Catalytic/Protective Properties of Martian Minerals and Implications for Possible Origin of Life on Mars
Abstract
1. Introduction
2. Martian Minerals
3. Organics Detected on the Surface of Mars and in Martian Meteorites
4. Preservation of Biosignatures
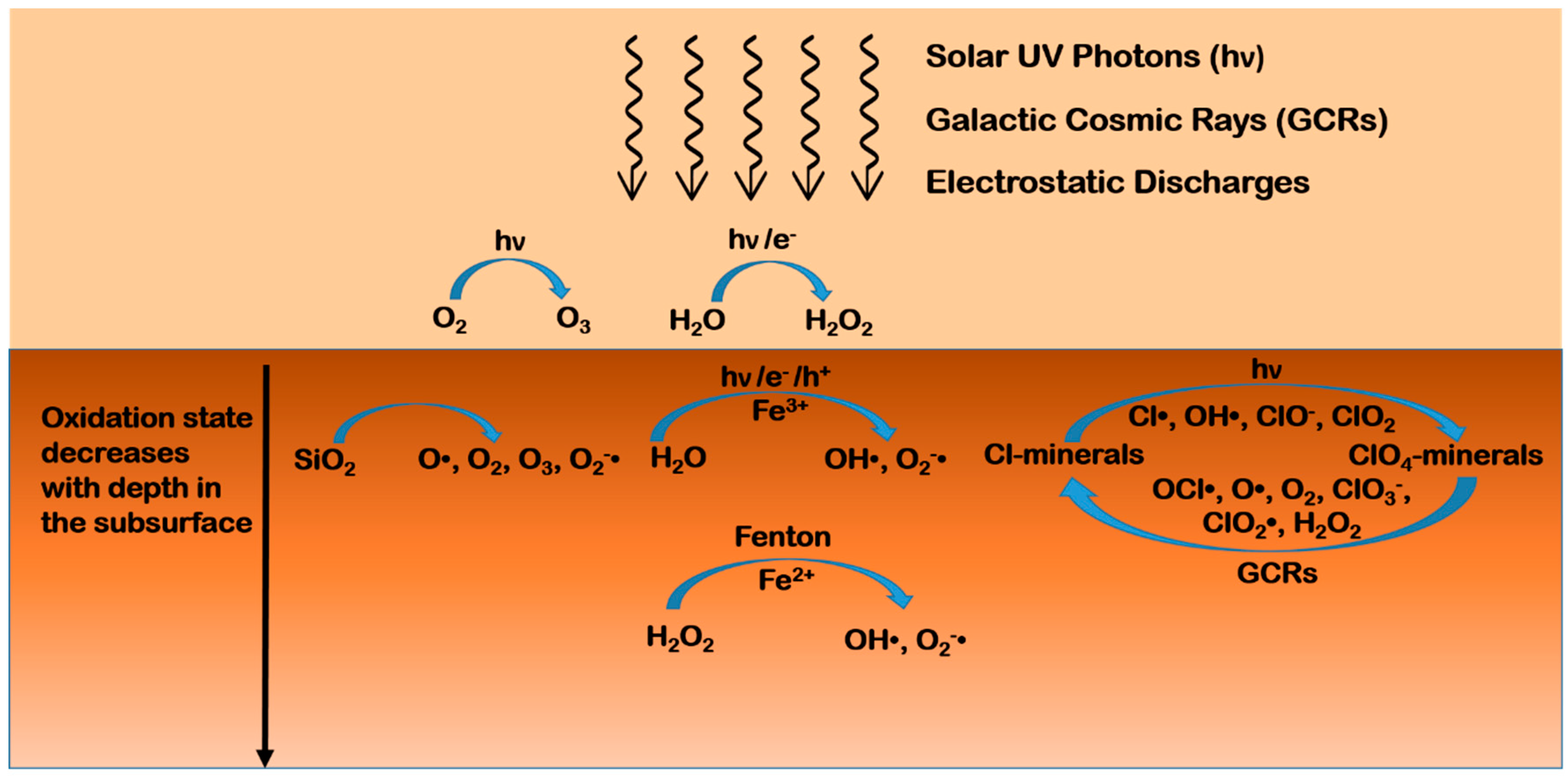
4.1. Oxidants on Mars
4.2. Photodegradation Processes of Organic Matter on Mars
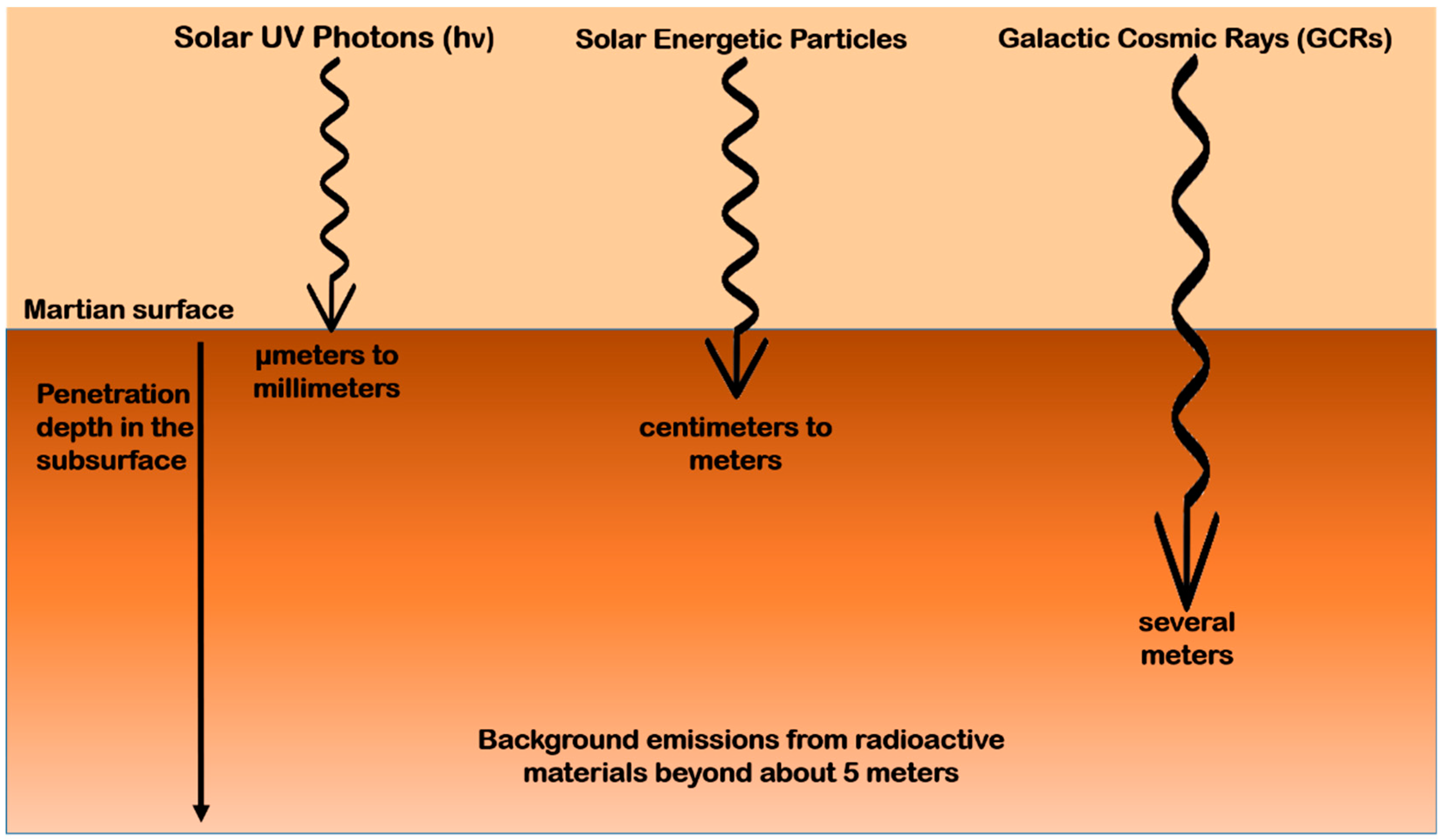
4.2.1. Radiation Environment on Mars
4.2.2. Laboratory Simulations of Martian Conditions
Early Investigations
Degradation Power of Water
Photocatalysis
Clay Minerals
Towards Realistic Simulations of the Martian Environment
Effects of Galactic Cosmic Rays and Solar Energetic Particles
5. Summary of the Results and Take-Home Messages
6. Implications for Future Martian Missions
Author Contributions
Funding
Conflicts of Interest
References
- Grotzinger, J.P.; Sumner, D.Y.; Kah, L.C.; Stack, K.; Gupta, S.; Edgar, L.; Rubin, D.; Lewis, K.; Schieber, J.; Mangold, N.; et al. A Habitable Fluvio-Lacustrine Environment at Yellowknife Bay, Gale Crater, Mars. Science 2014, 343, 1242777. [Google Scholar] [CrossRef] [PubMed]
- Arvidson, R.E.; Squyres, S.W.; Bell, J.F., III; Catalano, J.G.; Clark, B.C.; Crumpler, L.S.; de Souza, P.A., Jr.; Fairén, A.G.; Farrand, W.H.; Fox, V.K.; et al. Ancient Aqueous Environments at Endeavour Crater, Mars. Science 2014, 343, 1248097. [Google Scholar] [CrossRef] [PubMed]
- Sun, V.Z.; Milliken, R.E. Ancient and Recent Clay Formation on Mars as Revealed from a Global Survey of Hydrous Minerals in Crater Central Peaks. J. Geophys. Res. Planets 2015, 120, 2293–2332. [Google Scholar] [CrossRef]
- Carter, J.; Poulet, F.; Bibring, J.P.; Mangold, N.; Murchie, S. Hydrous Minerals on Mars as Seen by the CRISM and OMEGA Imaging Spectrometers: Updated Global View. J. Geophys. Res. Planets 2013, 118, 831–858. [Google Scholar] [CrossRef]
- Carter, J.; Poulet, F.; Bibring, J.-P.; Murchie, S.; Langevin, Y.; Mustard, J.F.; Gondet, B. Phyllosilicates and Other Hydrated Minerals on Mars: Global Distribution as Seen by MEx/OMEGA. In Proceedings of the 40th Lunar and Planetary Science Conference, Woodlands, TX, USA, 23–27 March 2009. [Google Scholar]
- Carter, J.; Loizeau, D.; Mangold, N.; Poulet, F.; Bibring, J.-P. Widespread Surface Weathering on Early Mars: A Case for a Warmer and Wetter Climate. Icarus 2015, 248, 373–382. [Google Scholar] [CrossRef]
- L’Haridon, J.; Mangold, N.; Meslin, P.-Y.; Johnson, J.R.; Rapin, W.; Forni, O.; Cousin, A.; Payré, V.; Dehouck, E.; Nachon, M.; et al. Chemical Variability in Mineralized Veins Observed by ChemCam on the Lower Slopes of Mount Sharp in Gale Crater, Mars. Icarus 2018, 311, 69–86. [Google Scholar] [CrossRef]
- Michalski, J.R.; Dobrea, E.Z.N.; Niles, P.B.; Cuadros, J. Ancient Hydrothermal Seafloor Deposits in Eridania Basin on Mars. Nat. Commun. 2017, 8, 15978. [Google Scholar] [CrossRef] [PubMed]
- Feldman, W.C.; Prettyman, T.H.; Maurice, S.; Plaut, J.J.; Bish, D.L.; Vaniman, D.T.; Mellon, M.T.; Metzger, A.E.; Squyres, S.W.; Karunatillake, S.; et al. Global Distribution of Near-Surface Hydrogen on Mars. J. Geophys. Res. Planets 2004, 109, E09006. [Google Scholar] [CrossRef]
- Milliken, R.E.; Mustard, J.F.; Poulet, F.; Jouglet, D.; Bibring, J.-P.; Gondet, B.; Langevin, Y. Hydration State of the Martian Surface as Seen by Mars Express OMEGA: 2. H2O Content of the Surface. J. Geophys. Res. Planets 2007, 112, E08S07. [Google Scholar] [CrossRef]
- Smith, M.D.; Wolff, M.J.; Lemmon, M.T.; Spanovich, N.; Banfield, D.; Budney, C.J.; Clancy, R.T.; Ghosh, A.; Landis, G.A.; Smith, P.; et al. First Atmospheric Science Results from the Mars Exploration Rovers Mini-TES. Science 2004, 306, 1750–1753. [Google Scholar] [CrossRef] [PubMed]
- Orosei, R.; Lauro, S.E.; Pettinelli, E.; Cicchetti, A.; Coradini, M.; Cosciotti, B.; Di Paolo, F.; Flamini, E.; Mattei, E.; Pajola, M.; et al. Radar Evidence of Subglacial Liquid Water on Mars. Science 2018, 361, 490–493. [Google Scholar] [CrossRef] [PubMed]
- Fisk, M.R.; Giovannoni, S.J. Sources of Nutrients and Energy for a Deep Biosphere on Mars. J. Geophys. Res. Planets 1999, 104, 11805–11815. [Google Scholar] [CrossRef]
- McKay, D.S.; Gibson, E.K.; Thomas-Keprta, K.L.; Vali, H.; Romanek, C.S.; Clemett, S.J.; Chillier, X.D.; Maechling, C.R.; Zare, R.N. Search for Past Life on Mars: Possible Relic Biogenic Activity in Martian Meteorite ALH84001. Science 1996, 273, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Morris, R.V.; Ruff, S.W.; Gellert, R.; Ming, D.W.; Arvidson, R.E.; Clark, B.C.; Golden, D.C.; Siebach, K.; Klingelhöfer, G.; Schröder, C.; et al. Identification of Carbonate-Rich Outcrops on Mars by the Spirit Rover. Science 2010, 329, 421–424. [Google Scholar] [CrossRef] [PubMed]
- Boynton, W.V.; Ming, D.W.; Kounaves, S.P.; Young, S.M.M.; Arvidson, R.E.; Hecht, M.H.; Hoffman, J.; Niles, P.B.; Hamara, D.K.; Quinn, R.C.; et al. Evidence for Calcium Carbonate at the Mars Phoenix Landing Site. Science 2009, 325, 61–64. [Google Scholar] [CrossRef] [PubMed]
- Ehlmann, B.L.; Mustard, J.F.; Murchie, S.L.; Poulet, F.; Bishop, J.L.; Brown, A.J.; Calvin, W.M.; Clark, R.N.; Des Marais, D.J.; Milliken, R.E.; et al. Orbital Identification of Carbonate-Bearing Rocks on Mars. Science 2008, 322, 1828–1832. [Google Scholar] [CrossRef] [PubMed]
- Eigenbrode, J.L.; Summons, R.E.; Steele, A.; Freissinet, C.; Millan, M.; Navarro-gonzález, R.; Sutter, B.; Mcadam, A.C.; Conrad, P.G.; Hurowitz, J.A.; et al. Organic Matter Preserved in 3-Billion-Year-Old Mudstones at Gale Crater, Mars. Science 2018, 360, 1096–1101. [Google Scholar] [CrossRef] [PubMed]
- Stewart, R.F.; Jensen, L.H. Redetermination of the Crystal Structure of Uracil. Acta Crystallogr. 1967, 23, 1102–1105. [Google Scholar] [CrossRef]
- Stern, J.C.; Sutter, B.; Freissinet, C.; Navarro-González, R.; McKay, C.P.; Archer, P.D.; Buch, A.; Brunner, A.E.; Coll, P.; Eigenbrode, J.L.; et al. Evidence for Indigenous Nitrogen in Sedimentary and Aeolian Deposits from the Curiosity Rover Investigations at Gale Crater, Mars. Proc. Natl. Acad. Sci. USA 2015, 112, 4245–4250. [Google Scholar] [CrossRef] [PubMed]
- Steele, A.; McCubbin, F.M.; Fries, M.D. The Provenance, Formation, and Implications of Reduced Carbon Phases in Martian Meteorites. Meteorit. Planet. Sci. 2016, 51, 2203–2225. [Google Scholar] [CrossRef]
- Fogel, M.L.; Steele, A. Nitrogen in Extraterrestrial Environments: Clues to the Possible Presence of Life. Elements 2013, 9, 367–372. [Google Scholar] [CrossRef]
- McCubbin, F.M.; Jones, R.H. Extraterrestrial Apatite: Planetary Geochemistry to Astrobiology. Elements 2015, 11, 183–188. [Google Scholar] [CrossRef]
- Powner, M.W.; Gerland, B.; Sutherland, J.D. Synthesis of Activated Pyrimidine Ribonucleotides in Prebiotically Plausible Conditions. Nature 2009, 459, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Szostak, J.W. Origins of Life: Systems Chemistry on Early Earth. Nature 2009, 459, 171–172. [Google Scholar] [CrossRef] [PubMed]
- Freissinet, C.; Glavin, D.P.; Mahaffy, P.R.; Miller, K.E.; Eigenbrode, J.L.; Summons, R.E.; Brunner, A.E.; Buch, A.; Szopa, C.; Archer, P.D.; et al. Organic Molecules in the Sheepbed Mudstone, Gale Crater, Mars. J. Geophys. Res. Planets 2015, 120, 495–514. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, J.; Savarino, J.; Jackson, T.L.; Thiemens, M.H. Evidence of Atmospheric Sulphur in the Martian Regolith from Sulphur Isotopes in Meteorites. Nature 2000, 404, 50–52. [Google Scholar] [CrossRef] [PubMed]
