Silicate-Promoted Phosphorylation of Glycerol in Non-Aqueous Solvents: A Prebiotically Plausible Route to Organophosphates
Abstract
:1. Introduction
2. Materials and Methods
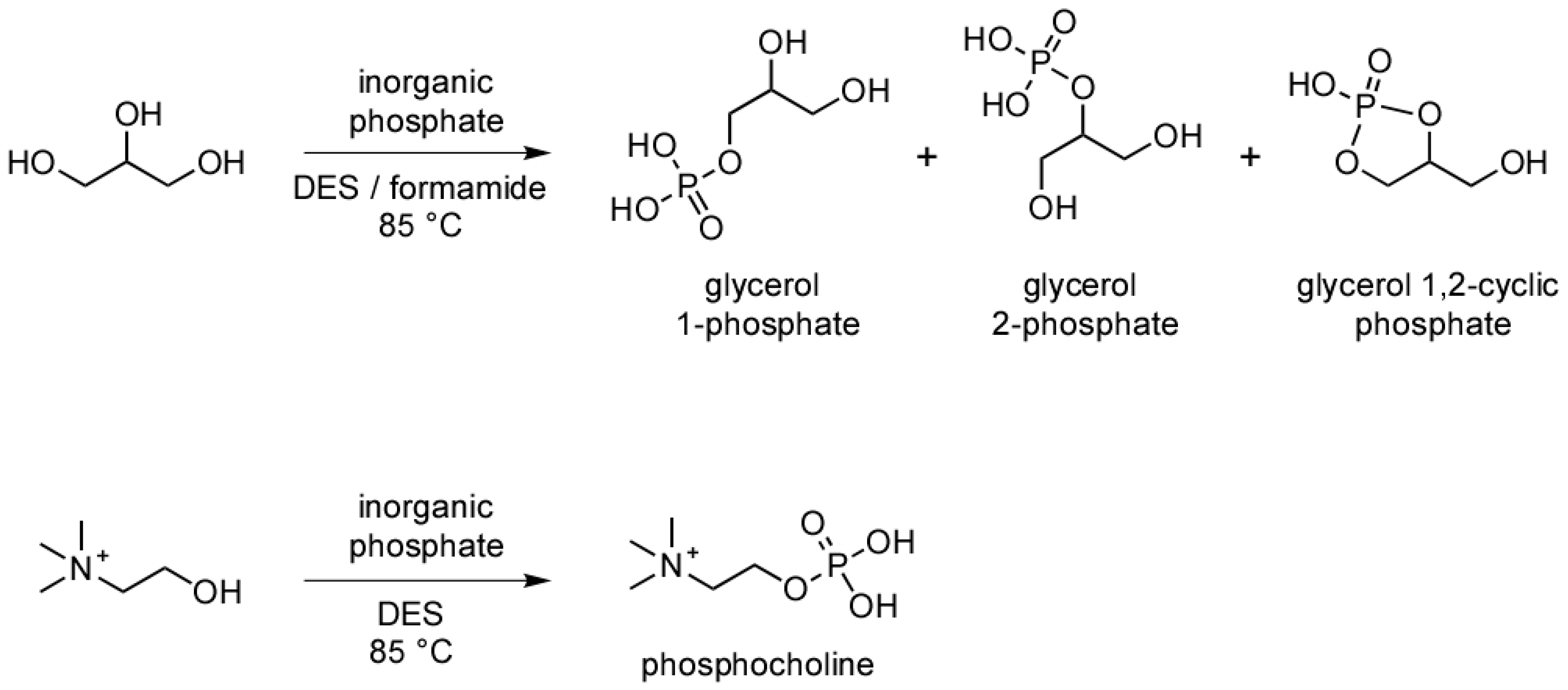
2.1. Phosphorylation of Glycerol in Formamide and a Deep Eutectic Solvent (DES)
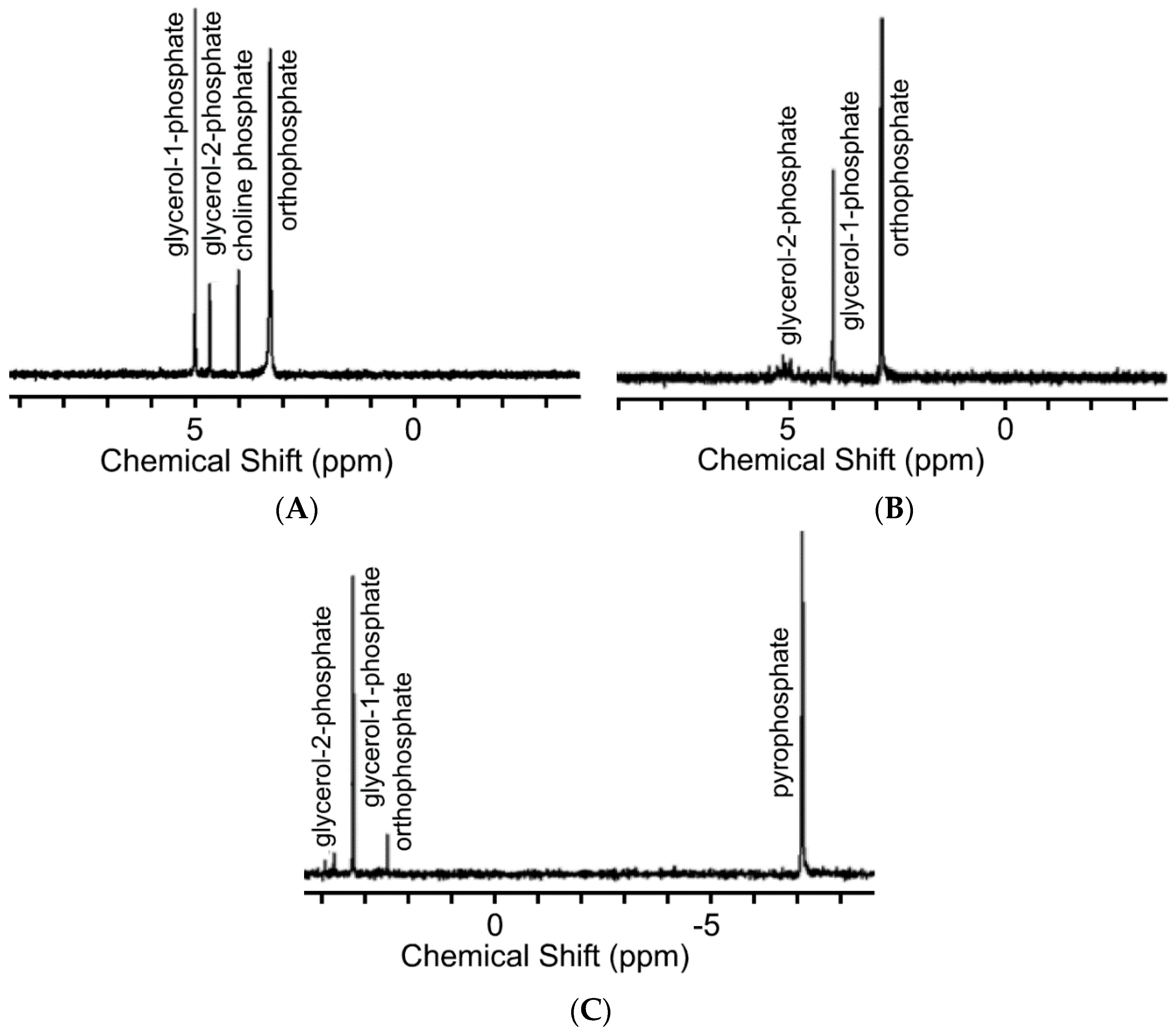
2.2. 31P-NMR and Mass Spectrometry (MS) Studies of the Phosphorylation Products
3. Results and Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pasek, M.A.; Dworkin, J.P.; Lauretta, D.S. A radical pathway for organic phosphorylation during schreibersite corrosion with implications for the origin of life. Geochim. Cosmochim. Acta 2007, 71, 1721–1736. [Google Scholar] [CrossRef]
- Pasek, M.A.; Kee, T.P. On the Origin of phosphorylated biomolecules. In Origins of Life: The Primal Self-Organization; Egel, R., Lankenau, D.-H., Mulkidjanian, A.Y., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 57–84. [Google Scholar]
- Schwartz, A.W. Phosphorus in prebiotic chemistry. Philos. Trans. R. Soc. B 2006, 361, 1743–1749. [Google Scholar] [CrossRef] [PubMed]
- Lombard, J.; López-García, P.; Moreira, D. The early evolution of lipid membranes and the three domains of life. Nat. Rev. Microbiol. 2012, 10, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Pohorille, A.; Deamer, D. Self-assembly and function of primitive cell membranes. Res. Microbiol. 2009, 160, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Joyce, G.F.; Schwartz, A.W.; Miller, S.L.; Orgel, L.E. The case for an ancestral genetic system involving simple analogues of the nucleotides. Proc. Natl. Acad. Sci. USA 1987, 84, 4398–4402. [Google Scholar] [CrossRef] [PubMed]
