3 (SO4)2−, 2 δ (OH) |
| 1634 | 1639 | δ (H2O) |
| 1445 | 1426 | 2ν3 (SO4)2−, δ (CH2O) |
| 1401 | ||
| 1301 | ||
| 1245 | Christiansen feature | |
| 1187 | ν3 (SO4)2− | |
| 1136 | δ (OH) | |
| 1053 | 1058 | δ (OH) |
| 1012 | δ (OH) | |
| 980 | 974 | ν1 (SO4)2− |
| 680 | 733 | ν4 (SO4)2− |
| 594 | 556 | γ (OH) |
| 510 | Fe-O | |
| 485 | 461 | Fe-O |
| 428 | Fe-O | |
| 410 | Fe-O | |
| Reflectance spectra (all band locations in nm) | ||
|---|---|---|
| Synthetic | Biological | Assignment |
| 437 | Fe3+ crystal field transition | |
| 602 a | 506 a | Fe3+ crystal field transition |
| 905 | 902 | Fe3+ crystal field transition |
| 1470 | 1427 | OH overtones |
| 1848 | Combinations of OH or H2O bending, stretching, and rotational fundamentals | |
| 1969 | 1941 | Combinations of OH or H2O bending, stretching, and rotational fundamentals |
| 2033 a | 2230 a | vOH/H2O + γ/δ OH/H2O |
| 2263 | Fe –O–H combination | |
| 2319 a | 3v3S-O or OH/H2O combination and overtone | |
| 2351 a | 3v3S-O or OH/H2O combination and overtone | |
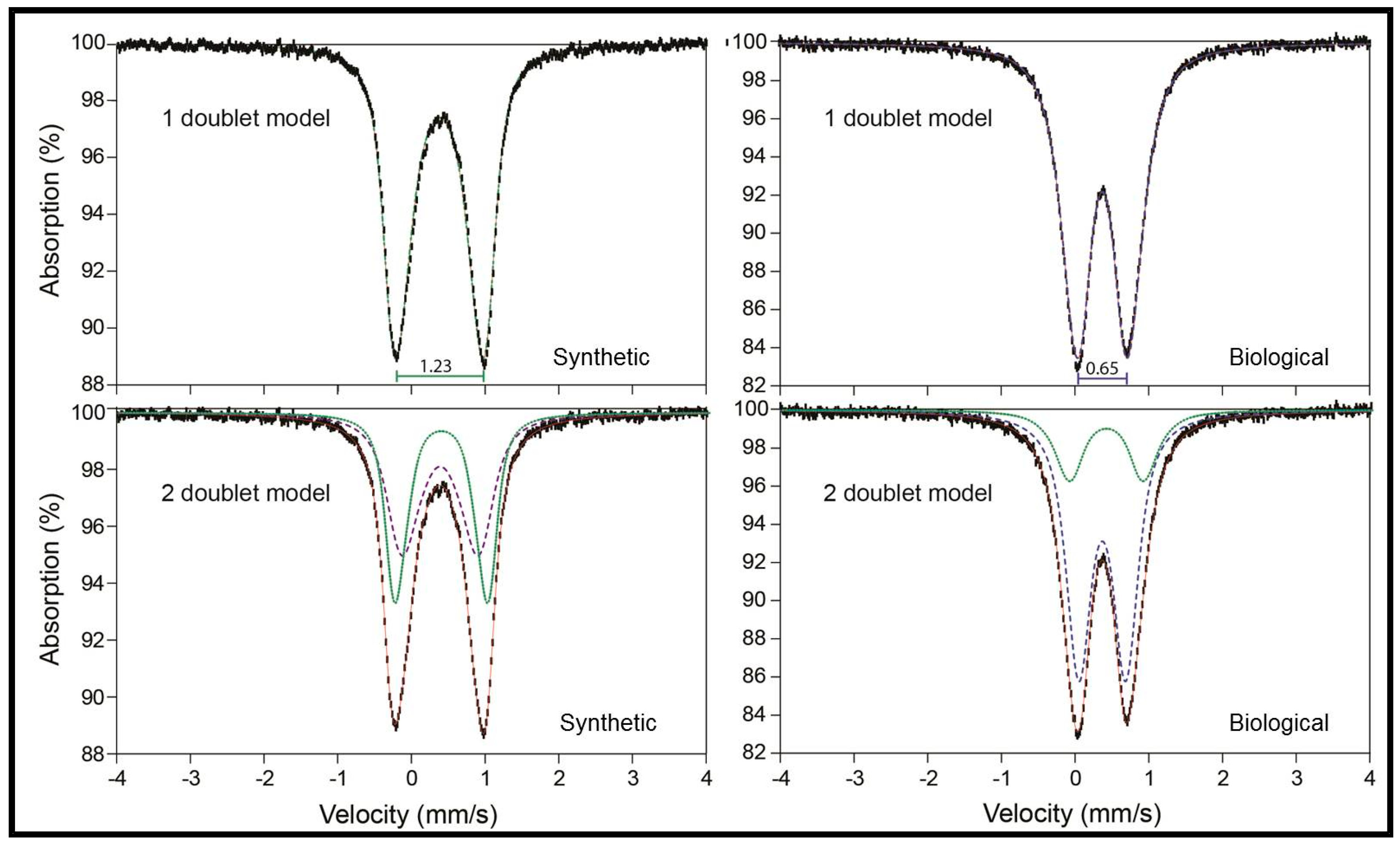
| Sample | δ | Δ | area | δ | Δ | area | |
|---|---|---|---|---|---|---|---|
| One-doublet model | |||||||
| Synthetic | 0.38 | 1.23 | 100 | ||||
| Biological | 0.37 | 0.65 | 100 | ||||
| Two-doublet model | |||||||
| Synthetic | 0.39 | 1.04 | 53 | 0.37 | 1.28 | 47 | |
| Biological | 0.39 | 0.96 | 23 | 0.37 | 0.61 | 77 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loiselle, L.; McCraig, M.A.; Dyar, M.D.; Léveillé, R.; Shieh, S.R.; Southam, G. A Spectral Comparison of Jarosites Using Techniques Relevant to the Robotic Exploration of Biosignatures on Mars. Life 2018, 8, 61. https://doi.org/10.3390/life8040061
Loiselle L, McCraig MA, Dyar MD, Léveillé R, Shieh SR, Southam G. A Spectral Comparison of Jarosites Using Techniques Relevant to the Robotic Exploration of Biosignatures on Mars. Life. 2018; 8(4):61. https://doi.org/10.3390/life8040061
Chicago/Turabian StyleLoiselle, Liane, Michael A. McCraig, M. Darby Dyar, Richard Léveillé, Sean R. Shieh, and Gordon Southam. 2018. "A Spectral Comparison of Jarosites Using Techniques Relevant to the Robotic Exploration of Biosignatures on Mars" Life 8, no. 4: 61. https://doi.org/10.3390/life8040061
APA StyleLoiselle, L., McCraig, M. A., Dyar, M. D., Léveillé, R., Shieh, S. R., & Southam, G. (2018). A Spectral Comparison of Jarosites Using Techniques Relevant to the Robotic Exploration of Biosignatures on Mars. Life, 8(4), 61. https://doi.org/10.3390/life8040061