- Vago, J.L.; Westall, F.; Cavalazzi, B.; Team, T.E.S.W. Searching for Signs of Life on Other Planets: Mars a Case Study. In Biosignatures for Astrobiology; Cavalazzi, B.W.F., Ed.; Springer: Cham, Switzerland, 2019; pp. 283–300. [Google Scholar]
- Westall, F.; Bost, N.; Vago, J.L.; Kminek, G.; Campbell, K.A. Biosignatures on Mars: What, Where, and How? Implications for the Search for Martian Life. Astrobiology 2015, 15, 998–1029. [Google Scholar] [CrossRef] [PubMed]
- Steele, A.; McCubbin, F.M.; Fries, M.; Kater, L.; Boctor, N.Z.; Fogel, M.L.; Conrad, P.G.; Glamoclija, M.; Spencer, M.; Morrow, A.L.; et al. A Reduced Organic Carbon Component in Martian Basalts. Science 2012, 337, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Glavin, D.P.; Freissinet, C.; Miller, K.E.; Eigenbrode, J.L.; Brunner, A.E.; Buch, A.; Sutter, B.; Archer, P.D.; Atreya, S.K.; Brinckerhoff, W.B.; et al. Evidence for Perchlorates and the Origin of Chlorinated Hydrocarbons Detected by SAM at the Rocknest Aeolian Deposit in Gale Crater. J. Geophys. Res. Planets 2013, 118, 1955–1973. [Google Scholar] [CrossRef]
- Flynn, G.J. The Delivery of Organic Matter from Asteroids and Comets to the Early Surface of Mars. Earth Moon Planets 1996, 72, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Frantseva, K.; Mueller, M.; ten Kate, I.L.; Van der Tak, F.F.S.; Greenstreet, S. Delivery of Organics to Mars through Asteroid and Comet Impacts. Icarus 2018, 309, 125–133. [Google Scholar] [CrossRef]
- Flynn, G.J.; Nittler, L.R.; Engrand, C. Composition of Cosmic Dust: Sources and Implications for the Early Solar System. Elements 2016, 12, 177–183. [Google Scholar] [CrossRef]
- Grady, M.M.; Verchovsky, A.B.; Wright, I.P. Magmatic Carbon in Martian Meteorites: Attempts to Constrain the Carbon Cycle on Mars. Int. J. Astrobiol. 2004, 3, 117–124. [Google Scholar] [CrossRef]
- Wright, I.P.; Grady, M.M.; Pillinger, C.T. Chassigny and the Nakhlites: Carbon-Bearing Components and Their Relationship to Martian Environmental Conditions. Geochim. Cosmochim. Acta 1992, 56, 817–826. [Google Scholar] [CrossRef]
- Vago, J.; Witasse, O.; Svedhem, H.; Baglioni, P.; Haldemann, A.; Gianfiglio, G.; Blancquaert, T.; McCoy, D.; de Groot, R. ESA ExoMars Program: The next Step in Exploring Mars. Sol. Syst. Res. 2015, 49, 518–528. [Google Scholar] [CrossRef]
- Mahaffy, P.R.; Webster, C.R.; Cabane, M.; Conrad, P.G.; Coll, P.; Atreya, S.K.; Arvey, R.; Barciniak, M.; Benna, M.; Bleacher, L.; et al. The Sample Analysis at Mars Investigation and Instrument Suite. Space Sci. Rev. 2012, 170, 401–478. [Google Scholar] [CrossRef]
- Millan, M.; Szopa, C.; Buch, A.; Coll, P.; Glavin, D.P.; Freissinet, C.; Navarro-Gonzalez, R.; Francois, P.; Coscia, D.; Bonnet, J.Y.; et al. In Situ Analysis of Martian Regolith with the SAM Experiment during the First Mars Year of the MSL Mission: Identification of Organic Molecules by Gas Chromatography from Laboratory Measurements. Planet. Space Sci. 2016, 129, 88–102. [Google Scholar] [CrossRef]
- Miller, K.E.; Eigenbrode, J.L.; Freissinet, C.; Glavin, D.P.; Kotrc, B.; Francois, P.; Summons, R.E. Potential Precursor Compounds for Chlorohydrocarbons Detected in Gale Crater, Mars, by the SAM Instrument Suite on the Curiosity Rover. J. Geophys. Res. Planets 2016, 121, 296–308. [Google Scholar] [CrossRef]
- Franz, H.B.; McAdam, A.C.; Ming, D.W.; Freissinet, C.; Mahaffy, P.R.; Eldridge, D.L.; Fischer, W.W.; Grotzinger, J.P.; House, C.H.; Hurowitz, J.A.; et al. Large Sulfur Isotope Fractionations in Martian Sediments at Gale Crater. Nat. Geosci. 2017, 10, 658–662. [Google Scholar] [CrossRef]
- Steele, A.; Benning, L.G.; Wirth, R.; Siljestrom, S.; Fries, M.D.; Hauri, E.; Conrad, P.G.; Rogers, K.; Eigenbrode, J.; Schreiber, A.; et al. Organic Synthesis on Mars by Electrochemical Reduction of CO2. Sci. Adv. 2018, 4, eaat5118. [Google Scholar] [CrossRef]
- Mustard, J.; Adler, M.; Allwood, A.; Bass, D.; Beaty, D.; Bell, J., III; Brinckerhoff, W.; Carr, M.; Des Marais, D.; Drake, B.; et al. Report of the Mars 2020 Science Definition Team. Space Safety Magazine, 15 July 2013. [Google Scholar]
- Córdoba-Jabonero, C.; Lara, L.; Mancho, A.; Márquez, A.; Rodrigo, R. Solar Ultraviolet Transfer in the Martian Atmosphere: Biological and Geological Implications. Planet. Space Sci. 2003, 51, 399–410. [Google Scholar] [CrossRef]
- Martínez, G.M.; Newman, C.N.; De Vicente-Retortillo, A.; Fischer, E.; Renno, N.O.; Richardson, M.I.; Fairén, A.G.; Genzer, M.; Guzewich, S.D.; Haberle, R.M.; et al. The Modern Near-Surface Martian Climate: A Review of In-Situ Meteorological Data from Viking to Curiosity. Space Sci. Rev. 2017, 212, 295–338. [Google Scholar] [CrossRef]
- Patel, M.R.; Bérces, A.; Kolb, C.; Lammer, H.; Rettberg, P.; Zarnecki, J.C.; Selsis, F. Seasonal and Diurnal Variations in Martian Surface Ultraviolet Irradiation: Biological and Chemical Implications for the Martian Regolith. Int. J. Astrobiol. 2003, 2, 21–34. [Google Scholar] [CrossRef]
- Patel, M.R.; Zarnecki, J.C.; Catling, D.C. Ultraviolet Radiation on the Surface of Mars and the Beagle 2 UV Sensor. Planet. Space Sci. 2002, 50, 915–927. [Google Scholar] [CrossRef]
- Vicente-Retortillo, Á.; Lemmon, M.T.; Martínez, G.M.; Valero, F.; Vázquez, L.; Martín, L. Seasonal and Interannual Variability of Solar Radiation at Spirit, Opportunity and Curiosity Landing Sites. Fís. Tierra 2016, 28, 111–127. [Google Scholar] [CrossRef]
- Vicente-Retortillo, Á.; Martínez, G.M.; Renno, N.O.; Lemmon, M.T.; de la Torre-Juárez, M. Determination of Dust Aerosol Particle Size at Gale Crater Using REMS UVS and Mastcam Measurements. Geophys. Res. Lett. 2017, 44, 3502–3508. [Google Scholar] [CrossRef]
- Vicente-Retortillo, Á.; Valero, F.; Vázquez, L.; Martínez, G.M. A Model to Calculate Solar Radiation Fluxes on the Martian Surface. J. Space Weather Space Clim. 2015, 5, A33. [Google Scholar] [CrossRef]
- Matthiä, D.; Ehresmann, B.; Lohf, H.; Köhler, J.; Zeitlin, C.; Appel, J.; Sato, T.; Slaba, T.; Martin, C.; Berger, T.; et al. The Martian Surface Radiation Environment—A Comparison of Models and MSL/RAD Measurements. J. Space Weather Space Clim. 2016, 6, A13. [Google Scholar] [CrossRef]
- Clark, B.C.; Kounaves, S.P. Evidence for the Distribution of Perchlorates on Mars. Int. J. Astrobiol. 2016, 15, 311–318. [Google Scholar] [CrossRef]
- Kounaves, S.P.; Chaniotakis, N.A.; Chevrier, V.F.; Carrier, B.L.; Folds, K.E.; Hansen, V.M.; McElhoney, K.M.; O’Neil, G.D.; Weber, A.W. Identification of the Perchlorate Parent Salts at the Phoenix Mars Landing Site and Possible Implications. Icarus 2014, 232, 226–231. [Google Scholar] [CrossRef]
- Georgiou, C.D.; Sun, H.J.; McKay, C.P.; Grintzalis, K.; Papapostolou, I.; Zisimopoulos, D.; Panagiotidis, K.; Zhang, G.; Koutsopoulou, E.; Christidis, G.E.; et al. Evidence for Photochemical Production of Reactive Oxygen Species in Desert Soils. Nat. Commun. 2015, 6, 7100. [Google Scholar] [CrossRef] [PubMed]
- Yen, A.S.; Kim, S.S.; Hecht, M.H.; Frant, M.S.; Murray, B. Evidence That the Reactivity of the Martian Soil Is Due to Superoxide Ions. Science 2000, 289, 1909–1912. [Google Scholar] [CrossRef] [PubMed]