- Schneider, K.C.; Benner, S.A. Oligonucleotides containing flexible nucleoside analogs. J. Am. Chem. Soc. 1990, 112, 453–455. [Google Scholar] [CrossRef]
- Hud, N.V.; Cafferty, B.J.; Krishnamurthy, R.; Williams, L.D. The origin of RNA and “my grandfather’s axe”. Chem. Biol. 2013, 20, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Cooper, G.; Kimmich, N.; Belisle, W.; Sarinana, J.; Brabham, K.; Garrel, L. Carbonaceous meteorites as a source of sugar-related organic compounds for the early Earth. Nature 2001, 414, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, R.I.; Maity, S.; Jones, B.M. Synthesis of Prebiotic Glycerol in Interstellar Ices. Angew. Chem. Int. Ed. 2015, 54, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Gull, M.; Ge, T.; Yingwu, W.; Chao, H.; Zhan, S.; Hongming, Y.; Shouhua, F. Resolving the enigma of prebiotic C–O–P bond formation: Prebiotic hydrothermal synthesis of important biological phosphate esters. Heteroat. Chem. 2010, 21, 161–167. [Google Scholar]
- Epps, D.E.; Nooner, D.W.; Eichberg, J.; Sherwood, E.; Oró, J. Cyanamide mediated synthesis under plausible primitive earth conditions. VI. The synthesis of glycerol and glycerophosphates. J. Mol. Evol. 1979, 14, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Gull, M.; Pasek, M.A. Is struvite a prebiotic mineral? Life 2013, 3, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Gull, M.; Zhou, M.; Fernández, F.M.; Pasek, M.A. Prebiotic phosphate ester syntheses in a deep eutectic solvent. J. Mol. Evol. 2014, 78, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Pasek, M.A.; Harnmeijer, J.P.; Buick, R.; Gull, M.; Atlas, Z. Evidence for reactive reduced phosphorus species in the early archean ocean. Proc. Natl. Acad. Sci. USA 2013, 110, 10089–10094. [Google Scholar] [CrossRef] [PubMed]
- Saladino, R.; Crestini, C.; Costanzo, G.; Negri, R.; Di Mauro, E. A possible prebiotic synthesis of purine, adenine, cytosine, and 4(3H)-pyrimidinone from formamide: Implications for the origin of life. Bioinorg. Med. Chem. 2001, 9, 1249–1253. [Google Scholar] [CrossRef]
- Costanzo, G.; Saladino, R.; Crestini, C.; Ciciriello, F.; Di Mauro, E. Formamide as the main building block in the origin of nucleic acids. BMC Evol. Biol. 2007, 7 (Suppl. 2), S1. [Google Scholar] [CrossRef] [PubMed]
- Saladino, R.; Carota, E.; Botta, G.; Kapralov, M.; Timoshenko, G.N.; Rozanov, A.Y.; Krasavin, E.; Di Mauro, E. Meteorite-catalyzed synthesis of nucleosides and of other prebiotic compounds from formamide under proton irradiation. Proc. Natl. Acad. Sci. USA 2015, 112, E2746–E2755. [Google Scholar] [CrossRef] [PubMed]
- Gull, M. Prebiotic Phosphorylation Reactions on the Early Earth. Challenges 2014, 5, 193–212. [Google Scholar] [CrossRef]
- Saladino, R.; Crestini, C.; Pino, S.; Costanzo, G.; Mauro, E.D. Formamide and the origin of life. Phys. Life Rev. 2012, 9, 84–104. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, J.S.; Voecks, G.E.; Hobby, G.L.; Ferris, J.P.; Williams, E.A.; Nicodem, D.E. Ultraviolet-gas phase and -photocatalytic synthesis from CO and NH3. J. Mol. Evol. 1975, 5, 223–241. [Google Scholar] [CrossRef] [PubMed]
- Adande, G.R.; Woolf, N.J.; Ziurys, L.M. Observations of interstellar formamide: Availability of a prebiotic precursor in the galactic habitable zone. Astrobiology 2013, 13, 439–453. [Google Scholar] [CrossRef] [PubMed]
- Schoffstall, A.M. Prebiotic phosphorylation of nucleosides in formamide. Orig. Life Evol. B 1976, 7, 399–412. [Google Scholar] [CrossRef]