- Sutter, B.; McAdam, A.C.; Mahaffy, P.R.; Ming, D.W.; Edgett, K.S.; Rampe, E.B.; Eigenbrode, J.L.; Franz, H.B.; Freissinet, C.; Grotzinger, J.P.; et al. Evolved Gas Analyses of Sedimentary Rocks and Eolian Sediment in Gale Crater, Mars: Results of the Curiosity Rover’s Sample Analysis at Mars Instrument from Yellowknife Bay to the Namib Dune. J. Geophys. Res. Planets 2017, 122, 2574–2609. [Google Scholar] [CrossRef]
- Brucato, J.B.; Fornaro, T. Role of Mineral Surfaces in Prebiotic Processes and Space-like Conditions. In Biosignatures for Astrobiology; Cavalazzi, B., Westall, F., Eds.; Springer: Berlin, Germany, 2018; pp. 183–204. [Google Scholar]
- Fornaro, T.; Brucato, J.R.; Branciamore, S.; Pucci, A. Adsorption of Nucleic Acid Bases on Magnesium Oxide (MgO). Int. J. Astrobiol. 2013, 12, 78–86. [Google Scholar] [CrossRef]
- Fornaro, T.; Brucato, J.R.; Feuillie, C.; Sverjensky, D.A.; Hazen, R.M.; Brunetto, R.; D’Amore, M.; Barone, V. Binding of Nucleic Acid Components to the Serpentinite-Hosted Hydrothermal Mineral Brucite. Astrobiology 2018, 18, 989–1007. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.-F. Origins of Life: From the Mineral to the Biochemical World. BIO Web Conf. 2015, 4, 14. [Google Scholar] [CrossRef]
- Hazen, R.M.; Sverjensky, D.A. Mineral Surfaces, Geochemical Complexities, and the Origins of Life. Cold Spring Harb. Perspect. Biol. 2010, 2, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Hazen, R.M. Mineral Surfaces and the Prebiotic Selection and Organization of Biomolecules. Am. Mineral. 2006, 91, 1715–1729. [Google Scholar] [CrossRef]
- Wächtershäuser, G. Evolution of the First Metabolic Cycles. Proc. Natl. Acad. Sci. USA 1990, 87, 200–204. [Google Scholar] [CrossRef] [PubMed]
- Hazen, R.M. Genesis: The Scientific Quest for Life’s Origin; Joseph Henry Press: Washington, DC, USA, 2005. [Google Scholar]
- Brandes, J.A.; Boctor, N.Z.; Cody, G.D.; Cooper, B.A.; Hazen, R.M.; Yoder, H.S. Abiotic Nitrogen Reduction on the Early Earth. Nature 1998, 395, 365–367. [Google Scholar] [CrossRef] [PubMed]
- Botta, L.; Bizzarri, B.M.; Piccinino, D.; Fornaro, T.; Brucato, J.R.; Saladino, R. Prebiotic Synthesis of Carboxylic Acids, Amino Acids and Nucleic Acid Bases from Formamide under Photochemical Conditions. Eur. Phys. J. Plus 2017, 132, 317. [Google Scholar] [CrossRef]
- Fornaro, T.; Boosman, A.; Brucato, J.R.; ten Kate, I.L.; Siljeström, S.; Poggiali, G.; Steele, A.; Hazen, R.M. UV Irradiation of Biomarkers Adsorbed on Minerals under Martian-like Conditions: Hints for Life Detection on Mars. Icarus 2018, 313, 38–60. [Google Scholar] [CrossRef]
- Ehlmann, B.L.; Edwards, C.S. Mineralogy of the Martian Surface. Annu. Rev. Earth Planet. Sci. 2014, 42, 291–315. [Google Scholar] [CrossRef]
- Bibring, J.P.; Langevin, Y.; Mustard, J. Global Mineralogical and Aqueous Mars History Derived from OMEGA/Mars Express Data. Science 2006, 312, 400–404. [Google Scholar] [CrossRef] [PubMed]
- Milliken, R.E.; Swayze, G.A.; Arvidson, R.E.; Bishop, J.L.; Clark, R.N.; Ehlmann, B.L.; Green, R.O.; Grotzinger, J.P.; Morris, R.V.; Murchie, S.L.; et al. Opaline Silica in Young Deposits on Mars. Geology 2008, 36, 847–850. [Google Scholar] [CrossRef]
- Mustard, J.F.; Murchie, S.L.; Pelkey, S.M.; Ehlmann, B.L.; Milliken, R.E.; Grant, J.A.; Bibring, J.; Poulet, F.; Bishop, J.; Dobrea, E.N.; et al. Hydrated Silicate Minerals on Mars Observed by the Mars Reconnaissance Orbiter CRISM Instrument. Nature 2008, 454, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Murchie, S.L.; Mustard, J.F.; Ehlmann, B.L.; Milliken, R.E.; Bishop, J.L.; McKeown, N.K.; Noe Dobrea, E.Z.; Seelos, F.P.; Buczkowski, D.L.; Wiseman, S.M.; et al. A Synthesis of Martian Aqueous Mineralogy after 1 Mars Year of Observations from the Mars Reconnaissance Orbiter. J. Geophys. Res. Planets 2009, 114, E00D06. [Google Scholar] [CrossRef]
- Negron-Mendoza, A.; Ramos-Bernal, S. The Role of Clays in the Origin of Life. In Origins. Cellular Origin, Life in Extreme Habitats and Astrobiology; Seckbach, J., Ed.; Springer: Dordrecht, The Netherlands, 2004; pp. 181–194. [Google Scholar]
- Ehlmann, B.L.; Mustard, J.F.; Fassett, C.I.; Schon, S.C.; Head, J.W., III; Des Marais, D.J.; Grant, J.A.; Murchie, S.L. Clay Minerals in Delta Deposits and Organic Preservation Potential on Mars. Nat. Geosci. 2008, 1, 355–358. [Google Scholar] [CrossRef]
- Ehlmann, B.L.; Mustard, J.F.; Swayze, G.A.; Clark, R.N.; Bishop, J.L.; Poulet, F.; Des Marais, D.J.; Roach, L.H.; Milliken, R.E.; Wray, J.J.; et al. Identification of Hydrated Silicate Minerals on Mars Using MRO-CRISM: Geologic Context near Nili Fossae and Implications for Aqueous Alteration. J. Geophys. Res. Planets 2009, 114, E00D08. [Google Scholar] [CrossRef]
- Ehlmann, B.L.; Mustard, J.F.; Murchie, S.L.; Bibring, J.-P.; Meunier, A.; Fraeman, A.A.; Langevin, Y. Subsurface Water and Clay Mineral Formation during the Early History of Mars. Nature 2011, 479, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Ehlmann, B.L.; Mustard, J.F.; Clark, R.N.; Swayze, G.A.; Murchie, S.L. Evidence for Low-Grade Metamorphism, Hydrothermal Alteration, and Diagenesis on Mars from Phyllosilicate Mineral Assemblages. Clays Clay Miner. 2011, 59, 359–377. [Google Scholar] [CrossRef]
- Bishop, J.L.; Dobrea, E.Z.N.; McKeown, N.K.; Parente, M.; Ehlmann, B.L.; Michalski, J.R.; Milliken, R.E.; Poulet, F.; Swayze, G.A.; Mustard, J.F.; et al. Phyllosilicate Diversity and Past Aqueous Activity Revealed at Mawrth Vallis, Mars. Science 2008, 321, 830–833. [Google Scholar] [CrossRef] [PubMed]
- Noe Dobrea, E.Z.; Wray, J.J.; Calef, F.J.; Parker, T.J.; Murchie, S.L. Hydrated Minerals on Endeavour Crater’s Rim and Interior, and Surrounding Plains: New Insights from CRISM Data. Geophys. Res. Lett. 2012, 39, L23201. [Google Scholar] [CrossRef]
- Adeli, S.; Hauber, E.; Le Deit, L.; Jaumann, R. Geologic Evolution of the Eastern Eridania Basin: Implications for Aqueous Processes in the Southern Highlands of Mars. J. Geophys. Res. Planets 2015, 120, 1774–1799. [Google Scholar] [CrossRef]
- Vaniman, D.T.; Bish, D.L.; Ming, D.W.; Bristow, T.F.; Morris, R.V.; Blake, D.F.; Chipera, S.J.; Morrison, S.M.; Treiman, A.H.; Rampe, E.B.; et al. Mineralogy of a Mudstone at Yellowknife Bay, Gale Crater, Mars. Science 2014, 343, 1243480. [Google Scholar] [CrossRef] [PubMed]
- Michalski, J.R.; Cuadros, J.; Bishop, J.L.; Darby Dyar, M.; Dekov, V.; Fiore, S. Constraints on the Crystal-Chemistry of Fe/Mg-Rich Smectitic Clays on Mars and Links to Global Alteration Trends. Earth Planet. Sci. Lett. 2015, 427, 215–225. [Google Scholar] [CrossRef]
- Bristow, T.F.; Bish, D.L.; Vaniman, D.T.; Morris, R.V.; Blake, D.F.; Grotzinger, J.P.; Rampe, E.B.; Crisp, J.A.; Achilles, C.N.; Ming, D.W.; et al. The Origin and Implications of Clay Minerals from Yellowknife Bay, Gale Crater, Mars. Am. Mineral. 2015, 100, 824–836. [Google Scholar] [CrossRef] [PubMed]
- Hurowitz, J.A.; Grotzinger, J.P.; Fischer, W.W.; McLennan, S.M.; Milliken, R.E.; Stein, N.; Vasavada, A.R.; Blake, D.F.; Dehouck, E.; Eigenbrode, J.L.; et al. Redox Stratification of an Ancient Lake in Gale Crater, Mars. Science 2017, 356, eaah6849. [Google Scholar] [CrossRef] [PubMed]