- Schoffstall, A.M.; Barto, R.J.; Ramos, D.L. Nucleoside and deoxynucleoside phosphorylation in formamide solutions. Orig. Life Evol. B 1982, 12, 143–151. [Google Scholar] [CrossRef]
- Costanzo, G.; Saladino, R.; Crestini, C.; Ciciriello, F.; Mauro, E.D. Nucleoside phosphorylation by phosphate minerals. J. Biol. Chem. 2007, 282, 16729–16735. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, Y.; Kim, H.-J.; Hutter, D.; Benner, S.A. Abiotic regioselective phosphorylation of adenosine with borate in formamide. Astrobiology 2015, 15, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Bada, J.L.; Chalmers, J.H.; Cleaves, H.J. Is formamide a geochemically plausible prebiotic solvent? Phys. Chem. Chem. Phys. 2016, 18, 20085–20090. [Google Scholar] [CrossRef] [PubMed]
- Burcar, B.; Pasek, M.; Gull, M.; Cafferty, B.J.; Velasco, F.; Hud, N.V.; Menor-Salván, C. Darwin’s warm little pond: A one-pot reaction for prebiotic phosphorylation and the mobilization of phosphate from minerals in a urea-based solvent. Angew. Chem. Int. Ed. 2016, 55, 13249–13253. [Google Scholar] [CrossRef] [PubMed]
- Abbott, A.P.; Harris, R.C.; Ryder, K.S.; Agostino, C.D.; Gladden, L.F.; Mantle, M.D. Glycerol eutectics as sustainable solvent systems. Green Chem. 2011, 13, 82–90. [Google Scholar] [CrossRef]
- Gállego, I.; Grover, M.A.; Hud, N.V. Folding and imaging of DNA nanostructures in anhydrous and hydrated deep-eutectic solvents. Angew. Chem. Int. Ed. 2015, 54, 6765–6769. [Google Scholar] [CrossRef] [PubMed]
- Ferris, J.P.; Ertem, G. Montmorillonite catalysis of RNA oligomer formation in aqueous solution. A model for the prebiotic formation of RNA. J. Am. Chem. Soc. 1992, 115, 12270–12275. [Google Scholar] [CrossRef]