- Rampe, E.B.; Ming, D.W.; Blake, D.F.; Bristow, T.F.; Chipera, S.J.; Grotzinger, J.P.; Morris, R.V.; Morrison, S.M.; Vaniman, D.T.; Yen, A.S.; et al. Mineralogy of an Ancient Lacustrine Mudstone Succession from the Murray Formation, Gale Crater, Mars. Earth Planet. Sci. Lett. 2017, 471, 172–185. [Google Scholar] [CrossRef]
- Bristow, T.F.; Rampe, E.B.; Achilles, C.N.; Blake, D.F.; Chipera, S.J.; Craig, P.; Crisp, J.A.; Des Marais, D.J.; Downs, R.T.; Gellert, R.; et al. Clay Mineral Diversity and Abundance in Sedimentary Rocks of Gale Crater, Mars. Sci. Adv. 2018, 4, eaar3330. [Google Scholar] [CrossRef] [PubMed]
- Osterloo, M.M.; Hamilton, V.E.; Bandfield, J.L.; Glotch, T.D.; Baldridge, A.M.; Christensen, P.R.; Tornabene, L.L.; Anderson, F.S. Chloride-Bearing Materials in the Southern Highlands of Mars. Science 2008, 319, 1651–1654. [Google Scholar] [CrossRef] [PubMed]
- Osterloo, M.M.; Anderson, F.S.; Hamilton, V.E.; Hynek, B.M. Geologic Context of Proposed Chloride-Bearing Materials on Mars. J. Geophys. Res. Planets 2010, 115, E10012. [Google Scholar] [CrossRef]
- Glotch, T.D.; Bandfield, J.L.; Wolff, M.J.; Arnold, J.A.; Che, C. Constraints on the Composition and Particle Size of Chloride Salt-Bearing Deposits on Mars. J. Geophys. Res. Planets 2016, 121, 454–471. [Google Scholar] [CrossRef]
- Squyres, S.W. In Situ Evidence for an Ancient Aqueous Environment at Meridiani Planum, Mars. Science 2004, 306, 1709–1714. [Google Scholar] [CrossRef] [PubMed]
- McAdam, A.C.; Franz, H.B.; Sutter, B.; Archer, P.D., Jr.; Freissinet, C.; Eigenbrode, J.L.; Ming, D.W.; Atreya, S.K.; Bish, D.L.; Blake, D.F.; et al. Sulfur-bearing Phases Detected by Evolved Gas Analysis of the Rocknest Aeolian Deposit, Gale Crater, Mars. J. Geophys. Res. Planets 2014, 119, 373–393. [Google Scholar] [CrossRef]
- Ruff, S.W.; Farmer, J.D. Silica Deposits on Mars with Features Resembling Hot Spring Biosignatures at El Tatio in Chile. Nat. Commun. 2016, 7, 13554. [Google Scholar] [CrossRef] [PubMed]
- Viviano-Beck, C.E.; Seelos, F.P.; Murchie, S.L.; Kahn, E.G.; Seelos, K.D.; Taylor, H.W.; Morgan, M.F. Revised CRISM Spectral Parameters and Summary Products Based on the Currently Detected Mineral Diversity on Mars. J. Geophys. Res. Planets 2014, 119, 1403–1431. [Google Scholar] [CrossRef]
- Gooding, J.L. Chemical Weathering on Mars Thermodynamic Stabilities of Primary Minerals (and Their Alteration Products) from Mafic Igneous Rocks. Icarus 1978, 33, 483–513. [Google Scholar] [CrossRef]
- Christensen, P.R.; Bandfield, J.L.; Clark, R.N.; Edgett, K.S.; Hamilton, V.E.; Hoefen, T.; Kieffer, H.H.; Kuzmin, R.O.; Lane, M.D.; Malin, M.C.; et al. Detection of Crystalline Hematite Mineralization on Mars by the Thermal Emission Spectrometer: Evidence for near-Surface Water. J. Geophys. Res. Planets 2000, 105, 9623–9642. [Google Scholar] [CrossRef]
- Christensen, P.R.; Morris, R.V.; Lane, M.D.; Bandfield, J.L.; Malin, M.C. Global Mapping of Martian Hematite Mineral Deposits: Remnants of Water-Driven Processes on Early Mars. J. Geophys. Res. Planets 2001, 106, 23873–23885. [Google Scholar] [CrossRef]
- Arvidson, R.E.; Poulet, F.; Morris, R.V.; Bibring, J.-P.; Bell, J.F.; Squyres, S.W.; Christensen, P.R.; Bellucci, G.; Gondet, B.; Ehlmann, B.L.; et al. Nature and Origin of the Hematite-Bearing Plains of Terra Meridiani Based on Analyses of Orbital and Mars Exploration Rover Data Sets. J. Geophys. Res. Planets 2006, 111. [Google Scholar] [CrossRef]
- Catling, D.C.; Moore, J.M. The Nature of Coarse-Grained Crystalline Hematite and Its Implications for the Early Environment of Mars. Icarus 2003, 165, 277–300. [Google Scholar] [CrossRef]
- Klingelhöfer, G.; Morris, R.V.; Bernhardt, B.; Schröder, C.; Rodionov, D.S.; de Souza, P.A.; Yen, A.; Gellert, R.; Evlanov, E.N.; Zubkov, B.; et al. Jarosite and Hematite at Meridiani Planum from Opportunity’s Mössbauer Spectrometer. Science 2004, 306, 1740–1745. [Google Scholar] [CrossRef] [PubMed]
- Souza-Egipsy, V.; Ormö, J.; Bowen, B.B.; Chan, M.A.; Komatsu, G. Ultrastructural Study of Iron Oxide Precipitates: Implications for the Search for Biosignatures in the Meridiani Hematite Concretions, Mars. Astrobiology 2006, 6, 527–545. [Google Scholar] [CrossRef] [PubMed]
- Bishop, J.L.; Fröschl, H.; Mancinelli, R.L. Alteration Processes in Volcanic Soils and Identification of Exobiologically Important Weathering Products on Mars Using Remote Sensing. J. Geophys. Res. Planets 1998, 103, 31457–31476. [Google Scholar] [CrossRef]
- Koch, C.B.; Mørup, S.; Madsen, M.B.; Vistisen, L. Iron-Containing Weathering Products of Basalt in a Cold, Dry Climate. Chem. Geol. 1995, 122, 109–119. [Google Scholar] [CrossRef]
- Schwertmann, U. The Effect of Pedogenic Environments on Iron Oxide Minerals. In Advances in Soil Science; Springer: New York, NY, USA, 1958; pp. 171–200. [Google Scholar]
- Fraeman, A.A.; Arvidson, R.E.; Catalano, J.G.; Grotzinger, J.P.; Morris, R.V.; Murchie, S.L.; Stack, K.M.; Humm, D.C.; McGovern, J.A.; Seelos, F.P.; et al. A Hematite-Bearing Layer in Gale Crater, Mars: Mapping and Implications for Past Aqueous Conditions. Geology 2013, 41, 1103–1106. [Google Scholar] [CrossRef]
- Ody, A.; Poulet, F.; Bibring, J.P.; Loizeau, D.; Carter, J.; Gondet, B.; Langevin, Y. Global Investigation of Olivine on Mars: Insights into Crust and Mantle Compositions. J. Geophys. Res. Planets 2013, 118, 234–262. [Google Scholar] [CrossRef]
- Ody, A.; Poulet, F.; Langevin, Y.; Bibring, J.P.; Bellucci, G.; Altieri, F.; Gondet, B.; Vincendon, M.; Carter, J.; Manaud, N. Global Maps of Anhydrous Minerals at the Surface of Mars from OMEGA/MEx. J. Geophys. Res. Planets 2012, 117, 1–14. [Google Scholar] [CrossRef]
- Bibring, J.-P.; Langevin, Y.; Gendrin, A.; Gondet, B.; Poulet, F.; Berthé, M.; Soufflot, A.; Arvidson, R.; Mangold, N.; Mustard, J.; et al. Mars Surface Diversity as Revealed by the OMEGA/Mars Express Observations. Science 2005, 307, 1576–1581. [Google Scholar] [CrossRef] [PubMed]
- Dehouck, E.; McLennan, S.M.; Meslin, P.-Y.; Cousin, A. Constraints on Abundance, Composition, and Nature of X-Ray Amorphous Components of Soils and Rocks at Gale Crater, Mars. J. Geophys. Res. Planets 2014, 119, 2640–2657. [Google Scholar] [CrossRef]
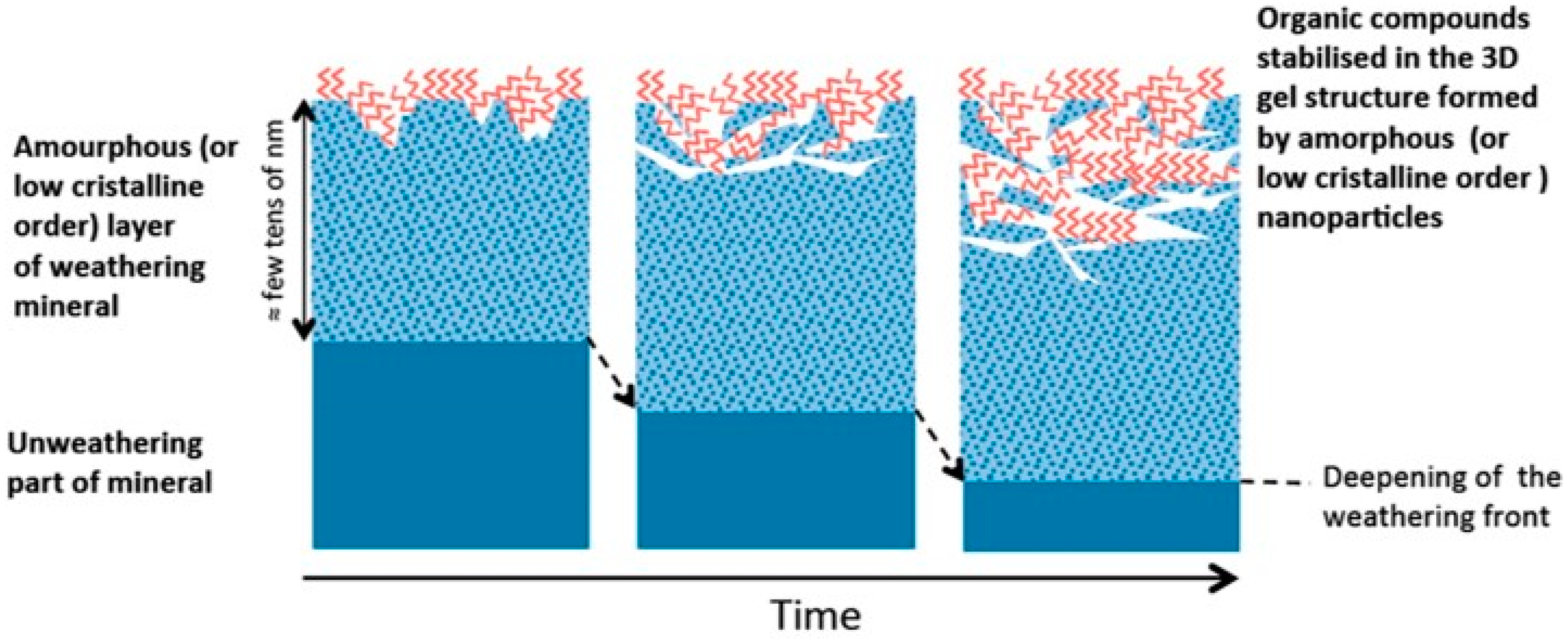
- Basile-Doelsch, I.; Balesdent, J.; Rose, J. Are Interactions between Organic Compounds and Nanoscale Weathering Minerals the Key Drivers of Carbon Storage in Soils? Environ. Sci. Technol. 2015, 49, 3997–3998. [Google Scholar] [CrossRef] [PubMed]
- Grady, M.M.; Wright, I.P.; Pillinger, C.T. A Carbon and Nitrogen Isotope Study of Zagami. J. Geophys. Res. Planets 1997, 102, 9165–9173. [Google Scholar] [CrossRef]
- Steele, A.; Fries, M.D.; Amundsen, H.E.F.; Mysen, B.O.; Fogel, M.L.; Schweizer, M.; Boctor, N.Z. Comprehensive Imaging and Raman Spectroscopy of Carbonate Globules from Martian Meteorite ALH 84001 and a Terrestrial Analogue from Svalbard. Meteorit. Planet. Sci. 2007, 42, 1549–1566. [Google Scholar] [CrossRef]
- Steele, A.; McCubbin, F.M.; Fries, M.D.; Golden, D.C.; Ming, D.W.; Benning, L.G. Graphite in the Martian Meteorite Allan Hills 84001. Am. Mineral. 2012, 97, 1256–1259. [Google Scholar] [CrossRef]
- Committee on an Astrobiology Strategy for the Exploration of Mars; National Academies Press: Washington, DC, USA, 2007; p. 130.
- Hedges, J.I.; Keil, R.G. Sedimentary Organic Matter Preservation: An Assessment and Speculative Synthesis. Mar. Chem. 1995, 49, 81–115. [Google Scholar] [CrossRef]
- Lalonde, K.; Mucci, A.; Ouellet, A.; Gélinas, Y. Preservation of Organic Matter in Sediments Promoted by Iron. Nature 2012, 483, 198–200. [Google Scholar] [CrossRef] [PubMed]
- Farmer, J.D.; Des Marais, D.J. Exploring for a Record of Ancient Martian Life. J. Geophys. Res. Planets 1999, 104, 26977–26995. [Google Scholar] [CrossRef]
- Biondi, E.; Howell, L.; Benner, S.A. Opal Absorbs and Stabilizes RNA–A Hierarchy of Prebiotic Silica Minerals. Synlett 2017, 28, 84. [Google Scholar]
- Grotzinger, J.P.; Crisp, J.; Vasavada, A.R.; Anderson, R.C.; Baker, C.J.; Barry, R.; Blake, D.F.; Conrad, P.; Edgett, K.S.; Ferdowski, B.; et al. Mars Science Laboratory Mission and Science Investigation. Space Sci. Rev. 2012, 170, 5–56. [Google Scholar] [CrossRef]
- Gaillard, F.; Michalski, J.; Berger, G.; McLennan, S.M.; Scaillet, B. Geochemical Reservoirs and Timing of Sulfur Cycling on Mars. Space Sci. Rev. 2013, 174, 251–300. [Google Scholar] [CrossRef]
- Sephton, M.A. Organic Compounds in Carbonaceous Meteorites. Nat. Prod. Rep. 2002, 19, 292–311. [Google Scholar] [CrossRef] [PubMed]
- Kotler, J.M.; Hinman, N.W.; Yan, B.; Stoner, D.L.; Scott, J.R. Glycine Identification in Natural Jarosites Using Laser Desorption Fourier Transform Mass Spectrometry: Implications for the Search for Life on Mars. Astrobiology 2008, 8, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Aubrey, A.; Cleaves, H.J.; Chalmers, J.H.; Skelley, A.M.; Mathies, R.A.; Grunthaner, F.J.; Ehrenfreund, P.; Bada, J.L. Sulfate Minerals and Organic Compounds on Mars. Geology 2006, 34, 357–360. [Google Scholar] [CrossRef]
- Fernández-Remolar, D.C.; Chong-Díaz, G.; Ruíz-Bermejo, M.; Harir, M.; Schmitt-Kopplin, P.; Tziotis, D.; Gómez-Ortíz, D.; García-Villadangos, M.; Martín-Redondo, M.P.; Gómez, F.; et al. Molecular Preservation in Halite- and Perchlorate-Rich Hypersaline Subsurface Deposits in the Salar Grande Basin (Atacama Desert, Chile): Implications for the Search for Molecular Biomarkers on Mars. J. Geophys. Res. Biogeosci. 2013, 118, 922–939. [Google Scholar] [CrossRef]
- Macko, S.A.; Engel, M.H.; Parker, P.L. Early Diagenesis of Organic Matter in Sediments. In Organic Geochemistry; Springer: Boston, MA, USA, 1993; pp. 211–224. [Google Scholar]
- Lasne, J.; Noblet, A.; Szopa, C.; Navarro-González, R.; Cabane, M.; Poch, O.; Stalport, F.; François, P.; Atreya, S.K.; Coll, P. Oxidants at the Surface of Mars: A Review in Light of Recent Exploration Results. Astrobiology 2016, 16, 977–996. [Google Scholar] [CrossRef] [PubMed]
- Chevrier, V.F.; Hanley, J.; Altheide, T.S. Stability of Perchlorate Hydrates and Their Liquid Solutions at the Phoenix Landing Site, Mars. Geophys. Res. Lett. 2009, 36, 1–6. [Google Scholar]
- Hecht, M.H.; Kounaves, S.P.; Quinn, R.C.; West, S.J.; Young, S.M.M.; Ming, D.W.; Catling, D.C.; Clark, B.C.; Boynton, W.V.; Hoffman, J.; et al. Detection of Perchlorate and the Soluble Chemistry of Martian Soil at the Phoenix Lander Site. Science 2009, 325, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Kounaves, S.P.; Carrier, B.L.; O’Neil, G.D.; Stroble, S.T.; Claire, M.W. Evidence of Martian Perchlorate, Chlorate, and Nitrate in Mars Meteorite EETA79001: Implications for Oxidants and Organics. Icarus 2014, 229, 206–213. [Google Scholar] [CrossRef]
- Smith, M.L.; Claire, M.W.; Catling, D.C.; Zahnle, K.J. The Formation of Sulfate, Nitrate and Perchlorate Salts in the Martian Atmosphere. Icarus 2014, 231, 51–64. [Google Scholar] [CrossRef]
- Carrier, B.L.; Kounaves, S.P. The Origins of Perchlorate in the Martian Soil. Geophys. Res. Lett. 2015, 42, 3739–3745. [Google Scholar] [CrossRef]
- Wang, A.; Jacson, A.; Yan, Y.C.; Houghton, J. (Per)Chlorate Formation Through Electrochemistry in Martian Atmosphere-Surface Interaction. In Proceedings of the 49th Lunar and Planetary Science Conference, Woodlands, TX, USA, 19–23 March 2018. [Google Scholar]
- Kolb, V.M. Oxidation of Organic Materials with Perchlorates: Relevance to the Chemistry on the Martian Surface; Hoover, R.B., Levin, G.V., Rozanov, A.Y., Retherford, K.D., Eds.; International Society for Optics and Photonics: Bellingham, WA, USA, 2009; Volume 7441, p. 74410E. [Google Scholar]
- Quinn, R.C.; Martucci, H.F.H.; Miller, S.R.; Bryson, C.E.; Grunthaner, F.J.; Grunthaner, P.J. Perchlorate Radiolysis on Mars and the Origin of Martian Soil Reactivity. Astrobiology 2013, 13, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Crandall, P.B.; Góbi, S.; Gillis-Davis, J.; Kaiser, R.I. Can Perchlorates Be Transformed to Hydrogen Peroxide (H2O2) Products by Cosmic Rays on the Martian Surface? J. Geophys. Res. Planets 2017, 122, 1880–1892. [Google Scholar] [CrossRef]
- Góbi, S.; Bergantini, A.; Kaiser, R.I. In Situ Detection of Chlorine Dioxide (ClO2) in the Radiolysis of Perchlorates and Implications for the Stability of Organics on Mars. Astrophys. J. 2016, 832, 164. [Google Scholar] [CrossRef]
- Góbi, S.; Bergantini, A.; Kaiser, R.I. Degradation of Adenine on the Martian Surface in the Presence of Perchlorates and Ionizing Radiation: A Reflectron Time-of-Flight Mass Spectrometric Study. Astrophys. J. 2017, 838, 84. [Google Scholar] [CrossRef]
- Góbi, S.; Abplanalp, M.J.; Kaiser, R.I. Effect of Perchlorates on Electron Radiolysis of Glycine with Application to Mars. Astrophys. J. 2016, 822, 8. [Google Scholar] [CrossRef]
- Turner, A.M.; Abplanalp, M.J.; Kaiser, R.I. Mechanistic Studies on the Radiolytic Decomposition of Perchlorates on the Martian Surface. Astrophys. J. 2016, 820, 127. [Google Scholar] [CrossRef]
- Sutter, B.; Quinn, R.C.; Archer, P.D.; Glavin, D.P.; Glotch, T.D.; Kounaves, S.P.; Osterloo, M.M.; Rampe, E.B.; Ming, D.W. Measurements of Oxychlorine Species on Mars. Int. J. Astrobiol. 2017, 16, 203–217. [Google Scholar] [CrossRef]
- Clancy, R.; Sandor, B.; Moriarty-Schieven, G. A Measurement of the 362 GHz Absorption Line of Mars Atmospheric H2O2. Icarus 2004, 168, 116–121. [Google Scholar] [CrossRef]
- Encrenaz, T.; Greathouse, T.K.; Lefèvre, F.; Atreya, S.K. Hydrogen Peroxide on Mars: Observations, Interpretation and Future Plans. Planet. Space Sci. 2012, 68, 3–17. [Google Scholar] [CrossRef]
- Encrenaz, T.; Greathouse, T.K.; Lefèvre, F.; Montmessin, F.; Forget, F.; Fouchet, T.; Dewitt, C.; Richter, M.J.; Lacy, J.H.; Bézard, B.; et al. Astrophysics Seasonal Variations of Hydrogen Peroxide and Water Vapor on Mars: Further Indications of Heterogeneous Chemistry. Astron. Astrophys. 2015, 578, A127. [Google Scholar] [CrossRef]
- Hunten, D.M. Possible Oxidant Sources in the Atmosphere and Surface of Mars. J. Mol. Evol. 1979, 14, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Atreya, S.K.; Wong, A.-S.; Renno, N.O.; Farrell, W.M.; Delory, G.T.; Sentman, D.D.; Cummer, S.A.; Marshall, J.R.; Rafkin, S.C.R.; Catling, D.C. Oxidant Enhancement in Martian Dust Devils and Storms: Implications for Life and Habitability. Astrobiology 2006, 6, 439–450. [Google Scholar] [CrossRef] [PubMed]
- Delory, G.T.; Farrell, W.M.; Atreya, S.K.; Renno, N.O.; Wong, A.-S.; Cummer, S.A.; Sentman, D.D.; Marshall, J.R.; Rafkin, S.C.R.; Catling, D.C. Oxidant Enhancement in Martian Dust Devils and Storms: Storm Electric Fields and Electron Dissociative Attachment. Astrobiology 2006, 6, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Bullock, M.A.; Stoker, C.R.; McKay, C.P.; Zent, A.P. A Coupled Soil-Atmosphere Model of H2O2 on Mars. Icarus 1994, 107, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Hartman, H.; McKay, C.P. Oxygenic Photosynthesis and the Oxidation State of Mars. Planet. Space Sci. 1995, 43, 123–128. [Google Scholar] [CrossRef]
- Zent, A.P. On the Thickness of the Oxidized Layer of the Martian Regolith. J. Geophys. Res. Planets 1998, 103, 31491–31498. [Google Scholar] [CrossRef]
- Huguenin, R.L.; Miller, K.J.; Harwood, W.S. Frost-Weathering on Mars: Experimental Evidence for Peroxide Formation. J. Mol. Evol. 1979, 14, 103–132. [Google Scholar] [CrossRef] [PubMed]
- Hurowitz, J.A.; Tosca, N.J.; McLennan, S.M.; Schoonen, M.A.A. Production of Hydrogen Peroxide in Martian and Lunar Soils. Earth Planet. Sci. Lett. 2007, 255, 41–52. [Google Scholar] [CrossRef]
- Davila, A.F.; Fairén, A.G.; Gago-Duport, L.; Stoker, C.; Amils, R.; Bonaccorsi, R.; Zavaleta, J.; Lim, D.; Schulze-Makuch, D.; McKay, C.P. Subsurface Formation of Oxidants on Mars and Implications for the Preservation of Organic Biosignatures. Earth Planet. Sci. Lett. 2008, 272, 456–463. [Google Scholar] [CrossRef]
- Zent, A.P.; Ichimura, A.S.; Quinn, R.C.; Harding, H.K. The Formation and Stability of the Superoxide Radical (O2−) on Rock-Forming Minerals: Band Gaps, Hydroxylation State, and Implications for Mars Oxidant Chemistry. J. Geophys. Res. Planets 2008, 113, E09001. [Google Scholar] [CrossRef]
- Möhlmann, D.T. Water in the Upper Martian Surface at Mid- and Low-Latitudes: Presence, State, and Consequences. Icarus 2004, 168, 318–323. [Google Scholar] [CrossRef]
- Möhlmann, D.T.F. The Influence of van Der Waals Forces on the State of Water in the Shallow Subsurface of Mars. Icarus 2008, 195, 131–139. [Google Scholar] [CrossRef]
- Benner, S.A.; Devine, K.G.; Matveeva, L.N.; Powell, D.H. The Missing Organic Molecules on Mars. Proc. Natl. Acad. Sci. USA 2000, 97, 2425–2430. [Google Scholar] [CrossRef] [PubMed]
- Mancinelli, R.L. Peroxides and the Survivability of Microorganisms on the Surface of Mars. Adv. Space Res. 1989, 9, 191–195. [Google Scholar] [CrossRef]
- McDonald, G.D.; de Vanssay, E.; Buckley, J.R. Oxidation of Organic Macromolecules by Hydrogen Peroxide: Implications for Stability of Biomarkers on Mars. Icarus 1998, 132, 170–175. [Google Scholar] [CrossRef]
- Foustoukos, D.I.; Stern, J.C. Oxidation Pathways for Formic Acid under Low Temperature Hydrothermal Conditions: Implications for the Chemical and Isotopic Evolution of Organics on Mars. Geochim. Cosmochim. Acta 2012, 76, 14–28. [Google Scholar] [CrossRef]
- Kuhn, W.R.; Atreya, S.K. Solar Radiation Incident on the Martian Surface. J. Mol. Evol. 1979, 14, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Huestis, D.L.; Bougher, S.W.; Fox, J.L.; Galand, M.; Johnson, R.E.; Moses, J.I.; Pickering, J.C. Cross Sections and Reaction Rates for Comparative Planetary Aeronomy. Space Sci. Rev. 2008, 139, 63–105. [Google Scholar] [CrossRef]