- Ferris, J.P.; Hill, A.R.; Liu, R.; Orgel, L.E. Synthesis of long prebiotic oligomers on mineral surfaces. Nature 1996, 381, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Hazen, R.M. Paleomineralogy of the Hadean Eon: a preliminary species list. Am. J. Sci. 2013, 313, 807–843. [Google Scholar] [CrossRef]
- Hargreaves, W.R.; Mulvihill, S.J.; Deamer, D.W. Synthesis of phospholipids and membranes in prebiotic conditions. Nature 1977, 266, 78–80. [Google Scholar] [CrossRef] [PubMed]
- Gull, M.; Yu, W.; Yingwu, W.; Zhan, S.; Ge, T.; Shouhua, F. Mimicking the prebiotic acidic hydrothermal environment: One-pot prebiotic hydrothermal synthesis of glucose phosphates. Heteroat. Chem. 2011, 22, 186–191. [Google Scholar]
- Dawley, M.M.; Scott, A.M.; Hill, F.C.; Leszczynski, J.; Orlando, T.M. Adsorption of formamide on kaolinite, a combined IR theoretical and experimental study. J. Phys. C 2012, 116, 23981–23991. [Google Scholar]
- Dai, Y.; Van Spronsen, J.; Witkamp, G.J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Analyt. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Pasek, M.A.; Kee, T.P.; Bryant, D.E.; Pavlov, A.A.; Lunine, J.I. Production of potentially prebiotic condensed phosphates by phosphorus redox chemistry. Angew. Chem. Int. Ed. 2008, 47, 7918–7920. [Google Scholar] [CrossRef] [PubMed]
- Bhuiyan, M.I.H.; Mavinic, D.S.; Beckie, R.D. A solubility and thermodynamic study of struvite. Environ. Technol. 2007, 28, 1015–1026. [Google Scholar] [CrossRef] [PubMed]