- Smith, D.S.; Scalo, J. Solar X-Ray Flare Hazards on the Surface of Mars. Planet. Space Sci. 2007, 55, 517–527. [Google Scholar] [CrossRef]
- Jain, R.; Awasthi, A.K.; Tripathi, S.C.; Bhatt, N.J.; Khan, P.A. Influence of Solar Flare X-Rays on the Habitability on the Mars. Icarus 2012, 220, 889–895. [Google Scholar] [CrossRef]
- Cockell, C.S.; Catling, D.C.; Davis, W.L.; Snook, K.; Kepner, R.L.; Lee, P.; Mckay, C.P. The Ultraviolet Environment of Mars: Biological Implications Past, Present, and Future. Icarus 2000, 146, 343–359. [Google Scholar] [CrossRef] [PubMed]
- Dartnell, L.R.; Desorgher, L.; Ward, J.M.; Coates, A.J. Modelling the Surface and Subsurface Martian Radiation Environment: Implications for Astrobiology. Geophys. Res. Lett. 2007, 34, L02207. [Google Scholar] [CrossRef]
- Dartnell, L.R.; Desorgher, L.; Ward, J.M.; Coates, A.J. Martian Sub-Surface Ionising Radiation: Biosignatures and Geology. Biogeosci. Discuss. 2007, 4, 455–492. [Google Scholar] [CrossRef]
- Matthiä, D.; Hassler, D.M.; de Wet, W.; Ehresmann, B.; Firan, A.; Flores-McLaughlin, J.; Guo, J.; Heilbronn, L.H.; Lee, K.; Ratliff, H.; et al. The Radiation Environment on the Surface of Mars—Summary of Model Calculations and Comparison to RAD Data. Life Sci. Space Res. 2017, 14, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Schuerger, A.C.; Golden, D.C.; Ming, D.W. Biotoxicity of Mars Soils: 1. Dry Deposition of Analog Soils on Microbial Colonies and Survival under Martian Conditions. Planet. Space Sci. 2012, 72, 91–101. [Google Scholar] [CrossRef]
- Fornaro, T.; Brucato, J.R.; Pace, E.; Cestelli-Guidi, M.; Branciamore, S.; Pucci, A. Infrared Spectral Investigations of UV Irradiated Nucleobases Adsorbed on Mineral Surfaces. Icarus 2013, 226, 1068–1085. [Google Scholar] [CrossRef]
- Gerakines, P.A.; Hudson, R.L. Glycine’s Radiolytic Destruction in Ices: First in Situ Laboratory Measurements for Mars. Astrobiology 2013, 13, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Kminek, G.; Bada, J.L. The Effect of Ionizing Radiation on the Preservation of Amino Acids on Mars. Earth Planet. Sci. Lett. 2006, 245, 1–5. [Google Scholar] [CrossRef]
- Poch, O.; Kaci, S.; Stalport, F.; Szopa, C.; Coll, P. Laboratory Insights into the Chemical and Kinetic Evolution of Several Organic Molecules under Simulated Mars Surface UV Radiation Conditions. Icarus 2014, 242, 50–63. [Google Scholar] [CrossRef]
- Poch, O.; Jaber, M.; Stalport, F.; Nowak, S.; Georgelin, T.; Lambert, J.-F.; Szopa, C.; Coll, P. Effect of Nontronite Smectite Clay on the Chemical Evolution of Several Organic Molecules under Simulated Martian Surface Ultraviolet Radiation Conditions. Astrobiology 2015, 15, 221–237. [Google Scholar] [CrossRef] [PubMed]
- Córdoba-Jabonero, C.; Zorzano, M.-P.; Selsis, F.; Patel, M.R.; Cockell, C.S. Radiative Habitable Zones in Martian Polar Environments. Icarus 2005, 175, 360–371. [Google Scholar] [CrossRef] [PubMed]
- France, J.L.; King, M.D.; MacArthur, A. A Photohabitable Zone in the Martian Snowpack? A Laboratory and Radiative-Transfer Study of Dusty Water-ice Snow. Icarus 2010, 207, 133–139. [Google Scholar] [CrossRef]
- ten Kate, I.L.; Garry, J.R.C.; Peeters, Z.; Quinn, R.; Foing, B.; Ehrenfreund, P. Amino Acid Photostability on the Martian Surface. Meteorit. Planet. Sci. 2005, 40, 1185–1193. [Google Scholar] [CrossRef]
- Stoker, C.R.; Bullock, M.A. Organic Degradation under Simulated Martian Conditions. J. Geophys. Res. Planets 1997, 102, 10881–10888. [Google Scholar] [CrossRef]
- Ten Kate, I.L. Organics on Mars? Astrobiology 2010, 10, 589–603. [Google Scholar] [CrossRef] [PubMed]
- Oró, J.; Holzer, G. The Photolytic Degradation and Oxidation of Organic Compounds under Simulated Martian Conditions. J. Mol. Evol. 1979, 14, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Scappini, F.; Casadei, F.; Zamboni, R.; Franchi, M.; Gallori, E.; Monti, S. Protective Effect of Clay Minerals on Adsorbed Nucleic Acid against UV Radiation: Possible Role in the Origin of Life. Int. J. Astrobiol. 2004, 3, 17–19. [Google Scholar] [CrossRef]
- Ciaravella, A.; Scappini, F.; Franchi, M.; Cecchi-Pestellini, C.; Barbera, M.; Candia, R.; Gallori, E.; Micela, G. Role of Clays in Protecting Adsorbed DNA against X-Ray Radiation. Int. J. Astrobiol. 2004, 3, 31–35. [Google Scholar] [CrossRef]
- Garry, J.R.C.; Ten Kate, I.L.; Martins, Z.; Nørnberg, P.; Ehrenfreund, P. Analysis and Survival of Amino Acids in Martian Regolith Analogs. Meteorit. Planet. Sci. 2006, 405, 391–405. [Google Scholar] [CrossRef]
- Biondi, E.; Branciamore, S.; Maurel, M.-C.; Gallori, E. Montmorillonite Protection of an UV-Irradiated Hairpin Ribozyme: Evolution of the RNA World in a Mineral Environment. BMC Evol. Biol. 2007, 7, S2. [Google Scholar] [CrossRef] [PubMed]
- Shkrob, I.A.; Chemerisov, S.D. Light Induced Fragmentation of Polyfunctional Carboxylated Compounds on Hydrated Metal Oxide Particles: From Simple Organic Acids to Peptides. J. Phys. Chem. C 2009, 113, 17138–17150. [Google Scholar] [CrossRef]
- Shkrob, I.A.; Chemerisov, S.D.; Marin, T.W. Photocatalytic Decomposition of Carboxylated Molecules on Light-Exposed Martian Regolith and Its Relation to Methane Production on Mars. Astrobiology 2010, 10, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Stalport, F.; Guan, Y.Y.; Coll, P.; Szopa, C.; Macari, F.; Raulin, F.; Chaput, D.; Cottin, H. UVolution, a Photochemistry Experiment in Low Earth Orbit: Investigation of the Photostability of Carboxylic Acids Exposed to Mars Surface UV Radiation Conditions. Astrobiology 2010, 10, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.P.; Pratt, L.M. Metal-Catalyzed Degradation and Racemization of Amino Acids in Iron Sulfate Brines under Simulated Martian Surface Conditions. Icarus 2010, 207, 124–132. [Google Scholar] [CrossRef]
- Johnson, A.P.; Pratt, L.M.; Vishnivetskaya, T.; Pfiffner, S.; Bryan, R.A.; Dadachova, E.; Whyte, L.; Radtke, K.; Chan, E.; Tronick, S.; et al. Extended Survival of Several Organisms and Amino Acids under Simulated Martian Surface Conditions. Icarus 2011, 211, 1162–1178. [Google Scholar] [CrossRef]
- Shkrob, I.A.; Marin, T.M.; Adhikary, A.; Sevilla, M.D. Photooxidation of Nucleic Acids on Metal Oxides: Physicochemical and Astrobiological Perspectives. J. Phys. Chem. C 2011, 115, 3393–3403. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, R.; Patel, M.; Cuadros, J.; Martins, Z. Influence of Mineralogy on the Preservation of Amino Acids under Simulated Mars Conditions. Icarus 2016, 277, 342–353. [Google Scholar] [CrossRef]
- Ertem, G.; Ertem, M.C.; McKay, C.P.; Hazen, R.M. Shielding Biomolecules from Effects of Radiation by Mars Analogue Minerals and Soils. Int. J. Astrobiol. 2017, 16, 280–285. [Google Scholar] [CrossRef]
- Ten Kate, I.L.; Garry, J.R.C.; Peeters, Z.; Foing, B.; Ehrenfreund, P. The Effects of Martian near Surface Conditions on the Photochemistry of Amino Acids. Planet. Space Sci. 2006, 54, 296–302. [Google Scholar] [CrossRef]
- Stalport, F.; Coll, P.; Szopa, C.; Raulin, F. Search for Organic Molecules at the Mars Surface: The “Martian Organic Material Irradiation and Evolution” (MOMIE) Project. Adv. Space Res. 2008, 42, 2014–2018. [Google Scholar] [CrossRef]
- Poch, O.; Noblet, A.; Stalport, F.; Correia, J.J.; Grand, N.; Szopa, C.; Coll, P. Chemical Evolution of Organic Molecules under Mars-like UV Radiation Conditions Simulated in the Laboratory with the “Mars Organic Molecule Irradiation and Evolution” (MOMIE) Setup. Planet. Space Sci. 2013, 85, 188–197. [Google Scholar] [CrossRef]
- Onoe, J.; Kawai, T.; Kawai, S. Peptide Formation from Amino Acid with Particulate Semiconductor Photocatalysts. Chem. Lett. 1985, 14, 1667–1670. [Google Scholar] [CrossRef]
- Saladino, R.; Brucato, J.R.; De Sio, A.; Botta, G.; Pace, E.; Gambicorti, L. Photochemical Synthesis of Citric Acid Cycle Intermediates Based on Titanium Dioxide. Astrobiology 2011, 11, 815–824. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xu, X.; Li, Y.; Ding, C.; Wu, J.; Lu, A.; Ding, H.; Qin, S.; Wang, C. Absolute Band Structure Determination on Naturally Occurring Rutile with Complex Chemistry: Implications for Mineral Photocatalysis on Both Earth and Mars. Appl. Surf. Sci. 2018, 439, 660–671. [Google Scholar] [CrossRef]
- Ehrenfreund, P.; Bernstein, M.P.; Dworkin, J.P.; Sandford, S.A.; Allamandola, L.J. The Photostability of Amino Acids in Space. Astrophys. J. 2001, 550, L95–L99. [Google Scholar] [CrossRef]
- McBride, M.; Kung, K.-H. Complexation of Glyphosate and Related Ligands with Iron (III). Soil Sci. Soc. Am. J. 1989, 53, 1668. [Google Scholar] [CrossRef]
- Yusenko, K.; Fox, S.; Guni, P.; Strasdeit, H. Model Studies on the Formation and Reactions of Solid Glycine Complexes at the Coasts of a Primordial Salty Ocean. Z. Anorgan. Allg. Chem. 2008, 634, 2347–2354. [Google Scholar] [CrossRef]
- Bada, J.L. Racemization of Amino Acids in Nature. Interdiscip. Sci. Rev. 1982, 7, 30–46. [Google Scholar] [CrossRef]
- Kavitha, V.; Palanivelu, K. Degradation of 2-Chlorophenol by Fenton and Photo-Fenton Processes—A Comparative Study. J. Environ. Sci. Health Part A 2003, 38, 1215–1231. [Google Scholar] [CrossRef]
- Zuo, Y.; Hoigne, J. Formation of Hydrogen Peroxide and Depletion of Oxalic Acid in Atmospheric Water by Photolysis of Iron (III)-Oxalato Complexes. Environ. Sci. Technol. 1992, 26, 1014–1022. [Google Scholar] [CrossRef]
- Gu, B.; Schmitt, J.; Chen, Z.; Liang, L.; McCarthy, J.F. Adsorption and Desorption of Natural Organic Matter on Iron Oxide: Mechanisms and Models. Environ. Sci. Technol. 1994, 28, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Williams, K.M.; Smith, G.G. A Critical Evaluation of the Application of Amino Acid Racemization to Geochronology and Geothermometry. Orig. Life 1977, 8, 91–144. [Google Scholar] [CrossRef] [PubMed]
- Möhlmann, D. Adsorption Water-Related Potential Chemical and Biological Processes in the Upper Martian Surface. Astrobiology 2005, 5, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Franchi, M.; Gallori, E. Origin, Persistence and Biological Activity of Genetic Material in Prebiotic Habitats. Orig. Life Evol. Biosph. 2004, 34, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Otroshchenko, V.A.; Vasilyeva, N.V. Formation of RNA Oligonucleotides over the Mineral Surface Preliminary Irradiated with UV Light. React. Kinet. Catal. Lett. 2009, 97, 151–156. [Google Scholar] [CrossRef]
- Horneck, G.; Reitz, G.; Rettberg, P.; Schuber, M.; Kochan, H.; Möhlmann, D.; Richter, L.; Seidlitz, H. A Ground-Based Program for Exobiological Experiments on the International Space Station. Planet. Space Sci. 2000, 48, 507–513. [Google Scholar] [CrossRef]
- Schuerger, A.C.; Mancinelli, R.L.; Kern, R.G.; Rothschild, L.J.; McKay, C.P. Survival of Endospores of Bacillus Subtilis on Spacecraft Surfaces under Simulated Martian Environments: Implications for the Forward Contamination of Mars. Icarus 2003, 165, 253–276. [Google Scholar] [CrossRef]
- Schuerger, A.C.; Richards, J.T.; Newcombe, D.A.; Venkateswaran, K. Rapid Inactivation of Seven Bacillus Spp. under Simulated Mars UV Irradiation. Icarus 2006, 181, 52–62. [Google Scholar] [CrossRef]
- Nir, E.; Kleinermanns, K.; Grace, L.; de Vries, M.S. On the Photochemistry of Purine Nucleobases. J. Phys. Chem. A 2001, 105, 5106–5110. [Google Scholar] [CrossRef]
- Ravanat, J.-L.; Douki, T.; Cadet, J. Direct and Indirect Effects of UV Radiation on DNA and Its Components. J. Photochem. Photobiol. B Biol. 2001, 63, 88–102. [Google Scholar] [CrossRef]
- Barbatti, M.; Aquino, A.J.A.; Szymczak, J.J.; Nachtigallová, D.; Hobza, P.; Lischka, H. Relaxation Mechanisms of UV-Photoexcited DNA and RNA Nucleobases. Proc. Natl. Acad. Sci. USA 2010, 107, 21453–21458. [Google Scholar] [CrossRef] [PubMed]
- Pavlov, A.K.; Blinov, A.V.; Konstantinov, A.N. Sterilization of Martian Surface by Cosmic Radiation. Planet. Space Sci. 2002, 50, 669–673. [Google Scholar] [CrossRef]
- Clark, B.C. Chemical and Physical Microenvironments at the Viking Landing Sites. J. Mol. Evol. 1979, 14, 13–31. [Google Scholar] [CrossRef] [PubMed]
- Gerakines, P.A.; Hudson, R.L.; Moore, M.H.; Bell, J.-L. In Situ Measurements of the Radiation Stability of Amino Acids at 15–140 K. Icarus 2012, 220, 647–659. [Google Scholar] [CrossRef]
- López-Esquivel Kranksith, L.; Negrón-Mendoza, A.; Mosqueira, F.G.; Ramos-Bernal, S. Radiation-Induced Reactions of Amino Acids Adsorbed on Solid Surfaces. Nucl. Instrum. Methods Phys. Res. Sect. A 2010, 619, 51–54. [Google Scholar] [CrossRef]
- Guzmán, A.; Negrón-Mendoza, A.; Ramos-Bernal, S. Behavior of Adenine in Na-Montmorillonite Exposed to Gamma Radiation: Implications to Chemical Evolution Studies. Cell. Mol. Biol. 2002, 48, 525–528. [Google Scholar] [PubMed]
- Góbi, S.; Förstel, M.; Maksyutenko, P.; Kaiser, R.I. A Reflectron Time-of-Flight Mass Spectrometric Study on the Degradation Pathways of Glycine on Mars in the Presence of Perchlorates and Ionizing Radiation. Astrophys. J. 2017, 835, 241. [Google Scholar] [CrossRef]
- Schoonen, M.; Smirnov, A.; Cohn, C. A Perspective on the Role of Minerals in Prebiotic Synthesis. AMBIO J. Hum. Environ. 2004, 33, 539–551. [Google Scholar] [CrossRef]
- Gallori, E.; Biondi, E.; Franchi, M. Mineral Surfaces as a Cradle of Primordial Genetic Material. In Life in the Universe; Seckbach, J., Chela-Flores, J., Owen, T., Raulin, F., Eds.; Cellular Origin and Life in Extreme Habitats and Astrobiology; Springer: Dordrecht, The Netherlands, 2004; Volume 7, pp. 145–148. [Google Scholar]
- Keppler, F.; Vigano, I.; McLeod, A.; Ott, U.; Früchtl, M.; Röckmann, T. Ultraviolet-Radiation-Induced Methane Emissions from Meteorites and the Martian Atmosphere. Nature 2012, 486, 93–96. [Google Scholar] [CrossRef] [PubMed]
- Okabe, H. Photochemistry of Small Molecules; Wiley: New York, NY, USA, 1978; Volume 431. [Google Scholar]
- Shemansky, D.E. CO2 Extinction Coefficient 1700–3000 Å. J. Chem. Phys. 1972, 56, 1582–1587. [Google Scholar] [CrossRef]
- Karaiskou, A.; Vallance, C.; Papadakis, V.; Vardavas, I.M.; Rakitzis, T.P. Absolute Absorption Cross-Section Measurements of CO2 in the Ultraviolet from 200 to 206 Nm at 295 and 373 K. Chem. Phys. Lett. 2004, 400, 30–34. [Google Scholar] [CrossRef]
- Vesela, A.; Wilhelm, J. The Role of Carbon Dioxide in Free Radical Reactions in Organism. Physiol. Res. 2002, 51, 335–340. [Google Scholar] [PubMed]
- Ehlmann, B.L.; Mustard, J.F. An In-Situ Record of Major Environmental Transitions on Early Mars at Northeast Syrtis Major. Geophys. Res. Lett. 2012, 39, L11202. [Google Scholar] [CrossRef]
- Goudge, T.A.; Mustard, J.F.; Head, J.W.; Fassett, C.I.; Wiseman, S.M. Assessing the Mineralogy of the Watershed and Fan Deposits of the Jezero Crater Paleolake System, Mars. J. Geophys. Res. Planets 2015, 120, 775–808. [Google Scholar] [CrossRef]
- Salvatore, M.R.; Goudge, T.A.; Bramble, M.S.; Edwards, C.S.; Bandfield, J.L.; Amador, E.S.; Mustard, J.F.; Christensen, P.R. Bulk Mineralogy of the NE Syrtis and Jezero Crater Regions of Mars Derived through Thermal Infrared Spectral Analyses. Icarus 2018, 301, 76–96. [Google Scholar] [CrossRef]
- Bridges, J.C.; Loizeau, D.; Sefton-Nash, E.; Vago, J.; Williams, R.M.E.; Balme, M.; Turner, S.M.R.; Fawdon, P.; Davis, J.M. Selection and Characterization of the ExoMars 2020 Rover Landing Sites. In Proceedings of the 48th Lunar and Planetary Science Conference, Woodlands, TX, USA, 20–24 March 2017. [Google Scholar]
- Amiri, S.; McGrath, M.; Al Awadhi, M.; Almatroushi, H.; Sharaf, O.; AlDhafri, S.; AlRais, A.; Wali, M.; AlShamsi, Z.; AlQasim, I.; et al. Emirates Mars Mission (EMM) 2020 Overview. In Proceedings of the 42nd COSPAR Scientific Assembly, Pasadena, CA, USA, 14–22 July 2018. [Google Scholar]
- Jiang, X.; Yang, B.; Li, S. Overview of China’s 2020 Mars Mission Design and Navigation. Astrodynamics 2018, 2, 1–11. [Google Scholar] [CrossRef]
- Haider, S.A.; Bhardwaj, A.; Shanmugam, M.; Goyal, S.K.; Sheel, V.; Pabari, J.; Prasad Karanam, D. Indian Mars and Venus Missions: Science and Exploration. In Proceedings of the 42nd COSPAR Scientific Assembly, Pasadena, CA, USA, 14–22 July 2018. [Google Scholar]
- Campagnola, S.; Yam, C.H.; Tsuda, Y.; Ogawa, N.; Kawakatsu, Y. Mission Analysis for the Martian Moons Explorer (MMX) Mission. Acta Astronaut. 2018, 146, 409–417. [Google Scholar] [CrossRef]
| Minerals | Region of Discovery on Mars | Mars Mission |
|---|---|---|
| Phyllosilicates, such as clay minerals (aluminium-rich dioctahedral and magnesium-rich trioctahedral smectites, kaolinite, illite, pyrophyllite-talc), micas (muscovite), and chamosite chlorite. | Echus Chasma, Mawrth Vallis, Eridania basin, Memnonia quadrangle, Elysium quadrangle, Nili Fossae, Argyre Planitia, Gale crater, and Marathon Valley. | ESA Mars Express, NASA Mars Exploration Rovers, NASA Mars Reconnaissance Orbiter, and NASA Mars Science Laboratory [1,4,71,72,73,74,75,76,77,78,79,80]. |
| Evaporites (chloride salts). | Southern highlands within Noachian-aged terrains. | NASA 2001 Mars Odyssey [87,88,89]. |
| Sulfates (magnesium sulfates, like kieserite, and calcium sulfates, like gypsum). | Gale crater, Endeavor crater, the North and South Poles of Mars. | ESA Mars Express and NASA Mars Science Laboratory [69,90,91]. |
| Opaline silica. | Valles Marineris, Gusev crater, and Gale crater. | NASA Mars Reconnaissance Orbiter and NASA Mars Science Laboratory [67,89,90]. |
| Ferric oxides, like hematite and goethite. | Chryse Planitia, Utopia Planitia, Ares Vallis, Gusev Crater, Meridiani Planum, Terra Meridiani, Valles Marineris, Aureum Chaos, Columbia Hills, Gale crater. | NASA Viking Landers, NASA Mars Global Surveyor, NASA Mars Pathfinder, NASA 2001 Mars Odyssey, and NASA Mars Exploration Rovers [66,92,93,101]. |
| Mafic minerals, such as pyroxene and olivine. | Mainly in older terrains, included within sand dunes, associated to ancient Noachian crustal rocks and early Hesperian volcanism in the southern hemisphere. | ESA Mars Express [105,106]. |
| Amorphous material. | Gale crater. | NASA Mars Science Laboratory [86,108]. |
| Reference | Sample/Preparation Method | Irradiation Source/Spectral Range | Temperature | Pressure/Atmospheric Composition | Oxidants | In Situ/Ex Situ Analysis | Analytical Techniques |
|---|---|---|---|---|---|---|---|
| Oro & Holzer 1979 [178] | Adenine, glycine, and naphthalene impregnated on powdered quartz at various concentration from 0.01% to 0.2%/Murchison meteorite | Mercury-Xenon lamp/200–300 nm | −10 to 25 °C | 1 mbar N2/various O2-content | None | Ex situ | Ion exchange chromatography for glycine, UV-vis spectrophotometry for adenine, Gas chromatography for naphthalene and Murchison |
| Stoker & Bullock 1997 [177] | Glycine powder mixed with palagonite at 1% concentration | Xenon lamp/210–710 nm | Room temperature | 100 mbar, 95.59% CO2, 4.21% Ar, 0.11% O2, 0.09% CO | None | In situ | Gas chromatography |
| Scappini et al. 2004 [179] | Aqueous suspension of DNA and montmorillonite and kaolinite (20 μg DNA and 2 mg clay in 2 mL water) | Nd:YAG pulsed laser/266 nm | Room temperature | Terrestrial ambient conditions | None | Ex situ | Biological transformation |
| Ciaravella et al. 2004 [180] | Aqueous suspension of DNA and montmorillonite and kaolinite (10 μg DNA and 2 mg clay in 1.4 mL water) | Electron impact X-ray source/Monochromatic X-rays of 1.49, 4.51, and 8.04 keV | Room temperature | Vacuum | None | Biological transformation | |
| Garry et al. 2006 [181] | JSC Mars-1 and Salten Skov Martian soil analogs containing native amino acids | Deuterium lamp/190–325 nm | Room temperature (Experiment I and II)/−63 °C (Experiment III) | 1 × 10−5 mbar (Experiment I and II)/7 mbar CO2 (Experiment III) | None | Ex situ | HPLC |
| Biondi et al. 2007 [182] | Aqueous suspension of RNA and montmorillonite (2.25 × 10−10 moles RNA and 1.3 mg montmorillonite in 75 μL water) | Atlas Germicidal Lamp (15 W)/254 nm | Room temperature | Terrestrial ambient conditions | None | Ex situ | Analysis of self-cleavage activity |
| Shkrob & Chemerisov 2009 [183] | Aqueous suspensions of carboxylic, hydroxycarboxylic, and aminocarboxylic acids, carboxylated aromatics, amino acids, and peptides with anatase, goethite, and hematite | Nd:YAG pulsed laser/355 nm | −196 to −73 °C/22 °C | 1 bar, N2 | None | Ex situ | EPR/transient absorption spectroscopy |