- Handschuh, G.J.; Lohrmann, R.; Orgel, L.E. The effect of Mg2+ and Ca2+ on urea-catalyzed phosphorylation reactions. J. Mol. Evol. 1973, 2, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, R.; Guntha, S.; Eschenmoser, A. Regioselective α-phosphorylation of aldoses in aqueous solution. Angew. Chem. Int. Ed. 2000, 39, 2281–2285. [Google Scholar] [CrossRef]
- Singh, B.; Lobo, H.; Shankarling, G. Selective N-alkylation of aromatic primary amines catalyzed by bio-catalyst or deep eutectic solvent. Catal. Lett. 2011, 141, 178–182. [Google Scholar] [CrossRef]
- Austin, S.M.; Waddell, T.G. Prebiotic synthesis of vitamin B6-type compounds. Orig. Life Evol. Biosph. 1999, 29, 287–296. [Google Scholar] [CrossRef]
- Miller, S.L.; Schlesinger, G. Prebiotic syntheses of vitamin coenzymes: I. cysteamine and 2-mercaptoethanesulfonic acid (coenzyme M). J. Mol. Evol. 1993, 36, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Pasek, M.A. Rethinking early Earth phosphorus geochemistry. Proc. Natl. Acad. Sci. USA 2008, 105, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2003, 1, 70–71. [Google Scholar] [CrossRef]
- Abbott, A.P.; Boothby, D.; Capper, G.; Davies, D.L.; Rasheed, R.K. Deep eutectic solvents formed between choline chloride and carboxylic acids: Versatile alternatives to ionic liquids. J. Am. Chem. Soc. 2004, 126, 9142–9147. [Google Scholar] [CrossRef] [PubMed]
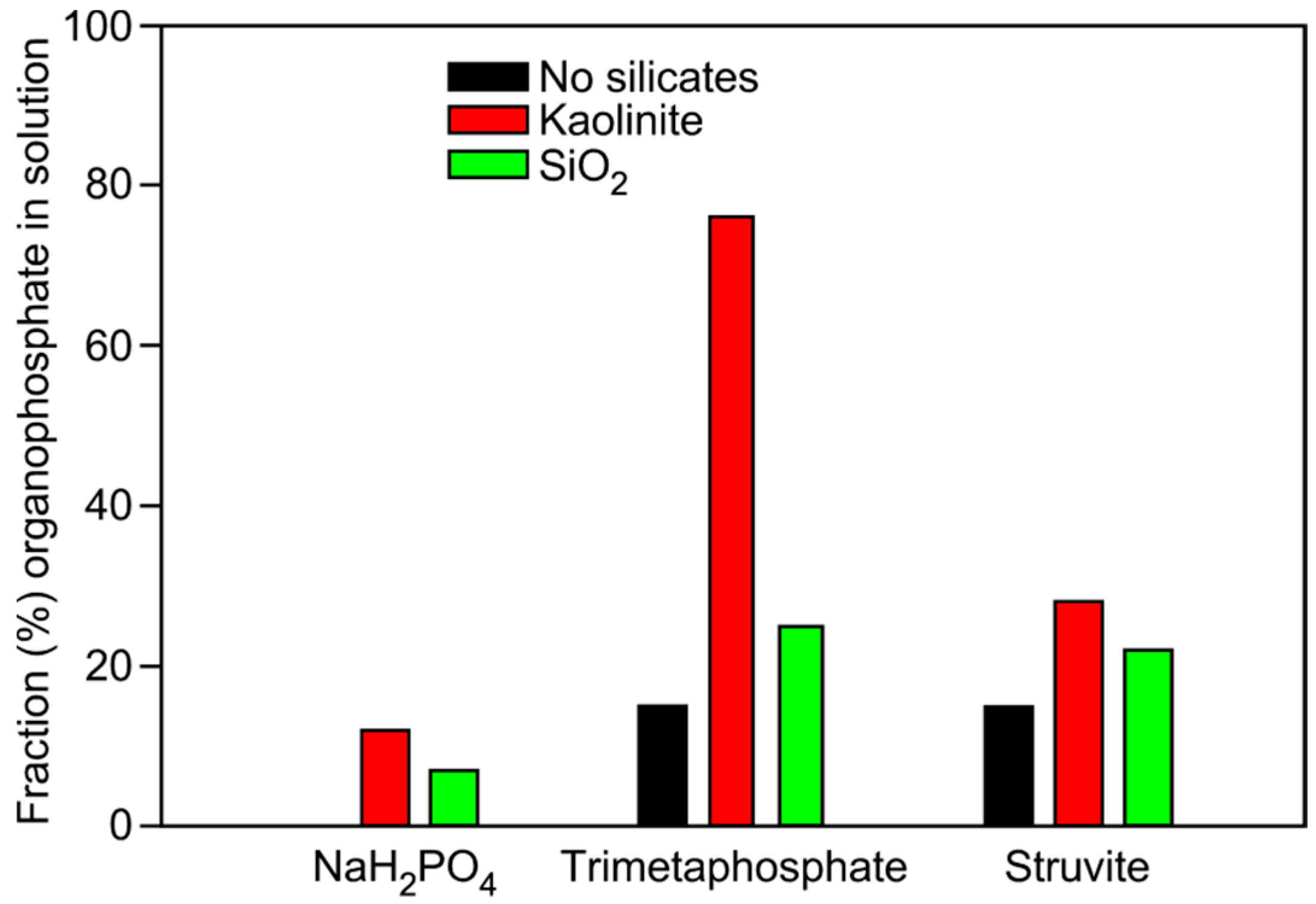
| Phosphate Source | Mineral Added | Glycerol-1-phosphate | Choline-phosphate | Glycerol-2-phosphate | Glycerol-1,2-cyclic phosphate | Total |
|---|---|---|---|---|---|---|
| NaH2PO4 | None | 0 | 0 | 0 | 0 | 0 |
| NaH2PO4 | Kaolinite | 8 | 3 | 1 | 0 | 12 |
| NaH2PO4 | Qtz Sand | 7 | 0 | 0 | 0 | 7 |
| TMP | None | 11 | 0 | 4 | 0 | 15 |
| TMP | Kaolinite | 35 | 10 | 12 | 5 | 76 |
| TMP | Qtz Sand | 18 | 3 | 3 | 1 | 25 |