| Shkrob et al. 2010 [184] | Aqueous suspensions of carboxylic, hydroxycarboxylic, and aminocarboxylic acids, carboxylated aromatics, amino acids, and peptides with anatase, goethite, and hematite | Nd:YAG pulsed laser/355 nm | −196 °C | 1 bar, N2 | None | Ex situ | EPR |
| Stalport et al. 2010 [185] | Carboxylic acids α-aminoisobutyric acid (AIB), mellitic acid, phthalic acid, and trimesic acid directly deposited on quartz windows or underneath a layer of JSC Mars-1 | Solar radiation >200 nm | Temperature at low Earth orbit | Pressure at low Earth orbit | None | Ex situ | IR spectroscopy |
| Johnson & Pratt 2010 [186] | Amino acids glycine, L-alanine, L-valine, L-glutamic acid, and L-aspartic acid in metal-rich sulfate brines (1 mM amino acid concentration) | Xenon lamp/250–700 nm | −40 to 20 °C | 7 to 15 mbar, 95.3% CO2, 2.7% N2, 1.6% Ar, and 0.13% O2 | None | Ex situ | XRD, HPLC |
| Johnson & Pratt 2011 [187] | Amino acids L-Alanine, L-valine, L-aspartic acid, L-glutamic acid, and glycine inoculated into I-MAR Martian regolith simulant at 0.01% concentration | Xenon lamp/210–900 nm | −40.4 to 24 °C (on average −17.6 °C) | 10−22 mbar (on average 13.3 mbar), 48.6% CO2, 50% Ar, 1.4% N2, 0.07% O2, 0.04% CO, 0.02% H2O, 0.01% H2 | None | Ex situ | HPLC |
| Shkrob et al. 2011 [188] | Aqueous suspensions of nucleic acid components with anatase, goethite, and hematite | Nd:YAG pulsed laser/355 nm | −196 °C | 1 bar, N2 | None | Ex situ | EPR |
| Fornaro et al. 2013 [168] | Nucleobases adenine, uracil, cytosine, and hypoxanthine adsorbed on magnesium oxide and forsterite at concentrations in the range 0.1–10% | Mercury-Xenon lamp/185–2000 nm | 25 °C | Vacuum (~10−2–10−3 mbar) | None | In situ | Diffuse Reflectance Fourier Transform Infrared (DRIFT) spectroscopy |
| Poch et al. 2015 [172] | Glycine, urea, and adenine co-deposited with nontronite with high molecule-mineral mass ratio (from 1.0 to 3.6) | Xenon lamp/190–400 nm | −55 °C | 6 ± 1 mbar, N2 | None | In situ | IR spectroscopy |
| dos Santos et al. 2016 [189] | 25 amino acids spiked onto augite, enstatite, goethite, gypsum, hematite, jarosite, labradorite, montmorillonite, nontronite, olivine, saponite, and a basaltic lava, at various concentration (approx. 0.001% to 0.1%) | Xenon lamp/200–400 nm | −80 to 20 °C | 6 mbar, 95% CO2, 5% N2 | None | Ex situ | GC-MS |
| Ertem et al. 2017 [190] | Purine, pyrimidine, and uracil impregnated on ferric oxide, calcite, calcium sulphate, kaolinite, and clay-bearing Atacama desert soil at 0.0025% concentration | Xenon lamp/200–400 nm (Experiment I); Gamma Cell 40 from a 137Cs source/Gamma rays 3 Gy (Experiment II) | −196 to 25 °C (Experiment I); 25 °C (Experiment II) | 15–25 mbar/Ambient pressure, 95.3% CO2, 2.7% N2, 0.13 O2 (Experiment I); Ambient pressure, 95.3% CO2, 2.7% N2, 0.13 O2 (Experiment II) | 0.6% NaClO4 | Ex situ | HPLC |
| Fornaro et al. 2018 [67] | AMP and UMP adsorbed on lizardite, antigorite, labradorite, natrolite, hematite, apatite, and forsterite at 5% concentration | Xenon lamp/200–930 nm (Experiment I); Xenon lamp/180–900 nm (Experiment II) | 25 °C (Experiment I); −20 °C (Experiment II) | Terrestrial ambient conditions (Experiment I); 6 mbar CO2 (Experiment II) | None | In situ (Experiment I); Ex situ (Experiment II) | Diffuse Reflectance Fourier Transform Infrared (DRIFT) spectroscopy |
| Mineral | Biomarker | Radiation | Protection/Preservation | Catalysis/Degradation | Supposed Mechanism |
|---|---|---|---|---|---|
| Quartz | Adenine | 200–300 nm | Under N2 | Under O2 | Photo-oxidation by O2 (Oro & Holzer 1979). |
| Quartz | Glycine | 200–300 nm | Under N2 | Under O2 | Photo-oxidation by O2 (Oro & Holzer 1979). |
| Quartz | Naphthalene | 200–300 nm | x | Under O2/N2 | Photo-oxidation by O2 (Oro & Holzer 1979). |
| Murchison meteorite | Indigenous organics | 200–300 nm | x | Under O2/N2 | Photo-oxidation by O2 (Oro & Holzer 1979). |
| Palagonite | Glycine | 210–710 nm | x | Under Martian-like atmosphere | Photolysis into CH4, C2H6, C2H4 (Stoker & Bullock 1997). |
| JSC Mars-1 and Salten Skov Martian soil analogs | Indigenous amino acids | 190–325 nm | x | Under Martian-like atmosphere | Decomposition induced by radicals produced by photolysis of water condensed onto minerals (Garry et al. 2006). |
| JSC Mars-1 Martian soil analog | Carboxylic acids α-aminoisobutyric acid (AIB), mellitic acid, phthalic acid, and trimesic acid | Solar radiation > 200 nm | x | Under low Earth orbit conditions | Decomposition induced by radicals/oxidants produced by TiO2–photocatalysis (Stalport et al. 2010). |
| I-MAR Martian regolith simulant | Amino acids L-Alanine, L-valine, L-aspartic acid, L-glutamic acid, and glycine | 210–900 nm | x | Under Martian-like atmosphere | Photolytic oxidation up to UV penetration depth, then decomposition induced by radicals formed from condensed atmospheric water vapor diffused into the regolith (Johnson & Pratt 2011). |
| Aqueous suspensions of anatase, goethite, and hematite | Carboxylic, hydroxycarboxylic, and aminocarboxylic acids, carboxylated aromatics, amino acids and peptides | 355 nm | x | Under N2 | Decarboxylation initiated by charge transfer from the metal oxide to the adsorbate. Specifically, anatase, goethite, and hematite feature a similar photocatalytic activity for aromatic, carboxylic, and hydroxycarboxylic acids, while for α-amino acids and peptides hematite has reduced activity (Shkrob et al. 2010). |
| Aqueous suspensions of anatase, goethite, and hematite | Nucleic acid components | 355 nm | Only for double-stranded oligoribonucleotides and DNA | Under N2 | Oxidation of purine nucleotides leads to formation of purine radical cations and sugar-phosphate radicals. In the case of pyrimidine nucleotides other than thymine only the sugar-phosphate moiety undergoes oxidation, while deprotonation from the methyl group of the base occurs for thymine derivatives. Single-stranded (ss) oligoribonucleotides and wild-type ss RNA are oxidized at purine sites, while double-stranded (ds) oligoribonucleotides and DNA show high stability against oxidation (Shkrob et al. 2011). |
| Aqueous suspensions of montmorillonite and kaolinite | DNA | 266 nm | Under terrestrial ambient conditions | x | Photoprotection due to specific molecule-mineral interactions; specifically, a change in DNA configuration from B to A when adsorbed on the mineral surface, which is more compact and its binding to the surface sites may take place through electrostatic and/or hydrogen bonds likely stabilizing the molecule (Scappini et al. 2004). |
| Aqueous suspensions of montmorillonite | RNA molecule ADHR1 | 254 nm | Under terrestrial ambient conditions | x | Photoprotection due to specific molecule-mineral interactions (Biondi et al. 2007). |
| Nontronite | Glycine and adenine | 190–400 nm | Under N2 | x | Photoprotection is not only due to mechanical shielding, but also stabilizing molecule-mineral interactions, such as electrostatic interactions of the molecules in the interlayers and/ or on the edges of nontronite allowing a more efficient energy dissipation and/or easier recombination for the fragments of the photo-dissociated molecules (Poch et al. 2015). |
| Nontronite | Urea | 190–400 nm | x | Under N2 | Catalysis in urea photo-oxidation and decomposition, maybe due to chelation with Fe3+ ions (Poch et al. 2015). |
| Smectites montmorillonite, nontronite and saponite | 25 Amino acids | 200–400 nm | Under Martian-like conditions | x | Photoprotection by mechanical shielding effect (dos Santos et al. 2016). |
| Sulfates gypsum and jarosite | 25 Amino acids | 200–400 nm | Under Martian-like conditions | x | Photoprotection due to low UV absorbance of sulfates or entrapment of amino acids upon recrystallization of partially dissolved sulfate (dos Santos et al. 2016). |
| Augite, enstatite, hematite and basaltic lava | 25 Amino acids | 200–400 nm | x | Under Martian-like conditions | Photocatalytic activity due to iron(II) reactions (dos Santos et al. 2016). |
| Calcite, calcium sulphate, kaolinite, clay-bearing Atacama desert soil + 0.6% NaClO4 | Purine, pyrimidine and uracil | 200–400 nm | Under Martian-like conditions | x | Photoprotection mechanism not specified (Ertem et al. 2017). |
| Ferric oxide + 0.6% NaClO4 | Purine, pyrimidine and uracil | 200–400 nm | x | Under Martian-like conditions | Complete decomposition before UV irradiation (Ertem et al. 2017). |
| Magnesium oxide and forsterite | Adenine, uracil, cytosine, and hypoxanthine | 185–2000 nm | x | Under vacuum | Catalysis likely due to a proximity effect (Fornaro et al. 2013). |
| Lizardite, antigorite and apatite | AMP and UMP | 200–930 nm | Under Martian-like conditions | x | Various photoprotection mechanisms: mechanical shielding/stabilizing molecule-mineral interactions for lizardite and antigorite, photo-luminescence for apatite (Fornaro et al. 2018). |
| Labradorite, natrolite, hematite, forsterite | AMP and UMP | 200–930 nm | x | Under Martian-like conditions | Remarkable catalytic activity of labradorite and natrolite, likely due to photo-ionization phenomena that may occur inside the mineral matrix promoting redox processes. For hematite and forsterite the catalytic activity is not so high, maybe due to the opacity of iron to UV radiation (Fornaro et al. 2018). |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fornaro, T.; Steele, A.; Brucato, J.R. Catalytic/Protective Properties of Martian Minerals and Implications for Possible Origin of Life on Mars. Life 2018, 8, 56. https://doi.org/10.3390/life8040056
Fornaro T, Steele A, Brucato JR. Catalytic/Protective Properties of Martian Minerals and Implications for Possible Origin of Life on Mars. Life. 2018; 8(4):56. https://doi.org/10.3390/life8040056
Chicago/Turabian StyleFornaro, Teresa, Andrew Steele, and John Robert Brucato. 2018. "Catalytic/Protective Properties of Martian Minerals and Implications for Possible Origin of Life on Mars" Life 8, no. 4: 56. https://doi.org/10.3390/life8040056
APA StyleFornaro, T., Steele, A., & Brucato, J. R. (2018). Catalytic/Protective Properties of Martian Minerals and Implications for Possible Origin of Life on Mars. Life, 8(4), 56. https://doi.org/10.3390/life8040056