| Struvite | None | 8 | 5 | 2 | 0 | 15 |
| Struvite | Kaolinite | 21 | 4 | 3 | 0 | 28 |
| Struvite | Qtz Sand | 12 | 10 | 0 | 0 | 22 |
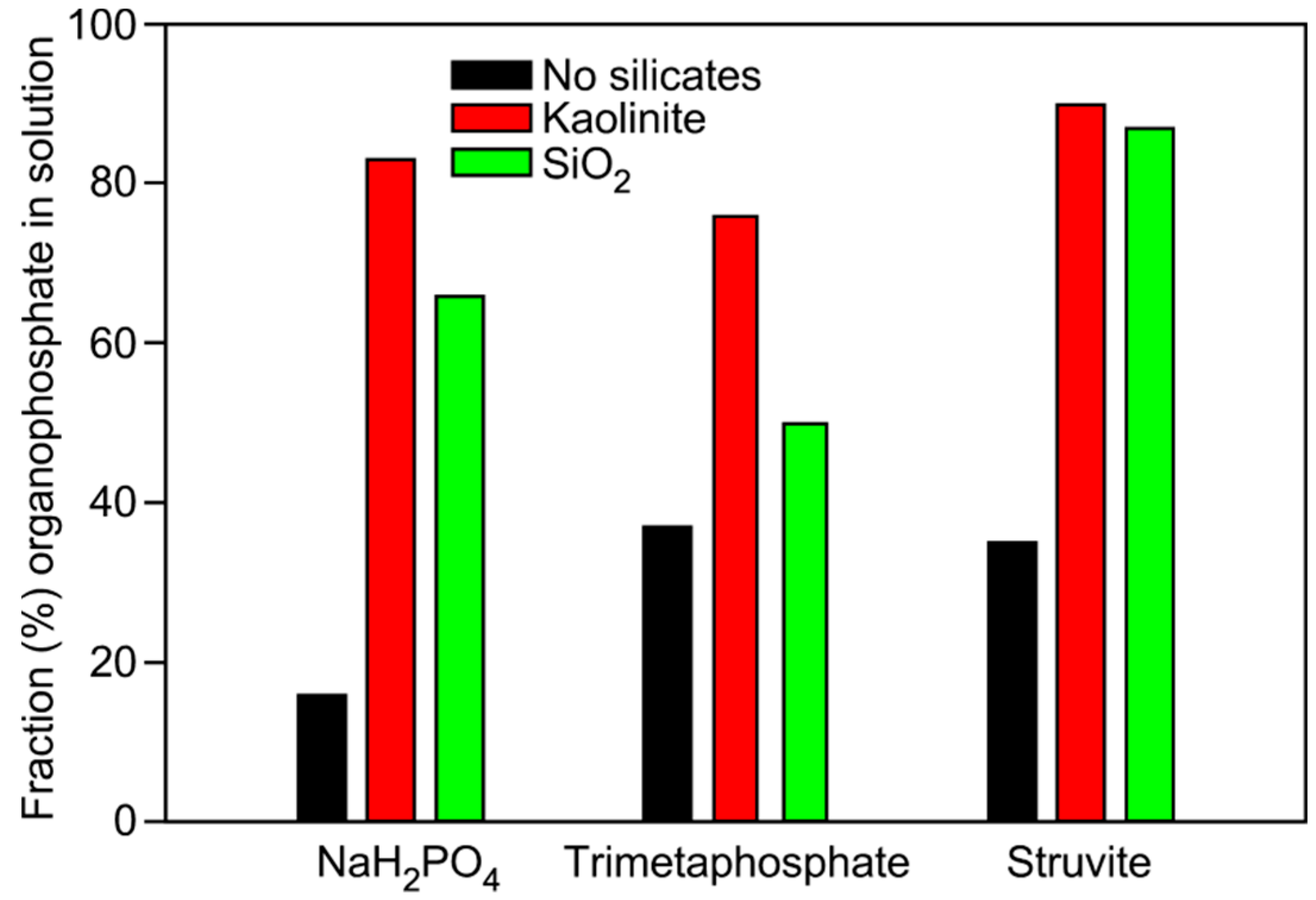
| Phosphate | Mineral Added | Glycerol-1-phosphate | Glycerol-2-phosphate | Glycerol-1,2-cyclic phosphate | Total |
|---|---|---|---|---|---|
| NaH2PO4 | None | 12 | 4 | 0 | 16 |
| NaH2PO4 | Kaolinite | 70 | 13 | 0 | 83 |
| NaH2PO4 | Qtz Sand | 56 | 10 | 0 | 66 |
| TMP | None | 35 | 2 | 0 | 37 |
| TMP | Kaolinite | 76 | 0 | 0 | 76 |
| TMP | Qtz Sand | 38 | 11 | 1 | 50 |
| Struvite | None | 30 | 5 | 0 | 35 |
| Struvite | Kaolinite | 78 | 11 | 1 | 90 |
| Struvite | Qtz Sand | 75 | 9 | 2 | 87 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gull, M.; Cafferty, B.J.; Hud, N.V.; Pasek, M.A. Silicate-Promoted Phosphorylation of Glycerol in Non-Aqueous Solvents: A Prebiotically Plausible Route to Organophosphates. Life 2017, 7, 29. https://doi.org/10.3390/life7030029
Gull M, Cafferty BJ, Hud NV, Pasek MA. Silicate-Promoted Phosphorylation of Glycerol in Non-Aqueous Solvents: A Prebiotically Plausible Route to Organophosphates. Life. 2017; 7(3):29. https://doi.org/10.3390/life7030029
Chicago/Turabian StyleGull, Maheen, Brian J. Cafferty, Nicholas V. Hud, and Matthew A. Pasek. 2017. "Silicate-Promoted Phosphorylation of Glycerol in Non-Aqueous Solvents: A Prebiotically Plausible Route to Organophosphates" Life 7, no. 3: 29. https://doi.org/10.3390/life7030029
APA StyleGull, M., Cafferty, B. J., Hud, N. V., & Pasek, M. A. (2017). Silicate-Promoted Phosphorylation of Glycerol in Non-Aqueous Solvents: A Prebiotically Plausible Route to Organophosphates. Life, 7(3), 29. https://doi.org/10.3390/life7030029






